Abstract
Our study aimed to investigate the effect of ultrasonic thermotherapy on the targeted delivery of liposomal doxorubicin to superficial tumors, local drug concentrations in tumor tissue, and the curative effect of chemotherapy. Twenty rabbits with VX2 tumors transplanted into the superficial muscle of the hind limb were randomly assigned to the following 4 treatment groups: (1) free doxorubicin, (2) liposomal doxorubicin hydrochloride, (3) liposomal doxorubicin hydrochloride plus 41°C thermotherapy, and (4) liposomal doxorubicin hydrochloride plus 43°C thermotherapy. Ultrasonic thermotherapy was delivered at 41°C to 43°C. Plasma, tumor, and organ/tissue homogenates were analyzed by high-pressure liquid chromatography to determine doxorubicin concentrations. The drug concentration in plasma and tumor tissue was significantly higher in the liposomal doxorubicin hydrochloride plus thermotherapy group than in the liposomal doxorubicin hydrochloride and free doxorubicin groups, but there were no significant differences among the 4 groups in the concentration in heart or kidney tissue. Combining thermotherapy with liposomal doxorubicin hydrochloride chemotherapy significantly increased the concentration of the drug in tumor tissue. The doxorubicin concentration was significantly higher in the liposomal doxorubicin hydrochloride plus 41°C thermotherapy group.
Introduction
Chemotherapy plays an important role in management of cancer. At present, chemotherapeutic drugs are commonly administered systemically. As a consequence, tissue selectivity is low, leading to significant side effects and low concentrations of drug in tumor tissue.
The use of drug delivery systems can help to solve these problems. Such systems have 2 stages: First, a drug carrier passes through permeable tumor vessels, and then the embedded drug is released into the intercellular space within the tumor. 1 In theory, a carrier can accumulate selectively in the region of a tumor (due to the greater permeability of new vessels and a retention effect). 2 If the embedded drug is then released within the tumor tissue, the efficacy of the treatment may be increased and the side effects reduced, thereby achieving the aims of targeted drug delivery.
Doxorubicin, an anthracycline antibiotic, is administered for the routine treatment of, for example, breast cancer, lung cancer, stomach cancer, thyroid cancer, and Hodgkin lymphoma, but due to its severe side effects its use is limited. 3 Embedding doxorubicin within liposomes can reduce the drug’s toxicity and increase the cycle time. However, studies have found that use of liposomes does not significantly improve the efficacy of chemotherapy due to the slow, passive release of the embedded drug. 4 Some studies have concluded that carrier size, limited flow of tissue fluid, and the compact nature of the tumor interstitial matrix limit the penetration of carriers into the extravascular extracellular space. 5 How to promote extravasation of drug carriers and the release of embedded drugs is the focus of our study.
Research shows a significant difference in sensitivity to raised temperatures between cancer cells and normal cells.
6
When a tumor is locally heated to 39°C to 40°C, temperature-triggered drug delivery, enhanced extravasation, and vasodilation will affect the cancer cells. Thermotherapy (application of mild hyperthermia) can be used to treat cancer. Thermotherapy significantly improves the efficacy of chemoradiotherapy and reduces its side effects so is often used in clinical practice.
7
Common methods of heating include hot water,
8,9
ultrasound,
10
and microwaves.
11
Kong
The VX2 tumor cell line, which is established after 70 passages of squamous cell carcinoma derived from Shope virus-induced rabbit papilloma, 14 has strong invasive and metastatic capacities and can be inoculated into rabbit liver, lung, kidney, bone, uterus, or other organs to produce in situ animal tumor models. The VX2 tumor is an invasive solid tumor with an abundant blood supply that is dominated by arteries; it is often used for interventional radiology, tumor thermal ablation, tumor chemotherapy, imaging, embolotherapy, and other experimental research.
Liposomal doxorubicin hydrochloride injection is the first nanodrug developed for clinical applications in China. In the present study, we used this drug combined with thermotherapy (41°C and 43°C) to treat rabbit VX2 tumor in the hind leg muscle layer. We aimed to determine the ability of thermotherapy to promote the release of liposomal doxorubicin into superficial tumor tissue and to increase the local concentration of the drug and to explore the feasibility of the use of thermotherapy to improve the curative effect of liposomal doxorubicin on superficial tumors.
Materials and Methods
Reagents
Reagents include liposomal doxorubicin hydrochloride injection (20 mg/10 mL bottle; Shanghai Fudan Zhangjiang Pharmaceutical Co, Ltd, Shanghai, China), doxorubicin hydrochloride injection (25 mg bottle; Sangon Bioteth Co, Ltd, Shanghai, China), ketamine, Sumianxin (2 mL/40 mg; Jilin HuaMu Animal Health Products Co, Ltd, Changchun, China), phosphate-buffered saline (PBS) solution, acetonitrile protein precipitation reagent, saline, and benzalkonium bromide.
Animals
All animal-related procedures were approved by and conducted under the guidelines of the Shanghai Jiaotong University Animal Care and Use Committee. New Zealand White rabbits aged 2 to 3 months and weighing 2.0 to 2.3 kg were obtained from the Experimental Animal Department of Shanghai Ninth People’s Hospital School of Medicine, Shanghai Jiaotong University. They were kept in the general environment at 50% to 60% humidity and 20°C to 22°C with a 12/12-hour light/dark cycle.
Instruments
A MyLab Twice ultrasound system (Esaote, Genova, Italy) was used for ultrasonography. The ultrasonic hyperthermia system was developed by biomedical instrument institute, school of Biomedical Instruments of Shanghai Jiao Tong University. This system has the following components: a controllable high-frequency power supply, an ultrasound applicator, a water cooling device, a multichannel temperature measurement circuit, a multichannel pulse width modulation generator circuit, an analog to digital converter card, a computer, and an electricity source.
The high-frequency power supply is developed and purpose built by biomedical instrument institute, School of Biomedical Engineering of Shanghai Jiao Tong University. The instrument has 2 driving frequencies, they are 1.1 and 3.4 MHz. The dimensions of the circular plane transducer are 40 mm and the thickness is 2 mm. The instrument has only 1 transducer. The structure of transducer is a plane, and the material of the transducer is piezoelectric ceramics. The cooling device makes use of Peltier effect, and making the semiconductor refrigeration on one face, and the other face product heat. The model number of the ADC card is PCI-1710 produced by Advantech (Taiwan). The card has 5 measure and control functions, including autoscan/gain scan, single and different analog input, PCI (Peripheral Component Interconnect) bus plug and play, first in first out (FIFO) buffer, and programmable counter.
The theory of a superficial tumor ultrasound hyperthermia therapeutic system is shown in Figure 1. Ultrasound is amplified by a power amplifier and sends energy to the target tumor by the ultrasound hyperthermia therapeutic head. There are 4 pinhead sensors in the system. In all, 1 pinhead sensor is to measure the cooling water temperature, and the other 3 pinhead sensors should be inserted into the target tumor, monitor point, and superficial cuticle during the therapy process. To control the temperature of target tumor, industry computer should measure the temperature and control the power output of ultrasound by Proportion Integration Differentiation (PID) feedback control algorithms. Figure 2 is the prototype of the ultrasound hyperthermia therapeutic system. This equipment has been tested by national medical device testing institutions designated by the Chinese State Food and Drug Administration (SFDA). The parameters and indicators comply with national standards.

Theory of superficial tumor ultrasound hyperthermia therapeutic system.

The prototype of ultrasound hyperthermia therapeutic system.
Preparation of Rabbit VX2 Tumor Model
VX2 frozen tumor cell suspension was provided by the Institute of Oral Medicine of Shanghai Ninth People’s Hospital School of Medicine. It was centrifuged at 800 rpm for 5 minutes following anabiosis according to general cell culture procedures. The PBS was added and the cells were centrifuged again for 5 minutes after removing the supernatant. Again PBS was added after removal of the supernatant, followed by stirring with a glass rod. Samples of the suspension were taken for Trypan Blue staining to count dead and live cells. The suspension was then adjusted to a concentration 106 living cells/mL and placed in several 1.5-mL bottles that were immediately put into an ice box and transported to the animal operating room for inoculation.
New Zealand White rabbits were anesthetized by intramuscular injection of ketamine (0.2 mL/kg) and Sumianxin (0.2 mL/kg), both lower extremities were shaved and depilated, and about 1.5 mL of the prepared tumor cell suspension was injected into the superficial muscle of the right hind leg under ultrasound guidance. Ultrasonography was used to monitor the growth of the tumors for 2 to 3 weeks after inoculation. Experiments were undertaken when the tumor size was greater than 1 cm in any dimension (Figure 3). For the donor rabbit, VX2 tumor cells were propagated and maintained by bilateral hind limb inoculation.

Ultrasound image from a rabbit VX2 transplanted tumor, where the tumor diameter was greater than 1 cm.
Treatment
Twenty VX2 tumor-bearing rabbits were randomly divided into the following 4 groups: (1) free doxorubicin hydrochloride (Free Dox), (2) liposomal doxorubicin hydrochloride (L-Dox), (3) liposomal doxorubicin hydrochloride plus 41°C thermotherapy (L-Dox + 41°C TT), and (4) liposomal doxorubicin hydrochloride plus 43°C thermotherapy (L-Dox + 43°C TT). The rabbits were anesthetized by intramuscular injection of ketamine (0.2 mL/kg) and Sumianxin (0.2 mL/kg) before treatment, and an electric blanket was placed on the leg without tumor to maintain a core body temperature of 34°C to 37°C after the anesthesia. The rabbits in 1 group received free doxorubicin hydrochloride and the rabbits in the other 3 groups received liposomal doxorubicin hydrochloride 5 mg/kg delivered by slow intravenous bolus injection into the ear vein; the duration of the injection was longer than 5 minutes, after which 1 mL saline was injected to wash out the ear vein. Body temperature, breathing, and heart rate were monitored.
In the L-Dox + 41°C TT and L-Dox + 43°C TT groups, thermotherapy was conducted immediately after the administration of liposomal doxorubicin hydrochloride. The thermotherapy instrument settings were as follows: treatment temperature 41°C and 43°C, respectively, water temperature 35°C, treatment frequency 1.1 MHz, and voltage duty ratio 100%. The output power was continuous. Treatment time was set to 40 minutes (allowing 5-10 minutes to reach the treatment temperature). The skin of the hind leg was depilated and disinfected with alcohol wipes. The thermal needle was then inserted into the central part of the tumor in the subcutaneous muscle layer guided by ultrasound, and the heating probe was placed on the skin over the tumor after the application of sound conducting gel, followed by continuous heating for 30 minutes.
Four hours after the end of treatment, the rabbits in all 4 groups were moved to the animal operating room, where 5 mL of blood was extracted before the animals were killed humanely. Liver, kidney, heart, and transplanted tumor tissue were dissected out, weighed, and stored at −80°C for high-pressure liquid chromatography (HPLC) and fluorescence analysis.
Quantification of Doxorubicin by HPLC
High-pressure liquid chromatography was used to quantify the drug content of rabbit tumor tissue and vital organs.
Preparation of reference solution
The reference solution was successively diluted with water to obtain standard solutions with concentrations of 1000, 500, 200, 1000, and 50 ng/mL.
Preparation of serum samples
Acetonitrile protein precipitation reagent of 100 μL was added to 100-μL samples of human serum, with vortex mixing and centrifugation at 12,000
Preparation of tissue samples
Tissue samples were weighed and added with twice the amount of saline (ie, 0.5 g/mL) and a stainless steel ball of 6 mm in diameter, then they were placed on a high-throughput tissue homogenate instrument (TL-48P/R; Wanbo Biological Technology Co, Shanghai, China) for 1 minute with 65 HZ and centrifuged at 12,000
HPLC-mass spectrometry analysis
An Acquity UPLC ZQ-2000 mass spectrometry (MS) system (Waters Corp, Milford, Massachusetts) was used with an Acquity UPLC BEH C18 column (1.7 μm, 2.1 × 50 mm2), a mobile phase of acetonitrile and 0.1% formic acid for linear gradient elution, a column temperature of 45°C, and an injection volume of 5 μL. For MS, ionization mode was used, with electrospray ionization (ESI+), capillary voltage 3.0 kV, ion source temperature 120°C, desolvent temperature 400°C, desolvent gas (nitrogen) flow rate 600 L/h, cone gas (nitrogen) flow rate 50 L/h, cone voltage 20 V, and retention time 2.52 minutes.
Statistical Analysis
SPSS 19.0 statistical software was used for analysis. Measurement data were expressed as the mean ± standard error of the mean. The Kruskal-Wallis test was used for overall comparison of the means of multiple groups and the Mann-Whitney test for pairwise comparisons.
Results
Doxorubicin Content of Rabbit Plasma Measured by HPLC
The concentration of doxorubicin in plasma was significantly higher in the L-Dox group than in the Free Dox group. Doxorubicin concentrations were 0.157 ± 0.017, 31.618 ± 7.722, 72.641 ± 20.899, and 64.312 ± 5.242 μg/mL in the Free Dox, L-Dox, L-Dox + 41°C TT, and L-Dox + 43°C TT groups, respectively (Figure 4). The concentration of doxorubicin in plasma was significantly higher in the L-Dox + 41°C TT and L-Dox + 43°C TT groups than in the L-Dox group (

Doxorubicin in rabbit plasma following treatment with free doxorubicin hydrochloride (Free Dox), liposomal doxorubicin hydrochloride (L-Dox), liposomal doxorubicin hydrochloride plus 41°C thermotherapy (L-Dox + 41°C TT), liposomal doxorubicin hydrochloride plus 43°C thermotherapy (L-Dox + 43°CTT), at a doxorubicin dose of 5 mg/kg. Data are shown as the mean doxorubicin concentration in plasma with the standard error of the mean (n = 5).
Doxorubicin Content of Rabbit Tumor Tissue and Vital Organs Measured by HPLC
There were no significant differences among the 4 groups in the concentrations of doxorubicin in heart and kidney tissues. The drug content of the liver was increased in the L-Dox, L-Dox + 41°C TT, and L-Dox + 43°C TT groups, but there were no significant differences among the 3 groups.
The concentration of doxorubicin in tumor tissue was significantly higher in the L-Dox + 41°C TT group than in the L-Dox group and the L-Dox + 43°C TT group (Table 1). Tumor doxorubicin concentrations were 0.187 ± 0.037, 0.700 ± 0.268, 3.210 ± 0.509, and 1.355 ± 0.492 μg/g in the Free Dox, L-Dox, L-Dox + 41°C TT groups, and L-Dox+43°C TT groups, respectively. The L-Dox + 41°C TT group exhibited 17-fold greater tumor drug delivery compared to the Free Dox group (
Drug Content (μg/g) of Different Tissues in Each Treatment Group.a
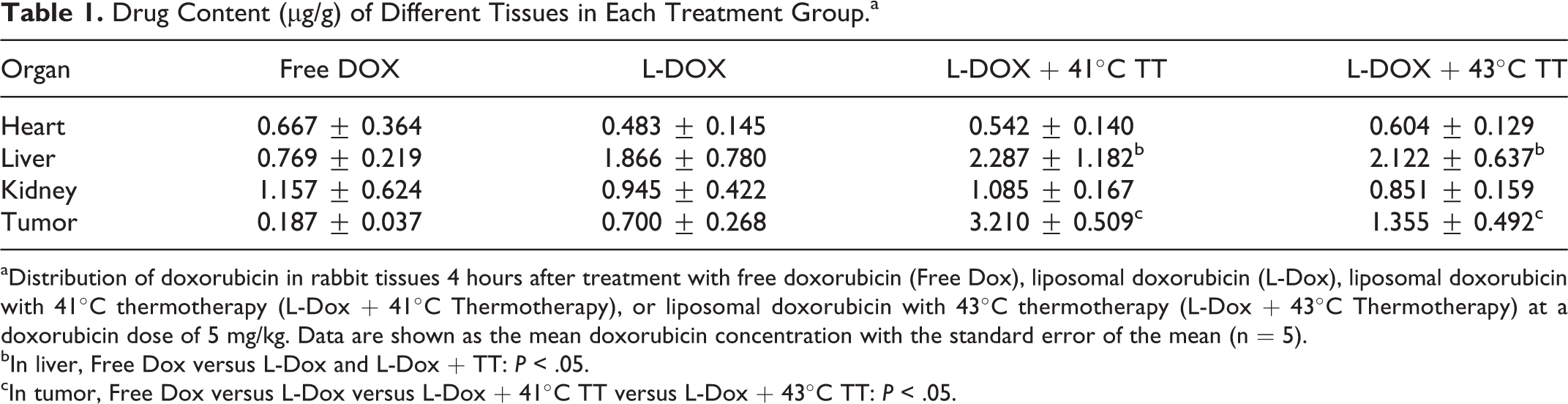
aDistribution of doxorubicin in rabbit tissues 4 hours after treatment with free doxorubicin (Free Dox), liposomal doxorubicin (L-Dox), liposomal doxorubicin with 41°C thermotherapy (L-Dox + 41°C Thermotherapy), or liposomal doxorubicin with 43°C thermotherapy (L-Dox + 43°C Thermotherapy) at a doxorubicin dose of 5 mg/kg. Data are shown as the mean doxorubicin concentration with the standard error of the mean (n = 5).
bIn liver, Free Dox versus L-Dox and L-Dox + TT:
cIn tumor, Free Dox versus L-Dox versus L-Dox + 41°C TT versus L-Dox + 43°C TT:
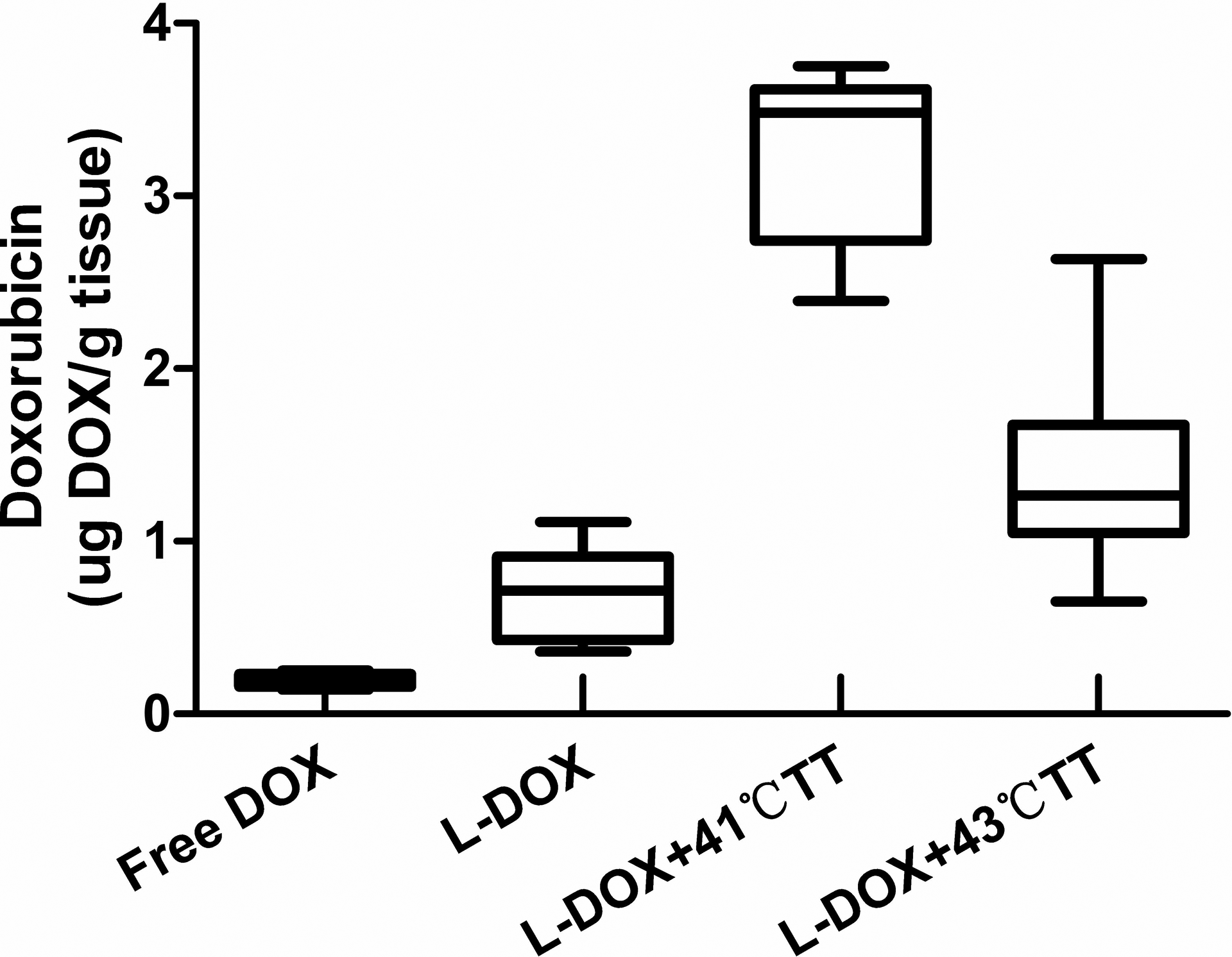
Doxorubicin in rabbit tumor tissue following treatment with free doxorubicin hydrochloride (Free Dox), liposomal doxorubicin hydrochloride (L-Dox), liposomal doxorubicin hydrochloride plus 41°C thermotherapy (L-Dox + 41°CTT), liposomal doxorubicin hydrochloride plus 43°C thermotherapy (L-Dox + 43°C TT), at a doxorubicin dose of 5 mg/kg. Data are shown as the mean doxorubicin concentration in tumor tissue with the standard error of the mean (n = 5).
Discussion
This study used the anticancer drug doxorubicin and a rabbit VX2 transplanted tumor model to explore the clinical feasibility of the use of thermotherapy to increase the efficacy of liposomal drug delivery in the treatment of superficial solid tumors.
Liposomal Drug Delivery
As a method of targeting anticancer drugs, liposomes have become a focus of research. Yatvin
Thermotherapy Settings
It is important to determine the optimal thermotherapy temperature and settings for the release of liposomal drugs. Kong
In the present study, thermotherapy was applied at 41°C or 43°C (Figure 6). For 30 minutes, 2 different thermal dose's effect on the extravasation of liposomes were studied. During treatment, the tumor was maintained at the required temperature by automatic adjustment of the high-frequency electrical power supply to the ultrasound transducer.

Temperature profile. The temperature fluctuated between 42°C and 44°C, and the mean temperature was 43°C.
The key issue in thermotherapy is how to achieve uniform heating of tumor tissue. The temperature should be maintained at 41°C to 43°C for about 30 to 60 minutes. With early high-intensity-focused ultrasound devices in which the temperature was measured by a thermocouple, the temperature within the target region was inferred according to the operator’s experience. Staruch
Drug Concentration in Tumor Tissue and Vital Organs Measured by HPLC
We determined the distribution of doxorubicin to evaluate the efficacy of treatment and the side effects on normal tissue. The plasma drug concentration was significantly higher in rabbits treated with liposomal doxorubicin hydrochloride than in those treated with free doxorubicin. According to our HPLC findings, the doxorubicin content of tumor tissue in the 4 treatment groups decreased in the following order: liposomal doxorubicin hydrochloride plus 41°C thermotherapy > liposomal doxorubicin hydrochloride plus 43°C thermotherapy > liposomal doxorubicin hydrochloride > free doxorubicin.
Chauhan
In addition, we found that the liposomes with hyperthermia group compared to the nonliposomal hyperthermia group, although drug concentrations in plasma increased significantly, showed no significant differences in the concentrations of doxorubicin in heart and kidney tissues among the 4 treatments. Use of liposomal doxorubicin hydrochloride increased the doxorubicin content of the liver, which may be related to its longer circulation time in the body.
It is worth noting that L-DOX + 41°C TT versus L-DOX + 43°C TT showed no difference in drug concentrations in plasma, heart, and liver and also no apparent damage to the surrounding tissue. However, drug concentration in the tumor for 41°C hyperthermia was significantly higher than that in the 43°C group, which may be because of bleeding stasis in the tumor blood vessels when the temperature is above 42°C 12 ; therefore, we believe that the best hyperthermia temperature should be 41°C.
Because of its serious side effects, the use of doxorubicin has been limited, and it has been suggested that liposomes with a long circulation time are safer. 22 Another study demonstrated that these liposomes reduce the extent of contact of the drug with normal tissue throughout the body, ensuring a high level of drug delivery. 23 Liposomal doxorubicin combined with thermotherapy achieves targeted drug delivery, further demonstrating that thermotherapy can play an important role in reducing drug side effects and improving the therapeutic window.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported from the Foundation of National Natural Science Foundation of China (No. 81272567).
