Abstract
Background
Doxorubicin (DOX) is a potent chemotherapeutic agent for breast cancer, but its effectiveness is often diminished by resistance mechanisms, particularly through p-glycoprotein (P-gp) mediated drug efflux. Clarithromycin (CAM), a macrolide antibiotic, inhibits multiple metabolic pathways including CYP3A and P-gp, potentially countering DOX resistance.
Objective
This study aimed to evaluate the potentiation of DOX and its effectiveness against the MCF-7 breast cancer cell line by encapsulating both DOX and CAM in PEGylated liposomes.
Methods
PEGylated liposomes containing DOX and CAM were prepared using the thin film hydration method. The physicochemical properties of the liposomes, including average particle size, polydispersity index (PDI), and zeta potential, were characterized. Encapsulation efficiencies for CAM and DOX were assessed, and stability of the liposomes was evaluated over 9 days at room temperature. Cell viability was measured using an IC50 assay, and P-gp expression levels were determined by ELISA.
Results
The CAM/DOX-PEGylated liposomes exhibited optimal average particle size (238 ± 26.7 nm), PDI (0.29 ± 0.107), and zeta potential (−20.9 ± 2.17 mV). These liposomes maintained good stability regarding size and charge over 9 days. Encapsulation efficiencies were 81.05% for CAM and 78.13% for DOX. The IC50 value for CAM/DOX-PEGylated liposomes was 0.13 µM, representing a significant reduction compared to the physical mixture of CAM and DOX (0.25 µM) and free DOX (0.21 µM) against MCF-7 cells. ELISA analysis showed a reduction in P-gp expression of approximately 5% with CAM/DOX-PEGylated liposomes compared to 1.61% with free DOX.
Conclusion
The results indicate that CAM encapsulated in PEGylated liposomes enhances the effectiveness of DOX against breast cancer cells, likely through the inhibition of p-glycoprotein. This approach may offer a promising strategy to overcome DOX resistance and improve chemotherapy outcomes.
Introduction
In 2022, nearly 20 million new cases of cancer and approximately 9.7 million deaths related to cancer were reported worldwide. By 2040, it is anticipated that the annual number of new cancer cases will increase to 29.9 million, with cancer-related deaths rising to 15.3 million. 1 In 2022, 2.3 million women worldwide were diagnosed with breast cancer, resulting in 670 000 deaths. Breast cancer affects women of all ages in every country around the globe. 2 Doxorubicin (DOX) (Figure 1A) is a member of the anthracycline class of chemotherapy medications. It is now used as a therapeutic intervention for many kinds of cancer. It is considered among the most efficient drugs against breast cancer, either on its own or in conjunction with other therapeutic agents.3–6

Chemical structure of (A) Doxorubicin, and (B) Clarithromycin.
The anticancer properties of DOX have been previously established and can be attributed to its capacity to impede topoisomerase II and intercalate into DNA, interfere mitochondrial function, and enhance the production of free radicals and oxidative harm leading to cell death.7–9 Resistance to DOX is a significant limitation, often resulting from its active extrusion by cells that overexpress P-glycoprotein (P-gp). P-gp acts as a transmembrane efflux pump for various cytotoxic substances, playing a crucial role in the ineffectiveness of cancer treatment strategies.10,11 P-gp is a member of the ATP binding cassette (ABC) superfamily, more precisely categorized under ABCB1. It plays a crucial role in efflux, contributing to multidrug resistance (MDR) in various cancers such as breast, ovarian, multiple myeloma, osteosarcoma, and lung cancer. 12 Studies consistently show that higher expression of P-gp correlates with increased resistance to chemotherapy across different cancer types. 13
Clarithromycin (CAM) (Figure 1B) is a widely used and effective semi-synthetic macrolide antibiotic known for its ability to inhibit bacterial protein synthesis. 14 There is substantial preclinical and clinical evidence supporting the potential use of CAM in combination with conventional treatments for managing various types of cancers.15,16 The anti-cancer effects of macrolides operate through multiple mechanisms that do not directly attack cancer cells. 17 These mechanisms encompass prolonged inhibition of inflammatory cytokines, suppression of autophagy, and inhibition of angiogenesis. 18 CAM has been identified as a P-gp inhibitor and has been extensively studied in clinical trials involving cancer patients. 19
A recent study by Amani et al verified that physical combination of CAM with DOX synergistically enhanced the anti-proliferative effect against MCF7 cells, leading to increased apoptotic cell death. Despite this synergy, CAM did not inhibit P-glycoprotein activity at a concentration of 100 μM. 20 Therefore, our objective in this study was to assess the potential effect of this combination using liposomes against the same cell line.
Liposomes improve the physicochemical properties of drugs and enhance their bioavailability. PEGylated liposomes, referred to as stealth liposomes, have shown increased accumulation at target sites and improved stability of anticancer drugs.4,21–27 The purpose of this study was to examine the synergistic effects of encapsulating CAM and DOX in PEGylated liposomes. Our objective was to increase cytotoxicity against MCF-7 breast cancer cells, potentially increasing therapy results. Furthermore, we wanted to see if the liposomal formulation might suppress P-glycoprotein function at lower drug doses, which could assist overcome multidrug resistance. This dual strategy provides the potential for more successful cancer treatment options.
Materials and Methods
Chemicals
Doxorubicin (>99%) was purchased from BIOSYNTH, UK. Clarithromycin (>99%) obtained from Dar AlDawa, Jordan, USA, Dipalmitoyl L-3- phosphatidylcholine (DPPC) (>99%), Polyethylene glycol DSPE-PEG −2000 (>99%) were obtained from Avanti Lipids, USA, Inc. Phosphate buffer saline (PBS) (99.9%), Cholesterol (CHOL) (>99%), and HPLC ethanol were purchased from Sigma Aldrich – USA. Promega – USA for MTT reagent, Dimethyl sulfoxide (DMSO) and Trypan Blue Dye from GCC-UK. P-gp kitt from (SUNLONG). Tedia-America (USA) provided methanol, and Acetonitrile. All other chemicals and solvents were of analytical grade. All reagents and chemicals were used without further treatment.
Cells
MCF-7 (ATCC number: HTB-22) were cultured in RPMI-1640 growth medium (Capricorn Scientific GmbH, Germany) supplemented with 10% (v/v) fetal bovine serum (FBS), 1% (v/v) 200 mM L-glutamine, and antibiotics; Penicillin–Streptomycin (100 IU mL−1-100 μg mL−1). Cells were passed once every three days. Cell counter (QuadCount™) with trypan blue staining was used to count viable cells. All experiments were performed for passages ranging from 3 to 10. Cells were maintained in humidified 5% CO2 incubator at 37 °C.
PEGylated Liposomes Formulation
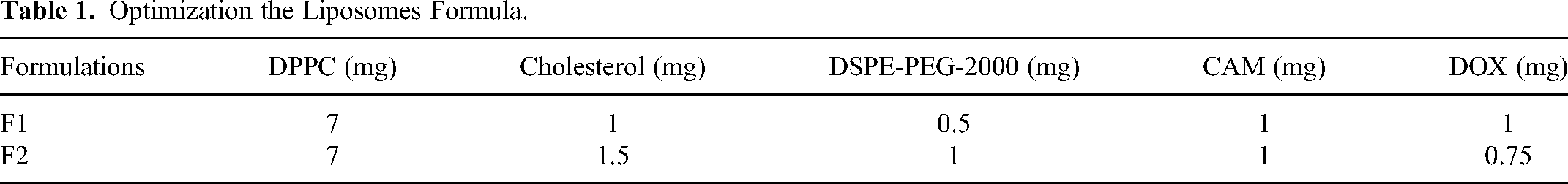
CAM/DOX PEGylated liposomes were prepared by the thin film hydration technique with some modifications.28,29 Briefly, DPPC, cholesterol, DSPE-PEG −2000, and CAM (Table 1) were dissolved in 3.0 mL of chloroform (HPLC grade) using a 50 mL round bottom flask (BUCHI, Germany). A thin film was formed by gentle evaporation of the organic solvent using a rotary evaporator (BUCHI, R-300) at 45 °C and 45 rpm, with an initial pressure of 450 mbar that gradually decreased with time (each 15 min) to 400, 300, 200, and 100 mbar. The evaporation was kept for another 30 min at 100 mbar. Two different formulas (F1 and F2) were examined utilizing different lipid ratios, bath and probe sonication time, and degree of filtration to optimize the best suitable liposomes in terms of appropriate average size, zeta potential charge, and poly dispersity index (PDI). DPPC, cholesterol, and DSPE-PEG −2000 ratios of (70:25:5 ± 5) are general guidelines and may need to be adjusted based on specific experimental conditions or desired membrane characteristics (eg, stability, fluidity, permeability). These ratios provides greater membrane rigidity, while cholesterol and DSPE-PEG-2000 maintain stability and functionality.30,31 It was observed that using probe sonication for (30, 10, 5, 5) seconds with a half-hour interval between each setting at 25% of 100 watts resulted in a reduction in particle size and achieved a low PDI value.5,32 All measurements were conducted in triplicate for each experiment. Formula F2 was determined to be the most optimized formulation based on these findings.
Optimization the Liposomes Formula.
Characterizations and Stability Evaluation
Dynamic Light Scattering (DLS) Measurements
The average particle size, zeta potential, and PDI were evaluated using Malvern Zetasizer® Instrument (Malvern, Model ZEN3600) for the prepared liposomes. Samples were diluted in the ratio of (10:990) in de-ionized water to obtain the appropriate counting number. All measurements were performed in triplicate.
In Vitro Stability of CAM/DOX PEGylated Liposomes
The stability of PEGylated liposomes loaded with drugs was assessed by monitoring changes in their particle size, polydispersity index (PDI), and zeta potential from the time of preparation over a period of 9 days.
Transmission Electron Microscopy
Transmission electron microscopy (TEM) analysis of various PEGylated liposomes utilized the Negative Staining Method.33,34 Initially, 200 mesh formvar copper grids were coated with carbon using low vacuum Leica EM ACE200 glow discharge coating equipment. Subsequently, the grids were treated with a 1.5% vinyl K Solution in chloroform and then a diluted suspension of liposomes in deionized water was applied and allowed to air-dry. Staining was achieved by incubating the grids with a 3% aqueous solution of uranyl acetate for 20 min at room temperature, followed by rinsing with distilled water and air-drying. The TEM images were obtained using a Versa 3D TEM system with a 30-kV acceleration voltage.
Determination of Encapsulation Efficiency and Drug Loading
Encapsulation efficiency (EE%) and drug loading (DL%) were calculated as described in the following equations:
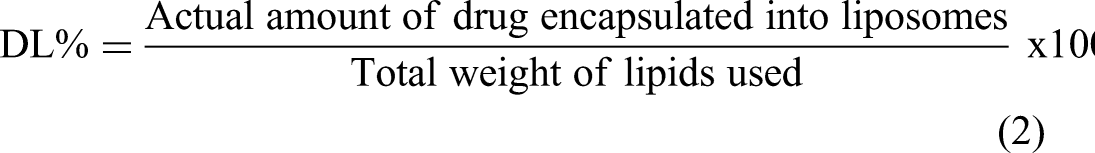
DOX in Vitro Release Assay
The in vitro release test involved placing 1 mL of either free DOX or CAM/DOX-PEGylated liposomes (333 μg/mL in PBS, pH 7.4) into a cellulose dialysis bag (50 KD cutoff), which was submerged in 30 mL of PBS. The release medium was shaken at 37 °C and 100 rpm. At specified time points (2, 4, 6, 24, 48, and 72 h), 1 mL of dialysate was sampled and replaced with fresh release medium. HPLC was used to quantify the amount of DOX in the dialysate samples.
Cell viability assay (MTT)
MCF-7 cells were seeded at a density of 5 × 103 cells per well in four 96-well plates (TPP, Switzerland). After 24 h, cells were treated with various concentrations of free DOX, CAM/DOX PEGylated liposomes, free CAM, and a physical mixture of DOX and CAM. An effective of DOX concentration range typically includes lower concentrations around 0.01–0.5 µM for minimal cytotoxicity and higher concentrations ranging from 5-10 µM where significant cell death occurs. A concentration starting from the highest concentration of 5 µM with a 1:1 serial dilution down to 0.0390 µM were used. Negative controls included untreated cells (media only) and PBs (5 μM). Following 24-h and 72-h incubation periods at 37 °C, treatments were removed, and 20 µL of 0.05 mg/mL MTT reagent (Promega, USA) was added per well. After 3.5 h of incubation with gentle vibration, the medium was aspirated, and formazan crystals were solubilized with 180 μL of DMSO per well. Absorbance of formazan was measured at 590 nm and 630 nm wavelengths using a Synergy HTX multi-mode microplate reader. Average absorbance from control wells was set as 100% survival, and IC50 values, representing concentrations inhibiting cell growth by 50%, were determined using equation (3).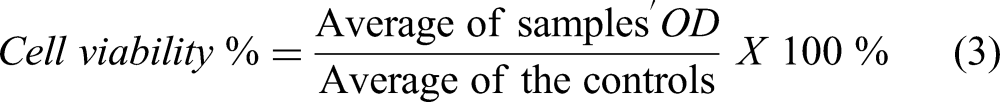
P-gp ELISA Assay
Samples Preparation
Six sterile T-25 flasks were seeded with 200 000 MCF-7 cells per flask and placed overnight in an incubator to promote cell adhesion. Different concentrations of free DOX and CAM/DOX PEGylated liposomes were applied to the cells as shown in Table 2. PBS treatment serves as negative controls. Treated cells and were incubated for 24 and 72 h. At the designated time point, the supernatant is carefully gathered into labelled tubes and stored at −20 °C.
Samples Concentrations of ELISA.

The Sanswich-P-gp kit (SUNLONG) was employed to quantify extracellular P-gp levels in culture media following treatment with free DOX and CAM/DOX PEGylated liposomes for 24 and 72 h. The microplate provided in the kit was pre-coated with a specific capture antibody targeting P-gp. Samples containing the antigen underwent a complex reaction within the wells. The kit was utilized in accordance with the manufacturer's instructions. The initial concentration of the standard (stock) provided was 5400 pg/ml. Five standards were prepared (3,600, 2,400, 1,200, 600, and 300 pg/ml) using the standard diluent supplied in the kit to establish a calibration curve. 50 μL of each standard, samples, and control are added to each well as shown in (Figure S1). Following sealing with a closure plate membrane, the incubator was shaken for 1 h at 37 °C as part of the procedure using the Sanswich-P-gp kit (SUNLONG). All wells were refilled with washing solution, which was then discarded after 5 wash cycles. Subsequently, 50 μl of Horseradish Peroxidase (HRP)-conjugated antibody specific to P-gp was added to each well (excluding the blank), followed by a 30-min incubation at 37 °C. Washing steps were repeated as previously described. To color the wells, 50 μl each of chromogen solution A and chromogen solution B were added, and the plate was incubated for 15 min while avoiding light. The reaction was stopped by adding 50 μl of stop solution to each well. Depending on the presence of P-gp, the color shifted from blue to varying shades of yellow. The optical density (OD) was measured at 450 nm using a microplate reader, with the procedure carried out in duplicate. The OD value was directly proportional to the concentration of P-gp in the samples.
Statistical Analysis
The results were presented as three independent experiments’ mean ± standard deviation. Statistical significance was determined using an unpaired two-tailed student's t-test with GraphPad Prism 8 (GraphPad Software Inc., USA). P < .05 was considered to assign a statistically significant difference.
Results and Discussion
Characterization and Stability of the CAM/DOX PEGylated Liposomes
Dynamic light scattering measurements using the Zetasizer® were employed to assess the characteristics of two different formulations of CAM/DOX PEGylated liposomes, including average particle size, Polydispersity Index (PDI), and Zeta Potential (charge) 36 (Table 3). It was observed that probe sonication for durations of (30, 10, 5, 5) seconds, with a half-hour interval between each cycle, at 25% MHz of 100 watts, resulted in a reduction in particle size and achieved a low PDI value of less than 0.3. Formula F2 was identified as the most optimized based on these findings.
Average Particle Size, Charge and PDI Results of CAM/DOX-PEGylated Liposomes.

The targeted average particle size of the prepared liposomes was aimed to facilitate effective accumulation in tumor tissue, leveraging the enhanced permeability and retention effect (EPR). Liposomes size up to 1000 nm have a heightened ability to interact with permeable blood vessels within tumors and penetrate tumor tissue. Our liposomes Both exhibited a favorable PDI less than 0.3, indicating well-dispersed liposomes. Additionally, PEGylated liposomes displayed a negative zeta potential averaging at −20.9 ± 2.17 mV, attributed to the presence of DSPE-PEG-2000. The optimized CAM/DOX-PEGylated liposomes demonstrated a particle size of 238 ± 26.7 nm with a PDI of 0.29 ± 0.11, alongside a zeta potential of −20.9 ± 2.17 mV (Figure 2A and B). PDI of 0.29 shows that the liposome population has a limited size distribution, implying that particle size is homogeneous. While a PDI less than 0.3 is typically deemed adequate for stability, numbers closer to 0.1 imply even more consistency. The present PDI implies that the liposomes are stable, however there may be some size fluctuation, which might affect the uniformity of drug release. 37
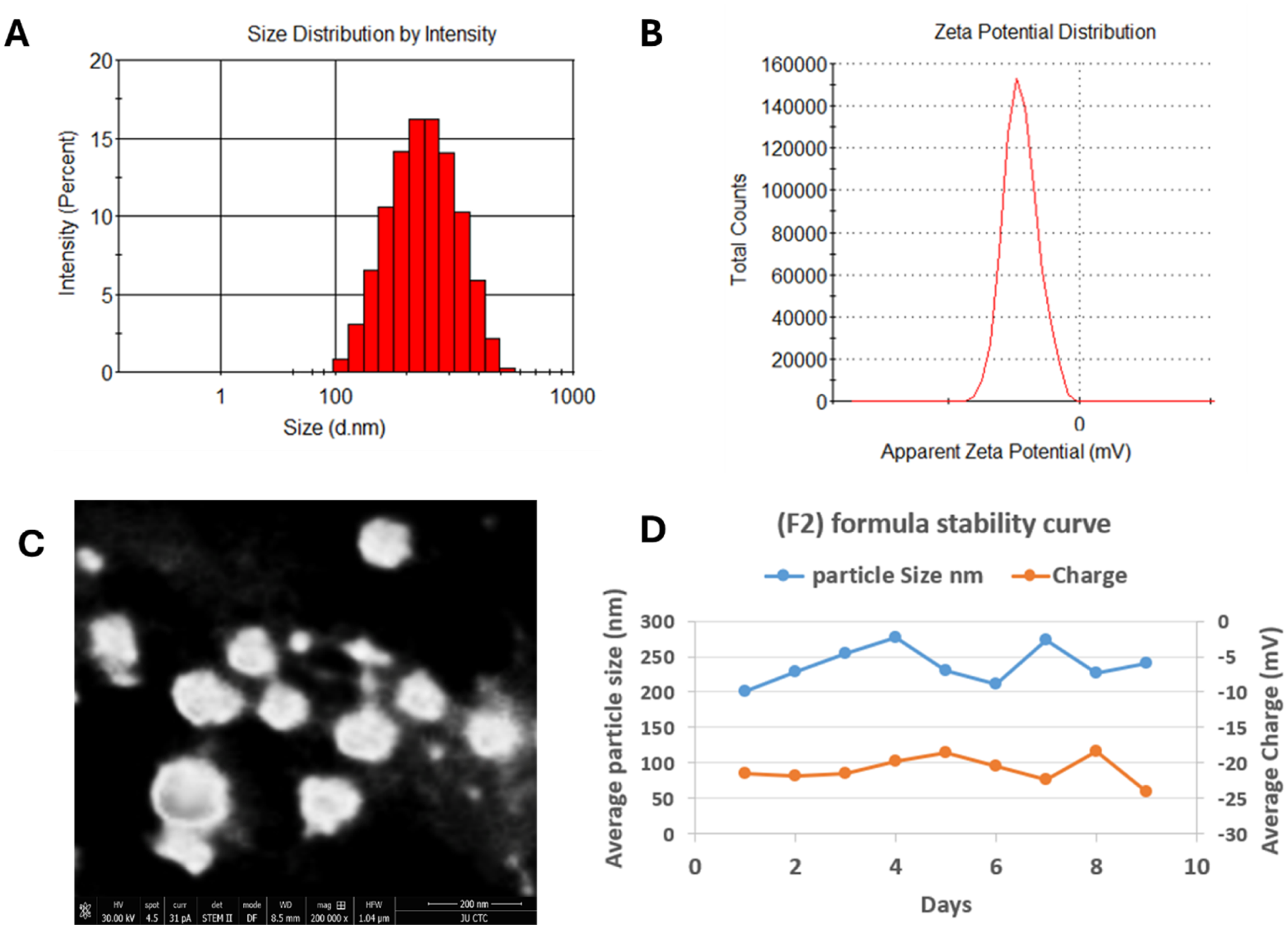
DLS measurements of F2 formula of CAM/DOX-PEGylated liposomes (A) Average particle size (B) Zeta potential, (C) TEM micrograph, and (D) CAM/DOX-PEGylated liposomes stability for 9 days.
While we aimed for a mean particle size of less than 200 nm for optimal passive targeting, we believe that even a size of 238 nm can still contribute to improved passive delivery. Larger liposomes may still benefit from EPR, particularly in the context of tumor microenvironments that exhibit leaky vasculature with fenestrations of 200 to 2000 nm.38,39 EPR-based drug delivery relies on a few characteristics, including circulation time, targeting, and the capacity to overcome barriers, all of which are determined by the size, shape, and surface features of drug particles. Subhan et al reported that nanoparticles ranging in size from 40 to 400 nm are ideal for ensuring lengthy circulation duration and increased accumulation in tumours with limited renal clearance. Shape and morphology are also key factors in passive targeting.40,41 Particles with a diameter less than 200 nm are more efficient but experiments with liposomes of different sizes suggest that the optimal size for escaping blood vessels and entering tumours is 400 nm.42,43 Kong et al found that heat around 42 °C able to increase the pore cutoff size in tumor vasculature to >400 nm, allowing all examined liposomes (varying from 100 nm to 400 nm) to extravasate into the tumor interstitium. 44 Li et al found that local hyperthermia increased tumor vasculature permeability and nanoparticle extravasation in all four tumor models tested. 45 PEGylated liposomes like Doxil, with a negative surface charge (∼-20 mV) and a size of 100-250 nm, enhance tumor accumulation by leveraging prolonged circulation and the leaky blood vessels of tumors. Our findings of a 238 nm particle size and negative zeta potential align with this, suggesting similar stability and effective tumor targeting. 46
TEM has been widely employed as the primary imaging technique for assessing the structural characteristics of nanosystems. In this study, TEM was utilized to confirm the size and structural properties of CAM/DOX-PEGylated liposomes (Figure 2C). The TEM images verified that the liposomes-maintained stability, exhibited a spherical shape, and were appropriately dispersed.
CAM/DOX-PEGylated liposomes demonstrated stable particle size and zeta potential over a 9-day period, as depicted in Figure 2D. At 4 °C, there were no significant changes observed in either the zeta potential or particle size, indicating excellent stability. Similar findings were reported by Nsairat et al, who highlighted that maintaining specific ratios of CHOL and DSPE-PEG-200 can effectively stabilize lipid liposomes for up to 7 days, aligning with the results observed here. 23
Encapsulation Efficiency and Drug Loading
The encapsulation efficiencies (EE%) and Drug loading (DL%) of DOX and CAM within CAM/DOX-PEGylated liposomes were calculated using equation (1) and equation (2), respectively, with calibration curves (R2 = 0.9958 and 0.9922, respectively). The EE% values were approximately 78.13 ± 4.5% for DOX and 82.05 ± 4.2% for CAM, indicating that both drugs were efficiently loaded into the CAM/DOX-PEGylated liposomes. This suggests a high level of drug encapsulation within the liposomal formulation. The DL% for DOX (6.16 ± 0.35%) and CAM (8.63 ± 0.72%) fall within the typical 5% to 20% range for liposomal formulations. While the liposomes successfully encapsulate both drugs, there is potential for improving drug loading efficiency through further optimization.
In Vitro Cumulative Release Assay
Figure 3 illustrates the release profiles of free DOX and DOX from CAM/DOX-PEGylated liposomes in Phosphate Buffered Saline (PBS, pH 7.4) at 37 °C over a 72-h period. Free DOX achieved nearly complete release after 72 h, while DOX release from CAM/DOX-PEGylated liposomes reached only 70% during the same period. This delayed and slower release of DOX from the CAM/DOX-PEGylated liposomes indicates the ability of the liposomal formulation to provide controlled drug delivery over a prolonged period and highlights the potential of the CAM/DOX-PEGylated liposome formulation as an effective carrier for chemotherapy drugs, offering a means of enhancing therapeutic effects while reducing the risk of toxicity associated with free drug administration. Additionally, the 70% release within 72 h may reflect a balance between encapsulation efficiency and the rate at which the drug is released from the liposomes.

Cumulative Release Profiles of free DOX, and DOX from CAM/DOX-PEGylated liposomes at pH 7.4 at 37 °C (Dialysis Method).
In Vitro Cytotoxicity Assay
MCF-7 cell lines were treated with CAM/DOX-PEGylated liposomes, free DOX, and a physical mix of CAM and DOX within 24 and 72 h. Figure 4 represents dose-response curves of these treatments at 72 h and the half inhibitory concentration (IC50) values during 24 and 72 h against MCF-7 cell line. CAM at concentrations ranging from 5 to 0.039 µM had no effect on the viability of MCF-7 cells.
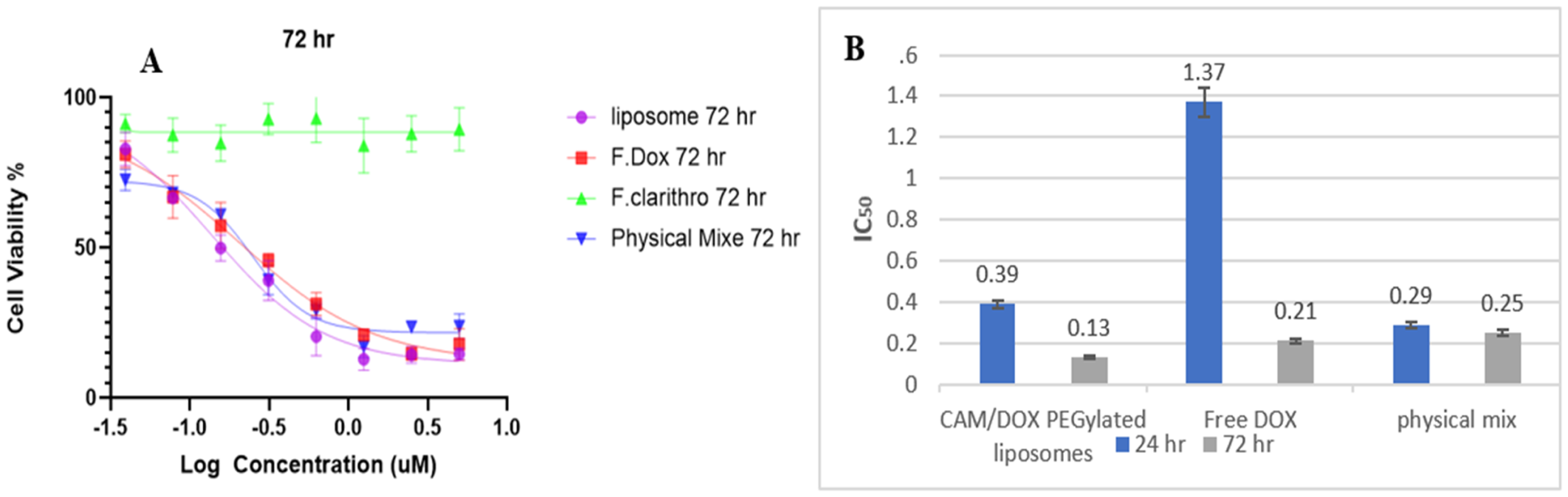
(A) represent the dose-response curves after 72 h and (B) The Half Inhibitory Concentration (IC50) values for CAM/DOX-PEGylated liposomes, free DOX, and a physical mix of CAM and DOX treatments against MCF-7 cell lines after 24 and 72 h.
Moreover, the physical mixture of DOX and CAM at equivalent concentrations to CAM alone did not significantly alter the cytotoxicity of DOX against MCF-7 cells after 72 h compared to free DOX alone. The cytotoxic activity of the physical mixture DOX/CAM against MCF-7 cells aligns with findings from Amani et al, 2020. 20 They tested the combined effects of CAM and DOX on MCF-7 cells exposed to varying concentrations (5, 100, and 500 µM) of CAM and (5, 200 µM) of DOX. Amani et al reported a 13% to 16% reduction in cell viability after 72 h with CAM concentrations of 100 µM and 500 µM, respectively. Interestingly, 5 µM CAM did not affect the IC50 value of DOX, suggesting further investigation is needed to elucidate the specific mechanisms underlying the CAM/DOX combination.
Based on our findings, treatment with CAM/DOX PEGylated liposomes significantly induced greater inhibition of cell growth compared to a physical mixture. This indicates that encapsulating CAM and DOX into PEGylated liposomes enhanced the cytotoxic effect of DOX, likely due to improved penetration and localization into tumor cells. The decrease in the IC50 value for CAM/DOX-PEGylated liposomes (0.13 µM) compared to the IC50 of the physical mixture (0.25 µM) after 72 h of treatment (Figure 4B) suggests enhanced potency in growth inhibition, which may be attributed to altered DOX efflux in MCF-7 cells. PEGylated liposomes protect encapsulated drugs, enhance cellular uptake, and reduce toxicity. Our results show that DOX and CAM-loaded PEGylated liposomes inhibit cell growth more effectively than physical mixtures, supporting these findings. 47 The mechanism underlying this potentiation between DOX and CAM needs further clarification. Specifically, CAM's potential to inhibit and reduce P-glycoprotein (P-gp) activity will be assessed using a P-gp ELISA assay, details of which will be provided subsequently.
P-Glycoprotein ELISA Assay
After measuring the optical density (OD) using a microplate reader as described in the materials and methods section, the concentration of P-gp in the samples was determined from the established calibration curve (Figure 5B). All results fell within the linear range of the calibration curve equation, ensuring the accuracy and reliability of the measurements.

(A) P-glycoprotein concentrations, (B) P-glycoprotein ELISA calibration curve.
Figure 4 shows the original concentrations of extracellular P-gp for free DOX and CAM/DOX-PEGylated liposomes for 24 and 72 h, multiplied by dilution factor 5. The OD value is directly related to the concentration of P-gp. Based on (Figure 5A) the concentration of P-gp for free DOX at 72 h did not differ significantly from that of free DOX at 24 h. However, the concentration of P-gp in CAM/DOX-PEGylated liposomes at 72 h was lower than that of CAM/DOX-PEGylated liposomes at 24 h, and the reduction was approximately 1.61% and 5.2% for free DOX and CAM/DOX-PEGylated liposomes, respectively. The ability of CAM to inhibit P-gp explains the reduction in P-gp concentration. Therefore, decreased concentrations of drug efflux pumps, such as P-gp, have been linked to a decrease in MDR and an increase in DOX accumulation in MCF-7 cells. CAM enhances rug accumulation and potentially overcoming multidrug resistance (MDR) in cancer therapy. Its ability to improve the efficacy of co-administered chemotherapeutic agents offers potential for synergistic drug combinations.19,48
Regarding the IC50 at 24 h treatment by physical mix group or CAM/DOX-PEGylated liposomes group. Amani et al, revealed that CAM can produce a synergistic cytotoxic effect when combined with Dox through dysregulation of autophagy (a high level of autophagy is associated with the development of DOX resistance). 20 Moreover, Komatsu et al, had confirmed that CAM is able within 24 or 48 h to potentiate the cytotoxic effect of Bortezomib against MCF7 through inhibition of autophagy. 49 Interestingly, this rapid potentiation effect is unlikely to be prominent, especially in the liposomal formulation, where it is expected that the release of free CAM will be slower in CAM/DOX-PEGylated liposomes than in physical mix group. On the other hand, our findings, at 72 h of CAM/DOX-PEGylated liposomal treatment, align with the significant down expression of extracellular P-glycoprotein as shown in the ELISA assay. Consequently, it seems that there is a time-dependent cytotoxic response for this combination through- at least- two different mechanisms, the inhibition of autophagy and P-glycoprotein down expression.
Conclusion
In this study, DOX, a potent chemotherapeutic agent effective against numerous solid tumors, is hindered by poor selectivity and multidrug resistance (MDR), limiting its anticancer efficacy. For the first time, CAM/DOX-PEGylated liposomes were prepared using thin film hydration, and probe sonication method. These liposomes exhibited high monodispersity and excellent encapsulation efficiency for both DOX and CAM. Colloidal stability assessments over 9 days demonstrated robust stability. Cell viability assays highlighted superior activity of CAM/DOX-PEGylated liposomes compared to both the physical mixture of CAM and DOX, as well as free DOX. Furthermore, CAM/DOX-PEGylated liposomes reduced the concentration of P-glycoprotein pumps compared to free DOX, as determined by P-gp ELISA assay. Future studies focusing on DOX-resistant cell lines are warranted to fully elucidate CAM's synergistic effect on enhancing DOX cytotoxicity. Additionally, in vivo investigations are essential to comprehensively evaluate CAM/DOX-PEGylated liposomes as a promising nanomedicine for anticancer therapy.
Supplemental Material
sj-jpg-1-tct-10.1177_15330338241312561 - Supplemental material for In Vitro Potentiation of Doxorubicin Cytotoxicity Utilizing Clarithromycin Loaded-PEGylated Liposomes
Supplemental material, sj-jpg-1-tct-10.1177_15330338241312561 for In Vitro Potentiation of Doxorubicin Cytotoxicity Utilizing Clarithromycin Loaded-PEGylated Liposomes by Islam Alfreahat, Hamdi Nsairat, Ibrahim Deeb Aldeeb, Ali Al-Samydai and Walhan Alshaer in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338241312561 - Supplemental material for In Vitro Potentiation of Doxorubicin Cytotoxicity Utilizing Clarithromycin Loaded-PEGylated Liposomes
Supplemental material, sj-docx-2-tct-10.1177_15330338241312561 for In Vitro Potentiation of Doxorubicin Cytotoxicity Utilizing Clarithromycin Loaded-PEGylated Liposomes by Islam Alfreahat, Hamdi Nsairat, Ibrahim Deeb Aldeeb, Ali Al-Samydai and Walhan Alshaer in Technology in Cancer Research & Treatment
Footnotes
Acknowledgements
Only the listed authors contributed to this work and no other individuals or collaborators were involved in this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Research Ethics and Patient Consent
This study did not involve any experiment on animal or human subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
