Abstract
Treatment uncertainties in radiotherapy are either systematic or random. This study evaluates the sensitivity of fractionated intensity-modulated proton therapy (IMPT) lung treatments to systematic and random setup uncertainties. Treatments in which single-field homogeneity was restricted to within ±20% (IMPT20%) were compared to full IMPT (IMPTfull) for 10 patients with lung cancer. Four-dimensional Monte Carlo calculations were performed using patient computed tomography geometries with ±5 mm systematic or random setup uncertainties applied over a 35 × 2.5Gy(RBE) treatment course. Fifty fractionated courses were simulated for each patient using both IMPT delivery methods with random setup uncertainties applied each fraction and for 3 energy-dependent spot sizes (big spots, σ≈18-9 mm; intermediate spots, σ≈11-5 mm; and small spots, σ≈4-2 mm). These results were compared to Monte Carlo recalculations of the original treatment plan assuming zero setup uncertainty. Results are presented as the difference in equivalent uniform dose (ΔEUD), V95 (ΔV95), and target dose homogeneity (ΔD1-D99). Over the whole patient cohort, the ΔEUD was 2.0 ± 0.5 (big spots), 1.9 ± 0.7 (intermediate spots), and 1.3 ± 0.4 (small spots) times more sensitive to ±5 mm systematic setup uncertainties in IMPTfull compared to IMPT20%. IMPTfull is 1.9 ± 0.9 (big spots), 2.1 ± 1.1 (intermediate spots), and 1.5 ± 0.6 (small spots) times more sensitive to random setup uncertainties than IMPT20% over a fractionated treatment course. The ΔV95 is at least 1.4 times more sensitive to systematic and random setup uncertainties for IMPTfull for all spot sizes considered. The ΔD1-D99 values coincided within uncertainty limits for both IMPT delivery methods for the 3 spot sizes considered, with higher mean values always observed for IMPTfull. The paired
Keywords
Introduction
In radiotherapy, setup uncertainties are typically classified as either systematic or random. 1 Systematic setup uncertainties remain constant over the treatment course, displacing the dose distribution relative to the intended target, whereas random setup uncertainties vary daily and lead to a blurring of the cumulative dose distribution.
The finite range of protons introduces an additional level of complexity and uncertainty compared to photons. The in-patient proton range can be influenced by both systematic and random setup uncertainties, the effects of which will be magnified in low-density lung tissue. Range uncertainties in proton therapy have been comprehensively presented in a previous review article. 2
Intensity-modulated proton therapy (IMPT) allows generation of highly conformal dose distributions by allowing individual fields to deliver an inhomogeneous dose to the target, such that the amalgamated dose meets the planning constraints.
3
This is in direct contrast to single-field uniform dose (SFUD) treatments that require individual beams to yield homogeneous target doses. There is no agreed upon value of target dose homogeneity that constitutes an “SFUD” field, posing the question how uniform is “
The intra-target dose gradients in IMPTfull plans can be steeper than those of IMPT20%, likely increasing their susceptibility to setup uncertainties. 6 Degradation of the dose distribution due to respiratory motion may exacerbate this susceptibility.
Motion effects are defined herein as the combination of dose blurring, which occurs in the target periphery, and interplay effects, which occur throughout the target volume. Increasing treatment planning margins can reduce the effect of dose blurring, at the expense of increased normal tissue irradiation. As interplay effects arise due to relative motion between the radiation beam and the patient anatomy, 7 increased margins will not counteract this effect. Due to their finite range in tissue, for protons (and particles in general) this relative motion can either be tumor motion or movement of the anatomy traversed by the beam proximal to the target, for example, ribs. Interplay effects are known to vary with spot size, 8,9 hardware parameters of the specific facility, and patient breathing characteristics. 10 Numerous investigators have previously analyzed either interplay effects, for example, 4,8,10 -14 or setup uncertainties, for example, 5,15,16 for particle lung treatments individually.
The work presented herein extends upon the previous studies of interplay and motion effects in proton therapy by applying a systematic or random setup uncertainty, while also modeling motion effects using 4D Monte Carlo simulations for each fraction over a clinically relevant treatment course. This investigation includes all specific motion effects investigated previously, that is, interplay and setup uncertainties, and further integrates the effect of fractionation.
Methods
Treatment planning
Retrospective IMPTfull and IMPT20% treatment plans were generated using ASTROID
17
for 10 patients with lung cancer whose details are shown in Table 1. All treatment plans consisted of 2 fields, with the beam angles chosen to minimize the dose to healthy lung and other critical tissues. Plans were generated on the average intensity projection computed tomography (CT), assuming delivery of 87.5Gy(RBE) in 35 × 2.5Gy(RBE) fractions in accordance with a multi-institutional clinical trial protocol active at our institution and the study of medically inoperable non-small cell lung cancer of Chang
Patient characteristics showing GTV and motion amplitudes along the SI, AP and LR axes. All motion amplitudes were obtained directly from patient 4DCT scans.

Abbreviations: GTV, gross tumor volume; SI, superior–inferior; AP, anterior–posterior; LR, left–right; 4DCT, 4-dimensional computed tomography.
As interplay and motion effects vary with spot size, 8,10 plans were generated and analyses performed for 3 distinct energy-dependent (90-230 MeV) spot sizes, namely, big spots (BS, σ ≈ 18-9 mm), intermediate spots (IS, σ ≈ 11-5 mm), and small spots (SS, σ ≈ 4-2 mm). These spot sizes were chosen to encompass the range available from current clinical systems.
Monte Carlo simulations
Monte Carlo simulations were performed using the TOPAS code (version beta8) 19 and Geant4 (version 4.9.6.p02). TOPAS has been used previously for Monte Carlo studies of proton therapy. 4,8,10,20 -22
The 4D simulations were performed on the 10 phases of the patient 4-dimensional computed tomography (4DCT) assuming the following timing properties of the delivery system: beam current: 2nA, energy switching time: 1s, spot settling time: 5ms, lateral scanning speeds: 30m/s (x) and 3m/s (y), and starting breathing phase: 0%
These specifications were chosen to represent those of a “
All spots within each of the proton fields were sorted in a time-dependent manner to simulate delivery to the corresponding phase of the patient 4DCT. These intermediate results were subsequently registered using the deformable registration framework “plastimatch” 23 to the 50% (end exhale) phase. All analyses of the 4D simulations were performed on the 50% phase clinical target volume (CTV) post-registration.
The focus of this work was not to evaluate the accuracy of analytical treatment planning calculations. Therefore, the 4D simulations were compared to a recalculation of the treatment plan using 3D Monte Carlo that assumed zero setup uncertainty. This 3D calculation is referred to throughout this article as the “
Treatment Setup Uncertainties
Systematic and random setup uncertainties arise from different circumstances and as such were investigated separately. The scenarios considered were ±5 mm shifts along the 3 coordinate axes (left–right, superior–inferior, and anterior–posterior) and nominal alignment. The magnitude of ±5 mm was selected based on the previous investigation of Nelson
To investigate the effects of systematic setup uncertainties, identical shifts were applied to each fraction of the treatment. As outlined previously, uncertainties were applied on a per-field basis, yielding 49 different combinations (nominal position and ±5 mm shifts along each axis = 7 scenarios per field) for the 2 field plans for each of the 10 patients.
To investigate random setup uncertainties, shifts were randomly applied for both fields for each of the 35 fractions in the treatment course. The randomization means that for some fractions, both fields experienced the same setup shift, while for others the shifts for the individual fields differed. These variations were studied based on the previous work of Nelson
The effect of treatment setup uncertainties was evaluated via the equivalent uniform dose (EUD), V95, and D1−D99 values. D1−D99 was preferred due to its heightened sensitivity compared to D5−D95. 10 The difference between the EUD in the 4D simulations (including setup variations) and in the TPR is defined as ΔEUD. Analogous definitions were used for ΔV95 and ΔD1−D99.



Taking the standard deviation of a given metric over all considered scenarios, that is, shifts along different axes, showed the variation arising from the different setup uncertainties. Taking the ratio of standard deviations (RSD) of the IMPTfull and IMPT20% standard deviations demonstrated which of the 2 techniques experienced more variation from these considered setup uncertainties.

From Equation 2, an RSD > 1 implies that IMPTfull was more sensitive to setup uncertainties than IMPT20%, as the range of variation in the given metric was higher. The paired
Results
Systematic Setup Uncertainties
Figure 1A-C shows the range of ΔEUD values when systematic ±5 mm setup uncertainties are applied throughout a treatment course for the different spot sizes and IMPT deliveries. For each of the 10 patients, the left (right) box represents IMPTfull (IMPT20%). The corresponding data for ΔV95 and ΔD1−D99 are shown in Figures 2A-C and 3A-C, respectively.

Box and whisker plots of the difference in EUD (ΔEUD) compared to the TPR for (A and D) BS, (B and E) IS, and (C and F) SS IMPTfull (blue, left in each patient column) and IMPT20% (red, right in each patient column) for 10 patients with systematic (A-C) and random (D-F) ±5 mm shifts in left–right, superior–inferior, or anterior–posterior axes. The horizontal line shows the median value, with the boxes bounding the 25th and 75th percentiles. The whiskers range from the minimum to maximum values and the black dots represent the ΔEUD with zero misalignment. EUD indicates equivalent uniform dose; TPR, treatment plan recalculation; BS, big spots; IS, intermediate spots; SS, small spots; IMPT, intensity-modulated proton therapy.
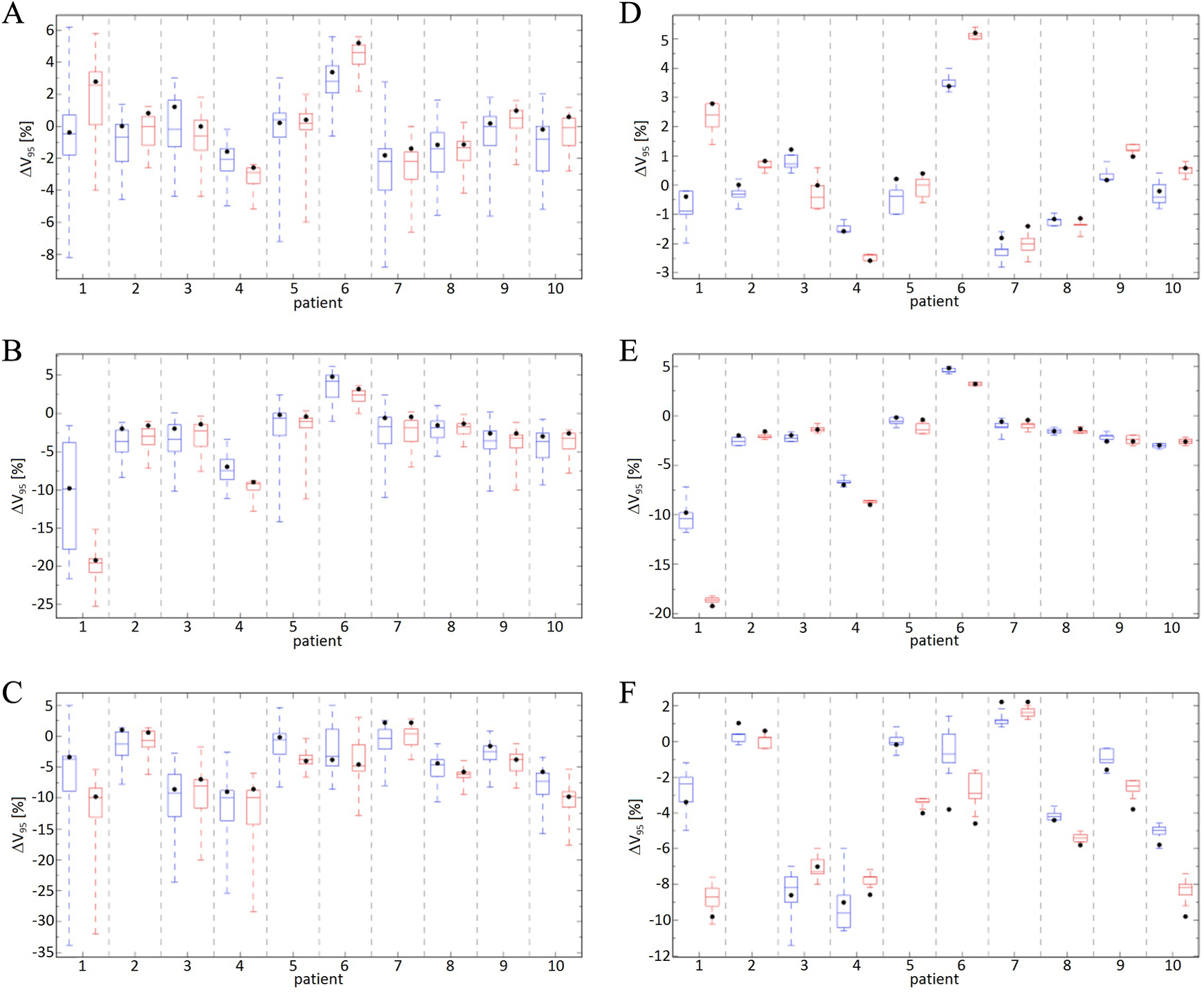
Box and whisker plots of the difference in V95 (ΔV95) compared to the TPR for (A and D) BS, (B and E) IS, and (C and F) SS IMPTfull (blue, left in each patient column) and IMPT20% (red, right in each patient column) for 10 patients with systematic (A-C) and random (D-F) ±5 mm shifts in left–right, superior–inferior, or anterior–posterior axes. The horizontal lines show the median value, with the boxes bounding the 25th and 75th percentiles. The whiskers range from the minimum to maximum values, and the black dots represent the ΔV95 with nominal alignment. TPR indicates treatment plan recalculation; BS, big spots; IS, intermediate spots; SS, small spots; IMPT, intensity-modulated proton therapy.
The RSDs for the ΔEUD averaged over the patient cohort are 2.0 ± 0.5 (BS), 1.9 ± 0.7 (IS), and 1.3 ± 0.4 (SS) for the different spot sizes. For the ΔV95, the corresponding values are 1.5 ± 0.3 (BS), 1.6 ± 0.5 (IS), and 1.4 ± 0.4 (SS). The ΔEUD and ΔV95 are more sensitive to systematic setup uncertainties for IMPTfull plans. There are specific patients for IS (#9) and SS (#4 and #6) that exhibit larger variations for ΔEUD for IMPT20%; however, the differences between IMPTfull and IMPT20% are <1% for all such occurrences. For ΔD1−D99, the differences between the IMPT deliveries are smaller, with RSDs of 1.3 ± 0.5 (BS), 1.1 ± 0.4 (IS), and 1.1 ± 0.2 (SS) obtained.
The paired
Random setup uncertainties
Figure 1D-F shows the difference in the ΔEUD between the simulated fractionated treatments and TPR defined in Equation 2. The RSDs for the different spot sizes averaged over all patients are 2.0 ± 0.9 (BS), 2.1 ± 1.1 (IS), and 1.5 ± 0.6 (SS). For the ΔV95, shown in Figure 2D-F, the RSDs are 1.4 ± 0.6 (BS), 1.8 ± 1.2 (IS), and 1.7 ± 1.2 (SS). As was observed for the systematic setup uncertainties, the ΔEUD and ΔV95 are more sensitive to random setup uncertainties for IMPTfull. The RSDs of 1.3 ± 0.5 (BS), 1.1 ± 0.6 (IS), and 1.2 ± 0.4 (SS) were observed for ΔD1−D99 (Figure 3D-F) for the different spot sizes, showing the target dose homogeneity has similar sensitivity to random uncertainties for the 2 IMPT techniques.
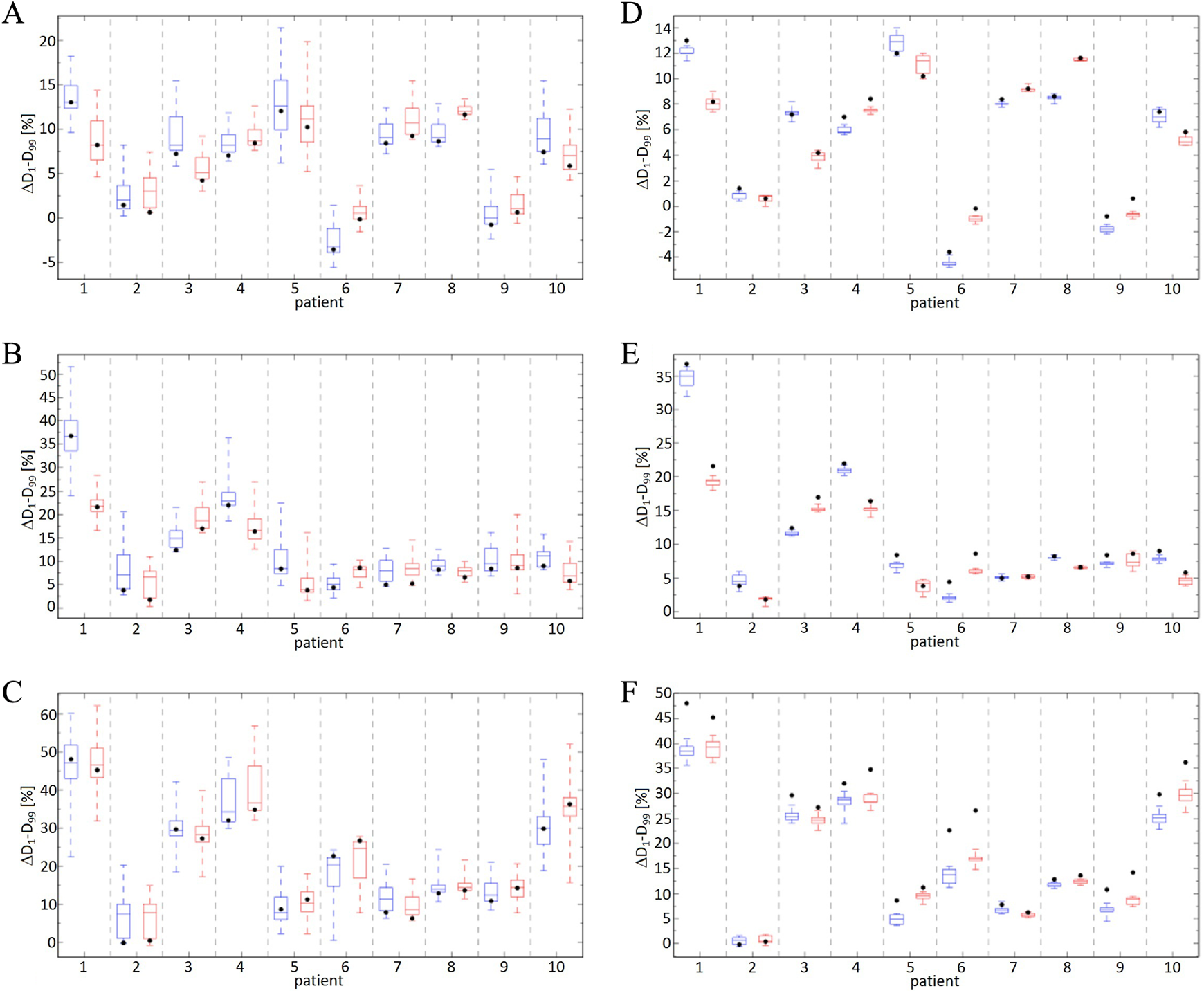
Box and whisker plots of the difference in D1−D99 (ΔD1−D99) compared to the TPR for (A and D) BS, (B and E) IS, and (C and F) SS IMPTfull (blue, left in each patient column) and IMPT20% (red, right in each patient column) for 10 patients with systematic (A-C) and random (D-F) ±5 mm shifts in left–right, superior–inferior, or anterior–posterior axes. The horizontal line represents the median value, with the boxes bounding the 25th and 75th percentiles. The whiskers range from the minimum to maximum values, and the black dots represent the ΔD1−D99 with zero misalignment. TPR indicates treatment plan recalculation; BS, big spots; IS, intermediate spots; SS, small spots; IMPT, intensity-modulated proton therapy.
The paired
Discussion
The IMPTfull and IMPT20% treatments were simulated for 10 patients having lung cancer with systematic and random setup uncertainties applied predelivery. Systematic uncertainties were simulated by applying the same 5 mm shift to each 2.5Gy(RBE) fraction of a 35 × 2.5Gy(RBE) treatment course. Random setup uncertainties were investigated by randomizing the setup uncertainties for each fraction of the treatment. All Monte Carlo simulations were 4D in that they included setup uncertainties and intrafractional motion effects. Fifty fractionated treatments, each 87.5Gy(RBE) in 35 × 2.5Gy(RBE) fractions were generated with random setup uncertainties for each of the 10 patients for each of the 3 spot sizes and both IMPTfull and IMPT20%. This increased the number of randomized treatment courses rather than only randomizing once, in case a particular randomization resulted in a particularly poor (or good) result. As can be seen when comparing the systematic to the random uncertainty data in Figures 1 to 3, the variation among 50 fractionated treatments with random setup errors is much smaller than among the systematic setup uncertainties. As there is a very small but finite probability that random uncertainties can present as systematic, the systematic uncertainties (left column of Figures 1 –3) can therefore be viewed as the (highly unlikely) worst-case scenario of a fractionated treatment with random setup uncertainties.
While the sensitivity to interplay effects alone does not differ significantly between IMPTfull and IMPT20%,
4
the sharper intra-target dose gradients in IMPTfull make this technique more susceptible to setup uncertainties. The calculated RSDs were always greater than 1, indicating that IMPTfull is more susceptible to both systematic and random setup uncertainties than IMPT20% for all conditions studied. For ΔD1−D99, the RSD values coincide within the uncertainty limits; however, the mean values are still all above 1. For RSD values ∼1, the paired
Beam angles were chosen to minimize the dose to normal lung. While this has obvious clinical implications, it may lead to the use of sub-optimal beam angles in terms of robustness against setup uncertainties. This is highlighted through examination of Figures 1 –3, which show that the delivery with nominal patient alignment does not give the smallest difference compared to the TPR for any patient. Increasing the number of fields in the treatment plans could also affect the susceptibility to setup uncertainties.
The results of this work are likely specific to the treatment-planning system used due to the differences in the optimization algorithms invoked in different platforms. The specific planning methodology and objectives could also be decisive. In this study, we ensured that our target objectives were met and then reduced lung and organ at risk (OAR) doses as much as possible. All normal tissue constraints specified by our institution were met for the lung and all OARs. If one accepted higher OAR doses with improved target conformity (or vice versa), this would likely alter the results observed. Increasing treatment-planning margins could reduce sensitivity to setup uncertainties; however, they will not mitigate interplay effects.
All analyses in this work were performed on a single 4DCT for each patient in the cohort, assuming a constant respiratory period of 5s. It is well known that patients will not necessarily breathe in such a consistent fashion. A variable respiratory period, either intrafractionally or interfractionally, increases averaging effects, 10 thereby reducing interplay effects and potentially any consequences of setup uncertainties. Averaging effects arise with fractionation due to variations in, for example, respiratory period, respiratory pattern, and/or setup position, which varies the position of hot and cold spots. This leads to a smearing of the dose distribution, with potentially improved target dose homogeneity compared to any single fraction. Increasing the number of beams used will increase averaging effects, 26 assuming they each cover the target volume quasi-homogeneously, which cannot be assumed for IMPT treatments, particularly IMPTfull.
Treatment planning using robust optimization against patient setup uncertainties would likely reduce the ramifications of setup uncertainties, at the expense of increased lung irradiation. Previous studies have outlined algorithms to include robust optimization in the treatment-planning process, for example 27 ; however such algorithms are not widely available in commercial (or in-house) platforms currently. Those platforms that have the ability to perform robust optimization execute the processes, and hence dosimetric and clinical evaluations, based on the planning CT and a 3D dose distribution, essentially neglecting intrafractional patient geometry changes and the time-dependent nature of pencil beam scanning proton fields. Full 4D optimization incorporating patient respiration, hardware timing parameters, and treatment-related uncertainties would be highly desirable.
Hypofractionated treatment courses (eg, 4 × 12Gy(RBE)) will likely yield different results compared to 35 × 2.5Gy(RBE) due to the lower number of fractions. Additionally, given the lower number of fractions in such treatment schemes, it is likely that rescanning and/or gating will need to be applied clinically.
The starting phase is the point in the patient respiratory cycle at which the beam delivery commences. Previous work 10 has shown the influence that the starting phase has on the interplay effect, showing large variations without a systematic trend. Assuming respiratory monitoring is not used, the starting phase will vary daily, diminishing the consequences of interplay on the cumulative dose distribution. 8 Given the variability introduced via the starting phase, we chose to perform this work using a single starting phase (0%). This allows us to be sure that the differences observed here are not influenced by variations in the starting phase. In a clinical setting, the daily variation in starting phase will increase averaging effects, thereby additionally smearing out interplay effects. 4,8,10
An energy switching time of 1 s and a spot settling time of 5 ms were assumed throughout this study. Varying the parameters of a proton scanning system alters the time structure of the field and hence the magnitude of interplay effects, 10 with longer delivery times generally reducing interplay via increased averaging effects.
Motion mitigation techniques, such as respiratory gating and rescanning, were not considered in this investigation. Adoption of motion mitigation techniques in this work could also have masked some of the effects from treatment uncertainties. The lack of clinical data for lung treatments with scanned proton beams means that there is no clear motion threshold above which such mitigation techniques would be implemented clinically. Interplay effects occur within the target volume and cannot be mitigated using increased margins. Rescanning techniques 26,28 can be used to reduce the interplay but not to reduce margins. Tracking 29 or respiratory gating 30 will allow the margins to be reduced but will not eliminate interplay effects altogether. A potential solution would be to combine rescanning and tracking into retracking, 31 which would reduce interplay effects and also allow reduced margins. Similarly, phase-controlled rescanning, 32 which combines gating and rescanning, could be a suitable treatment option.
Conclusion
Lung treatments utilizing IMPTfull are more sensitive to both systematic and random setup uncertainties than IMPT20%. Systematic setup uncertainties lead to larger deviations from the original treatment plan compared to random. Over the course of a fractionated treatment course (35 × 2.5Gy(RBE)), the differences between IMPTfull and IMPT20% remain statistically significant for the majority of this patient cohort. Furthermore, it has been shown that the impact of target motion combined with realistic random setup uncertainties over a conventionally fractionated treatment is surprisingly small, especially for large and intermediate spot sizes.
Footnotes
Acknowledgment
Thanks to Partners Research Computing for maintenance of the computing cluster.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Cancer Institute Grant R01CA111590.
