Abstract
Introduction:
We report our series of patients with large brain metastases, >3 cm in diameter, who received stereotactic radiosurgery (SRS) as a component of their treatment, focusing on survival and intracranial recurrence rates.
Materials and Methods:
The brain tumor database was queried for patients treated with SRS for large brain metastases. Local recurrence (LR) and distant brain recurrence (DBR) rates were calculated using cumulative incidence analysis, and overall survival (OS) was calculated using Kaplan-Meier analysis. Patients were classified into 1 of the 4 groups based on treatment strategy: SRS alone, surgery plus SRS, SRS plus whole-brain radiation therapy (WBRT), and salvage SRS from more remote WBRT and/or surgery.
Results:
A total of 153 patients with 164 lesions were evaluated. The SRS alone was the treatment approach in 62 lesions, surgery followed by SRS to the resection bed (S + SRS) in 33, SRS + WBRT in 19, and salvage SRS in 50. There was no statistically significant difference in OS between the 4 treatment groups (
Conclusion:
There were no significant predictors for local control. Keeping in mind that patient numbers in the SRS + WBRT group are small, the addition of WBRT to SRS did not appear to significantly improve survival or local control, supporting the delayed use of WBRT for some patients to prevent potential side effects provided regular imaging surveillance and salvage therapy are utilized. Prospective studies are needed to optimize SRS treatment regimens for patients with large brain metastases.
Introduction
Brain metastases are a significant clinical problem, occurring in up to 40% of patients with cancer. 1 Patients with brain metastases can be treated with stereotactic radiosurgery (SRS) alone, as an adjuvant to surgical resection or whole-brain radiation therapy (WBRT), or as salvage therapy. Large brain metastases, however, pose a unique problem for those undergoing treatment with SRS. Because the volume of normal tissue within the treatment area is larger, lower doses are prescribed to prevent adverse effects. 2 The Radiation Therapy Oncology Group (RTOG) protocol 90-05 established the maximum tolerated dose of 24 Gy for tumors <2 cm, 18 Gy for tumors 2.1 to 3.0 cm, and 15 Gy for tumors 3.1 to 4.0 cm. 3 However, a retrospective study from our institution found that tumors treated with 15 Gy (3.1-4.0 cm) or 18 Gy (2.1-3.0 cm) had a 3-fold increased risk of local failure compared to tumors treated with 24 Gy (<2.0 cm) with 1-year local control <50%. 4
Although there are published treatment guidelines for patients with brain metastases, 5 there is little evidence available for larger brain metastases, specifically those greater than 3 cm in maximum diameter. Additionally, there are no reported prognostic indicators for these patients, which would help determine the optimal treatment, especially since only patients with relatively good prognoses may survive long enough to benefit from aggressive treatment of their intracranial disease. Currently, patients with brain metastases tend to have a poor prognosis regardless of treatment modality—median survival is typically less than 1 year. 6 -10
Therefore, the purpose of our study was to compare survival and intracranial recurrence in patients with brain metastases larger than 3 cm in diameter who were treated with single-session SRS alone, WBRT and SRS boost, surgery followed by SRS to the resection bed, or salvage SRS from more remote prior WBRT and/or surgery.
Methods
Our institutional review board-approved brain tumor database was queried to identify patients treated with Gamma Knife (GK) SRS for large brain metastases (defined as those greater than 3 cm in largest diameter) between February 2007 and March 2013. In these patients, GK was given in a single treatment session. Computed tomography and magnetic resonance imaging (MRI) were used to define the brain metastases. Doses were prescribed based on the RTOG 90-05 guidelines, although tumors with diameters greater than 4 cm may have been dosed with less than 15 Gy at the discretion of the treating radiation oncologist. No margins were used, even when treatment included only the resection bed.
Patients were included in the study if they received single-session GK for a lesion >3 cm in largest diameter. Those who had staged SRS (SRS delivered over 2-3 sessions separated by 2-4 weeks) to the same lesion were excluded, as were those who had calvarial metastases or a tumor smaller than 3 cm in largest diameter prior to resection. Eligible patients were categorized into the following 4 groups based on treatment strategy: SRS alone, surgery plus SRS, SRS plus WBRT, and salvage SRS from more remote WBRT and/or surgery. Adjuvant therapies were distinguished from salvage therapies based on timing (occurring within 6 weeks of SRS) and/or the original intent of treatments as indicated by chart review. Primary end points include overall survival (OS), local recurrence (LR), and distant brain recurrence (DBR). Overall survival was defined from the date of SRS. Local recurrence was defined as recurrence (ie, increase in diameter of 20% from best response) within the original SRS treatment bed, and DBR was defined as recurrence (ie, new lesion) outside the original SRS treatment bed. Local recurrence and DBR rates were calculated using cumulative incidence analysis, and OS was calculated using Kaplan-Meier analysis. Competing risk regression analyses with death as a competing event were conducted for LR and DBR. Cox proportional hazards analysis was conducted for OS. Variables analyzed included age, gender, Karnofsky performance status (KPS), RPA class, GPA score, primary diagnosis and radioresistant histology (defined as renal cell carcinoma, melanoma, colon, or sarcoma), number of brain metastases treated with SRS, best response to SRS as defined by the Response Evaluation Criteria In Solid Tumors (RECIST) criteria 11 (complete response, partial response, stable disease, and progression), and incidence of radiation necrosis (as determined by tumor board review).
Results
A total of 153 patients with 164 lesions were eligible for analysis. There were 111 patients who died, 29 local failures, and 45 DBRs. Table 1 shows lesion characteristics. Median age was 61 (range 29-92). The RPA class was 1 in 12 patients, 2 in 116, and 3 in 25. The GPA score was 0 to 1.0 in 44 patients, 1.5 to 2.5 in 92, 3 in 11, and 3.5 to 4.0 in 6. The SRS alone was the treatment approach in 62 patients, surgery followed by SRS to the resection bed (surgery + SRS) in 33, SRS plus upfront WBRT (SRS + WBRT) in 19, and salvage SRS in 50. Median SRS dose was 15 Gy (range: 10-20). Median maximal tumor diameter was 3.5 cm (range: 3.0-6.8). Median MRI follow-up time was 2.7 months (range: 1-57); 68 (44%) patients had at least 4-month follow-up time. Overall, 9 (5.5%) patients developed radiation necrosis, 4 of which were in the SRS alone group (6.5% of patients in this group). There was a statistically significant difference between the 4 groups for the following variables: age at SRS, tumor size, and best response to SRS. Patients receiving SRS alone were older than patients in the other 3 treatment groups. Larger tumors were more commonly treated with surgery + SRS, and patients treated with surgery + SRS were more likely to achieve a complete response.
Characteristics of Brain Metastases.

Abbreviations: GK, Gamma Knife; GPA, graded prognostic assessment; KPS, Karnofsky performance status; mets, metastases; MRI, magnetic resonance imaging; NSCLC, nonsmall cell lung cancer; RPA, recursive partitioning analysis; SCLC, small cell lung cancer.
Overall survival approached, but failed to reach, a significant difference among the 4 groups (

Overall survival with metastases >3 cm treated with Gamma Knife (N = 153).
Analysis of Clinical Variables.
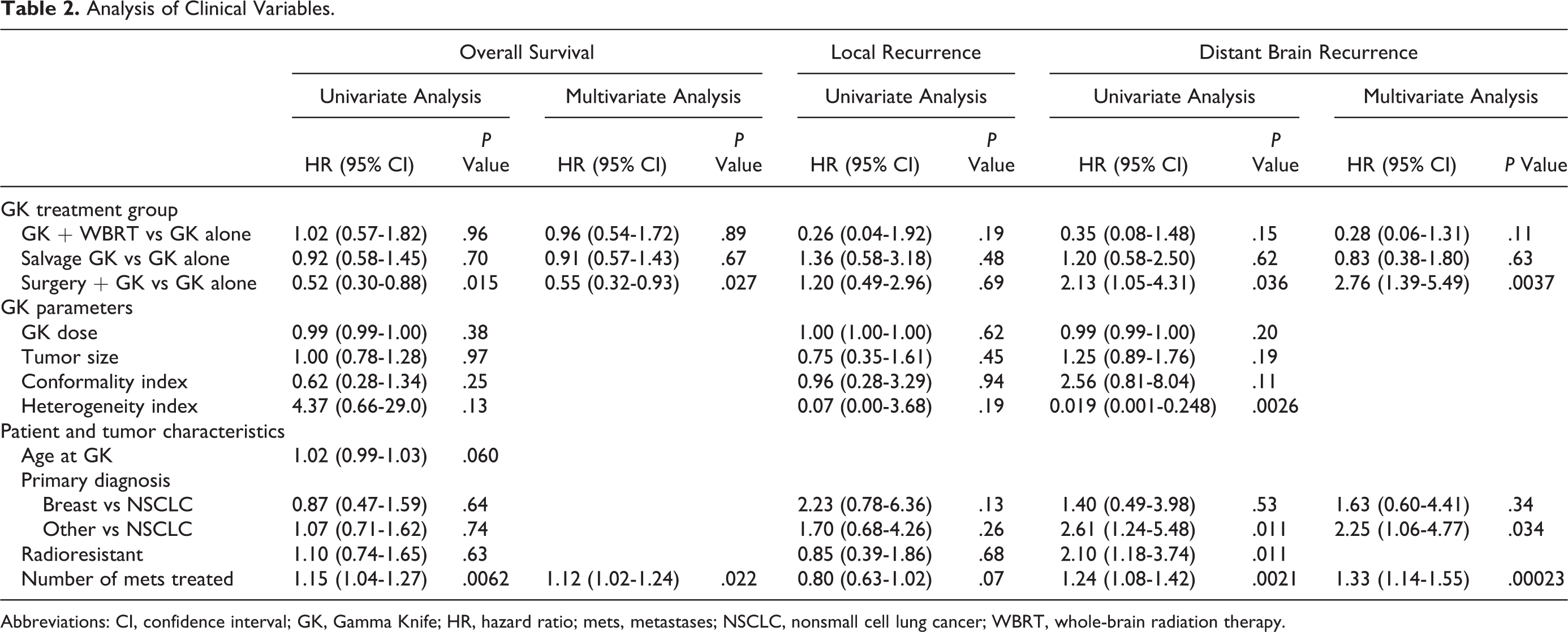
Abbreviations: CI, confidence interval; GK, Gamma Knife; HR, hazard ratio; mets, metastases; NSCLC, nonsmall cell lung cancer; WBRT, whole-brain radiation therapy.
There was no significant difference for local recurrence rates (

Cumulative incidence for local recurrence for patients with metastases >3 cm treated with Gamma Knife (N = 164).
There was a significant difference for DBR rates (
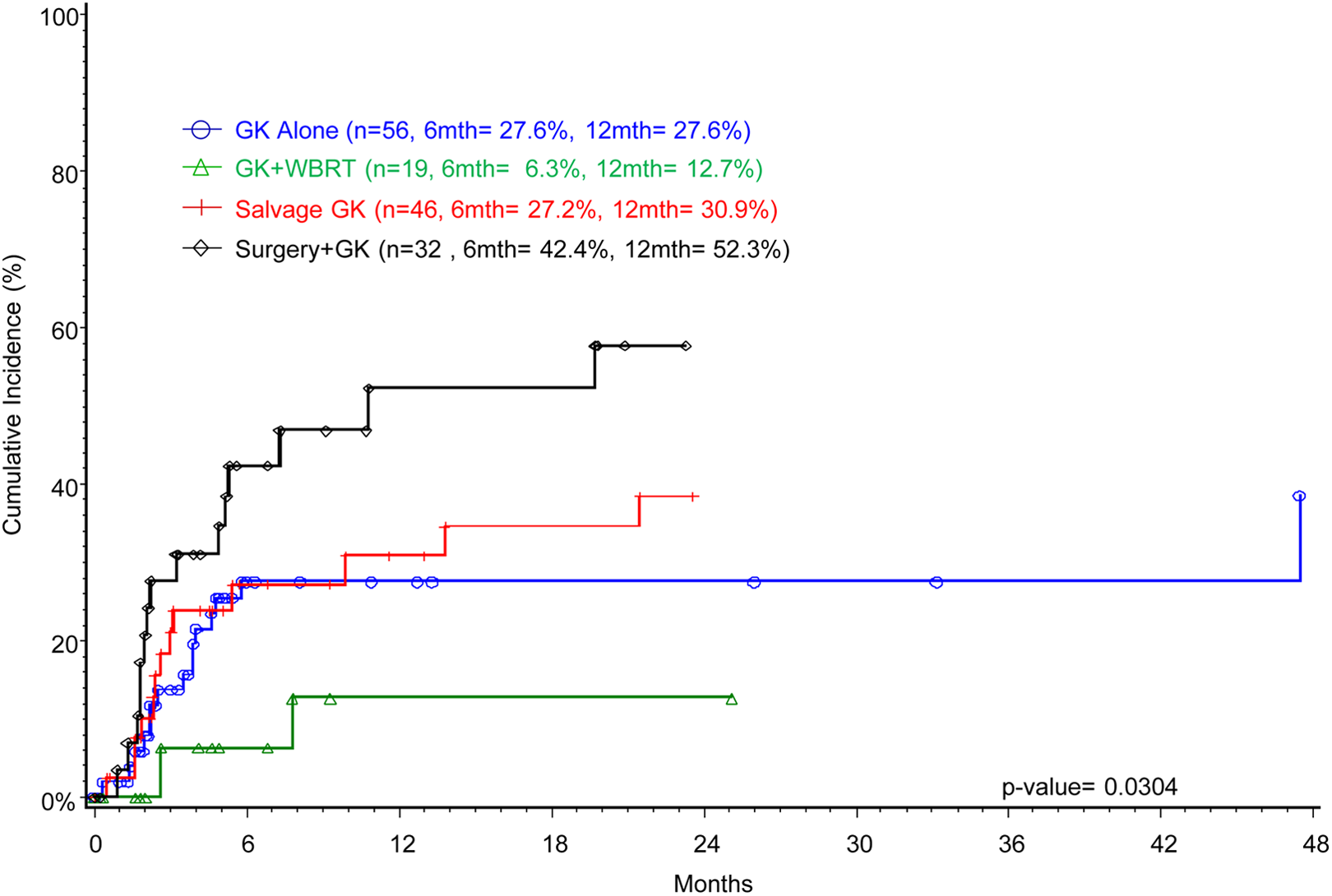
Cumulative incidence for distant brain recurrence for patients with metastases >3 cm treated with Gamma Knife (N = 153).
Discussion
Overall, we found no significant predictors for local control in patients with large brain metastases treated with SRS as part of the treatment strategy. However, a number of observations can be made.
SRS Alone
The body of literature regarding the use of SRS for large tumors (>3 cm in maximum diameter, roughly equivalent to 14 cm3 in volume) is growing and is summarized in Table 3. The studies using multisession SRS dosing schedules for large tumors appear to report better survival rates than those using a single session, including our study, which may be related to the higher overall dose that can be achieved with multiple sessions. A phase II clinical trial of multisession SRS included patients with larger metastases, but the median tumor size and volume were still relatively small. In that study, tumor volume (less than 6 cm3) was a significant prognostic indicator, 12 which is similar to the trend seen with single-session SRS studies. As expected, studies that include only patients with large metastases report worse outcomes.
Studies Examining SRS Alone for the Treatment of Large Brain Metastases.

Abbreviations: NR, not reported; SRS, stereotactic radiosurgery.
Four studies have examined multisession SRS for large tumors alone. One was a prospective cohort study examining staged SRS in 2 sessions for patients with metastases larger than 10 cm3 in volume with a resultant median survival of 11.9 months and a 1-year local control of 61%. 13 The remaining studies were retrospective in nature. 14 -16 Overall survival in our current single-session SRS study was less than that reported in the above-mentioned studies that used multiple sessions schedules, but comparable to the single-session SRS studies. 16,17 The other retrospective study of single-session SRS for patients with metastases larger than 3 cm in diameter, without a comparison group, had slightly better median survival (7.9 months) and 1-year local control, although this study used less strict criteria for local failure. 17 These studies are summarized in Table 3.
It is hard to make any specific recommendations of single versus multiple sessions for larger brain metastases as there has not been a randomized controlled trial (RCT) comparing the two. That said, given the limited dose that is typically given with single-session SRS, there is interest in multisession SRS at our institution.
Whole-Brain Radiation Therapy With SRS Boost
Whole-brain radiation therapy improves local and distant control of brain metastases but does not significantly improve OS. 18 Moreover, it is associated with an acute decline in various quality of life measures 19 as well as the potential for chronic neurocognitive changes. However, there is controversy regarding the relative effect of tumor progression versus WBRT itself on neurocognitive decline. 20,21 Despite this, WBRT remains the standard treatment for patients with multiple (>5) brain metastases. 2 Close imaging surveillance, allowing for possible salvage therapy, is recommended if WBRT is withheld. 19
As demonstrated in 2 RCTs, combination therapy with SRS boost to WBRT improves survival 22 and local control 22,23 versus WBRT alone for patients with fewer than 5 metastases. 5 One of these studies, which included patients with metastases up to 4 cm, found significantly improved survival (median 6.5 vs 5.3 months) for patients with metastases >2 cm when treated with combined therapy versus WBRT alone. The study also reported better local control in the combined therapy group, although tumor size was not analyzed as a variable. 22
A prospective cohort study comparing WBRT alone, SRS alone, and the two in combination for patients with a single metastasis from lung cancer included tumors up to 4.5 cm in diameter and found that both SRS alone and SRS boost to WBRT yielded significantly better median survival (9.3 and 10.6 months vs 5.7 months) and local control (6.9 and 8.6 months to local failure vs 4.0 months) than WBRT alone. Adding WBRT to SRS, however, did not confer a local control (
Interestingly, our results show that adding WBRT to SRS provided some benefit for local or distant control versus SRS alone, but this benefit failed to reach statistical significance. Our results, keeping in mind that our WBRT with SRS cohort is small and limits the power of this comparison, suggest that for patients with large brain metastases undergoing SRS, WBRT may be withheld as long as regular imaging surveillance is performed and salvage therapy is available for potential intracranial failures.
Stereotactic Radiosurgery and Surgery
Surgery followed by WBRT yields comparable survival to SRS boost to WBRT. 5 There are, however, no phase 3 trials comparing the 2 modalities because of the difficulty in randomizing patients to an invasive therapy (ie, surgery) compared to SRS. 2 Surgical resection followed by WBRT is often recommended for patients with tumors larger than 3 cm and causing a clinically significant mass effect. In addition, there is little specific evidence for the use of SRS for these large lesions 26 and SRS’s effect on a lesion is less predictable. A retrospective study, which included patients with metastases up to 4 cm, compared surgery plus WBRT to SRS alone and found no significant difference in either survival rates (38% vs 54% at 1 year) or local control rates (56% vs 64% at 1 year). 27 Several retrospective studies have compared surgery plus WBRT versus SRS boost to WBRT—all but one of them excluded patients with large brain metastases for SRS. One of these studies reported better survival and local control rates in the surgery group. 28 The other 3 studies found no difference in survival between the groups, whereas the SRS group had better local control. 29 -31 The study that included patients with metastases up to 4 cm in diameter found significantly better survival rates (56% vs 47% at 1 year) and local control rates (82% vs 66% at 1 year) in the SRS group. 32
In regard to SRS to the resection bed for large tumors, 1 prospective cohort study examined 112 patients with resected brain metastases with an average volume of 8.7 cm3 (up to 67 cm3) who were treated with a median SRS dose of 20 Gy (range 12-30 Gy) in a median 2 sessions (range 1-5 sessions). That study reported a median survival of 17 months with 1-year local and distant control rates of 91% and 46%, respectively. 33 Our group of patients who were treated with SRS to the resection bed had a lower median survival (12.2 months) than this prospective study that included patients with smaller tumors, which is not surprising. While our 6-month local control rates are comparable at 92%, our 12-month local control rates are much worse (66%). This difference can perhaps be partially explained by the higher average dose received in the prospective study (20 vs 15 Gy), which is dictated by the size of the resection bed in this cohort.
There are 3 retrospective studies that examined the role of SRS to the resection cavity of metastases with a median diameter greater than 3 cm or a median volume greater than 14 cm3. These studies report median survival from 10.9 to 15.6 months, with 6- and 12-month local control rates of 87% to 95% and 80% to 91%, respectively. 34 -36 Our results are comparable in terms of survival and 6-month local control. However, our 12-month local control is again worse.
Two retrospective studies completely excluded patients with smaller tumors, including only those with brain metastases greater than 3 cm in maximum diameter. One of these studies treated the metastases with 27 Gy in 3 sessions and found a median survival of 17 months, with a 1-year local control rate of 93%. 37 Both of these outcomes are better than our results, suggesting again that there is a dose–response relationship for SRS, even to a large resection bed. The other study used 24 Gy in 3 sessions, found a median survival of 5.5 months, and only reported a 6-month local control rate, which was 80%. The lower survival rates in this latter study may be partially accounted for by the larger tumor volumes included (average 29 cm3 and up to 81 cm3 vs an average 18 cm3 and up to 36 cm3 in the other study). 38
Collectively, the above-mentioned studies of surgery with adjuvant SRS—including our current study—reported poor distant control rates (ranging from 35% to 57% at 12 months). There may be a risk of leptomeningeal seeding of the tumor during resection, which localized treatment such as SRS does not address, but further studies are needed. 39 In our study, DBR occurred significantly more often in patients undergoing surgery with adjuvant SRS versus SRS alone (HR 2.13, 95% CI 1.05-4.31). Distant brain recurrence rates were higher in patients with more CNS metastases (HR 1.24, 95% CI 1.08-1.42) and radioresistant primary histology (HR 2.10, 95% CI 1.18-3.74), while patients with NSCLC primary tumors had lower rates of DBR. Despite the increased rates of DBR, our results indicate that surgery followed by SRS to the resection bed significantly improves survival versus SRS alone for these patients with large metastases (HR 0.52, 95% CI 0.30-0.88), although selection bias may be at play.
Prognostic Variables
There was a significant difference in patient age among the 4 groups in our study (62 vs 66 years for surgery vs SRS alone, respectively), and patients in the surgery group had fewer brain metastases on average. Other known prognostic factors, such as KPS (
In our study, none of the clinical variables, including treatment strategy, yielded a significant improvement in local control, which suggests that more studies are needed for optimization. The low rates of radiation necrosis in our study, however, suggest that dose escalation may be possible. A few strategies are being investigated at our institution, one of which includes staged SRS treatment in 2 to 3 sessions spaced within a month to intact metastases that are larger than 3 cm in diameter up to 5 cm in size. This technique has been previously described. 13,40 Four patients have received this treatment, and none have developed local failure at a median follow-up time of 4.6 months. This treatment technique is being investigated retrospectively at our institution and will be reported separately. Prospective studies, with dose escalation or neoadjuvant SRS, may be helpful to further clarify the best treatment strategies for patients with large brain metastases. 41 Neoadjuvant SRS, where SRS is delivered prior to resection of the brain metastasis, has a number of advantages, which include reducing the risk of leptomeningeal spread at the time of surgery and treating a tumor with an intact blood supply, which may improve control. Also, the true resection bed may be difficult to define for SRS planning. Having a target one can see at the time of SRS may reduce the risk of missing disease. This technique is being investigated prospectively at our institution.
Limitations of our study include its retrospective nature as well as a short follow-up time due to the poor prognosis for these patients. Also, there is an inherent selection bias in which patients are chosen for surgery; these patients are typically in better health, have good functional status, have well-controlled primary disease, and have fewer brain metastases. Another limitation is that we did not study a cohort of patients with surgical resection followed by WBRT. This was intentional since the study was designed to include contemporary patients since 2007. Given the institutional shift to do SRS postresection in ideal candidates, this group was the focus of this study.
Conclusions
Our single-institution, retrospective study compares outcomes for patients with large brain metastases (>3 cm) who were treated with single-session SRS radiosurgery alone, as an adjuvant to WBRT or surgical resection, or as salvage therapy. Although the addition of WBRT to SRS did modestly (but not significantly) reduce rates of local and distant recurrence, it did not appear to improve survival. This supports delayed use of WBRT for some patients to prevent potential side effects, so long as regular imaging surveillance and salvage therapy are utilized. Surgery followed by SRS appears to result in improved survival versus single-session SRS alone, although there was a higher incidence of distant failure. There were no significant variables for local control. Further studies are needed to optimize treatments for these patients.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J. H. Suh is consulting for Varian Medical Systems. G. H. Barnett is consulting and/or speaking for Varian Medical Systems, Monteris Medical, Inc.; Royalty payments for Mako Surgical Corp., Roche (Hoffman-La Roche, Ltd.). M. S. Ahluwalia is consulting and/or speaking for Monteris Medical, Inc.; PrIME Oncology, Inc. S. T. Chao is speaking bureau for Varian Medical Systems (honorarium).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
