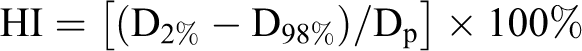Abstract
Purpose:
To evaluate 2 simultaneous integrated boost treatment planning techniques using helical tomotherapy for breast conserving therapy with regard to acute skin toxicity and dosimetry.
Methods:
Thirty-two patients were studied. The original approach was for 16 patients and incorporated a directional block of the ipsilateral lung and breast. An additional 16 patients were planned for using a modified approach that incorporates a full block of the ipsilateral lung exclusive of 4 cm around the breast. Dose–volume histograms of targets and critical structures were evaluated. Skin toxicity monitoring was performed throughout treatment and follow-up using the Common Terminology Criteria for Adverse Events.
Results:
Treatment was well tolerated with patients receiving a median dose of 59.36 Gy. Of the 16 patients in both groups, 8 had grade 2 erythema immediately after radiation. On 3-week follow-up, 10 and 7 patients in the original and modified groups showed grade 1 erythema. On 3- and 6-month follow-up, both groups had minimal erythema, with all patients having either grade 0 or 1 symptoms. No grade 2 or 3 toxicities were reported. Mean treatment time was 7.5 and 10.4 minutes using the original and modified methods. Adequate dose coverage was achieved using both methods (V95 = 99.5% and 98%). Mean dose to the heart was 10.5 and 1.8 Gy, respectively (P < .01). For right-sided tumors, the original and modified plans yielded a mean of 8.8 and 1.1 Gy (P < .01) versus 11.7 and 2.4 Gy for left-sided tumors (P < .01). The mean dose to the ipsilateral lung was also significantly lower in the modified plans (11.8 vs 5.0 Gy, P < .01).
Conclusions:
Tomotherapy is capable of delivering homogeneous treatment plans to the whole breast and lumpectomy cavity using simultaneous integrated boost treatment. Using the treatment methods described herein, extremely low doses to critical structures can be achieved without compromising acute skin toxicity.
Introduction
Breast conserving surgery followed by adjuvant whole breast irradiation (WBI) lowers the risk of ipsilateral breast tumor recurrence and improves overall survival. 1,2 The most common approach is conventionally fractionated radiation with a standard dose of 45 to 50 Gy given at 1.8 to 2.0 Gy/daily 5 days/wk followed by a sequential boost to the tumor bed. 3 –6 More recently, the use of a simultaneous integrated boost (SIB) has increased for patients with early-stage breast cancer, with the intent of reducing overall treatment time and cost without compromising local control. 7 –10 Studies have shown that, compared to a conventional sequential boost, a SIB delivered through 3-dimensional conformal radiotherapy (3D-CRT) or static-field intensity-modulated radiotherapy (IMRT) can improve target dose conformity with better sparing of normal tissues such as the heart and lungs. 7 –10 Furthermore, helical tomotherapy (Accuray, Sunnyvale, California) has emerged as a platform for delivering modulated helical treatment for both postlumpectomy and postmastectomy patients that were once treated with a combination of electron and static photon beams. 11 –14 A report from our institution first described the use of tomotherapy for breast conserving therapy with SIB, 15 followed by a comparative planning study showing that tomotherapy is capable of delivering a SIB with adequate tumor coverage and superior normal tissue sparing to lungs and heart in comparison with 3D-CRT. 16 Although the use of helical tomotherapy for WBI has been well reported, there are few clinical reports of acute and late toxicity using this radiation delivery modality to deliver WBI with a SIB.
In this study, we present a dosimetric evaluation and acute skin toxicity resulting from 2 treatment planning techniques for WBI with a SIB to the lumpectomy bed using helical tomotherapy. We compare a technique that we have previously described 15 to a modified treatment planning technique that attempts to further decrease the dose of radiation to normal structures including the heart and lungs.
Methods and Materials
Thirty-two patients with early-stage breast cancer treated with breast conserving therapy and recommended to undergo whole-breast radiotherapy without dedicated lymph node coverage were consecutively identified. This study was approved by the University of Wisconsin Institutional Review Boards. Half (n = 16) of the patients were treated with the original planning technique while the other half (n = 16) were treated with a modified technique as described subsequently.
Computed tomography (CT) simulation scans with a slice thickness of 2.5 mm were acquired for each patient with surgical clips and a radiopaque wire placed on the lumpectomy scar in order to assist in lumpectomy cavity delineation. Superior, inferior, medial, and lateral aspects of the entire breast were also outlined with a radiopaque wire based on clinical examination. The CT scans were imported to the ADAC Pinnacle treatment planning system (TPS; Philips Medical Systems, Cleveland, Ohio) for contouring. The treated breast was contoured with the assistance of the radiopaque wires placed during simulation and based on the radiographic findings of breast tissue with no skin retraction. The lumpectomy bed was contoured as a boost clinical tumor volume with a 1-cm expansion to the planning tumor volume (PTV). Critical organs including the ipsilateral and contralateral lung, heart, spinal cord, esophagus, trachea, and contralateral breast tissue were also delineated for dose constraint during optimization. The heart was contoured from its base to the branch point of the pulmonary trunk into the pulmonary arteries. The contralateral breast was defined per the consensus definitions of the anatomical borders from the Radiation Therapy Oncology Group (RTOG) Breast Atlas. The supraclavicular, internal mammary, and axillary lymph nodes were not purposefully contoured for treatment purposes. The CT images and related contours were exported to the TomoTherapy TPS (Version 4.2.3) for planning.
Planning parameters included field width, pitch, and modulation factor. Selecting appropriate values for these parameters is crucial to the plan dose distribution and treatment delivery time. 15,17 For the studied patients, TomoTherapy planning parameters include a 2.5-cm field width, pitch of 0.287, and modulation factor of 1.8 to 2.4. For most patients, the prescription dose was 50.4 Gy in 28 fractions to the whole breast with a SIB of 59.36 Gy over 28 fractions to the lumpectomy cavity. The goal of each plan was to deliver 100% of the prescription dose to 95% of the PTV.
Treatment planning for patients planned with the original technique used a complete block of the contralateral lung and breast and a directional block of the ipsilateral lung, heart, and spinal cord. Patients treated using the modified method were planned with a complete block for the contralateral lung, directional block for the heart, and no block for the contralateral breast. In addition, we generated a new complete block that included the ipsilateral lung exclusive of a 4-cm expansion around the breast PTV. The goal of this new region of interest was to control the dose to the ipsilateral lung and reduce the total lung dose. Although dose constraints were met in all cases, this modified treatment planning method was used in later patients in an attempt to further decrease dose to normal structures.
Dose volume histograms were computed for the PTV and critical structures. Target dose homogeneity index (HI) was defined as:
Reported doses include the prescription dose (Dp), and the dose received by 2% and 98% of the PTV (D2% and D98%, respectively). Other dosimetric indices include the percentage of the PTV receiving 107% (V107%) and 95% (V95%) of the prescription dose as well as the ratios of mean, maximum, and minimal doses to the prescription dose (Dmean/Dp, Dmax/Dp, and Dmin/Dp). In addition, a conformity index defined as the ratio of the volume covered by the 95% prescription isodose over the volume of the breast planning target volume as per the RTOG 1005 protocol was calculated. Normal structure dosimetric indices such as mean dose, volume receiving 30 Gy, and volume receiving 5 Gy were acquired for the heart, ipsilateral lung, contralateral lung, total lungs, and contralateral breast.
All patients were treated with helical tomotherapy with daily megavoltage CT (MVCT) guidance for setup verification. The clinical end point of this study was acute skin toxicity immediately following completion of the treatment. Patients’ acute skin toxicity monitoring was performed using the Common Terminology Criteria for Adverse Events (CTCAE; Version 3.0) upon completing treatment and was prospectively recorded for a period of 6 months. Statistical analysis was performed using STATA version 11.1 (StataCorp, College Station, Texas).
Results
Patient Characteristics
Seven patients in each group had right-sided tumors while the remaining 9 in each group had left-sided lesions. Patients’ ages ranged from 36 to 81 years at time of diagnosis, with a mean of 61.8 years. Patients planned using the original planning technique accrued from April 2009 to October 2010 with a median follow-up time of 50 months, while patients planned utilizing the modified technique accrued from December 2011 to October 2012 with a median follow-up time of 25 months. Patients had a mean body mass index (BMI) of 28.9 in the original planning method group and 28.6 in the modified planning method group (P = .87). Patients in the first group were more likely to be active smokers (5 of 16) versus patients in the modified group (2 of 16). The characteristics of the 32 patients are shown in Table 1.
Patient Characteristics.

Abbreviations: BMI, body mass index; ER, estrogen receptors; PR, progesterone receptor.
Treatment Delivery
The mean treatment delivery time using the original planning technique was 7.5 minutes with a range of 5.1 to 11.6 minutes. The median dose delivered to the whole breast PTV was 50.4 Gy (range 48.6-50.4) with median total dose to the lumpectomy PTV 59.36 Gy (range, 58.05-63.0; Table 2A).
Treatment Dosimetry for the Original (A) and Modified (B) Planning Methods.

Abbreviations: PTV, planning tumor volume; SD, standard deviation.
The mean treatment delivery time using the modified planning technique was 10.4 minutes with a range of 8.6 to 12.4 minutes. The median dose delivered to the whole breast PTV was 50.4 Gy (range 41.4-50.4) and the median total dose to the lumpectomy PTV was 59.36 Gy (range 48.99-61.6; Table 2B).
Target Volumes
Dosimetric evaluations of target volumes for both planning methods are summarized in Table 2. Target volumes were comparable between the 2 groups, with a mean breast PTV of 772.4 and 709.1 in the original and modified groups, respectively (P = .65), and mean lumpectomy PTV volumes of 54.1 and 35.4 (P = .13). For the whole breast PTV, the “cold spot” for the whole breast (V<95%) was slightly higher in modified plans as compared to the originals, 2.1% versus 0.5%. Patients treated with the original planning method had ratios of mean and minimum breast PTV dose to prescription dose average 106.7% and 80.7% with a HI of 17.5%. The modified plans’ ratios averaged 104.3% and 51.8%, respectively, with mean HI of 28.4% of the prescription dose. The conformity index per the RTOG 1005 protocol was also calculated. The original plans had a mean conformity index of 0.99 for the whole breast PTV, with a standard deviation of 0.01. Modified plans also had a breast PTV mean conformity index of 0.99 and standard deviation of 0.01 (P = .68).
For the lumpectomy PTV, the mean hot spot (V>107%) volume was low in both the original and modified plans, with a value of 0.7% and 0.2%. All hotspots were confined to the lumpectomy PTV regions. The mean cold spot volume (V<95%) was less than 0.5% in both cases. The ratios of mean, maximum, and minimum lumpectomy PTV dose to prescription dose averaged 102.6%, 106.9%, and 96.9% in the original plans and 101.7%, 105.4%, and 91.9% in modified plans. The mean HI of the lumpectomy PTV was 5.7% and 6.1% in original and modified plans, respectively. Lumpectomy PTVs were also similarly conformal, with a mean conformity index of 0.98 using the original planning method and 1.00 with the modified method (P = .02).
Organs at Risk
Dose distributions are shown for both right- and left-sided patients using both planning techniques (Figure 1). The dose indices for organs at risk are shown in Table 3. The mean dose to the heart was 10.5 Gy using original planning methods and 1.8 Gy using the modified technique (P < .01). For patients with right-sided tumors, the original and modified plans yielded a mean of 8.8 and 1.1 Gy, respectively (P < .01) and 11.7 and 2.4 Gy for left-sided tumors (P < .01). Heart V30 was similarly low, with a mean of 1.2% and 0.1% (P = .11).

Sample contours and dose distributions for both left- and right-sided patients using both the original and modified planning methods.
Dose to Organs at Risk Using the Original (A) and Modified (B) Planning Methods.

Abbreviation: SD, standard deviation.
The average mean lung dose (MLD) for the ipsilateral lung was also significantly lower in the modified plans, with a mean of 11.8 versus 5.0 Gy (P < .01). The MLD for the contralateral lung was significantly lower in modified plans, with a mean of 5.6 versus 0.5 Gy for the 32 patients (P < .01). Similarly the percentage of total lung receiving 20 Gy was 6.0% in original plans versus 4.8% in modified plans (P = .02). The mean dose to the contralateral breast was slightly higher in the modified plans, 4.4 versus 5.3 Gy, but the difference was not statistically significant (P = .08). The maximum dose and the percentage volume receiving 5 Gy were also slightly higher. Maximum spinal cord dose was lower in the modified plans. The absolute volume of the total body (the rest of unspecified normal tissue) receiving >5 Gy was 6700 cm3 in the original plans and 5200 cm3 in the modified plans (P < .01).
Acute Skin Toxicity
Overall, treatment was extremely well tolerated. Per CTCAE Version 3 definitions, 8 (50%) of the 16 patients treated using both the original and modified planning methods had grade 2 erythema immediately after treatment (Figure 2). On a 3-week follow-up, erythema had largely resolved with 10 patients showing grade 1 erythema in the original planning group and 7 showing grade 1 erythema in the modified treatment planning group.

Skin toxicity in patients treated using the original (A) and modified (B) treatment planning methods.
On both 3- and 6-month follow-up, both groups of patients had minimal residual erythema, with all patients either having grade 0 or grade 1 symptoms. There were no grade 2 or grade 3 toxicities reported at these end points.
Discussion
We have demonstrated that simple alterations in treatment planning techniques for breast and lumpectomy cavity radiation for early-stage breast cancer can improve dosimetric measures while achieving minimal skin toxicity. This was achieved with a mean treatment delivery time of 7.5 and 10.4 minutes, a savings of 25% to 50% as compared to published studies. 13,14,16 The slightly increased treatment time using the newer planning techniques highlights the trade-off between increasing dosimetric constraints and treatment time. Similarly, our relatively low rates of skin toxicity show that TomoTherapy can be used to effectively treat patients with acceptable acute skin toxicity; however, some care must be taken with planning. If the skin surface is included as an optimization target, inverse planning can result in solutions that favor tangential beam segments near the skin surface. 18 Although the combination of inverse planning and continuous rotational delivery allows TomoTherapy flexibility in delivering dose to superficial targets, high dose to the skin can result in significant skin desquamation and fibrosis. It is well known that if the “hot-spot” (>10% of prescribed dose) involves the skin surface, significant acute toxicity can develop. 19 A recent study of 158 patients with breast cancer treated with 3D-CRT has also suggested that lower doses can also correlate with toxicity. 20 Nevertheless, acute toxicity must be balanced with ensuring adequate tumor coverage. The RTOG 1005 protocol has an additional constraint, the conformity index, defined as the ratio of volume covered by the 95% isodose line divided by the volume of the breast, which serves as an aid in evaluating plans to ensure they are not too “cool” or too “hot.” Conformal plans are one of the strengths of TomoTherapy, with all of our plans having a Conformity index of nearly 1. We were also able to avoid severe skin toxicity with hot spot means of 107% and 105%, respectively. Similarly, Freedman et al treated patients with early-stage breast cancer having a 4-week hypofractionated IMRT course with a simultaneous electron boost and reported acceptable acute skin toxicity and quality of life. 21 Contouring the skin as an organ at risk for optimization has become one way to reduce skin dose and avoid toxicity. 18,22 Increasing breast size during treatment due to inflammation may lead to significant underdosing of the skin, while postsurgical changes may lead to contracture and overdosing. 23 Issues also exist with patient positioning, as a 5-mm shift may introduce dose variation of 3% to 9%. 24 Further complicating matters is that the TomoTherapy planning software itself may overestimate skin dose by anywhere between 3% and 13%. 25 –28 Despite these limitations, adequate breast surface dose has been shown to be achievable utilizing TomoTherapy in clinical practice both at our institution, 29 as well as others. 30,31 Special care must be taken when treating patients with breast cancer having IMRT. The inherent nature of TomoTherapy treatment allows for daily MVCT setup imaging, and extra attention must be paid to ensure that patient positioning is accurate. If patient positioning or setup is found to be inaccurate, our institutional practice is to use additional TLD measurements or dose recalculation on daily MVCT imaging to ensure adequate dosing. Although we have found daily patient setup to be satisfactory as reported previously, a robust approach has been described for volumetric modulated arc therapy utilizing an expanded optimization plan to mimic possible changes in size due to edema or breath variation. 32 A similar technique can be applied using TomoTherapy.
Finally while the focus of our study is on skin toxicity, skin dose is not the only factor found to be significantly associated with increased skin toxicity. The BMI, breast size, the use of breast IMRT, beam energy, maximum dose, and sagittal dose gradient have all been associated with increased toxicity. 19
With regard to other critical tissues, the techniques presented here achieve doses at least as good as those presented in previous studies. Our V20 lung doses were comparable to previously published data including those by Goddu et al (range, 5.8%-10.4%), while our total body dose was 33% lower than those achieved in the same study. 13 Although our heart doses using our original treatment planning parameters are comparable to previously published studies by Fogliata et al (range, 8.7-21.1 Gy) 33 as well as Goddu et al (mean, 12.2 Gy), 13 the heart doses achievable using our modified treatment planning techniques are significantly lower. This is especially relevant in enrolling patients on protocol. As practitioner experience with IMRT in the treatment of breast cancer improves, protocols have begun to implement more stringent guidelines. The recently closed RTOG protocol 1005, for example, requests that plans have a mean heart dose of less than 3.20 Gy. The original treatment planning methods described previously would not meet this constraint while the modified plans do so easily. For both heart and lung, our modified treatment planning approach allows us to achieve statistically significant improvements in radiation doses. With some evidence showing the risk of ischemic heart disease increasing linearly with dose delivered, 34 this may have implications for long-term cardiac morbidity.
We also acknowledge that although our doses to normal tissues achieved with the modified planning technique are significantly lower than that with the original method, they may in some cases be inferior to those that achievable with a tangential beam arrangement. This is especially true in cases of right-sided tumors and mean dose to the heart. Second, dose to the contralateral breast will necessarily be higher than with traditional opposed tangential beams, theoretically increasing the risk of secondary malignancy in a patient population with high long-term survival. These risks must be balanced by the realities of treatment delivery and patient convenience. The center where patients were treated in this study is at an academically affiliated TomoTherapy-only community center, where a single-treatment machine must serve a wide variety of radiotherapy needs. In this case, the closest standard linear accelerator is located over 60 minutes way. Although we acknowledge that a lower heart and contralateral breast dose may be desirable, the availability of treatment closer to home may increase the proportion of patients receiving recommended radiation as a component of their breast conserving therapy. Finally, patients in this study were accrued before newer limited-angle tomotherapy units were available, so this may be less of a factor in the future.
We acknowledge several limitations to our study. First, the limited number of patients limits our power to draw firm conclusions; however, our results are comparable to those previously published for breast IMRT. 21 Second, there is some heterogeneity in our treatment cohort, with a handful of patients receiving doses that were slightly higher or lower than originally planned. Furthermore, follow-up assessments did not extend beyond 6 months and thus late toxicity and cosmetic outcome were not assessed. Finally, patients treated on this study were treated using traditional helical TomoTherapy and were accrued before newer limited-angle TomoTherapy units were available.
Conclusion
To date, there have not been any published clinical evaluations of toxicity on helical TomoTherapy-based breast cancer treatment using the SIB dose regimen. In this study, we have shown that helical TomoTherapy is capable of delivering homogeneous treatment plans using SIB treatment within a reasonable treatment time, acceptable dose to critical organs, and with minimal skin toxicity.
Footnotes
Authors’ Note
All authors certify that this manuscript has not been published in whole or part nor is it being considered for publication elsewhere.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RJK is partially supported by NIH grant CA160639. The other authors received no financial support for the research, authorship, and/or publication of this article.