Abstract
Adjuvant platinum-based chemotherapy has developed its stability as the first-line treatment in advanced non-small cell lung cancer (NSCLC). The objective of this study was to investigate the prognostic value of meningioma-associated protein (MAC30) on adjuvant platinum-based chemotherapeutic response and survival in patients with NSCLC. A total of 174 retrospective stage III B to IV Chinese patients with NSCLC were enrolled in the study. Among all cases, 85 patients were given platinum-based chemotherapy and another 89 patients received molecularly targeted therapy. The expression of MAC30 in tumor samples was confirmed via immunohistochemical staining to correlate with the therapeutic response and survival of patients. Patients having NSCLC with MAC30 overexpression showed a poorer response to platinum-based chemotherapy, while there was no prognostic value of MAC30 expression on molecularly targeted therapy. Further, patients receiving platinum-based chemotherapy with enhanced MAC30 expression exhibited shorter survival. A multivariate analysis exhibited that increased MAC30 expression was an independent prognostic factor for overall survival in patients having NSCLC with platinum-based chemotherapy. In conclusion, patients having NSCLC with higher MAC30 expression resisted to platinum-based chemotherapy and exhibited worse survival.
Keywords
Introduction
Non-small cell lung cancer (NSCLC) accounts for 75% to 80% of lung cancer 1 and holds the first leading cause of malignancy-related deaths. 2 Indeed, upon first diagnosis, most patients with NSCLC were placed at advanced III B or IV stage cancer according to the diagnostic criteria. 3 Nowadays, it is widely accepted that patients with advanced local invasion and distant metastasis were appropriate to be candidates for systemic chemotherapy based on platinum as the standard first-line treatment. 4 -6 Actually, adjuvant platinum-based chemotherapy was established as a standard care in patients with NSCLC and was shown to prolong survival in patients with a performance status 0 to 2. 7 On the other hand, only 40% of patients achieved the effective clinical response to chemotherapy because of the undesirable chemoresistance to lung cancer. 8 Meanwhile, because the loss of organ reserve function made poor tolerance to chemotherapy, patients also tend to receive molecularly targeted therapy. Previous studies suggested the treatment of epithelial growth factor receptor (EGFR)-mutated NSCLC by EGFR tyrosine kinase inhibitors (TKIs) lead to remarkable tumor shrinkage and improvement in disease-free survival and quality of life compared to standard chemotherapy. 9,10 However, beyond these advances, clinically apparent acquired resistance to EGFR TKIs inevitably develops regardless of the line of therapy. 11 Overall, it is urgent to investigate the biological predictor of therapeutic response in NSCLC, which would be helpful to clarify the mechanisms of therapeutic resistance.
The gene of meningioma-associated protein (MAC30) on 17q11.2 was originally confirmed as an overexpressed gene in human meningioma. 12 Besides the regulation of cholesterol and lipid metabolism, 13 MAC30 as a member of the insulin-like growth factor-binding protein family (IGFBP) also played its roles on liver growth and differentiation. 14 Notably, the paradoxical expression of MAC30 was exhibited in different human cancer, with stronger expression in breast, esophagus, stomach, and colon cancer in contrast to lower levels in pancreatic and renal cancer. 15 -17 Based on the confused alterations, MAC30 may act as a suppressor in pancreatic and renal cancer while play as a promoter in breast, esophagus, stomach, and colon cancer. Unfortunately, the biological mechanism of MAC30 in human malignancies is still unclear. Although overexpression of MAC30 associated with short survival in NSCLC was reported, 18 there is no direct evidence to validate the correlation between MAC30 expression and response of adjuvant platinum-based chemotherapy or molecularly targeted therapy in patients with NSCLC.
Based on these considerations, via investigating the prognostic value of MAC30 expression on response to adjuvant platinum-based chemotherapy or molecularly targeted therapy, the primary aim of this current study was to identify the prognostic possibility of MAC30 on therapeutic response in patients with NSCLC.
Material and Methods
Patients and Tissue Samples
The present study enrolled a total of 174 Chinese patients diagnosed with NSCLC at stage III B or IV, while 85 of cases received adjuvant platinum-based chemotherapy, while other 89 patients were treated with oral molecularly targeted therapy at Yixing people hospital affiliated Jiangsu University between May 2004 and March 2012. This study was retrospectively performed and was approved by the Institutional Review Board of Jiangsu University Faculty of Medicine. The informed consent obtained from all patients was confirmed. And all tissue samples were collected via surgical biopsy. The histological diagnosis of tumor was based on the criteria. 3 None of the patients received adjuvant chemotherapy or radiotherapy before surgery. The histologic classification included squamous cell carcinomas (SQCLC), adenocarcinoma (AC), and adenosquamous (AS).
Immunohistochemistry Analysis
Immunohistochemistry staining of MAC30 protein was performed with 3-μm-thick sections cut from paraffin-embedded specimens. After pretreated at 60°C for 1 hour, the sections were dewaxed in xylene, hydrated, and washed in phosphate-buffered saline Tween solution (PBST). The sections were treated with 3% H2O2, then incubated with a polyclonal antibody against MAC30 (1:500; SC-139157, Santa Cruz, California) overnight at 4°C. After washing with PBST for 3 times every 15 minutes, the sections were incubated with corresponding secondary antibody at room temperature for 1 hour. The results were visualized with diaminobenzidine. In each immunohistochemistry run, the negative controls were stained without primary antibody.
Two independent pathologists with particular experience in immunohistochemistry evaluated MAC30 staining in all sections. The expression of MAC30 was quantified using a visual grading system based on the percentage of stained cells out of total number of tumor cells and divided from 0 to 3:0 = negative, 1+, 1% to 30%; 2+, 31% to 60%; and 3+, >60%. The intensity of staining was graded on a scale: 0, negative; 1, weak positive; 2, moderate positive; and 3, strong positive. The sums of extend score and intensity score were used to define the MAC30 expression levels, which were graded into 2 groups: low-level MAC30 expression (with a score ≤3) and high-level MAC30 expression (with a score>3).
Assessment
The patients selected in the current study were treated with platinum-based chemotherapy or molecularly targeted therapy. The response to therapy was assessed by clinical test, computerized tomography scanning of chest and serum tumor markers according to the Response Evaluation Criteria in Solid Tumor. 19 The response to therapy was defined as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). For the data analysis, CR and PR were presented as sensitive to therapy, while SD and PD were defined as nonresponse.
Statistical Analysis
The Kaplan-Meier analysis was used to calculate the relationships between MAC30 expressions with overall survival (OS). The data of response to therapy was analyzed by χ2 test. A Cox regression model was conducted to identify the independent prognostic factors that influenced the OS. All statistical analyses were operated using SPSS version 13.0. A
Results
Clinical Characteristics and Therapeutic Regimen
The age of all selected patients ranged from 43 to 82 years, and the median age was 63.7 years. The histopathological types were divided into SQCLC with 41 (23.6%) patients, AC with118 (67.8%) patients, and AS with 15 (8.6%) patients. Among all patients, 81 (46.6%) patients had stage III B and 93 (53.4%) patients exhibited stage IV. A total of 85 patients received platinum-based chemotherapy with other drugs as pemetrexed, paclitaxel, or docetaxel, while 89 patients were treated with oral molecular targeted drugs as gefitinib, erlotinib, or icotinib (Table 1).
Patients Characteristics Enrolled in this Study.

Abbreviations: SQCLC, squamous cell carcinomas; AC, adenocarcinoma; AS, adenosquamous.
MAC30 Expressions in Patients With Different Therapy
The expression of MAC30 was confirmed by immunohistochemistry shown in Figure 1. Table 2 shows patients with platinum-based chemotherapy ranged in age from 43 to 72 years with the median age of 54.7 years, while the other group of patients with age from 51 to 82 years, and the mean age was 61.6 years. In patients with platinum-based chemotherapy, overexpression of MAC30 was exhibited in 51 cases, while 48 patients in molecularly targeted therapy group showed the elevated MAC30 expression (Table 3). There was no significant difference between the therapeutic regimen and MAC30 expression (

Representative immunohistochemical staining for meningioma-associated protein (MAC30) expression in non-small cell lung cancer (NSCLC). A, High expression of MAC30. B, Low expression of MAC30. Original magnification, ×200.
MAC30 Expressions in Patients With Different Therapy.

Abbreviations: MAC30, meningioma-associated protein; SQCLC, squamous cell carcinomas; AC, adenocarcinoma; AS, adenosquamous.
Correlation Between MAC30 Expression and Therapeutic Response.

Abbreviations: MAC30, meningioma-associated protein; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease.
aStatistically significant difference.
Correlation Between MAC30 Expression and Therapeutic Response
In all patients with platinum-based chemotherapy, 12 patients showed a CR and 19 patients exhibited PR, while 37 patients were confirmed with SD and 17 patients were obtained with PD. Of the 51 patients with elevated MAC30, 9 (4%) patients showed a response to therapy, while 22 patients from 34 patients with reduced MAC30 exhibited response to platinum-based chemotherapy (
Table 3 shows the correlation between MAC30 expression and molecularly targeted therapeutic response. Complete response and PR were obtained in 11 and 40 patients, while SD and PD were obtained in 23 and 15 patients. As a general evaluation, there was no significant difference between the responses to targeted therapy in patients with different MAC30 expression (
Association Between MAC30 Expression and OS in Patients Having NSCLC With Platinum-Based Chemotherapy
In our present study, we identified the association between MAC30 and survival of patients with NSCLC receiving platinum-based chemotherapy via Kaplan-Meier analysis. Of the cases with platinum-based chemotherapy, patients with higher MAC30 expression displayed shorter OS than those in patients with lower MAC30 expression (

Kaplan-Meier survival curves for patients with non-small cell lung cancer (NSCLC). according to MAC30 expression. A, Overall survival (OS) of patients with non-small cell lung cancer (NSCLC) receiving platinum-based chemotherapy in high and low MAC30 expression groups. B, OS of patients with NSCLC receiving molecularly targeted therapy in high and low MAC30 expression groups.
Association Between MAC30 Expression and OS in Patients With Platinum-Based Chemotherapy.
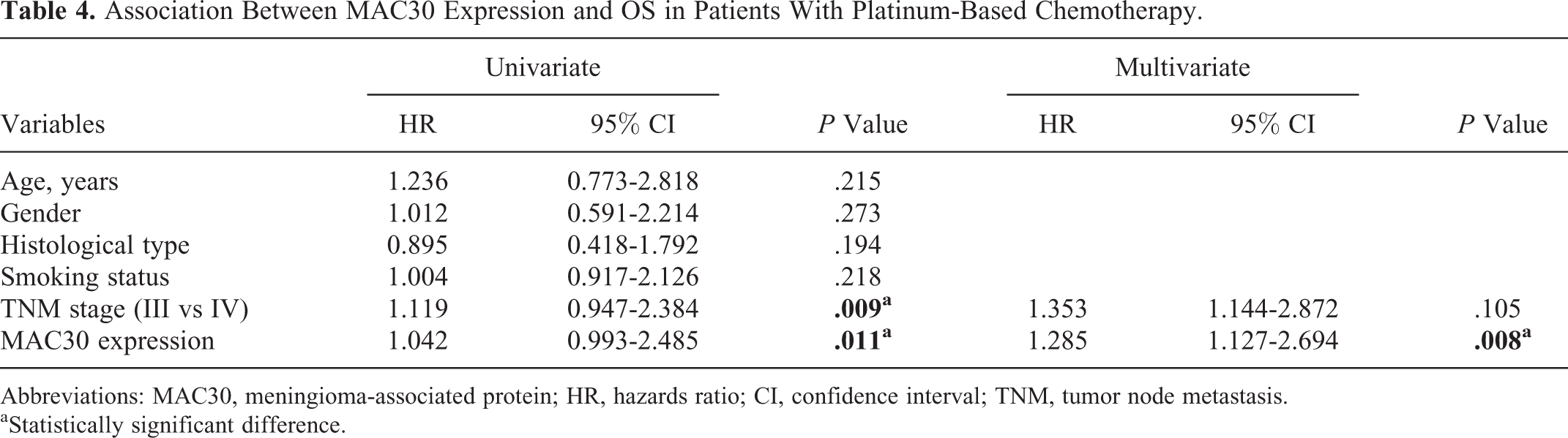
Abbreviations: MAC30, meningioma-associated protein; HR, hazards ratio; CI, confidence interval; TNM, tumor node metastasis.
aStatistically significant difference.
Discussion
Based on the biological and clinical advances on molecular mechanism in lung cancer, it is widely accepted that adjuvant chemotherapy became a standard manner in NSCLC therapy. 20 However, patients with NSCLC even diagnosed with the same histopathological stage of cancer may exhibit paradoxical response to chemotherapy and different prognoses. Meanwhile, with the advances of more specific characteristics of antitumor activity and reduced side effects, molecularly targeted therapy was used widely in patients with advanced NSCLC. 9 The previous data showed that patients having NSCLC with high expression of EGFR exhibited rapid progression and were not sensitive to chemotherapy, while more patients inclined to choose molecularly targeted therapy. 10 Indeed, the efficacy of prolonging survival extension and maintaining the organ function was unsatisfactory. The investigation of evidence recommending the valuable prediction of effective chemotherapy remains insufficient.
In our present study, we ushered a new idea on MAC30 in patients with NSCLC receiving platinum-based chemotherapy. And as a newly concerned protein, the role of MAC30-mediated tumor progression and invasion was less investigated. Zhang
On the other hand, we first found that there was no significant difference in responses to molecularly targeted therapy in patients having NSCLC with paradoxical MAC30 expression, to be followed by undifferentiated OS. The reason for the difference in these effects found in the 2 groups is unclear. Whatever, from the data in present study, we confirmed that patients having NSCLC with overexpression of MAC30 showed resistance to platinum-based chemotherapy but not to molecularly targeted therapy. And the interesting observation guides us to investigate the prognostic role of MAC30 in more patients having NSCLC treated with different molecularly targeted drugs, which should not be neglected.
By now, the investigation of MAC30 biological mechanism and function in tumor development and progression is limited. Xu
Conclusion
In the present study, we demonstrated that patients having NSCLC with overexpression of MAC30 showed resistance to platinum-based chemotherapy. Moreover, elevated MAC30 expression was a powerful predictor of OS in patients with NSCLC receiving platinum-based chemotherapy. Further studies will be required to investigate the molecular function of MAC30 in NSCLC.
Footnotes
Authors’ Note
YF, RH, and XB prepared the samples and carried out the data analyzed mostly. TL and ZW checked the data of patients. HD and HR both designed the project and wrote the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Grant No. YG201408 from Science Foundation of Health Department in Jiangsu Province of China and Grant No. 81470253, from National Natural Science Foundation of China.
