Abstract
Epithelial–mesenchymal transition (EMT) is associated with cancer metastasis and poor prognosis, but the exact mechanism has not been clarified. Centrosomal Aurora-A kinase gene is frequently overexpressed in a variety of cancers and plays a pivotal role in the growth and survival of cancer cells. However, its role in colorectal cancer metastasis has not been confirmed. Here we demonstrate that Aurora-A plays a crucial role in the progression and metastasis of colorectal cancer by regulating epithelial–mesenchymal transition. In our study, increased Aurora-A expression was detected in colorectal cancer clinical specimens compared to normal colorectal tissues. Moreover, overexpressed Aurora-A significantly promoted the proliferation, migration, and invasion capacity of colorectal cancer cells and then enhanced metastatic capacity of colorectal cancer in vitro and in vivo and eventually led to poor prognosis. Conversely, silencing Aurora-A expression in colorectal cancer cells decreased the capacity of proliferation, migration, and invasion and further reduced colorectal cancer metastasis. Mechanistically, we found that Slug was involved in Aurora-A–induced migration and invasion of colorectal cancer cells. Silencing Slug expression could block Aurora-A–induced migration, invasion, and metastasis of colorectal cancer cells. Furthermore, the expression of Aurora-A and Slug were positively correlated in colorectal cancer tissues and paired normal colorectal tissue. Taken together, our findings revealed a critical role of Aurora-A in colorectal cancer progression and metastasis by regulating epithelial–mesenchymal transition.
Introduction
Colorectal cancer is the second most common cause of cancer-related deaths; more than 1.4 million patients are diagnosed with colorectal cancer every year. 1,2 Tumor invasion and metastasis are the major cause of poor prognosis. 3 Nowadays, therapeutic methods for metastatic colorectal cancer are limited and insufficient. Therefore, effort concentrated on understanding the molecular mechanisms of colorectal cancer is urgent, and tremendous research is needed to identify effective molecular markers for patients with colorectal cancer.
Increasing evidence reveals that epithelial–mesenchymal transition (EMT) is correlated with tumor metastasis and plays a crucial role in providing mobility to cancer cells. 4 –6 The major feature of EMT is profound phenotypic changes, including loss of epithelial characteristics and acquisition of a mesenchymal phenotype, which finally decreases cell–cell adhesion and acquires invasive capacity to migrate to distant sites. 7 However, little is known about the mechanisms of EMT in colorectal cancer. Identification of EMT regulator may help us to understand the signaling programs and finding new therapeutic targets for metastatic cancers.
Aurora-A kinase belongs to the mitotic Aurora serine/threonine kinase family 8,9 and regulates centrosome maturation and separation, mitotic entry, bipolar-spindle assembly, chromosome alignment on the metaphase plate, and cytokinesis. 10 Its ectopic expression results in abnormal centrosome amplification and cellular transformation 11 and finally causes genetic instability and transformation in tumor cells by disrupting the proper assembly of the mitotic checkpoint complex. 12 Recently, it is reported that the expression of Aurora-A is dysregulated in various human tumors, such as breast cancer, nasopharyngeal cancer, and leukemia. 13 –15 But the function of Aurora-A in tumor development and metastasis has not been fully studied, and more work is needed to clarify the mechanisms by which Aurora-A promotes tumor progression.
In our study, we explored the role of Aurora-A in regulating proliferation, EMT, and metastasis of colorectal cancer cells. We verified Aurora-A promoted colorectal cancer progression and metastasis by activating Slug expression. Our findings not only detected the metastasis mechanism of colorectal cancer but also confirmed Aurora-A might serve as a potential molecular marker for advanced colorectal cancer.
Materials and Methods
Cell Lines and Cell Culture
The normal human colon epithelial cell line HCoEpiC and human colon cancer cell lines HCT116, HT-29, LoVo, and DLD-1 were purchased from the American Type Culture Collection. All cells were cultured according to the manufacturer’s protocol. All the cell lines were grown at 37°C in a 5% CO2/95% air atmosphere.
Plasmid Construction and Retroviral Delivery
pBabe.puro retroviral construct containing human Aurora-A complementary DNA (cDNA) and pSuper.retro.puro with short hairpin RNA (shRNA) against human Aurora-A (5′-GTCTTGTGTCCTTCAAATT-3′) were prepared to enhance or decrease Aurora-A expression. Viral particles were produced by transfection of the plasmids into Phi-NX cells using Lipofectamine 2000 (Invitrogen). Infected cells were selected with 2 μg/mL puromycin, and pBabe.puro and pSuper.retro.puro vectors were used as control, respectively.
RNA Isolation and Real-Time Quantitative Reverse Transcription Polymerase Chain Reaction
RNA was isolated using the TRIZOL Reagent (Invitrogen, California, USA). First strand cDNA was synthesized with the SuperScript II Reverse Transcriptase (Invitrogen, California, USA). The primers for quantitative reverse transcription polymerase chain reaction (qRT-PCR) are: Aurora-A, forward: 5′-CAAGTCCCCTGTCGGTTCC-3′; reverse: 5′-ATCAAATATCCCCGCACTCTGG-3′. GAPDH, forward: 5′-CGGAGTCAACGGATTTGGTCGTAT-3′; reverse: 5′-AGCCTTCTCCATGGTGGTGAAGAC-3′.
The qRT-PCR was performed using the SYBR Green PCR Master Mix (Thermo, Massachusetts, USA), and data collection was performed with an ABI PRISM 7900HT sequence detection system.
Antibodies and Chemicals
Antibodies against E-cadherin, α-catenin, vimentin, N-cadherin, and Slug were from Cell Signaling Technology (Boston, USA), and anti-Aurora-A antibody was from Abcam (Cambridge, UK). Lipofectamine 2000 transfection was purchased from Invitrogen.
Histologic and Immunohistochemical Analyses
Forty patients were included in our study. Tumors (n = 40) and paired tumor adjacent normal tissues (n = 10) were obtained from the Department of Pathology, Qilu hospital of Shandong university (Jinan, China). All patients signed informed consent for the experiment, and our study was approved by the medical ethical committee of Shandong University. The mean age of the patients was 59 ± 18 years. There were 22 patients with adenocarcinoma, 5 patients with mucinous carcinoma, and 3 patients with undifferentiated carcinoma. Among the 40 patients, there were 15 patients with tumor distant metastasis.
The immunohistochemical (IHC) analysis of Aurora-A expression was performed using the avidin–biotin–peroxidase complex method. Citrate buffer (0.01 mol/L, pH 6.0) was used for antigen retrieval. The endogenous peroxidase activity was blocked by 3% hydrogen peroxide. Primary antibody for Aurora-A (1:50 dilution) was incubated for 16 hours at 4 °C. The operation of remaining steps was in accordance with the manufacturer’s protocol. Five randomly high-power (400×) views were selected in each slide, and then the average positive rate of the 5 views was examined to account the proportion of stained cells.
In Vivo Studies
Nude mice (5-week-old) were purchased from Shanghai Slac Laboratory Animal Co. Ltd (Shanghai, China) and maintained in microisolator cages. The current experiments of animals were approved by the Use Committee for Animal Care. For metastasis assays, 2 × 106 cells were resuspended in phosphate-buffered saline with a volume of 0.1 mL. The suspended LoVo-shAurora-A, HCT116-Aurora-A, and their control cells were injected into the tail veins (5 mice/group). After 40 days, the mice were killed by chloral hydrate. Liver and lung tissues were dissected out, serial sections were made, and examined by hematoxylin and eosin staining.
Cell Proliferation Assay
A density of 1 × 103 cells (HCT116-Vector, HCT116-Aurora-A, LoVo-Vector, and LoVo-shAurora-A) were seeded in 96-well plates in triplicate. 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT; Promega, Wisconsin, USA) was used to detect cell proliferation at appropriate time points. Light absorbance of the solution was measured at 570 nm on a microplate reader.
Scratch-Healing Assay
HCT116-Vector, HCT116-Aurora-A, LoVo-Vector, and LoVo-shAurora-A cells were seeded in 6-well plates, respectively, sterile plastic 200μl micropipette tips scratched the cell monolayers, and phase-contrast microscopy was used to photograph the cells. The migration distance of the cells was measured with Photoshop files.
Migration and Invasion Assay
HCT116-Vector, HCT116-Aurora-A, LoVo-Vector, and LoVo-shAurora-A cells were seeded on Boyden chamber (8-μm pore size; BD, New Jersey, USA) with or without a thin layer of MATRIGEL Basement Membrane Matrix for in vitro migration and invasion assays. The chambers were then placed on 24-well plates containing 10% fetal bovine serum acting as chemoattractant. After 24 hours incubation, cells were fixed with 4% formalin and stained with Giemsa staining solution. The unmigrated cells on the surface of the membrane were removed using cotton swabs. Images were taken and analyzed by 3 independent experiments.
Statistical Analysis
Statistical analysis was done with SPSS/Win17.0 software (SPSS Inc, Chicago, Illinois). Results are presented as mean ± standard deviation and analyzed by Student 2-tailed t test. The criterion of statistical significance was P < .05.
Results
Upregulation of Aurora-A Expression in Patients With Colorectal Cancer
To investigate the role of Aurora-A in colorectal cancer development, IHC staining was used to detect the expression of Aurora-A in adjacent normal colorectal tissues and colorectal cancer tissues with or without distant metastasis. Consistent with previous studies, 16,17 our study confirmed the upregulation of Aurora-A in colorectal cancer tissues compared to adjacent normal tissues (Figure 1A). Most importantly, clinicopathologic feature analysis showed that higher Aurora-A expression was significantly correlated with colorectal cancer distant metastasis (Figure 1A). Overexpressed Aurora-A messenger RNA level and protein level were also detected in 6 colorectal cancer tumors relative to adjacent normal tissues (Figure 1B and C). Then, we analyzed Aurora-A expression in normal colorectal cells and 4 colorectal cancer cell lines; the results showed that Aurora-A was highly expressed in colorectal cancer cell lines, especially in invasive cancer cell lines compared to normal cells (Figure 1D and E).
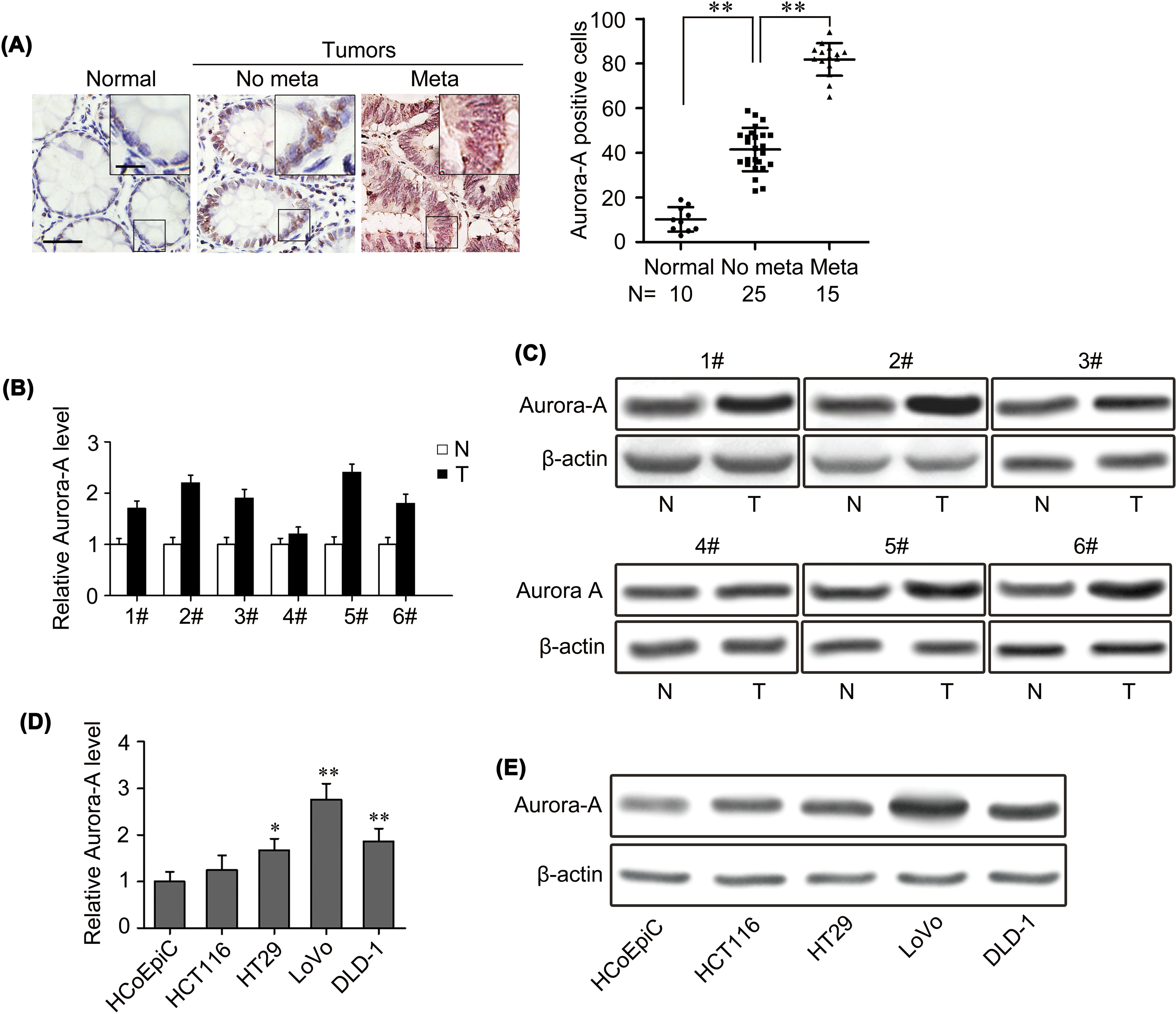
Aurora-A expression is upregulated in colorectal cancer. A, Representative images of Aurora-A immunohistochemical (IHC) staining in normal colorectal tissues and colorectal primary cancer tissues with or without metastasis. Normal, normal tissues; No meta, primary cancers without metastasis (in situ); Meta, primary cancers with metastasis. Scale bars: 50 μm and 20 μm in the insets. Corresponding semiquantification of Aurora A expression was shown in right panels. B and C, quantitative reverse transcription polymerase chain reaction (qRT-PCR; B) and Western blot (C) detected the expression of Aurora-A in 6 paired samples of colorectal cancer tissues versus normal colorectal tissues. N, Normal tissue; T, Tumor tissue. D and E, Aurora-A expression was analyzed by qRT-PCR (D) and Western blot (E) in normal colorectal cell and 4 human colorectal cancer cell lines.
Aurora-A Promotes Proliferative Capacity of Colorectal Cancer Cells
To ascertain the oncogenic activity of Aurora-A in colorectal cancer, we retrovirally established colorectal cancer cell lines stably silencing Aurora-A in LoVo cell (designated as LoVo-shAurora-A) and overexpressing Aurora-A in HCT116 cell (designated as HCT116-Aurora-A); the Aurora-A expression levels were quantified by qRT-PCR and Western blot (Figure 2A and B). Then, we evaluated the influence of Aurora-A on the proliferative capacity of colorectal cancer cells. As shown in Figure 2C and D, silencing the expression of Aurora-A significantly suppressed cell proliferation by MTT assay and generated less numbers and smaller colonies in colonies formation assay. In contrast, ectopic expression of Aurora-A dramatically increased cell proliferation and clonogenicity (Figure 2E and F).

Aurora-A promotes proliferative capacity of colorectal cancer cells. A and B, quantitative reverse transcription polymerase chain reaction (qRT-PCR) and Western blot analyzed the Aurora-A expression in the established cell lines. C-F, 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT) assay (C and E) and colony formation assay (D and F) detected cell proliferative capacity by silencing or overexpressing Aurora-A. *P < .05; **P < .01; based on the Student t test.
Aurora-A Promotes Migratory and Invasive Capacities of Colorectal Cancer Cells In Vitro
Migration and invasion is one of the characteristics of cancer cells, especially for malignant tumors. To test whether Aurora-A influences the migratory capacity of colorectal cancer cells, wound healing assay and Boyden chamber assay was carried out. The results showed that silencing Aurora-A expression in LoVo cells decreased the capacities of migration (Figure 3A and B), while overexpressing Aurora-A in HCT116 cells significantly increased migratory rate of colorectal cancer cells (Figure 3C and D). The similar results of invasion capacity were further confirmed with Matrigel chamber assay, silencing Aurora-A in LoVo cells reduced the degree of invasion (Figure 3B), while ectopic expression of Aurora-A in HCT116 cells dramatically enhanced the invasion capacity (Figure 3D). All these results revealed that Aurora-A acted as an oncogene influencing the migration and invasion behaviors in colorectal cancer cells.

Aurora-A promotes migratory and invasive capacity of colorectal cancer cells. LoVo-shAurora-A and HCT116-Aurora-A cells or control vector cells were subjected to wound healing assay (A and C), transwell migration assay (B and D, top), and Matrigel invasion assay (B and D, bottom). A and B, Decreased Aurora-A expression resulted in a inhibition in migratory and invasive ability of LoVo cells; quantification was carried out as a percentage of their vector controls. C and D, Overexpression of Aurora-A resulted in a significant increase in migratory and invasive ability of HCT116 cells; quantification was carried out as a percentage of their vector controls. Scale bars: 500 μm (A and C) and 50 μm (B and D). **P < .01; based on the Student t test.
Aurora-A Promotes Tumor Metastasis In Vivo
To determine whether Aurora-A regulates colorectal cancer metastasis in vivo, LoVo-shAurora-A, HCT116-Aurora-A, and their control cells were injected into the nude mice via tail vein, respectively. As expected, knockdown of Aurora-A in LoVo cells not only decreased the number of mice with distant metastasis (Figure 4A) but also decreased the number of metastatic tumors in lung and liver of each mouse (Figure 4B and C). In contrast, overexpression of Aurora-A in HCT116 cells dramatically enhanced the metastasis behavior, both in the number of mice with distant metastasis (Figure 4A) and the number of metastatic tumors in both lung and liver of each mouse (Figure 4D and E). These further demonstrate the pivotal role of Aurora-A in colorectal cancer progression.

Aurora-A promotes tumor metastasis of human colorectal cancer. A, The total number of mice with distant metastasis at 40 days after LoVo-Vector, LoVo-shAurora-A, HCT116-Vector, and HCT116-Aurora-A cells injected into tail vein. B and C, Representative images and the numbers of metastatic foci per section in lung (B) and liver (C) of individual mice injected with LoVo-Vector and LoVo-shAurora-A cells. D and E, Representative images and the numbers of metastatic foci per section in lung (D) and liver (E) of individual mice injected with HCT116-Vector and HCT116-Aurora-A cells. Scale bars: 50 μm (B-E) and 20 μm (insets in B-E). **P < .01 based on the Student t test.
Aurora-A Regulates the EMT Process in Colorectal Cancer Cells
Since EMT promotes the migratory and invasive potentials of cancer cells, the critical step of cancer metastasis, 18 we explored the role of Aurora-A on EMT. First, we analyzed the expression of E-cadherin, vimentin, and Aurora-A in colorectal cancer tissue and paired normal tissue. The results exhibited a significant loss of E-cadherin and an increased expression of vimentin and Aurora-A in colorectal cancer tissue compared to normal tissue (Figure 5A). In addition, we found that the expression of Aurora-A and E-cadherin were reversed in colorectal cancer tissues (Figure 5B), whereas the expression of Aurora-A was consistent with vimentin (Figure 5B). These results indicate that Aurora-A may be involved in the EMT of colorectal cancer.
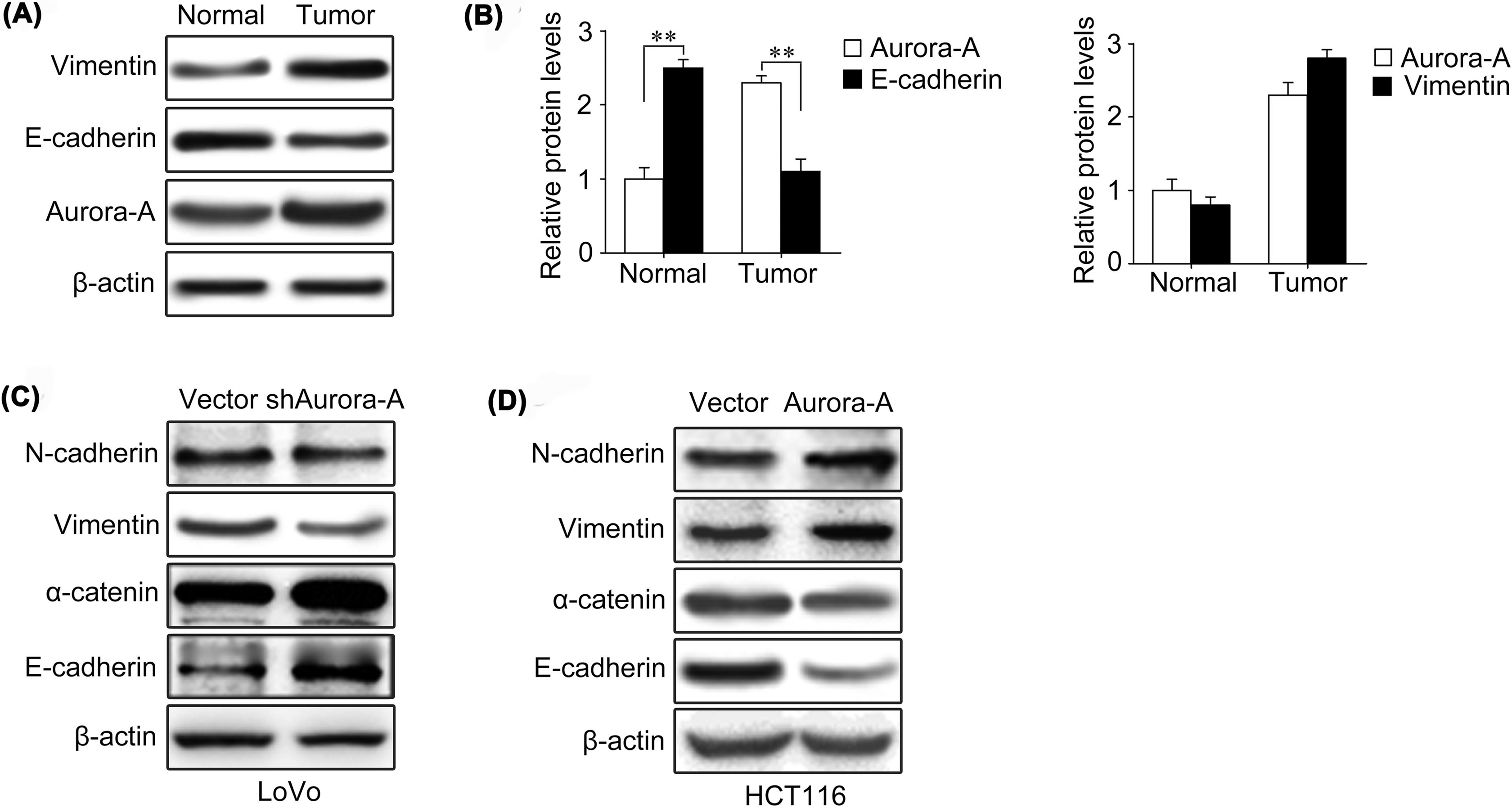
Aurora-A regulates the epithelial–mesenchymal transition (EMT) process in colorectal cancer cells. A, Expression of E-cadherin, vimentin, and Aurora-A was analyzed by Western blot in colorectal cancer tissue and paired normal tissues. B, Quantification analyses of the expression level between Aurora-A and E-cadherin and Aurora-A and vimentin. ** P < .01 based on the Student t test. C and D, Expression of epithelial and mesenchymal markers was analyzed by Western blot in the condition of silence or overexpression Aurora-A level.
To further verify the effect of Aurora-A on migration and invasion was mediated by EMT, we analyzed the epithelial and mesenchymal markers expression level on colorectal cancer cells. As shown in Figure 5C, silencing Aurora-A expression in LoVo cells increased the levels of epithelial markers (E-cadherin and α-catenin) and decreased the levels of mesenchymal markers (N-cadherin and vimentin). Conversely, overexpressed Aurora-A in HCT116 cells caused more mesenchymal markers expression (N-cadherin and vimentin), accompanied by decreased levels of epithelial markers (E-cadherin and α-catenin; Figure 5D).
Taken together, these findings suggest that Aurora-A may be an important regulator of EMT in colorectal cancer.
Slug Is Required for Aurora-A–Mediated EMT, Migration, and Invasion in Colorectal Cancer Cells
To better understand the mechanism of Aurora-A in regulating EMT and metastasis of colorectal cancer, we detected the expression of upstream transcription factors including Twist, Slug, and ZEB1, which play important roles in regulating EMT. The results showed that these EMT regulators were all decreased following Aurora-A knockdown in LoVo cells (Figure 6A) and were increased following Aurora-A overexpression in HCT116 cells (Figure 6B). These results indicate that Twist, Slug, and ZEB1 may be the target genes of Aurora-A in regulating EMT of colorectal cancer cells.

Slug-mediated Aurora-A–related migration and invasion of colorectal cancer cells. A and B, Western blot detected the protein levels of ZEB1, Slug, and Twist in LoVo-vector, LoVo-shAurora-A, HCT116-vector, and HCT116-Aurora-A cells. C, Silencing Slug restored the epithelial marker expression and decreased mesenchymal markers in HCT116-Aurora-A cells. D, Silencing Slug inhibited Aurora-A–induced migration and invasion in HCT116-Aurora-A cells.
Still now, the relation between Aurora-A and Slug in regulating EMT and metastasis have not been systematically investigated in colorectal cancer. To investigate the role of Slug in Aurora-A–mediated migration and invasion, we used shRNAs to knockdown Slug expression by transfecting HCT116-Aurora-A cells with Slug shRNA (Figure 6C). Slug deficiency in HCT116 cells led to increase in epithelial phenotypes expression and decrease in mesenchymal phenotypes expression at protein level (Figure 6C). Moreover, knockdown of Slug also reduced migratory and invasive capacities in HCT116-Aurora-A cells (Figure 6D). Collectively, the above-mentioned results imply that Slug may be required for Aurora-A–mediated EMT, migration, and invasion of colorectal cancer cells.
Discussion
Aurora-A plays an important role in maintaining the correct chromosome segregation during mitosis. It is overexpressed in variety of human cancers and associated with aneuploidy, high tumor grade, increased invasiveness, and poor prognosis. 19,20 Nowadays, great efforts have been made to illuminate the mechanism of Aurora-A in promoting cancer progression. 21 –23 Nevertheless, the detailed mechanisms between Aurora-A and colorectal cancer remain obscure.
In our study, we evaluated Aurora-A expression in colorectal cancer tissues and cell lines, we found that Aurora-A was overexpressed in colorectal cancer tissues and cell lines compared to nontumor tissues and cell lines. These results indicate that Aurora-A is related to colorectal cancer progression. Metastasis is general for patients with colorectal cancer which may cause poor prognosis. 24,25 Moreover, some studies reported that Aurora-A overexpression was closely correlated with metastasis in a variety of cancers, such as thyroid cancer, renal carcinoma, non-small cell lung cancer, and ovarian cancer. 26 –29 Therefore, we detected whether Aurora-A influences colorectal cancer progression and metastasis. Our study confirmed that ectopic expression of Aurora-A induced colorectal cancer cells proliferation, migration, and invasion and enhanced tumor progression and metastasis in vivo and in vitro. Conversely, silencing Aurora-A reversed cells proliferation and tumorigenesis capacity. Furthermore, our study clarified that Aurora-A promoted colorectal cancer invasion and metastasis by regulating EMT. Overexpressed Aurora-A significantly upregulated the expression of mesenchymal markers and decreased epithelial marker level in colorectal cancer cells; reduced Aurora-A expression reversed this effect. Our results are consistent with the reports that EMT plays a key role in tumor invasion and metastasis. 30,31
Mechanistically, the key finding of our study is that we detected the cross-talk between Aurora-A and Slug in promoting EMT, tumorigenesis, and metastasis in colorectal cancer. Slug is an important regulator of EMT and has been recognized as a transcriptional repressor of E-cadherin, which mediate the cell–cell contacts. 32 Some studies have reported the role of Slug in regulating EMT, migration, invasion, and metastasis in several types of cancer. 33 –35 Here we detected that the effect of Aurora-A in promoting colorectal cancer invasion and metastasis was mediated by Slug. Overexpression of Aurora-A could induce Slug expression, and knockdown of Slug could reverse Aurora-A–induced cell migration and invasion. This study suggested that Slug might be an important downstream target of Aurora-A. Our findings offer a potential therapeutic target and biomarker in colorectal cancer. However, the mechanisms regarding how Aurora-A regulates Slug expression in colorectal cancer has not been fully explored, and further studies are needed to investigate the detailed mechanisms.
In conclusion, our study suggests a pivotal role for Aurora-A in promoting EMT and tumor metastasis in colorectal cancer via regulating Slug expression. This provided a hypothesis for us that Aurora-A may serve as a potential molecular marker for colorectal cancer treatment and prognosis. Moreover, the observations that knockdown Slug expression dramatically blocked the tumor metastasis induced by Aurora-A provided us a therapeutic option by targeting Slug in Aurora-A overexpressed patients in clinical practice.
Footnotes
Abbreviations
Authors’ Note
Jin-Guang He and Luming Li contributed equally to this work
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
