Abstract
The aim of this study is to evaluate the effect of dose rate (DR) on lung tissue. The rats included in the study were randomly grouped into 3 groups: Group (G) 1 was defined as control group, and in this group rats were sham irradiated. G2 was the group receiving a single dose of 12 Gy in DR of 300 monitor unit (MU)/min. G3 was the group receiving a single dose of 12 Gy in DR of 600 MU/min. Radiotherapy (RT) was applied under general anesthesia with 6-MV photon beams to both lungs. At the 6th and 16th week of the RT, animals from each group were killed for light and electron microscopy evaluation. We evaluated the scores of each group in the 6th and the 16th week and found that in G2, there were significant increases in the perivascular fibrosis (P = .018), interstitial fibrosis (P = .002), total inflammation (P = .040), and total fibrosis (P = .003) scores. In G3, we found statistically significant increases in perivascular fibrosis (P = .001), interstitial fibrosis (P = .002), and total fibrosis scores (P = .029). There was no significant difference in the total inflammation score in G3 (P = .225). When we compare G2 and G3 in the 6th week, we found significant increase in the interstitial thickening (P = .039) and total inflammation (P = .035) scores in G3. Dose rate per fraction may have an impact on normal tissue toxicity. The prominent effect of increased DR in lung tissue is fibrosis which should be kept in mind, especially in cases where higher doses per fraction are used.
Introduction
The lung is one of the most sensitive organs to ionizing radiation, and damage to normal lung tissue remains a major dose-limiting factor for patients receiving radiation to the thorax. 1,2 Radiation-induced lung injury which is also named as “radiation pneumopathy” is a continuous process and regarded as the result of an abnormal healing response. Subclinical early damage in pneumocytes type I progress to an acute interstitial inflammation at 6 to 12 weeks after the onset of radiotherapy (RT) and further to lung fibrosis after many months and years. Inflammation is an essential part of the normal wound healing process. 1,3 Clinically, radiation-induced lung injury is typically divided into 2 phases: pneumonitis and fibrosis. Fibrosis is a kind of wound healing process and tends to manifest >3 months after treatment. This fibrosis is a form of chronic lung damage that usually evolves over 4 to 24 months after irradiation. 3
In parallel to the developments in the field of radiation oncology, the delivery of higher doses to the target while sparing the normal adjacent structures can be achieved successfully. However, this gives rise to an increase in beam numbers as well as adjustment of the intensities of the beams, which eventually increases the duration of each fraction time. Increase in the fraction time brings the concerns of its radiobiological efficiency. Recently, several in vitro studies pointed out the effect of fraction time on tumor control and emphasized the need for optimizing and decreasing the duration of each fraction. 4 –7 Novel intensity-modulated RT (IMRT) techniques and high-technology treatment machines have the ability to shorten the duration of each fraction by increasing the dose rate (DR). However, the effect of increase in DR on normal tissues with new treatment machines is still unknown.
The aim of this study is to evaluate the effect of increased DR on normal tissues, and lung tissue is selected for this purpose. To the best of our knowledge, this is the first study evaluating the effect of DR in the lung tissue of rats.
Methods and Materials
Study Design
This study included 48 young female Wistar-Albino rats (250-300 g), the use of which was approved by the Hacettepe University Animal Care and Use Committee. Animals were housed 4 per cage in a controlled animal holding room with a 12/12-hour light–dark cycle; temperature and relative humidity were continually monitored to provide standard laboratory conditions. Food and water were provided ad libitum. The rats included in the study were randomly grouped into 3 groups: group (G) 1 was defined as control group and in this group the rats were sham irradiated. G2 was the group receiving a single dose of 12 Gy in DR of 300 monitor unit (MU)/min. G3 was the group receiving a single dose of 12 Gy in DR of 600 MU/min. We determined the sample size according to sample size calculation formula. It was found that 48 rats would be enough to show significant difference. In the beginning of the study, we planned to include 16 rats to each group and kill 8 rats each time (6th and 16th week) from each group. However, there was no intervention in G1, and we knew that the animals in G2 and G3 had risk of death by time due to irradiation. Thus, we asked our statistician (DY) to arrange the groups without decreasing the statistical power of the study. As a result, the rats were distributed to groups as follows: G1: 8 rats, G2: 20 rats, and G3: 20 rats before the beginning of the study. At the 6th and 16th weeks of the RT, 4 animals from G1 and 8 animals from G2 and G3 were planned for killing for morphological examination.
Irradiation Protocol
Radiotherapy was applied under general anesthesia with intraperitoneally administered 90 mg/kg ketamine hydrochloride and 10 mg/kg xyalazine. A single dose of 12 Gy that has been shown to lead lung fibrosis 8 with 6-MV photon beams was applied via a single anterior field to 2 cm depth with source-axis distance technique. Elasto-gel bolus of 1 cm was used to build up the radiation dose on the lungs and to provide contour regularity. The field size was 4 × 4 cm and included both the lungs.
The total MUs were delivered in 8 fragments with 2 minutes of intervals in order to simulate the duration of IMRT treatment fraction. Thus, the total fraction durations in G2 (300 MU) and G3 (600 MU) were 18 minutes and 16 minutes, respectively. 4
Morphologic Studies
At the 6th week, 4 animals from G1 and 8 animals from G2 and G3 were killed for morphological examination. However, due to serial death of animals in G2 and G3 beginning after 9th week of RT, the number of killed animals in G2/G3 was 7/5, respectively. Majority of the deaths occurred after midnight, thus we could not perform postmortem analysis. The animals were anesthetized and killed by cervical dislocation, and both lungs were removed for light and electron microscopy evaluation.
Light Microscopy
The lungs were excised and fixed in 10% formaldehyde solution and embedded in paraffin for light microscopic examination. The slices obtained were stained with hematoxylin and eosin to evaluate the inflammation and with immunohistochemical triple staining to identify the fibrosis in the lung. Extent of the inflammation and congestion was graded on a scale of 0 (normal) to 4 (severe) as described in Table 1. 9 Fibrosis was defined as the thickened alveolar walls with superimposed collagen. As a quantitative end point, extent of the radiation-induced fibrosis was graded on a scale of 0 (normal lung or minimal fibrous thickening) to 4 (total fibrous obliteration of the field) as described in Table 2 by a pathologist blinded to the test groups. 10 After examining the whole sections for each rat in the special group, the mean value ± standard deviation (SD) was taken as the score of the group for each parameter.
The Scoring System For Lung Inflammation and Congestion.

The Scoring System For Lung Fibrosis.

Electron Microscopy
The tissue samples that were taken from the animals 6 and 16 weeks after RT were added into 2.5% glutaraldehyde for 24 hours for primary fixation. Then, these samples were washed with Sorenson phosphate buffer solution (pH: 7.4), and they were fixed in 1% osmium tetroxide. After which, they were washed with the same buffer and dehydrated in increasing concentrations of alcohol series. After dehydration, the tissues were washed with propylene oxide and embedded in epoxy resin embedding media. The semithin and ultrathin sections of the obtained tissue blocks were cut with an ultramicrotome (LKB Nova, Sweden). These semithin sections that were 2 μm in thickness were stained with methylene blue and examined under a light microscope (Nikon, Japan). Following this procedure, trimming was done to the tissue blocks and their ultrathin sections, which were about 60 nm in thickness were taken by the same ultramicrotome. These ultrathin sections were stained with uranyl acetate and lead citrate, and they were examined under Jeol JEM 1200 EX (Jeol, Japan) transmission electron microscope. The electron micrographs of the specimens were taken by the same microscope. This evaluation was done by a physician blinded to the test groups.
Statistical Method
The SPSS v. 13.0 was used for statistical analyses. As the pathological scores were ordinal in nature, the differences in pathological findings between the study groups were analyzed using the Kruskal-Wallis test. When an overall statistically significant difference was observed, pairwise comparisons were performed using the Mann-Whitney U test. Bonferroni correction was used for multiple comparisons. A 5% type-I error level was used for the statistical significance cutoff for overall comparisons.
Results
Light Microscopy Findings
The pathological scores of each group in the 6th and the 16th week are shown in Table 3. There was no significant difference in the control group (G1) in terms of inflammation and fibrosis due to aging of the animals. However, in G2, there were significant increases in the perivascular fibrosis (P = .018), interstitial fibrosis (P = .002), total inflammation (P = .040), and total fibrosis (P = .003) scores as time passed (Figure 1). In G3, similarly, we found statistically significant increases in perivascular fibrosis (P = .001), interstitial fibrosis (P = .002), and total fibrosis scores (P = .029). There was no significant difference in the total inflammation score in G3 (P = .225; Figure 2).

Hematoxylin and eosin (H&E) staining (×200) sections from the lungs of rats from group 2 in the 6th week (A) and 16th week (B). The scores for interstitial inflammation/interstitial thickening/perivascular fibrosis/interstitial fibrosis were 1/1/0/0 in (A) and 2/3/1/2 in (B), respectively.
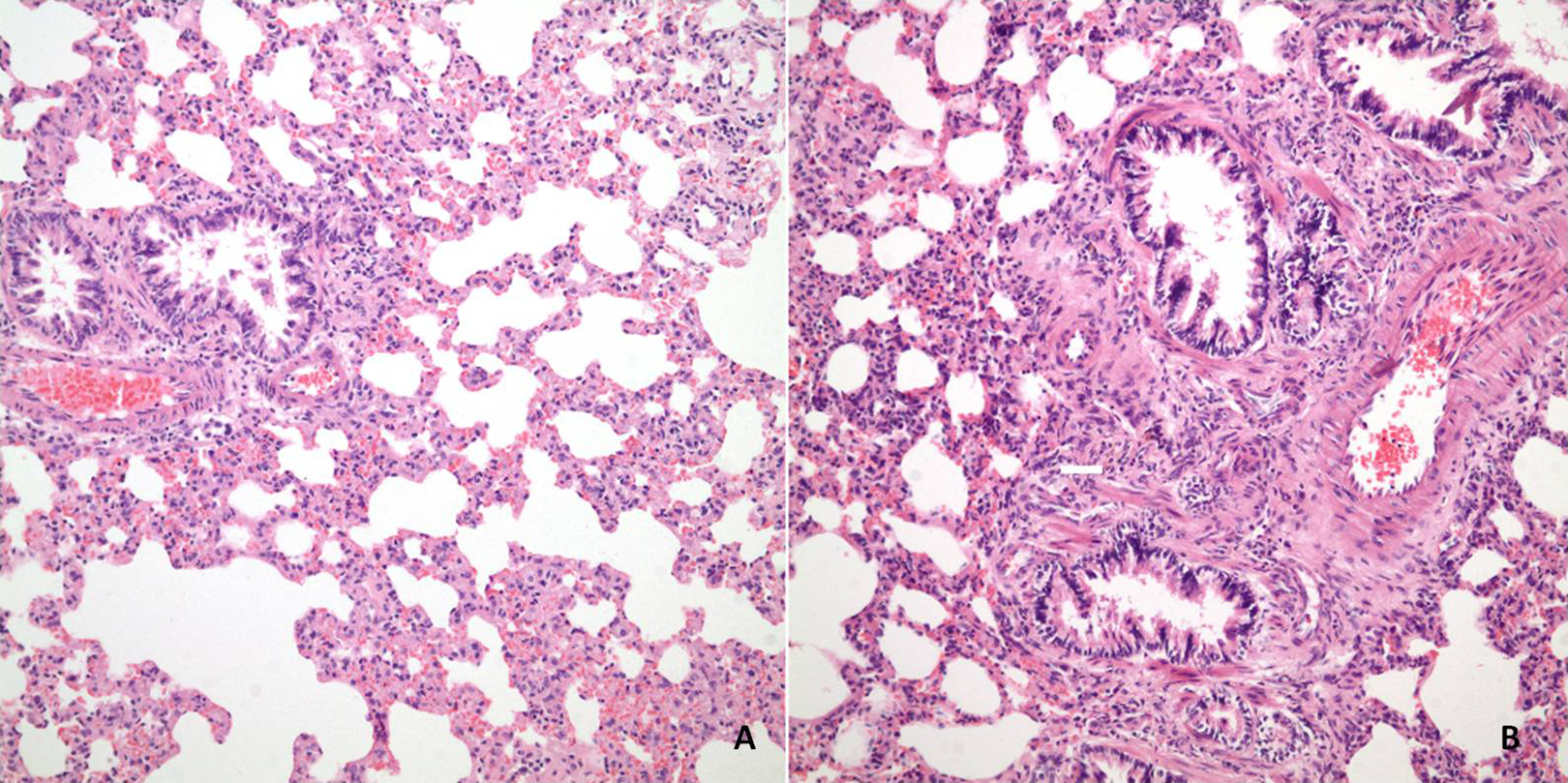
Hematoxylin and eosin (H&E) staining section from the lungs of rats from group 3 in the 6th week, ×100 (A) and 16th week, ×200 (B). The scores for interstitial inflammation/interstitial thickening/perivascular fibrosis/interstitial fibrosis were 2/2/1/1 in (A) and 3/3/3/3 in (B), respectively.
Comparison of Pathological Results For Each Group in the 6th and the 16th Week.
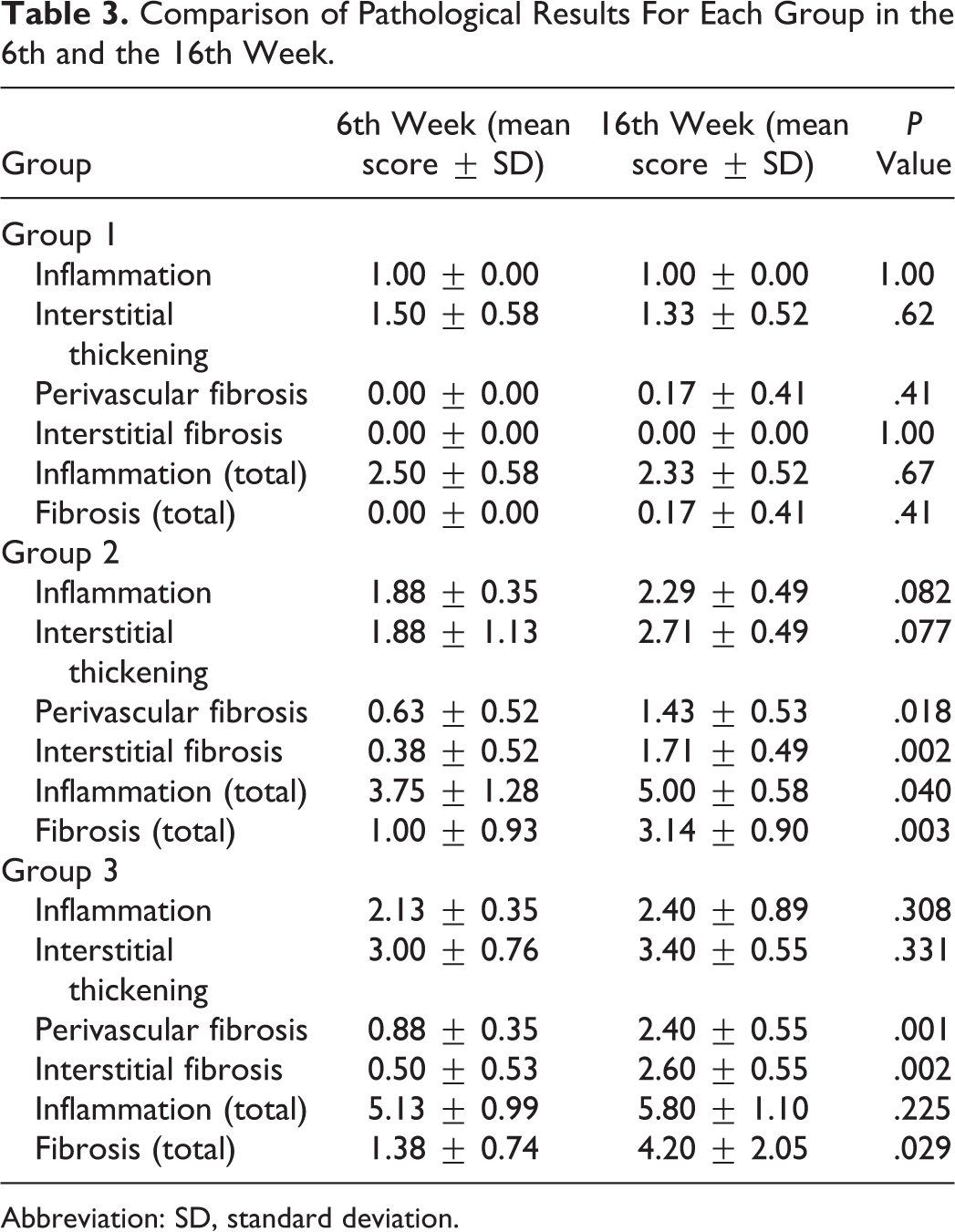
Abbreviation: SD, standard deviation.
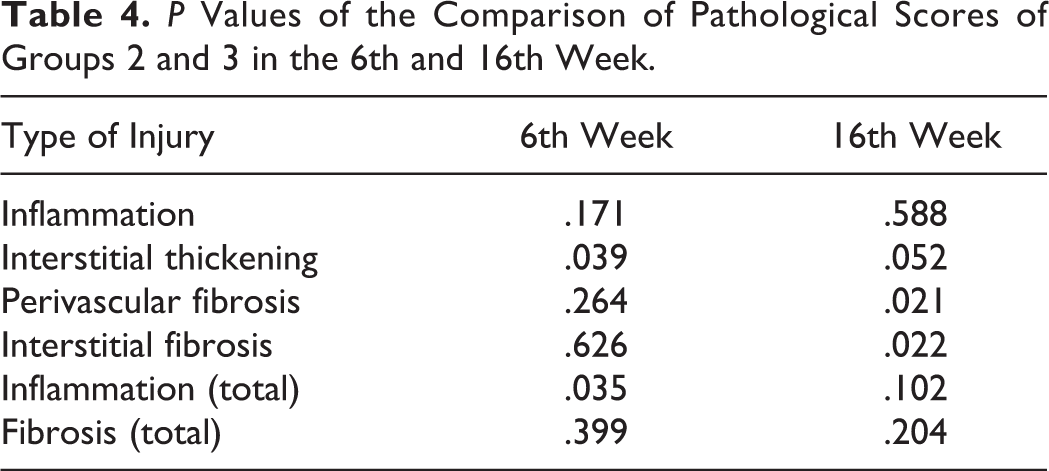
When we compared G2 and G3 in the sixth week, we found significant increase in the interstitial thickening (P = .039) and inflammation (P = .035) scores in G3. The 16th week evaluation demonstrated more prominent perivascular fibrosis (P = .021) and interstitial fibrosis (P = .022) in G3 which is statistically significant. However, inflammation scores did not show any significant difference at 16th week between the groups (Table 4).
P Values of the Comparison of Pathological Scores of Groups 2 and 3 in the 6th and 16th Week.
Electron Microscopy Findings
The sixth week transmission electron microscopic examination of the lung tissue revealed large vacuoles were observed in the alveolar epithelial cells in G2 and G3. Alveolocapillary membrane and capillary endothelium were normal ultrastructurally. Additionally in G3, there was mild degree of fibrosis (Figure 3A and B). Also in G3, there were vacuoles in the bronchiole epithelial cells, and intercellular edema was seen in between the bronchiole epithelial cells.

A, Electron micrograph showing large vacuoles (v) in alveolar epithelial cell in the 6th week examination of group 2. B, Electron micrograph showing large vacuoles (v) in alveolar epithelial cell and mild degree of fibrosis (f) in the 6th week examination of group 3. n indicates nucleus of alveolar epithelial cell; e, erythrocyte (original magnification, ×6000).
In the 16th week examination, G2 has ultrastructural findings similar to the 6th week. However, these vacuoles were more and larger in this week (Figure 4). In G3, on the other hand, the fibrosis was more prominent than in the 6th week. The vacuoles in alveolar epithelial cells were larger, and there were more huge vacuoles in this group when compared with the sixth week findings of the same group. The vacuoles in the bronchiole epithelial cells were larger and more prominent. Intercellular edema in between the bronchiole epithelial cells was more severe. Additionally, in this group, vacuoles were observed in the cytoplasm of capillary endothelial cells. However, alveolocapillary membranes were normal ultrastructurally (Figure 5A and B). The findings are summarized in Table 5. The amount of fibrosis observed in the study groups is scored in Table 6.

Electron micrograph showing larger vacuoles (v) inside the cytoplasm of type II alveolar epithelial cell in the 16th week examination of group 2. n indicates nucleus of alveolar epithelial cell; lb, lamellar body (original magnification, ×6000).

A, Electron micrograph showing larger vacuoles (v) inside the cytoplasm of alveolar epithelial cell and vacuoles (vv) inside the cytoplasm of capillary endothelial cell in the 16th week examination of Group 3. B, Electron micrograph showing fibrosis (f), type II lung cells (*) and nucleus of a type II lung cell (n) in the 16th week examination of group 3 (original magnification, ×6000).
The Summary of the electron microscopic examination of the lung tissue.

The amount of Fibrosis Observed in the Study Groups Scored in 4 Samples Using the Fibrosis Scoring.

Abbreviations: 0, no fibrosis; 1, mild fibrosis; 2, moderate fibrosis; 3, prominent fibrosis.
Discussion
The implementation of new techniques in RT assures the delivery of high doses to the target while sparing the normal adjacent structures. However, it is known that increased number of beams and adjustment of the intensities of the beams prolong the duration of each treatment fraction. The IMRT technique is one of these radiation techniques that improve the target dose conformity and normal tissue sparing. However, it is well known that the duration of 1 fraction of treatment also increases with the complexity of the treatment. A treatment fraction time in conventional RT is about 2 to 5 minutes compared to 15 to 45 minutes with IMRT. 5 Therefore, the prolongation of the treatment time may affect both the treatment outcome and radiation-related side effects in the healthy structures within or nearby the treatment field. On the other hand, high-technology treatment machines have the ability to shorten the duration of each fraction by increasing the DR of the treatment. The DR effect in brachytherapy is a well-known issue in radiobiology. Unfortunately, the long-term effect of using higher DR in external RT is still unknown. Our study was conducted to elucidate the effect of 2 different DRs on rats’ lung tissues. We delivered 12-Gy radiation dose in a single fraction to thoracic region of the rats with 2 different DRs. Our results give us an impression that increasing the DR from 300 MU/min to 600 MU/min leads to more prominent and earlier fibrotic response. The inflammation response on the other hand showed no difference between the groups.
Wang et al 5 explored the impact of prolonged fraction delivery time on tumor control. In that particular study, they have investigated the impact of fraction delivery time on equivalent uniform dose (EUD) and tumor control probability (TCP) using the linear-quadratic (LQ) formalism. A series of simulated and clinical IMRT plans with different fraction delivery times were evaluated in terms of EUD and TCP using the LQ parameters derived from compiled clinical data for prostate cancer. Their calculations indicated that fraction delivery times in the range of 15 to 45 minutes may significantly decrease cell killing. They concluded that total time to deliver a single fraction affects the outcome for tumors with low α–β ratio and short repair half-time and suggested increasing the prescription dose to compensate for the reduction in cell killing due to increased sublethal damage repair for treatments with delivery time more than 10 to 15 minutes.
Mu et al used Chinese hamster fibroblasts (V79-379-A) to study the effect of delivery time for each fraction. They concluded that prolonging the fraction time will spare tissues with fast DNA repair-like tumors. 4 This study and another study by Fowler et al 6 supported the idea of loss of biological effectiveness for fraction delivery times more than 15 to 30 minutes. Our study is different from the studies exploring DR effect of single fraction in tumoral tissue. In this particular study, we tried to evaluate the DR effect of single fraction in normal tissues like lung tissue. In order to simulate the IMRT fraction delivery time, we delivered the radiation in subfractions, thus the duration of each fraction was 18 and 16 minutes, respectively. 4 We found that when we increased DR, the response of injury such as fibrosis became more prominent and appeared earlier.
The DR studies in the literature were performed mainly on cell cultures using some biological models and recommended keeping the duration of fraction duration as short as possible in order not to lose the radiobiological effect on tumors. 5 –7 This can be accomplished in the clinic by adjusting the DRs in the new treatment machines capable of IMRT. Increasing the DR twice will half the duration of each fraction. However, the increase in DR brings the concerns about the biological effectiveness on normal tissues. As far as we know, there is no experimental or clinical data, searching for the efficacy of DR per fraction in terms of late tissue toxicity in external beam RT. This is the first experimental in vivo study regarding the possible toxic effect of increasing DR per fraction in lung tissue.
The alveolar epithelium consists of type I and type II epithelial cells. Type I cells cover approximately 90% of the alveolar surface and type II cells are the precursors of type I cells. In the steady state, the turnover time of the alveolar epithelium is approximately 4 to 5 weeks. However, after toxic injury, type I epithelial cells are denuded and the proliferation of type II cells is stimulated up to 10-fold. 4 Stimulation of type II cells promotes the secretion of growth factors and proteases and degradation of the extracellular matrix to allow removal of dead cells by normal process. 3 Pulmonary irradiation also reduces microvessel density and lung perfusion and promotes hypoxia. 5 All of these injuries stimulate the recruitment of a variety of inflammatory cells to the site of the injury, and it leads to establishment of chronic inflammation and fibrosis. In this phase, vascular damage and collagen deposition become apparent. Vascular injury and activation of coagulation cascade, cellular adhesion molecules, proinflammatory and profibrotic cytokines, and oxidative stress seem to play a vital role in the development of radiation-induced fibrosis. Clinically, radiation-induced lung injury is typically divided into 2 phases: pneumonitis and fibrosis. Subclinical early damage in pneumocytes type I progress to an acute interstitial inflammation at 6 to 12 weeks after the onset of RT. Radiation-induced lung fibrosis on the other hand is a form of chronic lung damage that usually evolves over 4 to 24 months after irradiation. 3 In this study, we sacrificed the rats at 6th and 16th weeks of RT in order to determine both the acute and the chronic phases of the lung injury. Our study shows us that the acute inflammatory effect of irradiation on lung tissue does not differ much according to DR. The fibrotic response after irradiation on the other hand was found to be closely related to DR.
The irradiation doses used in routine clinical practice are different from the ones used in animal studies. Most patients in routine practice are treated with conventional fractionation to a total dose of 50 to 70 Gy. However, in recent years, stereotactic radiosurgery and intraoperative RT that use single or 2 to 5 fractions of high-dose irradiation have become popular. It has been postulated that the LQ model is an appropriate methodology for determining isoeffective doses at large dose per fraction. 11 According to the LQ model, the 12-Gy single dose of RT, as used in our study, corresponds to 30 Gy, in the fractions, when α–β ratio of 3 to 4 is used.
In conclusion, our study showed that the DR per fraction can have an impact on normal tissue toxicity. The prominent effect of increased DR on fibrotic response may worsen the therapeutic ratio in clinical practice and should be kept in mind, especially in cases where higher doses per fraction are used.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
