Abstract
Purpose:
Current craniospinal irradiation (CSI) protocols do not include the parotid gland as an organ at risk, potentially leading to late effects of xerostomia and secondary parotid malignancies. We analyzed the effect of CSI treatment parameters on parotid dose.
Materials and Methods:
We retrospectively reviewed 50 consecutive patients treated with CSI to an intracranial dose >26 Gy. Parotid dose was compared to a Radiation Therapy Oncology Group (RTOG) dose constraint (at least 1 parotid with mean dose <26 Gy). The effects of CSI dose (≤24 Gy vs 24 Gy), volumetric-modulated arc therapy (VMAT) versus 3-dimensional (3D) CSI technique, boost dose (≤24 Gy vs 24 Gy), supratentorial versus infratentorial boost location, intensity-modulated radiation therapy (IMRT)-based versus 3D boost technique, supine versus prone position, and age on parotid dose were analyzed using multivariate regression analysis.
Results:
The RTOG parotid dose constraint was exceeded in 22 (44%) of 50 patients. On multivariate regression analysis, lower CSI dose and VMAT CSI technique were associated with reduced parotid dose for the CSI fields. For the boost fields, lower boost dose and supratentorial boost location were associated with lower parotid dose. All 5 patients who underwent VMAT CSI met dose constraints. Furthermore, for infratentorial lesions with a total (CSI plus boost) dose prescription dose >50 Gy (n = 24), 11 of 16 patients who received low-dose CSI (18-23.4 Gy) were able to meet dose constraints, when compared to only 2 of 8 patients who received high dose CSI (36 Gy).
Conclusion:
Given the large number of patients exceeding the parotid dose constraint, the parotid gland should be considered an organ at risk. CSI dose de-escalation and IMRT-based CSI techniques may minimize the risk of xerostomia.
Introduction
Craniospinal irradiation (CSI) is the first-line treatment for many patients with malignancies at risk of neuraxis spread. These patients, who are often within the pediatric age range, may be subject to severe late radiation-associated toxicities. With increasingly better clinical outcomes, recent efforts have focused on reducing the late effects of CSI. For example, 3-dimensional (3D) CSI dose de-escalation for patients with average-risk medulloblastoma has led to improved neurocognitive outcomes. 1 Cochlea-sparing intensity-modulated radiation therapy (IMRT) for the posterior fossa boost has reduced ototoxicity. 2 Comprehensive IMRT-based CSI techniques (eg, IMRT or volume-modulated arc therapy (VMAT)) have improved neuraxis dose conformity. 3 –5 Proton therapy CSI techniques can result in improved normal tissue sparing by minimizing exit dose. 6
The parotid gland represents a potentially underrecognized organ at risk (OAR) for patients undergoing CSI. No clinical protocol, to our knowledge, requires its delineation during treatment planning, despite evidence suggesting that parotid irradiation may increase the risks of xerostomia 7,8 and secondary parotid malignancies. 9 Although treatment planning studies suggest that comprehensive IMRT-based CSI may reduce parotid dose and xerostomia risk, 10,11 the effect of CSI radiation techniques on parotid dose has not been rigorously analyzed. The purpose of this study is to analyze the dosimetric parameters affecting the parotid gland in a retrospective cohort of CSI patients treated with traditional 3D and more modern IMRT-based techniques.
Materials and Methods
Patient Characteristics
We conducted an institutional review board-approved, retrospective analysis of 50 consecutive patients treated with CSI between 2005 and 2013 who had a total (CSI plus posterior fossa boost) intracranial dose prescription greater than 26 Gy. Patients with a dose prescription less than 26 Gy were excluded, since none would have exceeded a parotid dose constraint from the Radiation Therapy Oncology Group (RTOG) 0225 protocol (at least 1 parotid gland with a mean dose <26 Gy). 12 Patients’ characteristics are summarized in Table 1. Median age was 13 years (range, 3-55 years). In all, 37 (74%) patients were between ages 3 and 21.
Patient Demographics.

Abbreviation: PNET, primitive neuroectodermal tumor.
Treatment Planning
In all, 45 patients were treated with 3D CSI, defined as opposed lateral cranial fields matched to posterior–anterior (PA) spinal fields, and 5 patients underwent IMRT-based CSI using VMAT, in which a single VMAT plan covered the entire craniospinal axis. 5 Twelve patients were treated in the prone position, and 38 patients were treated in the supine position. 13
Following CSI, 47 patients had additional boost irradiation to infratentorial lesions (n = 27), supratentorial lesions (n = 17), or the whole brain (n = 3). For infratentorial lesions, the boost planning target volume (PTV) encompassed the entire posterior fossa (n = 5), the posterior fossa plus a tumor cone-down (ie, gross residual disease or postoperative tumor cavity; n = 4), or the tumor cone-down only (n = 18). Boost volumes were treated using 3D (n = 17) or IMRT-based (n = 30) techniques. All treatment planning was performed using the Eclipse planning system (Varian Medical Systems, Palo Alto, California). Parotid dose constraints were not implemented for any patient.
Radiation was delivered in daily fractions of 1.8 to 2 Gy. Median CSI, boost, and total (CSI + boost) prescription doses were 30.6 Gy (range, 14.4-39.6 Gy), 22.5 Gy (range, 9.0-37.8 Gy), and 54 Gy (range, 34.2-59.4 Gy), respectively.
Parotid Gland Dosimetry
The right and left parotid glands were manually contoured for all patients. The parotid dose was defined as the average dose within a given contour. Right, left, and combined (right + left) parotid doses were obtained for the CSI fields, the boost fields, and the total (CSI plus boost) fields. Given no statistical difference between right and left parotid doses for the total fields based on a 2-sided dependent t test (P = .26), combined parotid doses were used as parotid doses for individual patients.
Mean parotid doses and the percentage of patients exceeding the parotid dose constraint were computed for the entire cohort and the following stratifications: age (≤21 or >21 years), patient position (supine vs prone), CSI dose (≤24 or >24 Gy), CSI technique (VMAT vs 3D), boost location (supratentorial, infratentorial, whole brain, or none), boost dose (≤24 or >24 Gy), and boost technique (IMRT based vs 3D). The chi-square test of equal proportions tested whether the percentages of patients meeting dose constraints for a given stratification were different based on a P value ≤ .05.
Statistical Analysis
Univariate and multivariate linear regression models were applied to determine parameters that significantly affected parotid dose. For the CSI fields (n = 50), the effects of age (≤21 or >21 years), position (supine versus (vs) prone), CSI dose (≤24 or >24 Gy), and CSI technique (VMAT vs 3D) on CSI parotid dose were evaluated. For the boost fields to supratentorial/infratentorial lesions (n = 44), the effects of age (≤21 or >21 years), position (supine vs prone), boost location (supratentorial vs infratentorial), boost technique (IMRT-based vs 3D), and boost dose on boost parotid dose were evaluated. Factors were considered significant if P ≤ .05. Analysis was performed with R statistical software v3.0 (The R Foundation for Statistical Computing).
Subset Analysis of High-Dose Infratentorial Target Volumes
Due to the heterogeneous population of patients within this cohort, a subset of 24 patients with infratentorial lesions prescribed a high total (CSI + boost) dose greater than 50 Gy was analyzed. This subset included 22 patients with medulloblastoma treated definitively. Patients were stratified based on CSI dose (low: 18-23.4 Gy vs high: 36.0 Gy) and CSI technique (3D vs VMAT). Mean parotid doses, percentages of patients exceeding the parotid dose constraints, and mean dose–volume histogram (DVH) curves were obtained for these stratifications. Xerostomia risks were estimated using normal tissue complication probability (NTCP) analysis based on a previously validated Lyman-Kutcher-Burman (LKB) mean dose model (TD50 = 39.9 Gy; m = 0.4) from the adult xerostomia literature. 14
For low-dose CSI patients, the total (CSI + boost) doses from 3D CSI plans were compared with those from VMAT CSI plans for 3 structures (parotid, cochlea, and submandibular glands) using a 2-sided independent t test. This analysis was conducted in order to determine whether VMAT CSI increased dose to surrounding structures.
Results
Parotid Gland Dosimetry
Parotid doses ranged from 12.3 Gy to 43.2 Gy, with a median of 24.8 Gy. The mean parotid doses for the CSI, boost, and total fields were 21.6 Gy (95% confidence interval: 19.6-23.6), 4.3 Gy (3.1-5.5), and 25.9 Gy (23.9-27.9 Gy), respectively. Twenty-eight (56%) patients met the RTOG parotid dose constraints.
Mean parotid doses from the CSI, boost, and total fields for various parameter stratifications are shown in Table 2. High-dose CSI patients (>24 Gy) exhibited greater mean parotid doses than low-dose CSI patients (≤24 Gy) for the CSI fields (26.7 Gy [24.7-28.8] vs 15.6 Gy [14.1-17.1]) and for the total (CSI + boost) fields (29.3 Gy [26.8-31.8] vs 22.0 Gy [19.6-24.3]). The CSI dose was the only parameter predictive of the percentage of patients (41% for high dose vs 74% for low dose) achieving dose constraints (P = .04). The VMAT CSI technique, when compared to 3D CSI, had a lower mean parotid dose for the CSI (13.3 Gy [8.7-17.9] vs 22.6 [20.5-24.6]) and total fields (17.0 Gy [12.3-21.6] vs 26.9 Gy [25.0-28.9]). Furthermore, all 5 VMAT CSI patients met dose constraints.
Parotid Dose Contributions From the Craniospinal (CSI), Boost, and Total (CSI + Boost) Radiation Fields for Selected Parameter Stratifications.a

Abbreviations: 3D, 3-dimensional; IMRT, intensity modulated radiation therapy; VMAT, volume-modulated arc therapy.
aParotid doses are expressed as mean and 95% confidence interval (CI).
bParameter stratifications with statistically different percentages meeting dose constraints at a P value ≤ .05.
Infratentorial boost fields contributed a greater percentage of the mean total parotid dose (27.2% [22.5-31.9]) than supratentorial boost fields (2.8% [1.3-4.2]). However, boost technique did not influence mean boost parotid dose (3D: 4.2 Gy [2.8-5.6] vs IMRT-based: 4.8 Gy [3.1-6.5]). Not surprisingly, higher boost doses (>24 Gy) were associated with greater mean boost parotid doses (6.8 Gy [4.7-8.8]) versus lower (2.5 Gy [1.6-3.4]).
Regression Analysis
For the CSI fields (n = 50), statistically significant factors associated with CSI parotid dose on univariate analysis were CSI dose (P < .01) and CSI technique (P < .01). On multivariate analysis, both factors remained significant (P < .01). As shown in Table 3, CSI parotid dose increased by 0.7 Gy for every 1-Gy increase in CSI dose. The CSI parotid doses for patients treated with VMAT CSI were an average of 7.7 Gy less than those who received 3D CSI, when controlling for age, position, and CSI dose.
Multivariate Regression Models of CSI Parotid Dose and Boost Parotid Dose for Selected Parameters.a

Abbreviations: IMRT, intensity modulated radiation therapy; VMAT, volume-modulated arc therapy; CSI, craniospinal irradiation.
aP value less than .05 represents statistical significance. ‘-’ signifies that the corresponding parameter was not included in the model.
For the boost fields to supratentorial and infratentorial lesions (n = 44), statistically significant factors for parotid dose on univariate analysis were boost dose (P < .01) and boost location (P < .01). Boost technique (IMRT based vs 3D) was not statistically significant (P = .58). On multivariate analysis, boost dose (P = .03) and boost location (P < .01) remained significant. Boost parotid doses for patients with supratentorial lesions were 5.5 Gy less than those for patients with infratentorial lesions, when controlling for age, position, boost dose, and boost technique.
Subset Analysis of High-Dose Infratentorial Target Volumes
Table 4 tabulates parotid doses for patients with infratentorial lesions treated to a total dose of >50 Gy (n = 24). Of 16 patients who received low-dose CSI (18-23.4 Gy), 11 (69%) met parotid dose constraints, whereas only 2 (25%) of 8 patients who received high-dose CSI (36 Gy) met dose constraints. The total parotid doses for the low-dose versus high-dose CSI patients were 23.3 Gy (20.3-26.2) and 32.1 Gy (28.0-36.3), respectively, with corresponding xerostomia risks of 16.4% (12.1-20.7) and 32.2% (23.0-41.5) on NTCP analysis. With low-dose CSI, mean total parotid doses for the 3 VMAT CSI and 13 3D CSI patients were 14.2 Gy (11.8-16.7) and 25.4 Gy (23.0-27.7), respectively. The difference between these doses was statistically significant based on an independent 2-sided t test (P < .01). Respective xerostomia risks were 5.5% (3.8-7.2) and 18.9% (14.8-23.1). The mean DVH curves in Figure 1 show improved parotid gland dosimetry with low-dose CSI and VMAT CSI techniques.
Effects of Low-Dose (18-23.4 Gy) and High-Dose (36.0 Gy) CSI on Mean Parotid Dose for Patients With Infratentorial Planning Target Volumes Treated to Greater Than 50 Gya.

Abbreviations: CSI, craniospinal irradiation; LKB, Lyman-Kutcher-Burman; TD50, median toxicity dose; VMAT, volume modulated arc therapy.
an = 24. NTCP represents the normal tissue complication probability for xerostomia, based on the LKB mean dose model with a TD50 of 39.9 and an m of 0.4.
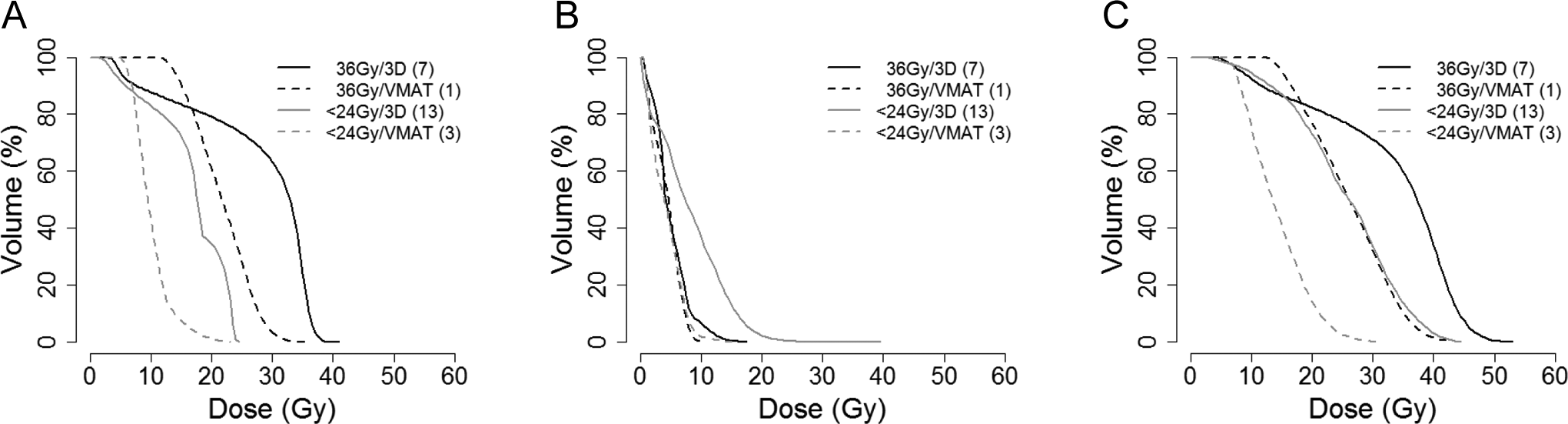
Mean dose volume histograms (DVHs) of craniospinal irradiation (CSI) fields (A), boost fields (B), and total fields (C) for patients with infratentorial planning target volumes treated to greater than 50 Gy (n = 24). DVHs are stratified by CSI dose (<24 Gy vs 36 Gy) and CSI technique (3D vs volume-modulated arc therapy [VMAT]).
There was no statistical difference in submandibular gland doses (P = .47) for the low-dose 3D CSI (8.2 [4.7-11.6]) and low-dose VMAT CSI (9.9 Gy [7.1-12.6]) subsets. There was also no statistical difference in cochlea doses (P = .62) for these same subsets (3D CSI: 37.7 Gy [35.6-39.7] and VMAT CSI: 38.3 Gy [36.6-40.0]).
Representative radiation isodose plans are shown in Figure 2. For the 11-year-old patient who received 23.4 Gy 3D CSI and a 32.4 Gy VMAT boost, the parotid glands were exposed to a higher radiation dose largely from the opposed lateral CSI fields (CSI parotid dose: 20.0 Gy; total parotid dose: 34.8 Gy). For the 13-year-old patient who underwent 23.4 Gy VMAT CSI and a 30.6 Gy VMAT boost, the parotid glands were exposed to much lower radiation dose (CSI parotid dose: 9.0 Gy; total parotid dose: 13.9 Gy).

Isodose plans of craniospinal irradiation (CSI), boost, and total (CSI + boost) fields for 2 patients with standard risk medulloblastoma. Parotid glands are contoured. A, Patient is an 11-year-old girl who underwent 23.4 Gy 3D CSI + 32.4 Gy VMAT boost. The CSI and total parotid doses were 20.0 and 34.8 Gy, respectively. B, Patient is a 13-year-old boy who underwent 23.4 Gy VMAT CSI + 30.6 Gy VMAT boost. The CSI and total parotid doses were 9.0 and 13.9 Gy, respectively. Isodose lines: orange, 50 Gy; yellow, 40 Gy; green, 30 Gy; blue, 20 Gy; pink, 10 Gy.
Discussion
In this study of unselected, consecutive patients treated with CSI, 44% of the patients exceeded the RTOG dose constraint for xerostomia. Patients at greatest risk of exceeding this constraint were those who received high-dose 3D CSI (36 Gy or greater) with boost irradiation to infratentorial target volumes. In this study, 6 of 7 patients with these attributes had mean parotid doses ranging between 29 and 43 Gy. Corresponding NTCP estimated xerostomia risks ranged from 25% to 58%. The high parotid doses are especially concerning, given the well-known association of parotid dose to xerostomia risk 14 and secondary parotid maliganicies 15 in this predominantly pediatric population.
This study demonstrated that recent trends of CSI dose de-escalation and novel IMRT-based CSI techniques, such as VMAT, have brought about marked reductions in both parotid dose as well as the estimated xerostomia risk. For 3D CSI fields, in which the parotid glands lie within the relatively fixed opposed lateral cranial fields (see Figure 2A), dose de-escalation has resulted in reductions in mean parotid dose from 33.0 Gy (28.7-37.3) to 25.4 Gy (23.0-27.7; 24% reduction) in a subset of 20 patients who underwent 3D CSI and infratentorial boost irradiation to a total dose greater than 50 Gy. For the small subset of 3 patients who underwent low-dose VMAT CSI, a further 45% reduction in mean parotid dose to 14.2 Gy (11.8-16.7) was observed. Unlike 3D CSI techniques, VMAT CSI was capable of modulating dose around the parotid glands as shown in Figure 2B. Similar dose reductions should be achievable with other IMRT-based CSI techniques including static-field IMRT and tomotherapy. Further dose sparing may be obtained by including the parotids as an OAR during IMRT optimization of the CSI fields.
The boost fields contributed a sizeable fraction of total parotid dose for infratentorial lesions (27.2% [22.5-31.9]) but not for supratentorial lesions (2.8% [1.3-4.2]). This result was expected, given the closer proximity of infratentorial target volumes to the parotid glands. For infratentorial lesions, boost dose was positively correlated with boost parotid dose. However, IMRT-based boost planning did not improve boost parotid dose. Although this result can be partially attributed to heterogeneous boost planning techniques (eg, differing 3D field arrangements, posterior fossa vs tumor-only boost fields), the parotid glands also were not designated as an OAR. Delineation of the cochlea as an OAR has led to a clinically significant reduction in cochlea dose and ototoxicity for IMRT posterior fossa boosts. 2 As such, we would expect at least a similar dose reduction if the parotid gland, an organ farther from the posterior fossa than the cochlea, were included as an avoidance structure. Parotid dose reduction for the boost field may be especially important for patients with medulloblastoma treated with low-dose CSI, since the boost field contributes a greater percentage of the total parotid dose for these patients (32.8% [27.8-37.7] compared to 16.0% [12.5-19.6] for high-dose CSI).
Our results are largely consistent with those reported in the literature. In an abstract by Kil et al, of 16 patients treated with CSI, the mean parotid dose was reduced from 34.3 ± 7.4 Gy with 3D technique to 17.2 ± 4.1 Gy with IMRT for CSI and boost. 11 In our study, the 5 patients who underwent VMAT CSI and boost exhibited a similar mean total parotid dose of 17.0 Gy (12.3-21.6). Although the radiobiological risk study by Brodin et al of 10 pediatric patients with medulloblastoma did not report parotid doses, the predicted risk of xerostomia was reduced from approximately 29% for 36 Gy 3D CSI and 25% for 36 Gy VMAT CSI to 14% for both 23.4 Gy 3D CSI and 23.4 Gy VMAT CSI. 10 Corresponding xerostomia estimates in our study were 34.1% (24.3-43.9), 19.2%, 18.9% (14.8-23.1), and 5.5% (3.8-7.2), respectively (see Table 4).
In this study, VMAT CSI did not significantly affect doses for 2 structures close to the parotid gland, the submandibular gland, and the cochlea. However, recent radiobiological risk studies estimated an increased risk of secondary cancer for VMAT CSI, due to greater exposure of organs including the breast, lungs, and stomach to low-dose radiation. 10,16 Yet, these studies also estimated decreased risks of myocardial infarction and heart failure due to more optimal cardiac sparing with VMAT CSI. Given that IMRT-based CSI may improve dosimetry for some organs but not others, determining whether a patient receives 3D CSI or IMRT-based CSI should be a clinical decision that takes into account a patient’s age, clinical status, and other comorbidities.
Proton therapy can achieve more optimal sparing of normal structures including the parotid gland than photon therapy. 6 This dosimetric advantage is largely due to minimal exit dose from proton beams. According to radiobiological risk estimates, intensity-modulated proton therapy CSI may minimize the risk of secondary cancers compared to both IMRT-based and 3D CSI techniques. 10,16 However, proton therapy remains limited in terms of availability compared to that of IMRT-based techniques.
This retrospective study has limitations. First, we did not prospectively capture the incidence of xerostomia after CSI. Instead, we estimated xerostomia risks after CSI using a well-validated NTCP model 14 derived from adult head and neck cancer data. Although the incidence of xerostomia after CSI has not yet been reported in the literature, xerostomia risk after CSI has been estimated in a radiobiological risk study, as discussed earlier. 10 Second, both the NTCP model and the dosimetric constraints used in this study were adapted from the adult patients with head and neck cancers. These adaptations may not be completely valid, given that (1) patients with head and neck cancer received submandibular gland doses (~60 Gy) 17 that were much greater than those for CSI patients (9.8 Gy in this study) and (2) recent studies have suggested that younger patients are less susceptible to xerostomia. 18,19 However, xerostomia remains a common side effect for pediatric patients after total body irradiation 20 and chemoradiation for nasopharyngeal carcinoma, 21 with 1 prospective protocol reporting a 50% rate. 22 Furthermore, the parotid dose constraints incorporated in the Children’s Oncology Group study ARAR0331 for nasopharyngeal cancer were adapted from an adult head and neck cancer protocol. 12
Given the relatively good prognosis for the large subset of CSI patients with standard risk medulloblastoma (5-year overall survival above 80% 23 ), minimizing treatment toxicity and late effects for long-term cancer survivors has become a priority. Numerous clinical studies have characterized how treatment adaptations including radiation dose de-escalation and advanced radiation therapy techniques have improved cognitive dysfunction 1 and ototoxicity. 2 However, no study has documented the late effects of parotid gland irradiation in CSI patients. Given the potential risk of xerostomia and secondary parotid malignancies in long-term survivors after CSI therapy, we recommend including the parotid glands as a critical OAR in future protocols, with subjective and objective measures of xerostomia prospectively collected and secondary cancers accurately documented. The clinical impacts of parotid irradiation in the context of dose de-escalation and modern radiation therapy techniques can then be fully realized.
Footnotes
Acknowledgments
The authors would like to acknowledge the Stanford Department of Radiation Oncology for supporting for this work.
Authors’ Note
Presented, in part, at the 54th Annual Meeting of the American Society for Radiation Oncology (ASTRO) in Boston, MA, October 28-31, 2012.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Clara Y. Choi previously consulted for Varian Medical Systems.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
