Abstract
Interleukins as immunomodulators are promising therapeutic agents for cancer therapy. Previous studies showed that there was an improved antitumor immunity in tumor-bearing mice using recombinant Newcastle disease virus carrying for interleukin-2. Interleukin-12 is a promising antitumor cytokine too. So we investigated and compared the antitumor effect of genetically engineered Newcastle disease virus strains expressing both interleukin-12 and/or interleukin-2 (rClone30–interleukin-2, rClone30–interleukin-12, and rClone30–interleukin-12–interleukin-2). In vitro studies showed that rClone30s could efficiently infect tumor cells and express interleukin-12 and/or interleukin-2. 3-(4,5-Dimethylthiazol-2-y)-2,5-diphenyl-tetrazolium bromide results showed rClone30s possessed strong cytotoxic activities against multiple tumor cell lines (U251, HepG2, A549, and Hela). Animal studies showed that rClone30–interleukin-12–interleukin-2 was more effective in inhibition of murine hepatoma carcinoma tumors, with the mean tumor volume (day 14) of 141.70 mm3 comparing 165.67 mm3 of rClone30–interleukin-12 group, 210.47 mm3 of rClone30–interleukin-2 group, 574.70 mm3 of rClone30 group, and 1206.83 mm3 of phosphate-buffered saline group. Moreover, the rClone30–interleukin-12–interleukin-2 treated mice secreted more interferon γ (333.518 pg/mL) and its downstream cytokine interferon-γ induced protein 10 (16.006 pg/mL) in tumor than the rClone30–interleukin-12 group (interferon γ: 257.548 pg/mL; interferon-γ induced protein 10: 13.601 pg/mL), rClone30–interleukin2 group (interferon γ: 124.601 pg/mL; interferon-γ induced protein 10: 9.779 pg/mL), or rClone30 group (interferon γ: 48.630 pg/mL; interferon-γ induced protein 10:1.650 pg/mL). For the survival study, rClone30–interleukin12–interleukin2 increased the survival rate (12 of 16) of the tumor-bearing mice versus 11 of 16 in rClone30–interleukin–12 group, 10 of 16 in rClone30–interleukin-2 group, 7 of 16 in Clone30 group, and 0/16 in phosphate-buffered saline group, respectively. To determine whether the mice treated with recombinant virus developed protective immune response, the mice were rechallenged with the same tumor cells. The results showed that viral-treated mice were significantly protected from rechallenge. These results suggest that expressing both interleukin-2 and/or interleukin-12 could be ideal approaches to enhance the antitumor ability of Newcastle disease virus, and rClone30–interleukin-12–interleukin-2 is slightly superior over rClone30–interleukin-12 and rClone30–interleukin-2 alone.
Introduction
The most common types of cancer treatment are surgery, chemotherapy, and radiation therapy. Traditional therapies frequently lead to severe side effects and easily recurrent and not much improved the survival of the patients with metastases. 1,2 Other treatments such as immunotherapy, hyperthermia, photodynamic therapy, and virotherapy had been applied in clinically.
Virotherapy using oncolytic viruses was proposed as a potent cancer therapy. Over the past century, a number of oncolytic viruses have been discovered, such as mumps, measles, adenovirus, reovirus, Newcastle disease virus (NDV), and others. 3,4 Newcastle disease virus as a vector for gene therapy is under clinical evaluation. 5 Newcastle disease virus is a member of the Avulavirus genus in the Paramyxoviridae family. The 15186, 15192, or 15198 nucleotide-negative, single-stranded RNA genome of NDV encodes the nucleocapsid protein, phosphoprotein (P), matrix protein, fusion protein (F), hemagglutinin-neuraminidase (HN), and RNA-dependent RNA polymerase (L). 6 In addition, nonstructural protein (V), which acts as an interferon (IFN) α/β antagonist, and possibly a second one (W), are generated by RNA editing during P gene transcription. 3,5,6 The first report of NDV in antitumor efficacy was published in 1964. 7 Newcastle disease virus is an ideal candidate for cancer therapy due to the characteristics of NDV to selectively replicate within tumor cells and lead to apoptosis. The inherent viral oncolytic properties are believed to be derived from defective IFN signaling pathways in tumor cells. 3,5,8 The development of reverse genetics technology for NDV enable us to modify the NDV genome as well as introduce foreign sequences to refine the antitumor activity. 9 -13 Previous studies in our laboratory showed that recombinant NDVs (rNDVs) expressing Trail, interleukin (IL) 15, and IL-2 are promising antitumor agents. 14 -16 Other reports also showed that rNDVs carrying granulocyte-macrophage colony-stimulating factor and tumor necrosis factor α were significantly enhanced with the antitumor efficacy compared to parental virus. 10,17
Interleukins as the regulator of immune response are widely applied in clinical study, including cancer. Among the ILs, IL-2 is the only one approved by the Food and Drug Administration for the treatment of cancer. 18,19 Interleukin-2 is a pleiotropic cytokine that drives T-cell growth, augments natural killing (NK) cytolytic activity, promotes antibody production, and induces the proliferation and differentiation of B cells and regulatory T cells. 20,21 Although some promising results are achieved, in clinical studies, some serious side effects occur in almost all patients treated with IL-2 protein. 22 -24
Interleukin-12 is a promising candidate for tumor immunotherapy. 25,26 Extensive research suggested that IL-12 promotes an effective destruction of cancer cells by inducing proliferation of NK cells and T cells and production of IFN-γ, and IL-12 enhances the generation and activity of cytotoxic T lymphocytes (CTLs) through activation of STAT4. 27 -30 It is important to note that IFN-γ is able to induce a powerful protection against the development of tumors by inducing the production of interferon-γ–induced protein 10 (IP-10) and monokine induced by gamma-interferon (MIG), which promote antiangiogenic activity. In addition, several other mechanisms of IL-12 strongly contribute to antitumor activities. 25,26,31 Although encouraging results have been shown in preclinical studies, in clinical investigations, often severe side effects and toxicities are associated with systemic administration of IL-12 protein, and this fact markedly dampens hopes of IL-12 use in patients with cancer directly. 32 -34 Nevertheless, several clinical studies have shown bright results with IL-12 administration by gene therapy, and currently studies based on IL-12 are ongoing with the key focus of reducing toxicities and side effects. 35 -38 In addition, the antitumor activity of IL-12 can be improved by its combination with various therapeutic modalities. 39,40 Previous studies have shown that a combination of IL-12 and IL-2 would result in a synergistic effect for inducing antitumor response. The synergistic mechanism is mainly induced by the enhancement of the production of IFN-γ, the activation of cytolytic activity of lymphokine-activated killer (LAK) cells, and the induction of antigen-specific CTL activity. 40 -43 Furthermore, IL-12 increases the expression of IL-2R, whose expression at a sufficient quantity is necessary for the development of a proper anticancer immune response, especially in IL-2 immunotherapy. 44,45
In this study, we introduced sequences encoding IL-12 and/or IL-2 into the genome of lentogenic NDV Clone30 strain at the position between F and HN genes, generated 3 rNDVs (rClone30–IL-12, rClone30–IL-12–IL-2, and rClone30–IL-2), and tested and compared the antitumor efficacy of these viruses both in vitro and in vivo. Our results show that both rClone30s encoding IL-2 and/or IL-12 are promising agents for hepatoma carcinoma therapy, and the rClone30–IL-12–IL-2 is slightly superior over rClone30–IL-12 or rClone30–IL-2 alone.
Materials and Methods
Animals, Specific Pathogen Free Embryos, and Biochemical Reagents
Six-week-old ICR mice were purchased from Changchun Yisi (Changchun, China; approval number: SCXK-2011-0004) and housed in a pathogen-free environment on a standard diet. Specific pathogen free (SPF) embryos were purchased from Harbin Veterinary Research Institute (Harbin, China). All restriction (RE) enzymes, ampicillin, pMD18-T simple vector, and DNA ligase, and so on were purchased from Takara Biotechnology Co, Ltd (Dalian, China). All enzyme-linked immunosorbent assay (ELISA) kits were purchased from R&D (Minneapolis, MN, USA).
Plasmids and Cell Lines
The plasmids of prClone30, pTM-np, pTM-p, and pTM-l were cloned from the NDV lentogenic strain Clone30 (pBrClone30) by our laboratory. The plasmid containing IL-12 gene (Porf-mIL-12) was kindly provided by Dr Yijie Li (XinJiang university, Xinjiang, China). The plasmid pMD18-T–IL-2 was kept in our laboratory. Human hepatocarcinoma cells (HepG2), Mouse hepatocarcinoma cells (H22), A549, Hela, U251, Vero, and BHK-21 were kept in our laboratory. All cell lines were grown at 37°C under 5% CO2. HepG2, Vero, and BHK-21 cells were maintained in Dulbecco modified Eagle medium (DMEM; GIBCO [Grand Island, NY, USA]) supplemented with 10% fetal bovine serum (FBS), 1% penicillin/streptomycin, 1% nonessential amino acids, and 1% sodium pyruvate. A549 were cultured in Roswell Park Memorial Institute 1640 medium, supplemented with 10% FBS, 1% penicillin/streptomycin, 1% nonessential amino acids, and 1% sodium pyruvate. Hela and U251 cells were maintained in DMEM supplemented with 10% calf serum, 1% penicillin/streptomycin, 1% nonessential amino acids, and 1% sodium pyruvate.
Construction and Rescue of the Recombinant Viruses
The IL-2 (462 bp) and IL-12 (1623 bp) genes were cloned into pMD18-T simple vector by polymerase chain reaction (PCR) method. The following primer sequences were used for PCR: PIL-2F: GGTTAACCGCCACCATGTACAGGATGCAACTCCTGTCT PIL-2A: CGACGCGTCGCAATTAAGTCAGTGTTGAGATGATGCT PIL-12F: GGTTAACCGCCACCATGTGTCAATCACGCTACCTCCTCT PIL-12A: CGACGCGTCGCTAGGATCGGACCCTGCAGGGAACAC PIL-2F’: GGCCTGAGAGGCCTTAAGAAAAAATACGGGTAGAAGGCCACCATGTACAGGATGCAACT PIL-2A’: GGCCTCTCAGGCCTTAAGTCAGTGTTGAGATGATGCT
Sequencing was performed to confirm that no mutation occurred. The correct IL-2 and IL-12 fragments were inserted into the genome of pBrClone30 between F and HN genes by HpaI and/or MluI sites. The integrity of the entire foreign sequence was confirmed by sequencing. The resulting plasmids were named prClone30–IL-2, prClone30–IL-12, and prClone30–IL-12–IL-2, respectively. The rescue procedure for obtaining the recombinant virus has been established in our laboratory. 14 -16 All recombinant viruses were grown in embryonated SPF eggs. The rescued viruses were named rClone30–IL-2, rClone30–IL-12, and rClone30–IL-12–IL-2, respectively. The genomic RNA of the viruses was extracted using TRIzol reagent (Invitrogen [Carlsbad, CA, USA]), and the RNA was reverse transcribed using ThermoScript reverse transcriptase. The antigenomic-sense primer was 5′-CACAGATGAGGAACGAAGGT-3′. Amplification of the complementary DNAs was performed by PCR and sequenced for inserting fidelity.
Determination of Virus Growth
The growth curves of the recombinant viruses were assessed by a multistep growth assay in Vero cells. Vero cells were infected at multiplicity of infection (MOI; virus particles per tumor cell) of 1 and incubated at 37°C in DMEM containing 5% FBS and 1 µg of acetyl-trypsin/mL. Supernatants containing viruses were harvested at 24 intervals and titrated by plaque assay in Vero cells. Titrations were performed in duplicate. The concentrations of viruses were determined on Vero cells and were expressed as mean log10 50% tissue culture infective dose per mL.
Enzyme-Linked Immunosorbent Assay
HepG2 and U251 cells were infected with recombinant viruses at an MOI of 5 or phosphate-buffered saline (PBS) as control, the infection supernatants were harvested at 48 hours later, and assayed by IL-12 and IL-2 ELISA kits (R&D Systems) following the supplier’s protocol.
In Vitro Cytotoxicity Assay
The human HepG2, A549, Hela, and U251 cell lines were tested for the sensitivity to recombinant viruses in vitro by the 3-(4,5-dimethylthiazol-2-y)-2,5-diphenyl-tetrazolium bromide (MTT) staining method in 96-well plate. Cells were plated at 2 × 104 cells/well in 96-well plates, after 24 hours later, and cells were infected with recombinant viruses or medium as control at MOIs of 0.01, 0.1, 1, and 10, respectively. Forty-eight hours after infecting, 20 µL (5 mg/mL) of MTT was added into each well and incubated for 4 hours. After being incubated, the media were aspirated and 150 µL dimethyl sulfoxide was added into each well and gently shaken for 10 minutes. The optical density (OD) of all sample was detected by microplate reader at OD 490 nm. Results were expressed as the cytotoxicity% of cells as determined by the measured absorbance of each sample relative to control. Cytotoxicity% was calculated using the following formula: Cytotoxicity% = 100 × (OD control − OD sample)/OD control. All samples were analyzed in quintuplicate.
The Expression of Foreign Genes in the Tumors of Recombinant Virus Treated Mice
The mouse hepatocarcinoma cells (H22) were injected into the 6-week-old ICR mice by intraperitoneal injection. The ascites were harvested at sterile condition after 6 days postinjection, and the ascites containing H22 cells was diluted to 2 × 106/mL with PBS. The 2 × 105 H22 cells per mouse were injected at right groin area of the mouse in a total volume of 100 µL by hypodermic injection. Ten days after injection, mice with tumor reaching to 12 mm were treated by intratumoral injection of 1 × 107 recombinant viruses or PBS, in total volume of 100 µL. Fifteen mice were used in each group. The treatments were repeated on days 2, 4, and 6 after initiation of treatment, for a total of 4 treatments. On days 3, 6, and 10 after completion of treatment, the tumors of the animal were dissected and manually dissociated with scissors and forceps into PBS, and the sera of animals were also collected. Following centrifugation, the supernatants were collected and analyzed by IL-12, IL-2, IFN-γ, and IP-10 ELISA kits. The concentration of the samples was determined and plotted against a standard curve. The standard curve was generated by serially diluting the stock standard in dilution buffer supplied by the kit.
Animal Studies
In tumor-bearing mice described earlier, 5 days after injection, solid tumors were formed, and the diameter of tumors between 3 and 6 mm was selected and randomly divided into different groups, 16 mice were included in each group. The mice were treated by intratumoral injection of a dose of 1 × 107 pfu of recombinant viruses or PBS, in total volume of 100 µL. The treatments were repeated on days 2, 4, and 6 after treatment initiation for a total of 4 treatments. Tumor volumes were monitored every other day before treatment, using digital calipers in 2 dimensions. Tumor volume was calculated using the following formula: tumor volume (V) = 4/3π × S2/2 × L/2, where S is the smallest measured diameter and L is the largest diameter. According to the institutional protocols, the animals were euthanized when tumor size reached 18 mm in any dimension or at defined experimental time points. Sixty days after treatment initiation, all of the mice that had undergone tumor regression were rechallenged with another injection of 2 × 105 H22 cells into the left groin area in a total volume of 100 µL, and 6 naive age-matched mice were used as control. The remaining mice in each group were observed for 120 days with measurement of tumor sizes every other day.
Statistical Analysis
For comparison of the statistical significance between different treatment groups, SPSS version 19 software (SPSS-IBM, Chicago, Illinois) was applied. All graphs were performed in utilizing GraphPad prism version 5.01 software (GraphPad Software Inl., La Jolla, California). All values were expressed as mean ± standard deviation; *P < .05 were considered statistically significant.
Results
Generation of Recombinant Viruses
The IL-12 and IL-2 genes with GS (gene start) and GE (gene end) sequences were correctly inserted into the newly created HpaI and/or MluI position between the F and L genes of rClone30 genome, and the resultant virus plasmids were named after prClone30–IL-2, prClone30–IL-12, and prClone30–IL-12–IL-2, respectively (Figure 1A). Sequencing was performed to confirm that no mutation occurred in the resultant plasmids. These recombinant viruses were rescued with helper plasmids in BHK-21 cell line, and all the supernatant of the transfected monolayers could be grown to high titer in the allantoic fluid of 10-day-old SPF embryonated chicken eggs. After incubation for 3 days, the allantoic fluids were harvested and analyzed in a rapid plate hemagglutination (HA) test using chicken erythrocytes. The RNAs of resultant viruses were extracted and, the presence of the inserted genes in the genome was verified by real-time PCR (Figure 1B) and confirmed by sequencing.
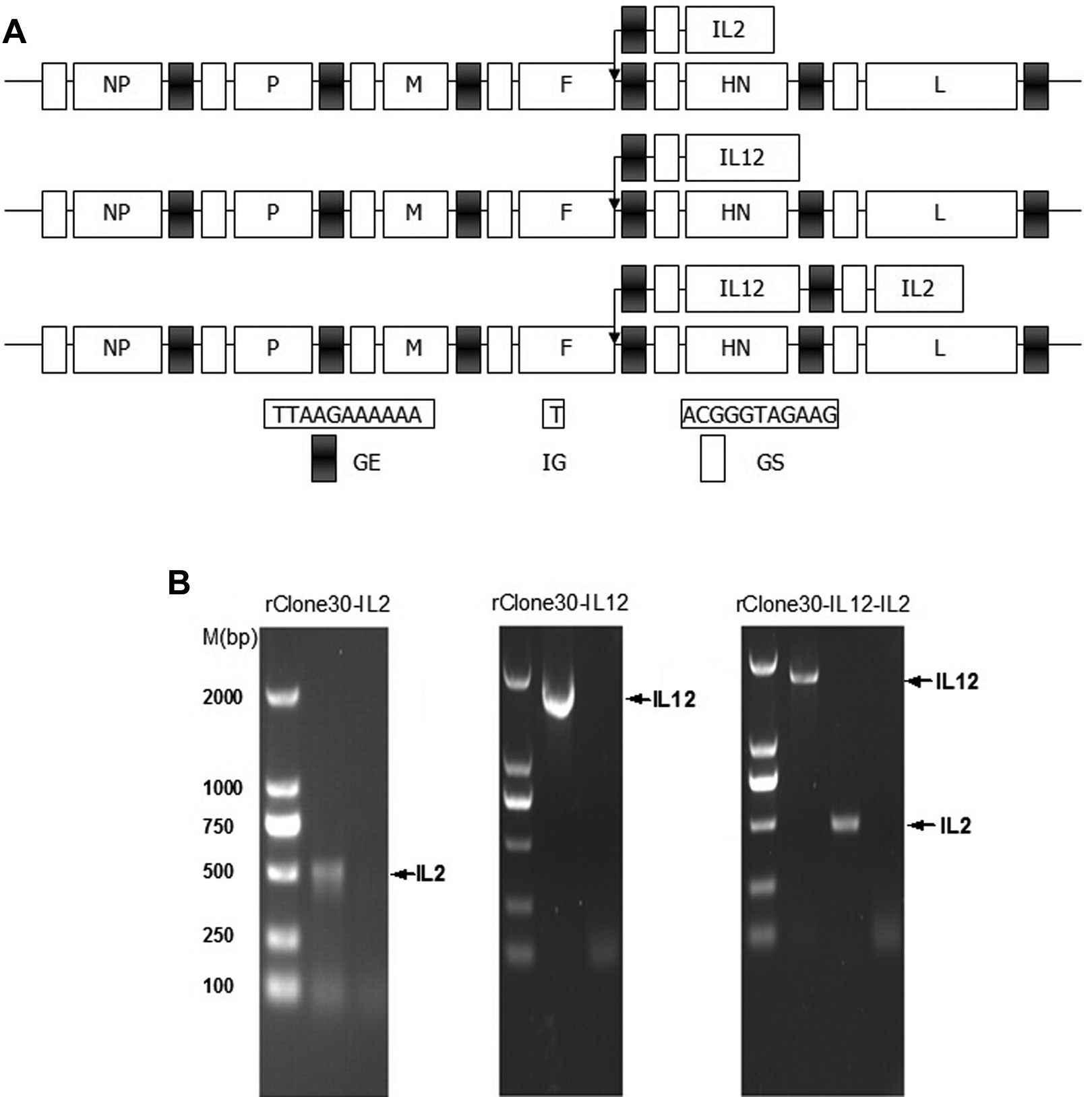
Construction and identification of the rClone30s with insert IL-2 and/or IL-12. A, Schematic diagrams of insertion of the IL-2 and/or IL-12 genes into the NDV genome at the position between the F and HN genes. Black box indicated conserved GE sequence. White box indicated conserved GS sequence. B, The IL-2 (462 bp) and IL-12 (1623 bp) genes were amplified by RT-PCR using RNA template extracted from allantoic fluid infected with the rClone30s and confirmed by DNA sequencing. IL indicates interleukin; NDV, Newcastle disease virus; F, fusion protein; HN, hemagglutinin-neuraminidase; GE, gene end; GS, gene start; RT-PCR, real-time polymerase chain reaction.
Growth Curves of the Recombinant Viruses
To compare the growth characteristics of the recombinant viruses with wild-type virus, Vero cells were infected with viruses at an MOI of 1, and the supernatants were harvested at 24, 48, 72, and 96 hours postinfection, and the supernatant titers were determined in duple. Six days after infection, all the tested viruses were shown the similar kinetics and magnitude of replication (Figure 2).
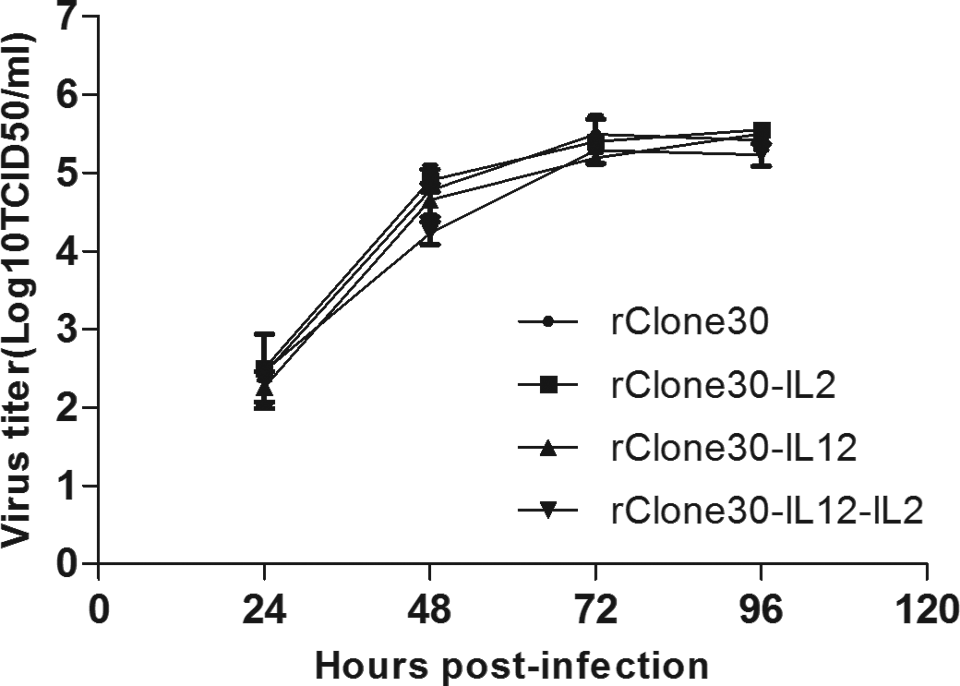
Growth curves of rClone30s. Vero cells were infected with the recombinant viruses at the MOI of 1, and the viral titers in the supernatants were assessed by plating 10-fold dilution on Vero cells at 24, 48, 72, and 96 hours postinfection and were shown as log10 TCID50/mL. The results represent mean values from 2 independent experiments. Results are means ± SD. MOI indicates multiplicity of infection; TCID50, tissue culture infectious dose 50; SD, standard deviation.
Expression of IL-12 and IL-2 by Tumor Cells Infected With Recombinant Viruses
Tumor cell lines HepG2 and U251 were used to confirm that IL-12 and/or IL-2 were expressed in the recombinant virus-infected tumor cells. Tumor cells were infected with viruses at an MOI of 5, and the supernatants were collected and assayed by ELISA kit at 72 hours postinfection. The results showed that each cell line infected with recombinant viruses expressed the desired gene, while no IL-12 or IL-2 could be detected in the control group that was infected with the rClone30 (Figure 3).

The expression levels of IL-2 and IL-12 in vitro. HepG2 and U251 cells were infected with rClone30s at MOI of 5, and the supernatants were collected at 72 hours postinfection. The concentrations of the IL-2 and IL-12 in the supernatants were analyzed by ELISA kit. Results are means ± SD. IL indicates interleukin; MOI, multiplicity of infection; SD, standard deviation.
The Cytotoxicity of Recombinant Viruses to Tumor Cells
Tumor cell lines of U251, HepG2, Hela, and A549 were used to investigate the cytotoxicity of recombinant viruses in vitro. The results showed that after infected with different MOIs of recombinant viruses for 48 hours, recombinant viruses showed the capacity of inducing tumor cell death, and only a slight difference has been observed between different viruses at the same MOI. Moreover, the U251 cell line showed the most sensitive for viruses, which lead to the highest percentage of cell death and the Hela cell line against that (Figure 4).

Cytotoxicity of the rClone30s in U251, HepG2, A549, and Hela cell lines. The cell lines were infected with rClone30s at MOIs of 0.01, 0.1, 1, and 10, the control was treated with medium. Cytotoxicity was assessed at 48 hours by MTT method. Results are means ± SD. MOI indicates multiplicity of infection; SD, standard deviation; MTT, 3-(4,5-dimethylthiazol-2-y)-2,5-diphenyl-tetrazolium bromide.
The Expression Levels of IL-12, IL-2, IFN-γ, and IP-10 in the Tumors of Recombinant Viruses-Treated Mice
In order to test the expression levels of IL-12, IL-2, IFN-γ, and IP-10 within the tumors, after treatments finished, tumors were dissected on days 3, 6, and 10, and the sera were also collected. The sera and the supernatants of tumor homogenates were tested for the in situ presence of IL-12, IL-2, IFN-γ, and IP-10 by ELISA. On day 3, tumor homogenates from the mice treated with rClone30–IL-2, rClone30–IL-12, and rClone30–IL-12–IL-2 had detectable levels of IL-12 and/or IL-2 and nearly negligible level in sera (Figure 5). As shown in Figure 6, the concentration of IFN-γ and IP-10 within tumors of rClone30–IL-12 and rClone30–IL-12–IL-2 treated groups was higher than others and nearly undetectable level in the PBS group. After treatment finished for 10 days, those were still detectable levels of IFN-γ and IP-10 within the tumors (Figure 7).

The expression levels of IL-2 and IL-12 within tumor and serum. Tumor homogenate and serum from rClone30s-treated mice removed and collected on day 3 were tested for production of IL-2 and IL-12 by ELISA. Results are means ± SD. **P < .01. IL indicates interleukin; ELISA, enzyme-linked immunosorbent assay; SD, standard deviation.
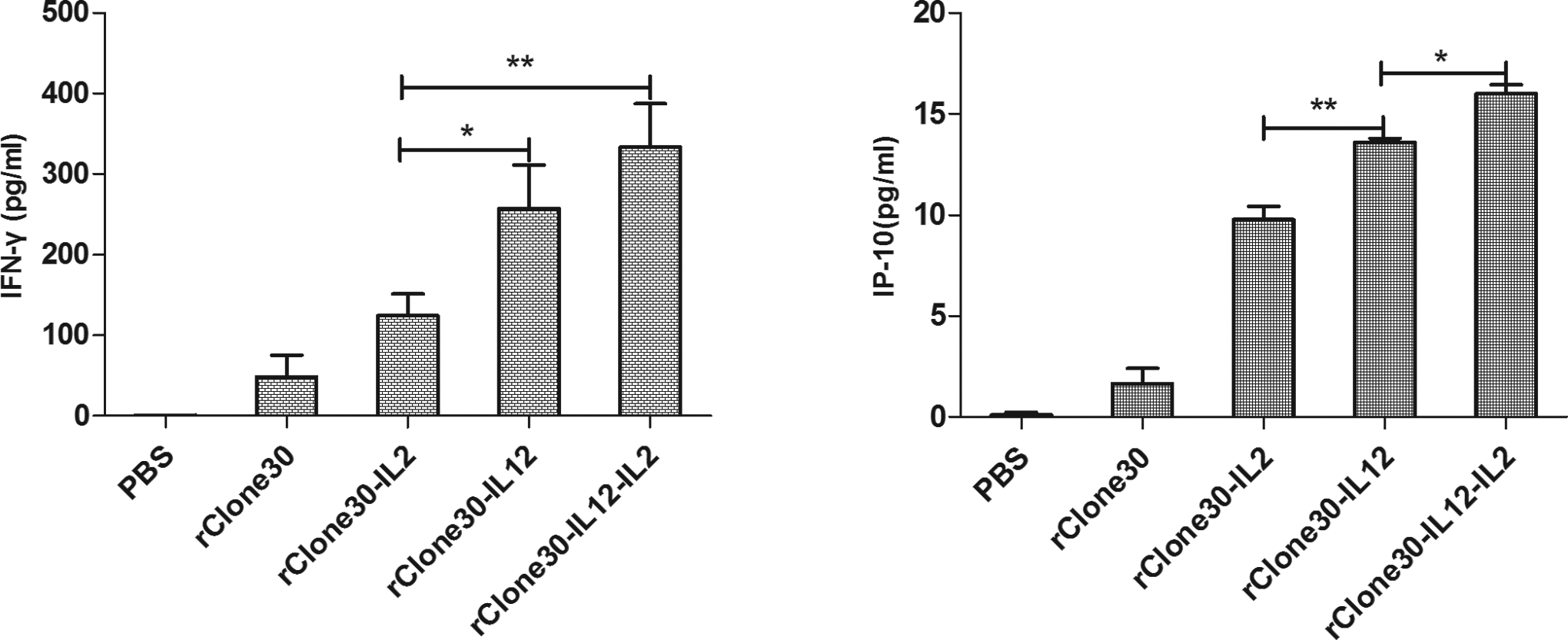
The expression levels of IFN-γ and IP-10 in tumor. Tumor homogenate from treated mice removed on day 3 was tested for production of IFN-γ and IP-10 by ELISA. Results are means ± SD. **P < .01, *P < .05. IFN, interferon; ELISA, enzyme-linked immunosorbent assay; SD, standard deviation; IP-10, interferon-γ induced protein 10.
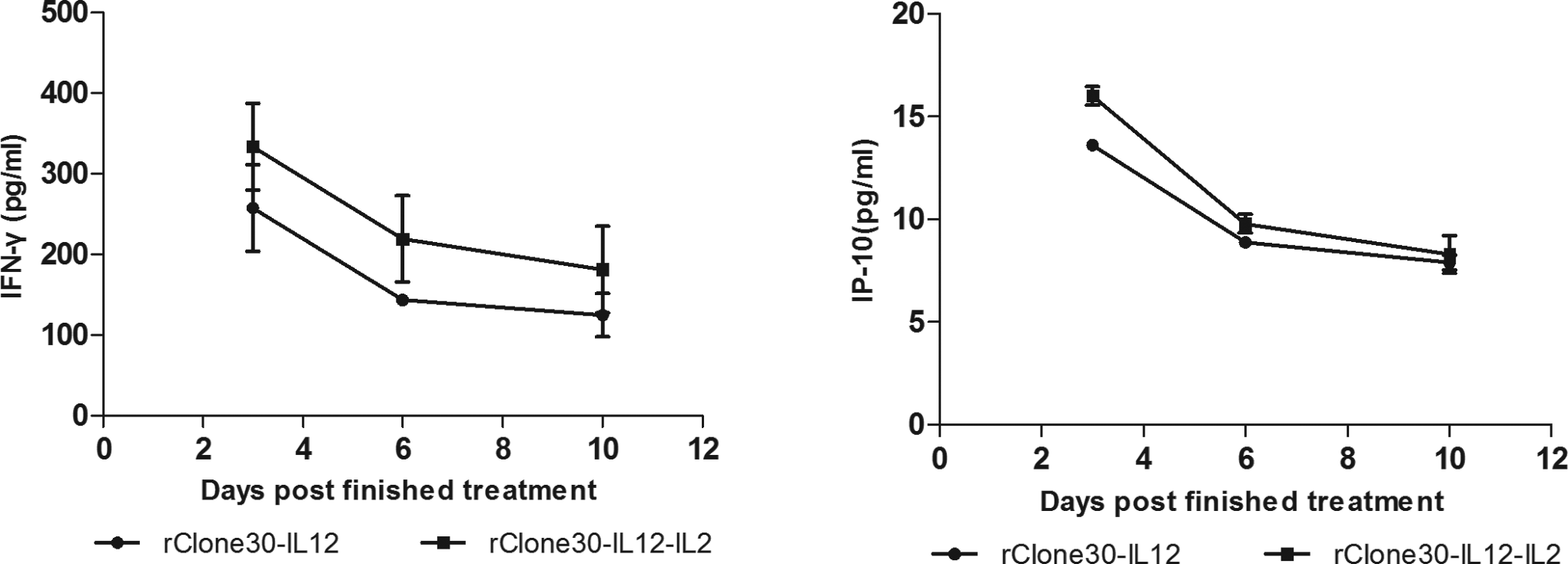
The concentration curves of IFN-γ and IP-10 within tumor after treatment finished. Tumor homogenates from rClone30–IL-12 and rClone30–IL-12–IL-2 treated mice removed on days 3, 6, and 10 were tested for the concentrations of IFN-γ and IP-10 by ELISA. Results are means ± SD. IFN, interferon; ELISA, enzyme-linked immunosorbent assay; SD, standard deviation; IL, interleukin; IP-10, interferon-γ induced protein 10.
Treatment of H22 Tumor-Bearing Mice With Recombinant Viruses Result in Enhancement of Tumor Regression
In order to assess and compare the antitumor effects of recombinant viruses encoding IL-12 and/or IL-2 in vivo, H22 groin tumor-bearing mice model as described earlier was selected and tested. The tumor volume was calculated every other day before treatment. Mice treated with rClone30 (574.70mm3) resulted in a significant reduction in tumor volume compared to the PBS group (1206.83 mm3; **P < .01). Mice treated with rClone30–IL-12–IL-2 (141.70 mm3), rClone30–IL-12 (165.67 mm3), and rClone30–IL-2 (210.47 mm3) exhibited a clear enhancement in tumor growth inhibition compared with rClone30 group (*P < .05; Figure 8). Mice were killed when the tumor size reached to 18 mm. No complications or toxicity from intratumoral injection of NDV were observed.

Recombinant viruses encoding IL-2 and/or IL-12 effectively suppress tumor growth in H22 tumor-bearing mice model. A, Sixteen 6-week-old ICR mice were injected in the right groin area with 2 × 105 H22 cells. When the tumor diameter was reached to 3 to 6 mm, the tumors were intratumoral injection treatment with 107 pfu of rClone30s or PBS as control (day 0). The treatments were repeated on days 2, 4, and 6 for a total of 4 treatments. Tumors volume were recorded every other days. B, Mean tumor volume of 5 groups. Results are means ± SD. **P < .01, *P < .05. IL indicates interleukin; PBS, phosphate-buffered saline; SD, standard deviation.
Recombinant Viruses Efficiently Enhanced the Mice Survival Rate and Protected the Mice From Rechallenge With H22 Cells
After treatment initiation, PBS-treated mice began to succumb to tumor progression (d > 18 mm) on day 15, and all had died by day 45. Mice treated with rClone30 had a significant effect on the enhancement of survival rate with 7 of 16 of mice undergoing tumor regression compared with 0 of 16 mice in the PBS group (**P < .01), and further enhanced in rClone30–IL-2(10 of 16), rClone30–IL-12(11 of 16), and rClone30–IL-12–IL-2(12 of 16), respectively (Figure 9 and Table 1, panel A).To determine whether the mice treated with recombinant viruses translate to an enhancement in survival rate and developed protective systemic immune response against rechallenge with the same tumor cells, 60 days posttreatment initiation, the remaining mice were rechallenged with a second dose of H22 tumor cells on the contralateral flank of the mouse. Six naive age-matched mice were used as control (data not shown). Over the next 60 days, 3 of 7 mice in the rClone30 group, 4 of 10 of the rClone30–IL-2 group, 3 of 11of the rClone30–IL-12 group, and 4 of 12 of rClone30–IL-12–IL-2 group initially developed into visible tumors (Table 1, panel B), but all needed not to be sacrificed (d < 6 mm) and eventually regressed or did not changed in size until the completion of this study.

Percentage survival of tumor-bearing mice treated with rClone30s or PBS and the remaining mice protect from rechallenge with the same tumor cells. Sixty days posttreatment initiation, 12/16 of mice treated with rClone30–IL-12–IL-2 underwent tumor regression comparing 11/16 of rClone30–IL-12 group, 10/16 of rClone30–IL-2 group, 7/16 of rClone30, and 0/16 of PBS group, respectively. Mice were sacrificed when the diameter of volume was more than 18 mm in any dimension. All of the surviving animals were rechallenged with a new injection of 2 × 105 H22 cells (arrows) and observed for another 60 days. Over the next 60 days, viral-treated mice were significantly protected from rechallenge (d < 6 mm) and not changed the survival rate. Results are means ± SD. **P < .01;*P < .05. IL indicates interleukin; PBS, phosphate-buffered saline; SD, standard deviation.
Complete Remission of Tumors Observed in 2 Independent Experiments and Tumor Progress After Rechallenged With the Same Dose of Tumor Cells.

AExperiment 1 was done with total of 7 animals per group. Experiment 2 was corresponding to animals in Figure 8.
BThe numbers of remainder mice (experiment 2) developed into visible volume of tumor.
Discussion and Conclusion
The use of NDV vectors to deliver foreign genes is an ideal method for cancer therapy, particularly in solid tumors. Promising results were noted in multiple models of murine or human cancers using different strains of NDV. 8 -16 In this study, we chose the NDV lentogenic strain Clone30 which is safe to avian species (not cause serious disease) as the parental skeleton to construct the recombinant viruses. The shortcoming of Clone30 is that the cell lysis ability is inferior to velogenic and mesogenic strains.
Our results showed that the IL-12 and/or IL-2 genes were correctly inserted into the genome of Clone30 at the position between F and HN genes, and the growth curves of rClone30s were similar to parental virus. The rClone30s were capable of infecting tumor cell lines and expressing IL-12 and/or IL-2, and the expression level of IL-12 and/or IL-2 differed in different tumor cell lines. In vivo, IL-12 acts in a form of a heterodimeric cytokine comprising the light chain p35 (IL-12A) and the heavy chain p40 (IL-12B). 46 However, the P40-P40 homodimer acts mostly as a competitive suppressant of IL-12 actions. 47 Hence, we chose the fusion gene of IL-12 (P70) as the candidate for this study. In the future study, it is necessary to investigate the optimum site of NDV for insertion gene, which leads to highest expression level of inserted gene and minimum influence of the replication characteristic of NDV.
The MTT results showed that both strains of viruses could induce cell death in a dose-dependent manner, and only a slight difference has been observed between different viruses at the same MOI, which indicate the cell death induced in vitro is a result of viral killing more than the function of ILs. The U251 cell line showed the most sensitive for viruses which lead to the highest percentage of cell death, and the Hela cell line against that. These results indicate that NDV as an antitumor agent could be investigate for some specific cancer which sensitive for NDV most. In the future study, it is necessary to try to build U251 tumor-bearing mice and test the sensitive of rNDVs for this model.
To investigate the antitumor effect of recombinant viruses in vivo, we chose the HCC tumor-bearing mice as the model. Our study showed that both recombinant viruses encoding IL-12 and/or IL-2 were significantly suppressed tumor growth when compared with rClone30 (*P < .05) and PBS groups (**P < .01). Although no statistically significant difference was apparent, mice treated with rClone30–IL-12–IL-2 had a slightly superior than the rClone30–IL-12 group and the rClone30–IL-2 group in tumor regression and enhancement of survival rate. This result might owing to the expression of IL-12 and IL-2 within tumors. Linking previous studies from our laboratory and other research institutes, we speculate that the immunostimulatory properties of IL-12 and IL-2 resulted in the enhancement of LAK, NK, and CTL cells activity and increased tumor infiltration with CD4+ and CD8+ cells were important factors for tumor regression. 40 -43 Moreover, IL-12 and IL-2 are known to synergize in the induction of IFN-γ expression. 48 The ELISA results showed that the mice from rClone30–IL-12–IL-2 treatment group induced more IFN-γ and IP-10 released, both of these molecules have been shown to be antiangiogenestic, which might be another important factor for resulting in inhibition of tumor growth. 25 -28 The high expression level of the inserted genes within tumor and negligible in serum highlight the safety and efficiency of NDV serve as a vector for delivering human IL-12 and IL-2 into tumor cells in the future application. At the end of this study, virus-treated mice were rechallenged with the same tumor cells, and results showed that viral-treated mice were efficiently protected from rechallenged. This result may due to the induction of inflammatory immune infiltrates in distant tumors by localized therapy with NDV. 49 In the future study, it is necessary to do further investigation to test the superiority of rClone30–IL-12–IL-2 in antitumor efficacy compared with rClone30–IL-12 or rClone30–IL-2 alone.
For virotherapy, the presence of preexisting neutralizing antibodies is a major concern for their future development, such as measles virus and herpes simplex virus. 4 Newcastle disease virus as an avian paramyxovirus making it immunogenic to the general human population and without the concern of preexisting neutralizing antibodies. 9 However, the antiviral immune responses associated with long-term therapy may cause some limitation in clinical applications. 3,50 In the future study, it is necessary to investigate the optimum injection time and the number of injections to optimize the therapeutic effect. In addition, NDV could cause HA and bind to free sialic acid in blood by intravenous injection, then it could attenuate therapeutic efficacy of NDV, so we chose intratumoral injection route as our delivery method. In the future study, it is important that characterizing the NDV vector apply to long-term therapy and developing some intravenous injection methods serve for clinical applications.
In summary, the efficiency of NDV to infect tumor cells and locoregional expression of the IL-12 and IL-2 genes, the combination of IL-12 and IL-2 genes had the greatest influence on enhancement of antitumor efficacy of rNDV compared with IL-12 or IL-2 alone, and NDV as a traditional antitumor agent had been widely investigated both in preclinical and in clinical studies with no severe symptoms occurred. All of these highlight the potential of this vector for carrying human IL-12 and/or IL-2 genes as an efficacy and safety candidate for hepatoma carcinoma therapy in clinical application.
Conclusion
The results of this study warrant further investigations of rClone30 encoding human IL-12 and/or IL-2 in clinical application.
Footnotes
Authors’ Note
Guiping Ren and Guiyou Tian contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funds of Science and Technology Planning Program of Heilongjiang Province (GC13C104), Educational Committee of Heilongjiang Province (12521z004), Research Fund for the Postdoctoral Program of Heilongjiang Province (LBH-Q09162), National Natural Science Foundation of China (J1210069/J0116), the Heilongjiang Province Project of Applied Technology Research and Development (GC13C105), and the National Natural Science Fund Biologic Science Base Improve Program of Research Training and Capacity(J1210069/J0124).
