Abstract
The purpose of this research is to establish a process of irradiating mice using the Gamma Knife as a versatile system for small animal irradiation and to validate accurate intracranial and extracranial dose delivery using this system. A stereotactic immobilization device was developed for small animals for the Gamma Knife head frame allowing for isocentric dose delivery. Intercranial positional reproducibility of a reference point from a primary reference animal was verified on an additional mouse. Extracranial positional reproducibility of the mouse aorta was verified using 3 mice. Accurate dose delivery was validated using film and thermoluminescent dosimeter measurements with a solid water phantom. Gamma Knife plans were developed to irradiate intracranial and extracranial targets. Mice were irradiated validating successful targeted radiation dose delivery. Intramouse positional variability of the right mandible reference point across 10 micro-computed tomography scans was 0.65 ± 0.48 mm. Intermouse positional reproducibility across 2 mice at the same reference point was 0.76 ± 0.46 mm. The accuracy of dose delivery was 0.67 ± 0.29 mm and 1.01 ± 0.43 mm in the coronal and sagittal planes, respectively. The planned dose delivered to a mouse phantom was 2 Gy at the 50% isodose with a measured thermoluminescent dosimeter dose of 2.9 ± 0.3 Gy. The phosphorylated form of member X of histone family H2A (γH2AX) staining of irradiated mouse brain and mouse aorta demonstrated adjacent tissue sparing. In conclusion, our system for preclinical studies of small animal irradiation using the Gamma Knife is able to accurately deliver intracranial and extracranial targeted focal radiation allowing for preclinical experiments studying focal radiation.
Keywords
Introduction
Targeted radiation therapy (RT) led to a paradigm shift in cancer therapy. Unfortunately, it remains difficult to model targeted RT in small animals. Early small animal studies of RT involved significant radiation dose to large portions of animals instead of conformal RT. 1,2 While treating tumor effectively, these studies are unable to provide useful information about localized tissue toxicities experienced in targeted RT. 3
As stereotactic body radiation therapy (SBRT) has become a paradigm in radiation treatment, 4 little is known about normal tissue response to large doses of conformal RT. The SBRT doses are fatal to small animals unless given conformally. Multiple dedicated systems have been developed to fill the need for conformal small animal RT. These include the small animal radiation research platform, 5,6 the micro-RT system, 7 the X-Rad 225CX, 8 and the GE RS120 micro-computed tomography (CT). 9 These have been effectively used in multiple small animal studies by Tuli et al, 10 Ford et al, 11 Baumann et al, 12 Kiehl et al, 13 and others. Unfortunately, many academic institutions lack such dedicated small animal irradiators.
We have reported on the use of CyberKnife for the delivery of conformal RT in mice. 14 A 3-dimensional (3D)-printed stereotactic body mold was used, and 3 fiducial marks were placed on the mold. The positional accuracy between mice was 1.41 ± 0.73 mm, which was added to the target volume to generate a planning target volume (PTV). Treatment times with CyberKnife are long as compared to Gamma Knife because CyberKnife requires beam-by-beam dose delivery, whereas Gamma Knife can deliver treatment dose without repositioning; this is significant if many mice have to be treated.
Using a stereotactic head frame, Gamma Knife delivers conformal and accurate doses to tumors with beams measuring 4, 8, and 16 mm in diameter. The planning process is straightforward, and a highly conformal plan may be developed in minutes. The small beams are suitable for small animals, as these beam sizes relative to mice are proportional to linac beam sizes relative to humans. Additionally, dose plans developed on Gamma Knife for mice are analogous to conformal radiation plans in humans. Thus, Gamma Knife is an ideal system used to develop an animal model for studying clinical patient treatment schema. In fact, Wiant et al 15 and Charest et al 16 have previously used Gamma Knife for small animal RT. Wiant et al 15 developed a restraint system with a surrogate fiducial system to be docked on the Gamma Knife head frame for such studies. In a similar spirit, we developed an adaptor for Gamma Knife Perfexion (Manufactured by Elekta AB, Stockholm, Sweden) and a target positioning system with laser guidance for small animal irradiation by devising a stereotactic body immobilizer using a 3D printer for the treatment of mice, demonstrating positional reproducibility of this immobilizer, designing the adaptor for adaptable use for different tumor locations (implanted under the skin) without additional image-based planning, validating dose delivery using a phantom for radiation quality assurance (QA), and delivering intracranial and extracranial conformal RT to mice as a model for normal tissue radioprotective studies.
Materials and Methods
Developing a Stereotactic Base Plate and Body Mold System
An Institutional Animal Care and Use Committee protocol (protocol #2013-0063) was approved prior to initiating animal experiments. We previously developed a custom stereotactic immobilization device with an accuracy of 2 mm 14 and now demonstrate an improved device with an accuracy of 1 mm. The new device immobilizes the mouse with a spring-loaded bite block as the mouse is stretched to full extension in prone position (Figure 1). This body frame was created using a body contour developed with the in-house UNIVIEW Version 2.0 software. The body contour in a Digital Imaging and Communications in Medicine (DICOM)-RT format was exported and converted using the software to stereolithography format. This contour was smoothened, and the rest of the structures (bite block and base plate) were created using Solidworks Professional Version to ensure intermouse utility. The mold was then printed on a 3D printer (Project3000; Manufactured by 3D Systems, Rock Hill, South Carolina), which has a resolution of 0.068 × 0.068 × 0.38 mm (x, y, z) in 32-μm layers and an accuracy of 0.025 to 0.05 mm per 2.54 cm. The material used was an ultraviolet curable acrylic plastic, which was a solid acrylate-based photopolymer resin (USP Class VI certified for approved medical applications, ∼$250/kg). Printing time was several hours (unattended).

This stereotactic system consists of the following 3 major parts: a body mold having 4 holes for mouse legs, a base plate where the body mold locks on, and a spring-loaded bite piece. After positioning a mouse in the body mold, the mouse’s upper teeth are hooked to the bite piece. Finally, the body mold is locked on to the base plate. Except the spring and bite ring, all parts are printed out using a 3D printer.
Positional Reproducibility of Intracranial and Extracranial Immobilization
For imaging on micro-CT (X-SPECT; Gamma Medica-Ideas, Inc, Northridge, California), the base plate was taped down on the CT couch, so all imaged mice were in the same coordinate space. In positioning each mouse to be scanned, only the body mold was detached from the base plate.
To verify intracranial immobilization, a single mouse was placed in the stereotactic device and was scanned in the micro-CT scanner. The mouse was removed and repositioned into the frame 9 more times with repeat scanning to assess reproducibility. The most superior point of the right mandible as a surrogate for the right brain hemisphere was selected on 10 micro-CT scans, and the deviation from the original reference image was calculated. Intermice positional accuracy was measured with 2 different mice (22 and 29 g) placed in the stereotactic immobilizer, and micro-CT scans were obtained. The micro-CT scans were registered based on the frame. The most superior point of the right mandible was selected on each of the 3 micro-CT scans, and the deviation from the position on the first mouse reference point was measured to assess the positional setup error in irradiating the right hemisphere of the brain.
To verify extracranial immobilization, 3 different mice (16.9, 20.0, and 24.9 g) were placed in the stereotactic body mold and were scanned in the micro-CT scanner. The mouse aorta was contoured on micro-CT scans of these mice. These micro-CT images were rigidly fused to the micro-CT images from the middle-weighted mouse using the skin surface at a distance of 43 mm from the reference point on the body frame as the point of fusion as a surrogate for the distal aorta at the level of the iliac bifurcation.
Development of Adapter for Stereotactic Base Plate to Gamma Knife Perfexion System
The stereotactic base plate and mold were docked to a uniquely designed adapter to fit the Gamma Knife Perfexion system to serve multiple purposes: provide a self-enclosed system in the event that a small animal was to awaken during the midst of radiation delivery, provide isolation of small animal secretions from the Gamma Knife, connect to the head frame locking mechanisms of the Gamma Knife, and provide a laser-guided mechanism and 3D stage to accurately deliver radiation to a stereotactic point on the mouse body in a methodology analogous to linear accelerator isocentric setup.
This adapter was custom built using 1/4-inch acrylic (Figure 2A). The laser-guided system was composed of 3 lasers (2 lateral and 1 vertex) to serve as an isocenter for radiation delivery. To verify the laser convergence to a point, a QA phantom was designed (Figure 2B).

A transparent box built out of 1/4-inch acrylic is attached to Gamma Knife Perfexion couch. The transport box functions to attach to the Perfexion system and contains a 3D stage placed in the box to accommodate the stereotactic system that holds the mouse. This stereotactic system is composed of the base plate that attaches to the 3D stage, the body mold that holds the mouse in place, and the bite block that provides extra immobilization (A). This laser-guided 3D stage has an aluminum frame and is equipped with 3 lasers (2 lateral and 1 vertex) and a battery pack with laser beam collimated to a 1-mm diameter (B).
Planning and Dose Delivery Process
After scanning individual mice in the base plate on the micro-CT scanner, DICOM-formatted images were imported into the Gamma Knife planning system. Since we could not define an image set in the planning system without the head frame in the image and the frame would not fit in the micro-CT scanner, DICOM images were read into the planning system as a preplan image set.
Radiation shots were placed to develop a conformal treatment plan, but this plan could not be exported directly to the Gamma Knife patient positioning system (PPS), since the head frame was not defined in the image set. Thus, the shot coordinates were not directly transferable. Instead, a reference point in the image was selected (typically on the frame but not restricted to it). The relative distance in x, y, and z was recorded between shot (a) and the reference point (s). In the Gamma Knife PPS, we adjusted the 3D stage in our customized box in the x, y, and z directions by dialing along those axes, so that the reference point coincided with the laser-guided system. The laser-guided system position was fixed to the box, so that it was designed to be coincident with a coordinate of (100, 100, 0) in the PPS. For the treatment, the actual shot(s) was(were) manually created in physics mode by typing the shot coordinates. The shot(s) coordinate(s) in PPS coordinate(s) was(were) calculated relative to the point of the laser-guided system.
Validation of Dose Delivery
For dosimetric and positional verification testing, we developed 2 cylindrical mouse phantoms out of solid water (Figure 3A), and setup is shown. (Figure 3A) The dimensions of each phantom were 9 cm in length and 2.7 cm in diameter. The first phantom was cut across the longitudinal axis along the diameter to serve as a tool for QA of coronal and sagittal dose delivery. The second phantom was cut across the transverse axis at 2.5 cm from the base surface to serve as a tool for QA of accurate axial dose delivery. Both thermoluminescent dosimeters (TLDs) and radiographic films were inserted into the phantoms to accurately measure dose as previously described. Sample films are shown in Figure 3C. 14 For the treatment planning, we verified the micro-CT data and measured Hounsfield number versus density in our previous publication. 14 Using our UNIVIEW software, we resampled micro-CT images to make them compatible with normal clinical image format (512 × 512 pixels).
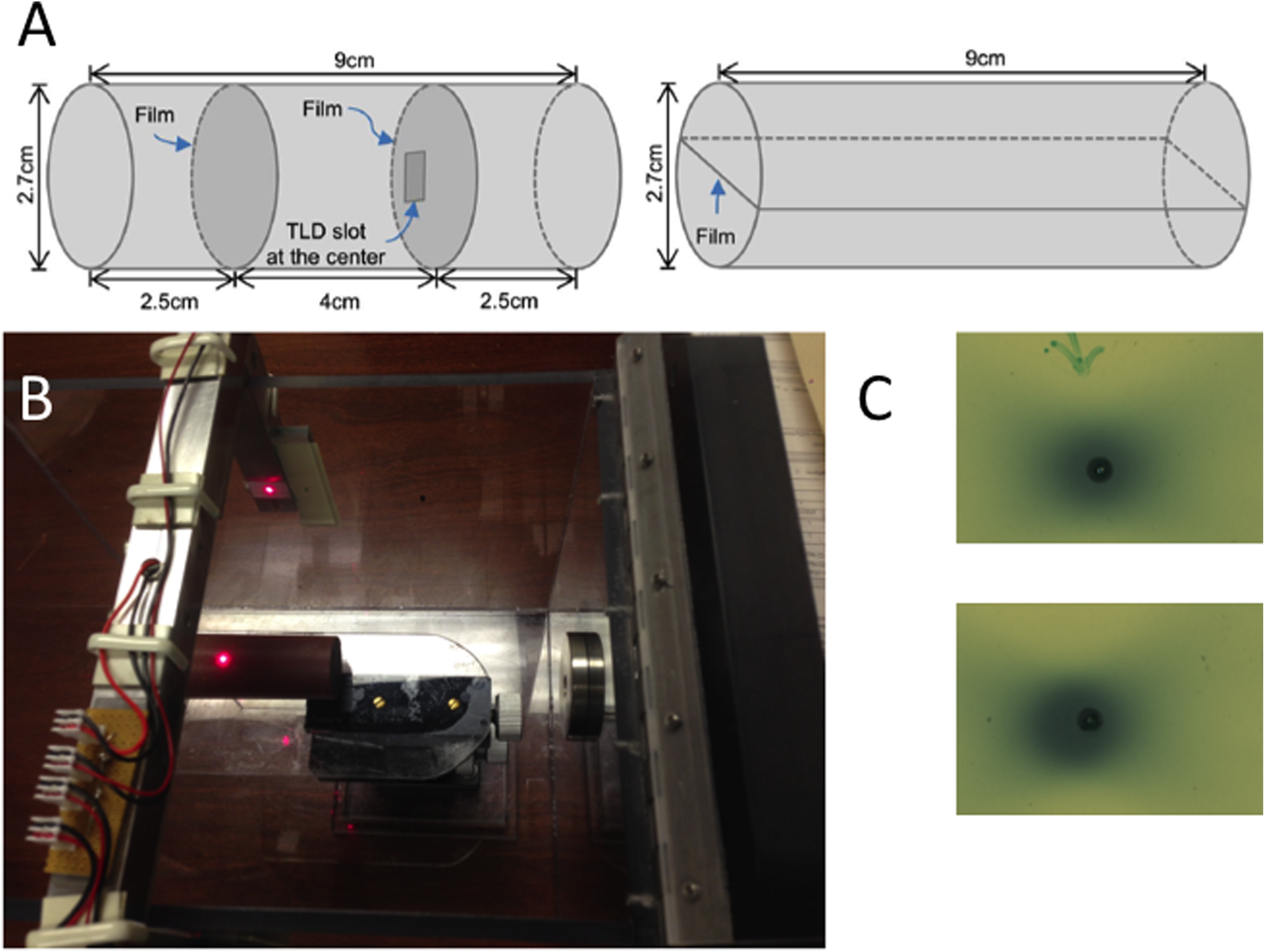
A, The schematic of cylindrical mouse phantoms used for quality assurance (QA). The phantom on the left allows for placement of film or thermoluminescent dosimeter (TLD) in an axial plane, and the phantom on the right allows for film placement in coronal or sagittal planes. B, This figure shows the phantom setup in the transport box. C, Films showing 2 examples of sample shot accuracy. The 4-mm shot was placed on the center of phantom, and the laser was set to the pinprick mark on the film.
Absolute dose verification was performed with dose delivery through the Gamma Knife. A CT scan of the solid water phantom was obtained. All dose calculations used the TMR 10 algorithm implemented in the planning system. We placed an 8-mm shot of radiation to a total dose of 2 Gy at the 50% isodose to the position corresponding to the TLD in each phantom. This was repeated 10 times to ensure accurate dose delivery. We also created a TLD response curve relative to various doses in order to validate our measurement.
Positional reproducibility of dose delivery was performed by measuring radiochromic films. For both axial and sagittal positional verification, 10 films each were placed in the phantom plane to verify positional delivery of dose. Using the adaptor’s laser-guided system, we matched the laser to the center of a 1-mm hole placed in the center of each film (Figure 3B). We placed an 8-mm shot of radiation to a total dose of 2 Gy at the 50% isodose to the position corresponding to the laser center.
Accurate In Vivo Intracranial and Extracranial Delivery of Conformal Radiation
In order to demonstrate the feasibility of using our system for conformal radiation delivery, we conducted an intracranial and extracranial in vivo study. For intracranial delivery, an 8-mm shot was delivered to the right hemisphere of the brain of 1 mouse to a dose of 3.0 Gy at the 50% isodose. The mouse was killed within 30 minutes of dose delivery, and the phosphorylated form of member X of histone family H2A (γH2AX) staining was performed. Similarly, for extracranial delivery, an 8-mm shot of radiation to a total dose of 14 Gy at the 50% isodose was delivered to the distal aorta at the level of the iliac bifurcation of 1 mouse. The mouse was killed within 30 minutes of dose delivery, and the γH2AX staining was performed.
Results
Positional Reproducibility of Cranial Immobilization
For intramice positional accuracy, the most superior point of the right mandible as surrogate for the right brain hemisphere was selected on each of the 10 micro-CT scans, and the deviation from the original reference image was calculated at a mean of 0.65 ± 0.48 mm. The most superior point of the right mandible was selected on each of 3 micro-CT scans, and the deviation from the position on the first mouse was calculated at a mean of 0.76 ± 0.46 mm.
Positional Reproducibility of Extracranial Immobilization
Figure 4A shows spinal alignment between 2 mice after image fusion at the skin surface 43 mm from the head holder to assess reproducibility of extracranial immobilization at the level of the mouse aorta in a sagittal plane. The difference in central aortic depth from the skin surface at midline at a distance of 43 mm from the reference point of the body between the 3 mice was measured at a maximum of 0.75 mm. The difference in the craniocaudal distance from the reference point of the body frame and the most superior part of the distal aorta between the lightest and heaviest mouse was measured at 3.90 mm. The fused aortic contours are visualized in Figure 4B, showing excellent interanimal reproducibility for extracranial positioning and a generated PTV used for treatment delivery.
A, The spinal alignment between 2 mice after image fusion at the skin surface 43 mm from the head holder. Despite different weights, the lumbar spines, which are at the level of the radiation target, are relatively well aligned, and fusion to the skin surface can be used for radiation delivery at this level. Although the thoracic spine does not appear to be well aligned, the radiation target is not at this level. B, The image fusion of the 3 mouse aorta contours (pink, blue, and green) on the body of 1 mouse as well as a generated PTV derived from a uniform expansion upon the union of the contours (teal). PTV indicates planning target volume. Note: The color version of the figure is available at journals.sagepub.com/home/tct
Accuracy of Laser-Guided Radiation Delivery
Phantom measurements using films were taken in coronal and sagittal planes for positional verification. Mean deviation from the radiation center to the laser-guided setup point in the coronal plane was 0.67 ± 0.29 mm and in the sagittal plane was 1.01 ± 0.43 mm.
Dosimetric Verification
Using the phantom, 2 Gy doses at the 50% isodose were delivered to the TLD placed in the axial plane as planned on the Gamma Knife. The dose enclosing the TLD was 3.8 Gy. The mean measured TLD dose was 2.9 ± 0.3 Gy.
In Vivo Verification of Dose Delivery
The γH2AX staining of the irradiated mouse brain at 3.0 Gy is shown in Figure 5 and of the irradiated mouse distal aorta at 14.0 Gy is shown in Figure 6, demonstrating effective 8-mm shot delivery to small animals in the planned treatment position.

The phosphorylated form of member X of histone family H2A (γH2AX) staining of an irradiated brain demonstrates hemibrain targeting. Immediately following 3 Gy of irradiation with Gamma Knife, an animal was killed and the brain was harvested for immunohistochemistry. The γH2AX staining revealed nuclear foci in the right hemibrain (A) but not in the left hemibrain (B).
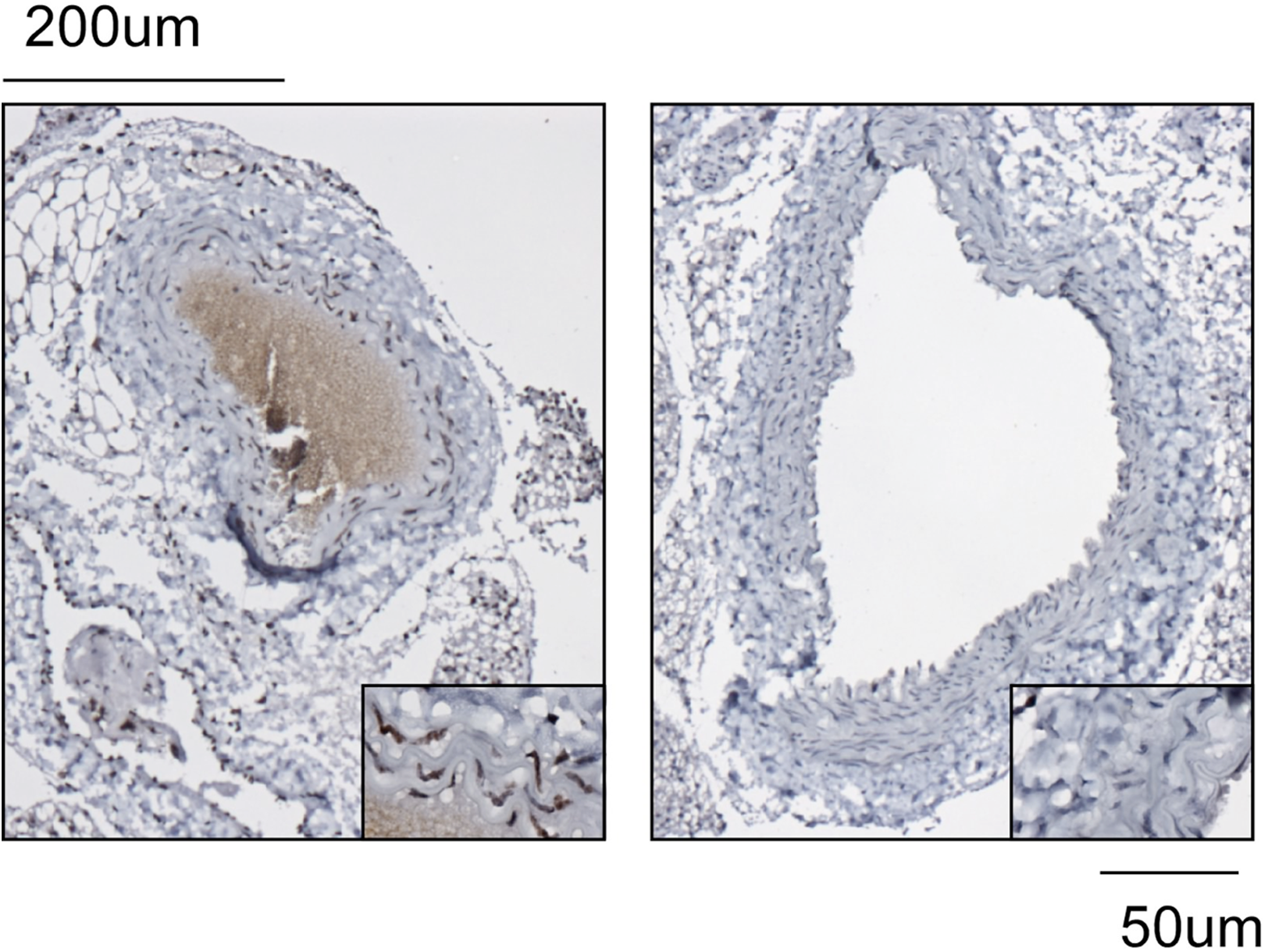
Immunochemistry for the phosphorylated form of member X of histone family H2A (γH2AX) reveals intense nuclear staining after delivery of 14 Gy to the mouse distal aortic wall (A) but not the unirradiated proximal aortic wall (B).
Discussion
We present a precise, reproducible, and versatile application of the Gamma Knife Perfexion for the development of models studying radiation effects in small animals for preclinical studies. For worldwide clinical centers without dedicated small animal irradiators, this system is a comparable tool producing experimentally relevant conformal treatment plans.
Similarly, weighted mice were successfully irradiated using the same setup to the same region. This versatility can be used for normal organ studies as the same target organs can be mapped out once and the same radiation plan can be delivered to the same organ in multiple mice. Any minor positional adjustments can be made using the 3D stage similar to daily positional setup for clinical treatments. A single plan can be used for subcutaneous or orthotopic tumor injections if tumor sizes are the same. Even if the location of tumor varies, beams can be guided by the laser system by adjusting the 3D positioning stage. Delivering accurate radiation doses to a tumor and sparing normal structures as opposed to conventional larger field animal irradiation allow the study of long-term effects of localized radiation without immediately threatening the life of the small animal. Additionally, if the mice are of drastically different weights or for use with other small animals, custom immobilizers can be easily developed using the 3D printing technique described, and individualized treatment plans for each immobilization system can be developed.
We previously reported the design of a system using CyberKnife for small animal RT. 14 Unlike the CyberKnife system, Gamma Knife planning is much faster and does not require the computationally intensive inverse planning or long delivery of CyberKnife. Single shots can be placed to encapsulate tumors or conform around tumors using the Gamma Knife. Furthermore, our system uses an isocentric-based planning system mimicking a linear accelerator setup. Unlike the CyberKnife, which restricts planning relative to fiducial markers, our device allows for changes in tumor location by dialing the 3D stage, allowing for fast and accurate delivery of radiation to multiple different small animals. This is more readily adaptable than the CyberKnife for subcutaneous or orthotopic tumor injection.
Conversely, a limitation of our system is a reliance on an external laser-guided system for radiation delivery as opposed to anatomic imaging. The upcoming Gamma Knife Icon has a cone beam CT for image guidance, which will further improve the accuracy of radiation delivery. This additional technology may allow elimination of the complicated 3D stage and laser-guided system from our device, but a simpler stage using the stereotactic base plate and body mold would still be necessary.
Although we used a micro-CT scanner, it is not necessary for this process but was used for finer dose and image resolution. The small field of view available on most CT scanners for brain scanning may be used instead. This is readily available on any clinical scanner.
Small animal irradiation with megavoltage irradiation requires special considerations from the physics perspective. The ability to deliver adequate dose was verified using solid water phantoms. The measured dose from our TLD experiments (2.9 Gy) was lower than the planned dose, enclosing the TLD (3.8 Gy). This discrepancy likely originates from the differences in attenuation due to the transparent box (measured at 5.7%), enclosing the system and a smaller phantom relative to a human head. Currently, we are working on developing a system for Monte Carlo dose calculation by modeling Perfexion with our treatment adaptor.
Secondary scattered radiation may cause significant dose delivery to normal tissues, particularly in small animals for which dose falloff is not insignificant relative to organ size. However, we were able to demonstrate significant dose falloff as verified by immunohistochemical studies demonstrating adequate tissue sparing of the contralateral brain hemisphere and proximal portion of the mouse aorta in our in vivo studies.
Three recent studies have reported the use of stereotactic radiosurgery for irradiation of small animals in preclinical studies. A study from Washington University showed effective intracranial single-hemisphere delivery of radiation to healthy mice as a model for radiation necrosis. 17 The details of the couch designed for attachment were not elaborated in the report from Washington University, so comparisons between systems are limited. The authors did report successful intracranial radiation using their system, but unlike our study, the study did not show successful targeted extracranial radiation. A study from Sherbrooke University reported their methodology and application of Gamma Knife to target rat brains. 18 They used the methodology described by Charest et al. 16 In that methodology, a mold was made for the mouse head, and radiation was delivered to a fixed point in the frame, which corresponded to the mouse brain. In our system, the irradiation position can be dynamically adjusted using the laser-guided 3D stage to deliver radiation to any area of the mouse body. Thus, our system is more adaptable. Finally, a study from the University of Florida used frameless stereotactic radiosurgery for the treatment of intracranial tumors in dogs. 19 In their methodology, a frameless system was used for much larger animals (dogs). This was done using conventional linear accelerator-based planning rather than the Gamma Knife system. This methodology would be difficult to apply to small animals such as mice.
In conclusion, we have developed a solution for small animal irradiation using Gamma Knife allowing for conformal, reproducible, and efficient radiation delivery for preclinical studies without a dedicated small animal irradiator.
Footnotes
Abbreviations
Acknowledgments
The authors would like to acknowledge George Daher for his technical advice that assisted in the development of our image guidance using laser technology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was received through philanthropic donations from the Kimble Foundation, the DC Austin Foundation, the Case Comprehensive Cancer Center, and the Adult Brain Tumor Foundation.
