Abstract
This acute study was aimed at exploring the ability of a cryoablative lesion to drive the distribution of a concomitant in situ injection of a free epirubicin–ethanol–ethiodol–methylene blue mixture. We report the feasibility and safety of this new percutaneous computed tomography-guided combinatorial ablative procedure on VX2 tumors. Eight New Zealand white rabbits bearing 16 tumors on both side of the back muscle were randomly selected and treated on the same day with the following procedures: (1) 8 concomitant cryoablation and interstitial chemotherapy and (2) 8 intratumor marginal chemotherapy. For the latter, an injection needle was positioned at the inner distal margin of a first selected tumor side, where the chemotherapy was delivered during 5 serial sequences. For the concomitant therapy, a single cryoneedle maintained the ice front at the tumor margin, where a needle delivered the drug dose during 5 freeze–injection–thaw sequences. Enhanced computed tomography scans on days 3, 7, and 10 assessed the tumor contours and the tracer localization. Two rabbits were killed on days 0, 3, 7, and 10 for gross and histopathological analyses. During the concomitant therapy, ioversol was distributed at the tumor and iceball margins along with the methylene blue. Enhanced computed tomography on days 3, 7, and 10 showed a focal enlarging defect of the tumor marginal enhancing rim. The rim coincided with focal necrosis at histopathology. During the intratumor chemotherapy procedure, computed tomography showed that the tracers distributed mostly over the tumor mass. No marginal necrosis was detected at histopathology. On day 10, the tumor size for the intratumor chemotherapy group was twice that of the concomitant therapy group. No adverse events were observed. In this VX2 tumor model, our image-guided concomitant therapy is feasible and may enhance the effectiveness of a free epirubicin tracer mixture at the tumor margin.
Keywords
Introduction
Image-guided tumor ablation 1 is the direct application of chemical or thermal therapy to a specific focal tumor. Percutaneous interstitial cancer therapy uses needles or catheter-like devices that allow the direct intratumoral (IT) application of energy and/or the instillation of injectable diagnostic and/or therapeutic substances. The intratumor instillation uses drugs or biologic substances such as chemicals, cytotoxics, radioactive agents, gene, vaccine, or proteins which come in various desirable formulations. The goal is to distribute the drugs at a therapeutic concentration to the whole target, including a safety margin, while preventing drug leakage or reflux. Intratumoral chemotherapy (ITCH) and/or immunotherapy 2,3 is a promising method, which allows the delivery of the active substances directly into the tumor interstitial compartment at a high concentration, with little to no side effects compared to systemic administration. However, ITCH suffers a lack of technical standardization in terms of dose, localization, and timing as well as in association with locoregional or systemic therapies.
Cryoablation (CA), a thermal deprivation ablative procedure, has been used extensively in patients presenting with primary or metastatic tumors as a curative or palliative method or as a tumor localization or manipulation technique before resection. 4,5 Modern instrumentation allows a percutaneous, image-guided approach at a single or multiple primary and/or metastatic tumor sites in the liver, lung, prostate, kidney, bone, brain, skin, adrenals, lymph node, and soft tissue. 6 Breast and pancreas are more recent applications. Cryoablation is a well-standardized and effective technique. However, although a safe and effective method for relatively small tumors, typically ranging from 3 to 4 cm, CA has major limitations related to large size and/or irregular tumor and to the intraoperative control of the kill margin location that cannot be assessed with current imaging technology. 6 Despite these drawbacks and owing to recent advances in the treatment of cancer, it becomes increasingly evident that quality of life and prolonged survival can be obtained with repeat cryoablative sessions and combinatorial therapies. Many clinical studies show that combination therapies with CA (local, regional, and/or systemic) may be more effective than each therapy used alone. 6 Cryochemotherapy is a combination that has been investigated with variable results since the investigative works of Benson 7 and Ikekawa et al. 8 Drugs such as peplomycin, 5-fluorouracil, bleomycin, and doxorubicin in free form have been shown to enhance cryosurgery of prostate, liver, and lung tumors in vitro and at moderate subzero temperatures. 9,10 However, it is not known which timing, route of administration, formulation, or dosing for the associated cytotoxic drugs is best for improving the effectiveness and predictability of the cryoablative ice margin. 11 –13 Many studies use drugs formulated with vehicles such as a matrix, capsule, or nanoparticle that facilitate tumor targeting, drug residence, slow release rate, and intracellular uptake. Promising combination therapy associated with nanoparticle-encapsulated doxorubicin and CA 14 demonstrate enhanced eradication of cancer stem cells (CSTs) in vitro. We have demonstrated in vivo that intratumoral injection deposition of injectable microencapsulated chemotherapeutics at the tumor and iceball margin during conservative CA is feasible, safe, and effective at inhibiting the growth of prostate tumor xenografts 15 or at eradicating a few tumors. However, such formulations are complex, expensive, and still need to be homogeneously distributed into the target. In vitro, 16 we have demonstrated that low-molecular-weight (LMW <1000) free drug in solution or emulsion can be controllably spatially distributed, that is, driven within various tissue equivalent media, with concomitant serial freezing and local injection. Our present work describes a new ablative procedure: computed tomography (CT)-guided concomitant CA and interstitial chemotherapy (Co-CACH) on a VX2 tumor model in vivo. We first report the feasibility, safety, and initial efficacy of the Co-CACH procedure at targeting a solution of free epirubicin at the tumor margin. A secondary objective was to demonstrate on sequential CT and enhanced CT (E-CT) imaging that planning, targeting, monitoring, controlling, and assessing early treatment response are feasible.
Materials and Methods
Tumors
The animals, tumor, and study protocol were provided and approved by the Chinese People’s Liberation Army General Hospital and animal ethics committee. Ten healthy New Zealand white adult rabbits, weighing 2.8 to 3.5 kg (mean 3.2 ± 0.4 kg), were used. Under CT guidance, a VX2 tumor was implanted on each side of the back muscle.
A successful tumor take was confirmed by a nodular enhancement at E-CT 1 week after implantation. At 3 weeks, E-CT revealed the tumor as a regular round or ovoid-shaped mass, with volume ranging from 3.6 to 4.5 cm3 (mean 4 ± 0.5 cm3), marginal ring enhancement (tumor-enhanced rim [Trm]), and clear boundary. 17 Metastatic-free animals that presented with a well-delineated tumor were entered into the protocol.
Drug Formulation, Preparation, and Dosage
We are using an injectable therapeutic fluid solution (the injectate) made of low-molecular-weight (LMW) drug tracers and a cytotoxic drug, in an ethanol–saline solution. The injectate comprises the cytotoxic epirubicin (EPI; Zhejiang Hisun Pharmaceutical Co Ltd, Zhejiang, China), a radio-opaque contrast agent, ioversol (IO), 320 mg/mL (Tyco Healthcare, Shanghai, China [molecular weight, MW:807.11]), absolute ethanol 95% (EtOH; HongzhiWeida Industry & Trade Co. Ltd, Beijing, China), methylene blue (MB) 1% (Jiangsu Jumpcan Pharmaceutical Co Ltd, Taizhou, China), and 0.9% saline solution (NaCl). The EPI is a time- and dose-dependent, DNA-binding, permeant, vesicant cytotoxic used for IT chemotherapy. 18 The formula for the tumor volume is as follows: Vt = (L × W × W)/2, where L and W are the greatest length and width. The volume of injectate Vi (mean 2 ± 0.2 mL) is calculated with the formula Vi = Vt/2.3 The EPI dosing per tumor is 0.5 mg. Each animal receives an EPI total dose of 1 mg, that is, 0.3 mg/kg (body weight). 19
Preparation of the injectate is made in the operating room according to safety standards. The following proportion (v/v) of components was prepared: IO, 40%; MB, 12%; EtOH, 8%; and NaCl, 12%. A 3-mL plastic syringe contains the prepared dose. The needle directly connected to the syringe is filled with the solution before insertion, CT guidance, and intratumor positioning. The drugs were selected based on the following parameters: they had to be readily available, LMW (<1000), and low cost. The injectate had to be a cytotoxic solution of EPI that would be easy to prepare as well as easy to inject and monitor during CT imaging. The MB dye has no dark toxicity and has been used for histopathological determination of nonlethal margin during renal CA. 20 Ioversol has been used free or formulated for evaluation of intratumor injection distribution, 18 and its concentration (40%) is adjusted to keep the iceball CT imageable at all times during injection. Absolute EtOH is used at low concentration (8%) as a drug diffusion facilitator and cell membrane permeant. A recent study has confirmed that even lower EtOH concentrations (5%) mixed with cisplatin and injected intratumorally can kill tumors previously resistant to the cisplatin systemic regimen through the inhibition of membrane proteins in CSTs. 21 The EPI is a cytotoxic widely used for systemic, regional, or local chemotherapy. 2 It is also a vesicant drug, and it has a vascular toxicity when administered in a single dose. 22
Instruments
Computed tomography was performed using the Imager (Philips Brilliance CT Big Bore, Philips, Amsterdam, Netherlands; 16 slice, and slice thickness 5 mm). The cryosurgical unit consisted of Cryo-Hit (Galil Medical, Yokneam, Israel). We used a single 17G (1.47 mm, IceRod) cryoneedle for core tumor ablation and a 20G (0.9 mm), 9 cm long, Chiba needle for tumor injection connected to a plastic 2.5-mL syringe.
Overall Experimental Design
The study was performed as follows: 3 weeks after intramuscular implantation, we selected 10 animals bearing tumors presenting with a spheroid appearance, a regular circular or ovoid ring enhancement on E-CT images, and a mean volume of 4 ± 0.5 cm3.
For each animal, the tumors were randomly divided into treatment group (ITCH, 4 animals and Co-CACH, 4 animals), each group bearing 8 tumors, and control group comprising 2 animals and 4 tumors. Each animal of the treatment groups had both tumors injected on the same day. Additionally, 1 side was also randomly selected for concomitant conservative CA. The ablative effectiveness of a combination cryoablative freezing with simultaneous tumor margin injection of the injectate (Co-CACH) was analyzed in conjunction with IO localization distribution at sequential intraoperative CT scans, postoperative E-CT imaging, and MB dye localization at macroscopic and microscopic examination. The findings were compared with the changes following the intratumor marginal injection of the therapeutic fluid alone (ITCH) from days 0 to 10 after the operation.
Protocol for treatment series
The experiments included a total of 16 tumors in 8 animals (Table 1). Each animal is operated on under general anesthesia induced with amiodarone (Ketamine, Su-Mian-Xin) 0.5mg/kg, injected intramuscularly, and placed in the prone position under a CT imager. Each operation is conducted under sterile conditions. Both tumors are simultaneously treated. After the operation, the animals that aren’t killed are returned to their cages, where they have free access to food and water. No antibiotics or pain relievers are administered before or after the operation.
Treatment Groups and Tracers Distribution.a

Abbreviations: F-IN, Freezing injection sequence; T, Tumor; Tc, tumor core; Tm, tumor margin; ICEm, ice margin; CT, computerized tomography; E-CT, enhanced CT; Trm, tumor-enhanced rim; IO, ioversol; MB, methylene blue; DIST, distribution; NA, not available; ITCH, interstitial chemotherapy; CO-CACH, concomitant cryoablation and interstitial chemotherapy.
a Tracers distribution is described according to localization, coverage in percentage of target area, and IO intensity on day 0 (weak+, medium++, and high+++). n is the number of tumors. Five short time (30 seconds) sustained injection sequences of small volume. One-fifth of the calculated dose of mixture are effectuated at slow flow rate, 0.6 to 1.2 mL/min. Each intratumor injection (ITCH) or concomitant freeze-injection-thaw (Co-CACH) sequence is repeated every 3 to 5 minutes. The procedure allows the ice ball margin to be maintained at tumor margin. At each time point, 2 animals are euthanized for gross examination and histopathology.
Co-CACH Treatment Group, concomitant CA and interstitial chemotherapy, comprising 8 tumors: First a 0.9 mm diameter, 20G open end needle was E-CT guided and positioned to the inner distal side edge of the tumor. Second, a 17G cryoneedle was inserted into the tumor center, and the cooling tip was positioned 5 to 10 mm away from the tip of the injection needle (Figure 1A-1Br)

Intraoperative enhanced computed tomography (E-CT) and CT imaging of the decentered needle (NDL), the cryoneedle (cryoablation [CA]), and monitoring of the ioversol (IO) contrast distribution during the intratumoral chemotherapy (ITCH; left) and the concomitant cryoablation and interstitial chemotherapy (Co-CACH; right) procedures. On day 3, the E-CT allows early assessment of marginal response: the contrast gap (arrowhead) indicates a kill zone that enlarges on days 7 and 10.
Serial CT images of the growing iceball and injectate distribution were taken every 3 to 5 minutes. The freezing of the cryoneedle initiates the Co-CACH procedure; after 3 minutes the first amount of injectate—one-fifth of the total dose—is injected for 30 seconds. The injection flow rate ranged from 0.6 to 1.2 mL/min (mean 0.9 ± 0.3 mL/min; Figure 2). Freezing was conducted at the system’s lowest temperature and adjusted with the goal of “covering” the whole tumor with ice, but no further, that is, a conservative freezing without safety margin. The ice margin progression estimate was ∼2 mm/min, and it took between 2 to 4 minutes for it to reach the marginal location of the needle tip (Figure 1Br-1Cr).

A procedural diagram of the interstitial cryoablation and concomitant drug injection. The technique uses a single probe (CA), a single needle (NDL), a single injection site, and 2 freeze–injection–thaw (FRZ-INJ-TH) cycles. The drug mixture is injected during the first freeze sequence at the tip of a needle inserted at inner tumor margin (Tm, dotted curve). A drug dose is delivered during each FRZ or following thaw sequence (TH). The CA is in tumor center and the NDL is adjusted for position and distance from the tip of the CA. The FRZ-INJ-TH sequence can be repeated at will along with the delivery of a fraction of the drug dose, that is, up to 5 times in the present study. S is the probe activation start time.
The injectate distribution was imaged by CT during the 5 repeated sequences of 3 minutes freezing and simultaneous injection, lasting 30 seconds each. The freezing injection sequences were alternated with passive thaw sequences (FRZ-INJ-TH) lasting from 1 to 3 minutes. Upon completion of the procedure, the cryoneedle and frozen tumor were left to spontaneous thaw, and then the injection needle was removed before the cryoneedle (Figure 1D). No compression or occlusive material was applied on the puncture sites.
ITCH Treatment Group, intratumor marginal chemotherapy, comprising 8 tumors: This was performed on the tumor located on the opposite side of Co-CACH site. Under E-CT, the needle was positioned at the tumor inner distal edge, and the injectate was slowly injected. The injectate distribution was imaged at each of the 5 sequences of injection (Figure 1A-1Dl), which was made similar to the dose and timing of that in the Co-CACH procedure. Sequential intraoperative CT scans assessed the distribution pattern and localization of the injectate and allowed 3D reconstruction imaging of the contrast agent within the tumor (Figure 3).
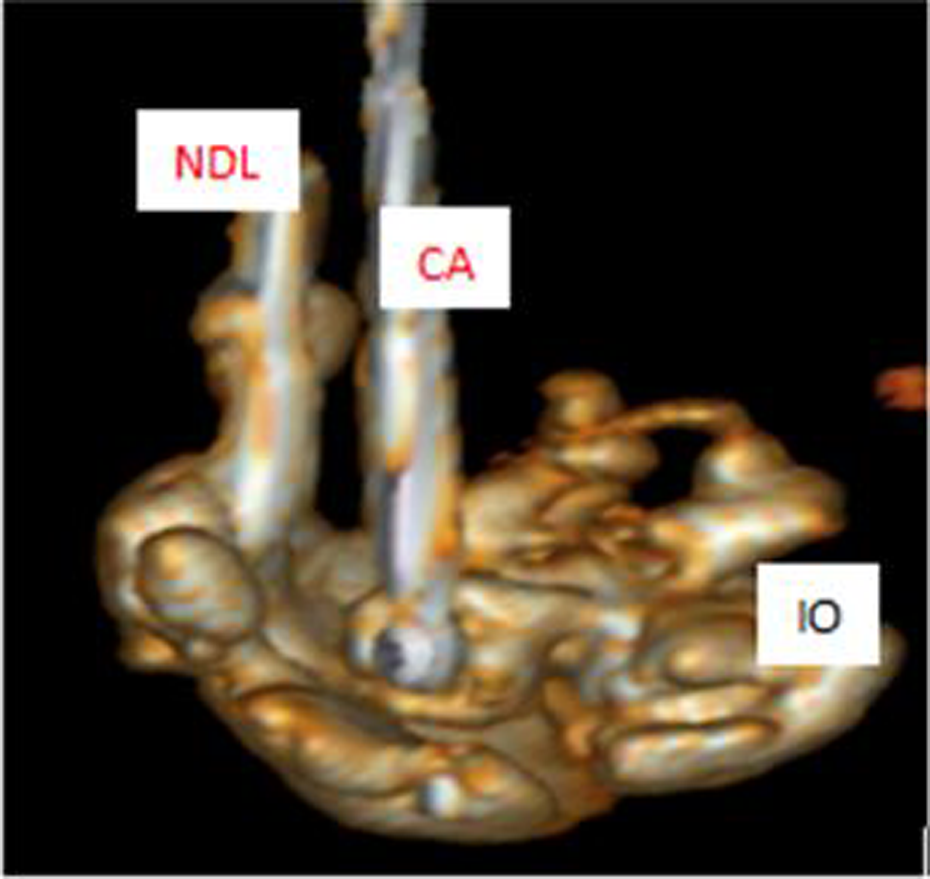
A 3-dimensional (3D)-reconstructed image of the ioversol (IO) distribution during a freeze-injection sequence of the concomitant cryoablation and interstitial chemotherapy (Co-CACH) procedure. Note that the cryoneedle (CA) and the injection needle (NDL) are located into a concave radio-opaque zone (IO), which matches with the nonopaque imprint of the impervious convex ice ball margin.
Two rabbits were euthanized postoperation, according to the approved method, after 1 hour (d0), 3 days (d3), 7 days (d7), and 10 days (d10; Figure 4). The E-CT scans were performed at d3, d7, and d10 before killing (Figure 5). Images of the tumors were compared with macroscopic examination and gross pathological section to evaluate tumor blue staining, necrosis, and coagulation from therapy.

Macroscopic aspect of the methylene blue (MB) dye distribution after the intratumoral chemotherapy (ITCH; (eft) compared to the concomitant cryoablation and interstitial chemotherapy (Co-CACH; right) observed on the first 72 hours. The dye distributes inhomogeneously into the ITCH-treated tumor but not at the margin, like the ioversol (IO) tracer does on day 0; Figure 1D (left). The margin viability is confirmed by the enhanced chemotherapy (E-CT) tumor rim enhancement of Figure 1E (left). One hour after the Co-CACH procedure, the tumor section passing by the long axis of the cryoneedle (white double arrow) shows the MB dye distributed at the tumor margin (yellow arrows) up to the needle track (black arrow). Given that the ice ball margin was located at the tumor margin, the therapeutic mixture containing the MB tracer has permeated the peripheral region of the ice ball like the IO tracer as shown in Figure 1D (right) [Color version of the figure available online].
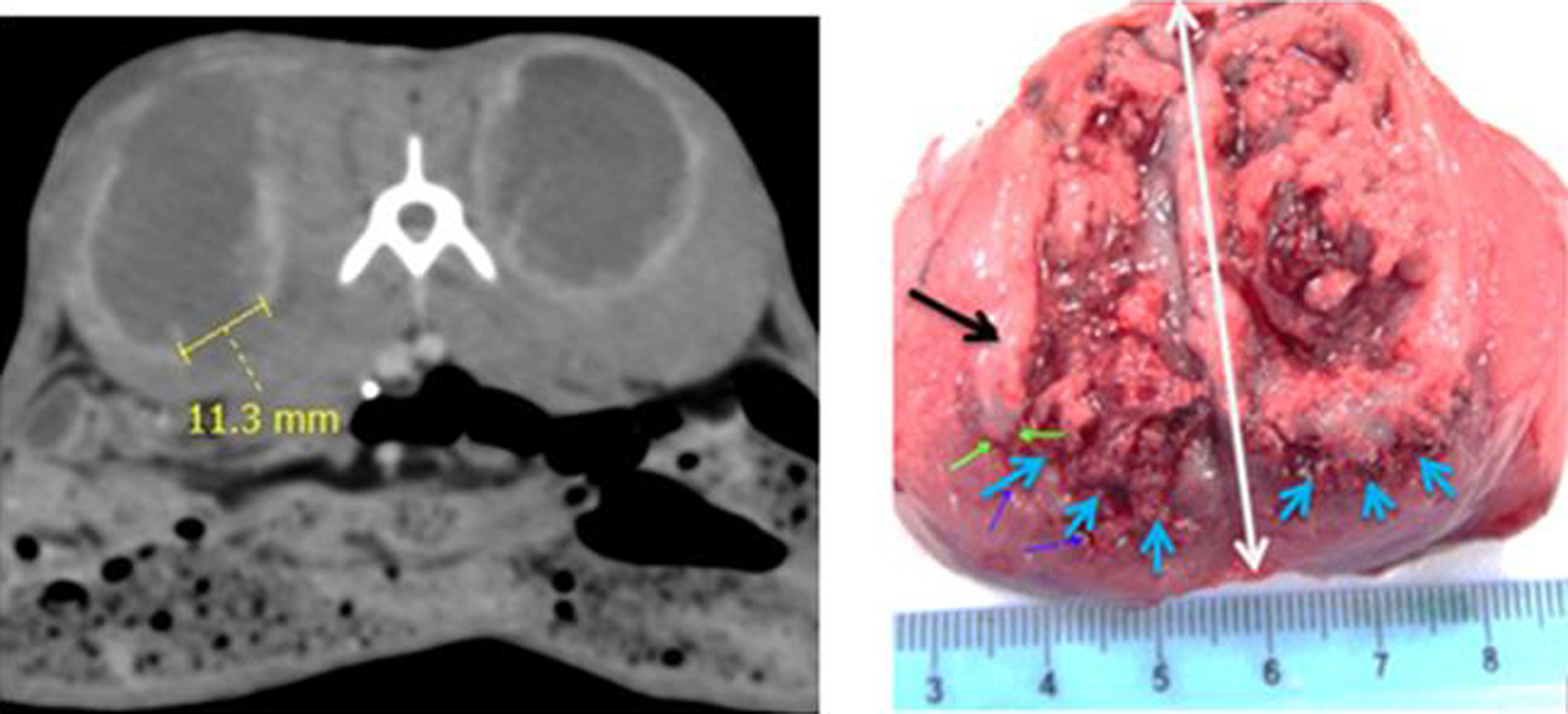
Ten days after the concomitant cryoablation and interstitial chemotherapy (Co-CACH) procedure, the tumor margin evidences a kill zone on the enhanced computed tomography (E-CT) image (left) and at macroscopic examination (right). The necrotic gap (blue arrows) at the tumor margin (green arrows) corresponds to the lack of ring enhancement measured on the E-CT. The black arrow shows the tumor margin of fish-like consistency [Color version of the figure available online].
Control group, untreated animals, comprising 4 tumors: The control group animals were observed for tumor growth until the end of observation period.
Results
The Iceball Margin Drives the Flow and Distribution of the Therapeutic Fluid Concomitantly Injected
During the freezing injection sequence of the Co-CACH procedure, the radio-opaque contrast agent, IO, of the injectate was visible on CT along the low-density ice ball margin but not within. The injectate was located at the edge of tumor margin, adopting an arc-, or cup-like pattern matching the ice margin on the 3D reconstruction. During passive thaw, the IO contrast spread a little within the inner tumor margin. The macroscopic examination of the tumor slices showed the blue stain (MB) distributed evenly within the tumor margin but not within the tumor core on day 0. Frostbite tissue was observed at the top skin edge of the tumor. Microscopic observation at low magnification (hematoxylin and eosin [HE], 40×) revealed multifocal carcinoma foci that invaded the tumor interface with normal tissue. These cancerous foci had a funicular or adenoid pattern, and mitotic cells were obvious at high magnification (HE, 400×).
From days 3 to 10, 2 zones, on the top and bottom of the tumor margin, show a lack of contrast (contrast gap) on the E-CT scan. The bottom contrast gap shows progressive enlargement of the enhancing ring circumference from 13% to 20% on days 3 and 10 (Figure 6). Gross examination shows a whitish tissue of fish-like consistency on top and bottom of the tumor along with necrotic tissue at the bottom tumor margin. Histopathology of the latter confirms coagulation necrosis with cell membranes disappearance, nuclear fragmentation, karyolysis, and cytoplasmic collapse (Figure 7). Conversely, during the same period, for the intratumor chemotherapy only group, a large number of viable tumor cells and no necrotic foci are visible at the tumor margin.
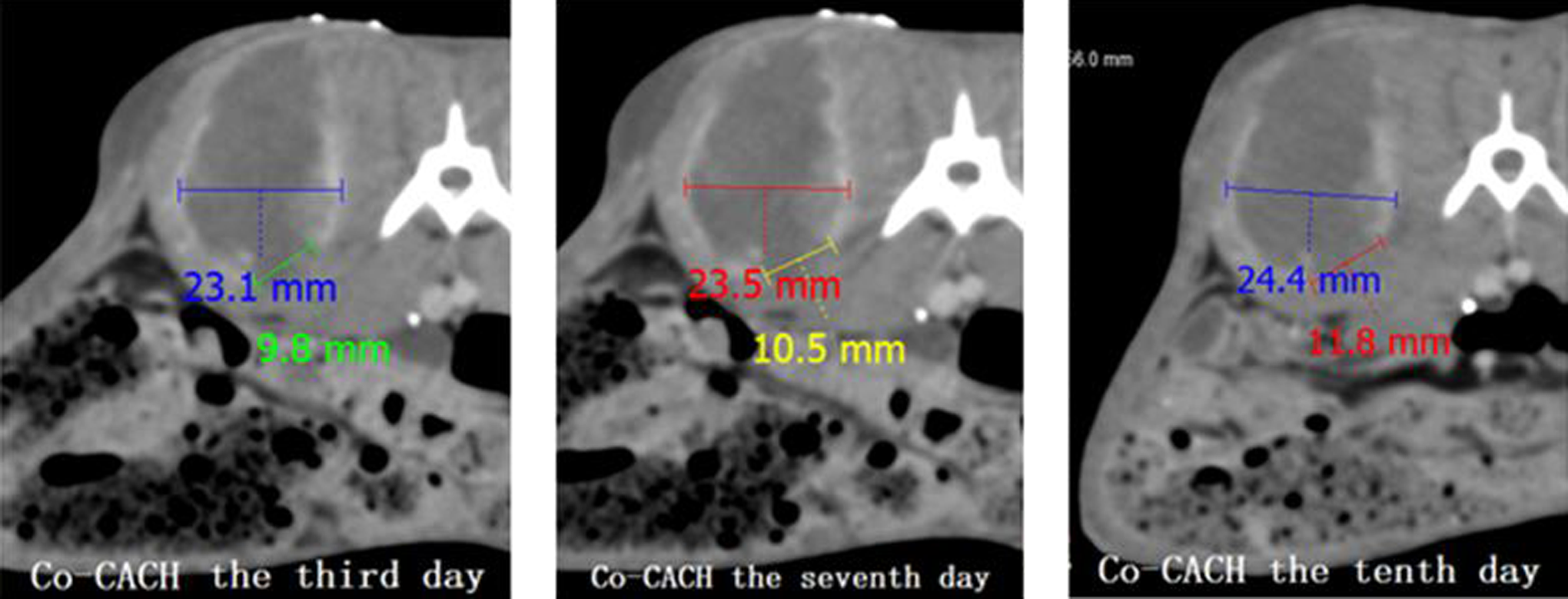
The gap of enhancement at the tumor rim on serial enhanced computed tomography (E-CT) from day 3 to day 10 was increasing in size, while tumor growth was progressing. Such evolution confirmed the limited kill efficacy of the conservative cryoablation on the tumor margin and the marginal kill effectiveness of the focalized cryo-assisted drug delivery.

Tumor margin histology (hematoxylin and eosin [HE],×400) 10 days after the concomitant cryoablation and interstitial chemotherapy (Co-CACH) procedure. Multiple zone of coagulation necrosis (white arrows) with nuclear fragments (blue arrows), absence of cells structures, coexist with tumor cell swelling and early signs of coagulative necrosis (green arrows) [Color version of the figure available online].
Injection at Inner Tumor Margin Distributes the Therapeutic Fluid into the Tumor
For the ITCH group, during injection and intraoperative CT imaging only a little amount of injectate was located at the tumor margin, while most of it was distributed to the tumor center after the injection. One hour after operation, gross pathological examination of the excised and sliced tumors showed leakage of the light blue-stained liquid with necrotic debris from the tumor along with a blue-stained tumor margin. The histopathology of the sample was similar to the Co-CACH group. From days 3 to 10, the tumor size was 50% larger than that of the Co-CACH group. A little amount of the contrast agent remained in the center of the tumor on the third day which was washed out later, and a full ring enhancement corresponding to a viable tumor margin was seen on days 7 and 10. Only a small amount of tumor cell nucleus fragmentation and karyolysis were observed between muscle fiber cells and tumor cells on the pathological section (Figure 8).

Tumor margin histology (hematoxylin and eosin [HE],×400) 10 days after the interstitial chemotherapy (ITCH) procedure. Hyperchromatic nuclei and nuclear fragments (green arrows) coexist with small, scattered zones of coagulation necrosis (white arrows) and multiple viable tumor cells. [Color version of the figure available online]
Histopathological aspects remained unmodified from days 0 to 10 and revealed scattered foci of cell alterations within tumor. The MB dye coloration was fading over the 10-day period.
Control Group Demonstrates Unabated Tumor Growth
Nodular enhancement on E-CT scan 1 week and 3 weeks after the implantation evidenced tumor take and growth with a regular round or oval mass, a peripheral ring enhancement (Trm), and a clear boundary. Growth continued until killing at the end of the observation period.
Discussion
In the recent years, CA has been used in conjunction with multiple drugs injected systemically, regionally, or locally, in order to help improve the kill efficacy of a cryolesion, that is, the death of the cancer cells in the peripheral zone of an ice ball. 23 Various adjuvants such as chemicals, chemotherapeutics, or biologics have been used in preclinical and clinical applications as “cryosensitizers” before or after CA. 11 The goal was to bring the edge of the kill zone (cryolesion lethal margin at ∼−25°C) that cannot be imaged intraoperatively closer to the edge of the ice ball (cryolesion nonlethal margin ∼−0.5°C) that can be imaged intraoperatively. Such realization would allow an improved monitoring of the cryoablative zone, especially for large, irregular, and/or ill-located tumors. Cryochemotherapy, a well-known ablative modality has raised much interest, but results have been inconsistent so far. Currently, it is recognized that CA has the potential for increasing a drug bioavailability 23 to a solid tumor and that intratumor chemotherapy allows a much higher concentration of the drug within the target and a quasi-absence of side effects. 2 However, cryochemotherapy suffers a poor control over the drug distribution 23 and a short half-life of free drugs within the target. 20 The past 2 decades have seen many unsuccessful attempts at “optimizing” the CA and chemotherapy combination likely due to complex and multiple interactions. Most drug injection and CA strategies involve systemic or locoregional injection before or after CA. 11,23 They miss the potential “driving” role of an intervening ice ball on a locally coinjected fluid.
For such cryochemotherapeutic optimization, we propose a novel procedure for facilitating drug targeting at the tumor margin during a percutaneous cryoablative procedure. This procedure is the interstitial delivery of an epirubicin traceable therapeutic solution at the margin of a large VX2 tumor during repeated short sequences of simultaneous freezing–injection–passive thaw (FRZ-IN-TH). The tumors were frozen without safety margin in order to evaluate the effect of the therapeutic fluid.
Technique Feasibility and Targeting Efficacy
We wanted the injectate to be intraoperatively distributed at the margin of the tumor at a therapeutic concentration and we hypothesized that the ice ball would be impervious to a concomitantly injected solution. Our strategy consisted of (1) formulating the injectate for tracing the drugs during CT without obscuring the ice ball image, (2) positioning the injection needle first with its tip located at the tumor inner distal margin and the cryoneedle located at the tumor center so that images have less metal needle-related artifacts, and (3) timely activating the cryoneedle and injection sequences so that injection can be performed at a slow flow rate with simultaneous CT monitoring during tumor freezing. The present results demonstrate that the injection and simultaneous freezing are possible and controllable since the ice ball is CT identifiable and the injectate flow is simultaneously visible at all times during the Co-CACH procedure.
Feasibility
Our concomitant CA and interstitial chemotherapy technique is unique in that it includes the fractionated injection of a single dose of therapeutic fluid in the course of a freezing cycle in the vicinity of a cryoneedle and it uses 5 repetitive short sequences of FRZ-IN-TH to deliver the therapeutic fluid. The Co-CACH procedure worked quite well at driving the injectate around the distal margin of the ice ball and tumor as evidenced on intraoperative CT scan, provided that the injection needle tip is properly positioned as is done for any image-guided interstitial targeting devices. 1 The central positioning of the cryoneedle tip within the tumor following that of the marginal injection needle located 5 to 10 mm apart were crucial steps for minimizing CT image distortion and for the ice ball to controllably reach the injection needle tip. We used this time period for doing the initial injection. The timely energy and fluid administration allowed us to repeatedly inject the calculated dose at a slow flow rate. Indeed, the 5 serial short sequences of FRZ-INJ-TH (3 min–0.5 min–2.5 min) did deliver the injectate, between 0.6 and 1.2 mL/min, without interruption, while still being able to image and maintain the ice ball at the tumor margin. Such injection rate minimizes the risk of leakage or reflux back through the needle tract. 24 However, we couldn’t totally prevent reflux from occurring either during injection or after needle removal. Indeed, some intraoperative reflux was intervening for either therapy but more frequent—50% vs 13%—and greater—0.3 vs 0.1 mL—for the ITCH compared to the Co-CACH procedure. Overall, the reflux to total dose ratio was estimated as minimal, that is, 5.5% (Co-CACH) and 15% (ITCH), and we didn’t compensate for this loss contrary to some authors. 3 It is well known that during IT chemotherapy, reflux is quite common, which many techniques try to prevent 24 –27 to avoid side effects or complications. The additional use of an injection needle during CA raises the question of an increased risk of tract seeding. Our acute study couldn’t assess this parameter. However, we think that the therapeutic injectate would kill the detached cells occasionally flowing back into the needle tract. Overall, the risk is quite low <0.14% on a large series of patients undergoing intratumoral needling. 28 –30
Targeting
Our Co-CACH technique shows a striking advantage at targeting the drug to the tumor margin compared to intratumor injection only. On intraoperative CT images of injectate distribution during the procedure, we observe a localized marginal flow. The expanding and/or steady ice ball margin seems impervious to the injectate IO tracer during the serial freezing sequences, becomes permeant when ice melts, and until killing 1 hour later. Conversely, the ITCH shows the injectate IO tracer immediately permeating the tumor with little amount distributing at the margin. We hypothesized that the ice margin extrudes the injected solution that is flowing in a bulk flow pattern along the interface between frozen and unfrozen tissue. Remarkably, the injectate doesn’t leak out into surrounding tissue during the freezing period but rather spreads into the melting ice ball and cryotreated tumor. Such containment is likely related to the vasoconstriction occurring in the hypothermal region, 6 preventing the outer spread of the drug. The blue coloration of the margin sparing the tumor center as well as the outer healthy muscular tissue clearly points to the deposition of the MB dye in the slush ice region of the ice ball. We hypothesize that the EPI drug with an MW of 579 which lies between that of IO (MW: 807) and MB (MW: 319) would settle down in the tracer region. Those results were expected from previous findings in vitro and on tissues ex vivo. 16
Safety
Both procedures were safe. No local or systemic adverse events (AEs) were observed. This is a confirmation of the known safety of intratumor chemotherapy 2,3 that was preserved by the ITCH as well as the Co-CACH procedures. Remarkably, the predominant marginal distribution of the injectate up to 1 hour after the Co-CACH operation didn’t entail any intra- or postoperative AEs. We think that the Co-CACH intraprocedural targeting efficacy is the essence of safety. To us, the ice margin assists with the deposition of the selected LMW free drugs mixture and with a marginal fluid flow to which factors such as a peripheral increased hydraulic conductivity, an augmented extracellular fluid fraction triggered by the freezing, the volume of injectate, 12,13,31 and a more central fluid-impervious hard ice 6,16,32 contribute. The slush ice region whose outer rim is CT imageable would act as a tunable fluid-channeling network that can be switched on and off with time-adjusted freeze and thaw sequences. This “water–ice” structural and spatial tuning had to be and was adequately set for our EPI solution, the selected conventional delivery devices, and the operative conditions. Additionally, the hypothermal region surrounding the frozen tumor has a protective effect on healthy cells. Hypothermia not only helps with targeting tumor cells with cytotoxics 33 but also prevents the uptake of EPI into healthy cells by inhibiting a transport protein. 34 Although our ITCH technique of serial, slow, small-dose marginal, and single-site injection diverges from conventional intratumoral chemoablative methods, 35 –38 it insured an adequate monitoring and good IT distribution of the injectate to more than 70% of the target tumor with moderate leakage. Larger series are needed to confirm those results.
Effectiveness
The Co-CACH procedural effectiveness is multiparametric and based on the following assumptions: (1) the intratumoral transport of IO, MB, and EPI would be similar; (2) the EPI penetration into its intracellular target—the DNA—would be facilitated by the conjunction of freezing 12,31 and the injectate-associated EtOH 8%; (3) the injectate would distribute over the target area for a sufficient period of time; and (4) a single dose of injectate would be sufficient to possibly image and detect an initial effectiveness, that is, coagulative necrosis.
All components of the fluidic injectate are LMW (less than 1000), and we were assuming they would be transported together with the bulk flow resulting from the procedure. Boucher et al have shown that such transport mechanism works for high-molecular-weight and LMW drugs as well. 39 Since we didn’t label the EPI molecule, we couldn’t directly evidence its transport or localization along with the tracers. However, our results point to its implication in the local observed effects.
Indeed, the ITCH procedure doesn’t kill the marginal cells, a fact probably attributable to the rapid intraoperative distribution 40,41 and dilution of the EPI solution within the tumor and its subsequent low concentration at the tumor margin. Contrast-enhanced CT scans on days 7 and 10 confirm the limited effectiveness of the injection alone with an integral peritumoral enhancing rim, known to be hypervascular. 17,42 This region exhibits viable cancer cells and cell clusters at microscopic examination. Conversely, a localized marginal kill (delayed coagulation necrosis) is observed at bottom of cryoablated and injected tumors, which matches with the contrast gap on serial E-CT appearing on day 3 and enlarging until day 10 (Table 1). The histopathology confirms that this zone has no more viable cells on day 3 compared to day 10, while viability was observed immediately after the operation. We infer that such delayed coagulation necrosis is due to the marginalized, concentrated EPI, which was rapidly distributed to target cells 40,41 in the sublethal, slush ice region 32 of the ice ball (non-CT imageable) during freezing but CT visible during melting. A similar necrotic effect has been described in previous studies using 5-fluorouracil-loaded microcapsules deposited at ice ball margin during passive thaw. 15 Strikingly, while slowly released low-dose 5-FU from microcapsules was cytotoxic for a prostatic tumor vasculature, it is a bolus injection and single exposure to free EPI that is apparently cytotoxic to the VX2 tumor target. This is an observation that is in line with Zhang et al study 43 and may pave the way for our technique to possibly become a cryo-assisted antivascular chemotherapy. For the Co-CACH technique to be effective at whole tumor ablation, the injectate should have been distributed to the whole ice ball and tumor margin. We noted that the ITCH was different from the Co-CACH procedure with regard to the percentage of tumor area where the tracers distributed. The IO marginal coverage was always incomplete at termination of the freezing injection sequences, over about 50% of the perimeter, and located at bottom of melted tumor. Thus, a similar volume of injectate distributing within more than 70% of a tumor with the ITCH technique was permeating about 50% of the perimeter of a frozen tumor of similar volume. It is likely that an augmented dose of injectate, drug concentration, and/or an additional site of injection could have been used, 25,38 with improved efficacy. 36 The effectiveness of the Co-CACH versus the ITCH procedure on tumor growth retardation was greater but not statistically quantifiable due to the limited number of tumors and the short time of observation.
Effectiveness is also linked to the EPI dosing. The total dose injected 1.57 mg, that is, 6 mg/m2, for a human equivalent dose 44 was decided from previous works 18 on a similar tumor model. At completion of injection, the EPI concentration per mL of tumor in the ITCH group was estimated at ∼0.138 mg/mL on an average, assuming a homogeneous distribution of a 2 mL injectate within a 4-mL tumor volume. Clearly, this dose is at least 1 order of magnitude inferior to a conventional human dosage for an intravenous route of administration. 41 However, our “low” dosage, that is, 25% to 50% of the dose used for an intra-arterial route of administration, 45,19 was effective at killing marginal cells starting 3 days after the cryo-assisted cytotoxic delivery but not after the intratumoral administration of the cytotoxic alone. We think that a higher marginal localization of EPI, with the Co-CACH procedure compared to the ITCH procedure, is responsible for such augmented effectiveness. 40 A contribution to effectiveness may also stem from a prolonged intratumor residence of the drug, up to 1 hour, at the tumor margin resulting from the Co-CACH procedure, the accompanying vasoconstriction, and vascular stasis. The prolonged retention of IO, greater than 0.5 hour, points to the simultaneous retention of EPI. 18 Of utmost interest was our ability to monitor the drug distribution and its early effectiveness with conventional CT imaging techniques. In our study, the localization, residence time, and wash out of the IO contrast allowed an improved monitoring and early assessment of marginal chemotherapy effectiveness during the Co-CACH procedure. Those results must be confirmed on a larger series.
We are aware of some limitations of this preliminary in vivo acute study: the limited number and large size of tumors did not allow for statistical analysis or assessing the long-term evolution of the marginal coagulation necrosis and the effectiveness of the combined procedure. Additionally, we did not evaluate the risk of needle tract seeding discussed earlier. Our study is qualitative rather than quantitative with regard to the IO coverage and MB-staining percentages and because of the presence of small reflux that we purposely didn’t compensate for, no exact relation of drug dose-to-kill effect was possible. Although we have related the localized necrosis to the EPI distribution during the Co-CACH procedure, we didn’t actually label the EPI and therefore we could have used a colored anthracycline cytotoxic. 46 Finally, we didn’t compare the ablative effect of “conservative” CA with our Co-CACH technique, since CA without safety margin has long been demonstrated unable to fully eradicate a solid tumor. 47,48 We purposely didn’t use a prefreezing IT administration of the therapeutic fluid, given that previous in vitro 16 and clinical applications had shown that an LMW cytotoxic-tracer drug solution or emulsion-injected intratumoral immediately before a cryoablative procedure is not displaced by the ice ball. 49
The VX2 tumor model has been extensively used for CA and chemical ablation as well. It is a fast-growing, highly aggressive tumor, 17 with no capsule that could have somehow retained the injected mixture. The intraoperative demonstration of the ice margin localization and tumor margin persistence of the injectate for a few hours (IO) and days (MB) raises much hope, with the potential use of our combinatorial cryo-assisted drug deposition therapy for ill-delineated lesions or metastases that usually lack any peripheral capsular barrier.
Conclusion
We are demonstrating for the first time that a CT-guided percutaneous cryoablative procedure combining concomitant short serial sequences of freezing and slow, sustained interstitial injection of an epirubicin–ioversol–ethanol solution is feasible. In our study, this cryo-assisted drug deposition technique is safe, controllable, and has a superior marginal targeting efficacy over intratumor chemotherapy used alone. The intraoperative imaging of the drug distribution and retention correlates with an augmented marginal kill at histopathology that is also imageable. These preliminary results need to be confirmed on larger sample, smaller tumors, and longer observation time. Our technique is a simple and immediately usable method that has the potential to increase the kill ratio at the margin of large, irregular, ill-defined, or otherwise incompletely cryoablated lesions. It may also impact the current intraoperative strategy of tumor cryolocalization or CA and extirpation by combining therapeutic injections aimed at improving local or locoregional treatment strategies.
Footnotes
Authors’ Note
PLP, XH, and YX conceived and designed the experiments. XH, XZ, PD, and XZ performed the experiments. XH and PLP analyzed the data. PLAGH animal Lab contributed to reagents/materials/analysis tools. JL and YA contributed to histopathology. PLP and XH wrote the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to Dr Patrick Le Pivert from Interventional Drug Delivery Systems & Strategies (ID2S2), Medical Cryogenics, Jupiter33458, Florida, for his assistance in the refinement of both the language and content of this work. This work was largely supported by the Animal experimental Center of Chinese PLA General Hospital.
