Abstract
Introduction
Cervical cancer was the fourth leading cause of cancer-related morbidity and mortality among women worldwide in 2020.1,2 Currently, it is considered an entirely preventable cancer due to the efficacy of human papillomavirus (HPV) vaccines and screening measures. However, there is still a glaring gap between the underlying incidence rate and social resources required. In countries with low or medium Human Development Index (HDI), cervical cancer remains the leading cause of cancer incidence and mortality in women. 1 The nonmetastatic advanced stage is associated with very high mortality; almost half of these cancers are diagnosed at a nonmetastatic advanced stage with an overall 5-year survival of approximately 65%. 3 With the aging of the population, elderly patients account for the majority of oncology patients, and it is estimated that patients over 65 years of age will account for 70% of newly diagnosed cancer cases. 4 Cervical cancer is traditionally diagnosed in younger women, but it has a second incidence peak between 60 and 70 years of age. 5 In China, 23.8% of cervical cancer patients are older than 60 years, and the incidence in elderly patients is still increasing. 6
Older patients are often excluded from clinical trials because of their poor compliance and comorbidities, making clinical data difficult to obtain. According to a resource-stratified clinical practice published by the National Comprehensive Cancer Network (NCCN), scaled-up treatments, including surgery, chemotherapy, radiotherapy (RT), and targeted therapy, are beneficial for improving patients’ survival .
7
However, physical fragility in elderly women results in more frequent posttreatment complications which worsen the peri-treatment mortality. An American study showed that among 8200 patients who underwent open hysterectomy between 1998 and 2010, postoperative complications systematically increased with age.
8
Elderly patients also differ in their tolerance to various treatments. Tolerance to chemotherapy is a specific concern in elderly women due to hypoalbuminemia, degeneration of renal function, and improved redistribution of body mass.
9
The side effects of RT may exacerbate preexisting problems. In the Gynecologic Oncological Group (GOG) clinical trials, radiation protocol deviations and failure to complete brachytherapy (BT) increased with age (
Many previous studies have revealed a correlation between clinical characteristics and overall survival (OS) or disease-specific survival (DSS) of patients with cervical cancer. These characteristics include age, pathology, tumor microenvironment, and therapeutic methods.11–16 However, most of these studies had a small sample size, were single-centered, and included patients of all ages. Moreover, there is currently no prognostic model for elderly patients with nonmetastatic cervical cancer based on the Surveillance, Epidemiology, and End Results (SEER) database.17–20 Most studies only focused on mortality as a single endpoint, and traditional analytical methods tend to exaggerate the association between survival outcomes and risk factors. Consequently, the prognostic factors for elderly patients with cervical cancer should be confirmed using larger cohorts and more advanced models. The SEER database contains the data for a great number of patients and is a widely used database. 21 For patients with cervical cancer, a nomogram to predict the survival of elderly patients has not been reported, no previous studies have noted the effect of competitive risk events on the probability of patients’ survival. 22 This study aimed to construct and validate a competitive risk model (CRM) nomogram to predict survival time for patients over 65 years of age diagnosed with nonmetastatic cervical cancer.
Materials and Methods
Data Source
This study retrospectively analyzed data extracted from the SEER database using the SEER*Stat software (version 8.3.6). The SEER program gathered the clinical data of approximately 34.8% of the US population from 18 registries covering all regions of the country (http://www.seer.cancer.gov) and is publicly available. SEER data are population-based, with a good representation of racial/ethnic minorities and foreign-born individuals. The data include sociodemographic characteristics (age, race, education, and marital status), geographic information (urban/rural designation), cancer-specific factors (site, stage, and lymph node status), pathologic variables (margin status, perineural invasion), and treatment (surgery, radiation, and order of treatment). Women aged over 65 years with histologically confirmed primary cervical cancer (ICD-O-3, C53.9) and positive follow-up records were assessed for eligibility. All included cases were diagnosed between 2010 and 2015, since the SEER database began to collect information about molecular subtypes and distant metastasis sites in 2010, and the follow-up records were last updated on December 31, 2015. The population information in this study is collected from SEER database and involves no identifying information throughout the analyses. The study was exempted from approval by the Institutional Review Board at Air Force Medical University because the study was deemed not to constitute human subject research.
Patient Enrollment
A total of 95,234 patients diagnosed with cervical cancer were identified in the US SEER population between 2010 and 2015. Of these, 16 cases were excluded because the primary sites were not in the cervix. Patients with ambiguous staging such as blank, not available, or unknown stage (n = 76,368), those with unavailable surgery (n = 31), or RT (n = 8224) records, those who are < 65 years old (n = 8298), and those without definite survival months (n = 23) were excluded. Patients with distant metastasis (n = 418) were also excluded. Finally, 1856 cases from the SEER database were included in the analysis cohort. The included data were divided into “Training” and “Validation” sets at a ratio of 5:5 for subsequent analysis, as shown in Figure 1.

Flowchart of case inclusion and exclusion.
Variables Definition
We manually selected variables of interest from the SEER database, including age, race, year of diagnosis, primary site, treatment(s) received, marital status, histology, number of in situ malignant tumors, tumor stage (refer to the International Federation of Gynaecologists and Obstetricians/FIGO, American Joint Committee on Cancer (AJCC) seventh edition staging system), number of primary tumors, and subsequent malignant lesions (sequence number). The primary sites were classified as cervix uteri and others. Nonmetastatic patients were defined as patients with M0 stage in the TNM classification system. The following 2 criteria were used to exclude patients: blanks, not available, or unknown stage and M1 patients in the TNM staging variable. Then we screened the data using “dplyr_1.0.7” package of R software (version 3.6.1, http://www.r-project.org). The age variable was classified into groups as follows: 65 to 70, 71 to 75, 76 to 80, and 81 to 98 years according to quartile and median values (taking an integer when a boundary value was encountered). The race was converted into 3 categories: White, Black, and Others. Diagnosis represents the confirmation time of the diagnosis. The treatments were mainly surgery (Yes, No), RT (Yes, No), and chemotherapy (Yes, No). Marital status was categorized into single/unmarried, married, divorced/separated, and widowed/others. Histology was categorized as adenocarcinoma (ADC), squamous cell carcinoma (SCC), and others. The number of in situ malignancies was classified as n = 1 or n > 1. The FIGO stage was reported as I, II, III, or IV. The primary outcomes were OS and DSS. Competitive events were defined as deaths not caused by cervical cancer.
Statistical Analyses
The dataset was bisected into the training and validation sets. Continuous variables are described as median and interquartile range (IQR) and compared by group using Student's
Results
Comparison of Baseline Between Training and Validation sets
A total of 1856 patients diagnosed between 2010 and 2015 were eligible and enrolled in the cohort (Figure 1). Then, patients were randomly assigned to the training and validation sets at a 5:5 ratio. Table 1 shows the patients’ demographic and clinical characteristics. The median (IQR) survival time of all enrolled patients was 24 (13-46) months, and the median survival times for patients in the training and validation sets were 24 (13-48) and 24 (13-44) months, respectively. A total of 833 patients (45%) died during the follow-up period, and among these, 497 deaths (27%) were due to cervical cancer. All patients were over 65 years old, and the median age was 73 (68-79) years. Most of the patients were White (72%), aged 65 to 70 years old (41%), and widowed/other (41%). The primary site was the cervix uteri (ICD-O-3, C53.9) in 1516 (82%) patients, and 394 (21%) patients had multiple in situ malignant tumors. The proportion of patients who underwent surgery, RT, and chemotherapy was 29%, 98%, and 71%, respectively. The histological findings included SCC (n = 311, 74%), ADC (n = 1376, 17%), and others (n = 169, 9%). The FIGO stages were I (n = 475, 26%), II (n = 565, 30%), III (n = 731, 39%), and IVa (n = 85, 5%). In addition, there were no significant differences in clinical characteristics between the training and validation sets, indicating that the patients were adequately randomized (
Demographic and Clinical Characteristics of Patients with Primary Cervical Cancer.

Note: Diagnosis: Year at diagnosis; Primary site: The site of the tumor originated from; Number: In situ malignancy number.
Abbreviations: ADC, adenocarcinoma; FIGO, the International Federation of Gynaecologists and Obstetricians; IQR: interquartile range; SCC, squamous cell carninoma.
Survival Analysis of Training set
Kaplan–Meier analysis and log-rank test were performed to explore the influence of all demographic and clinical variables on OS and DSS. The results showed that younger patients with higher FIGO stage, receiving chemotherapy, RT, and surgery had an improved OS and DSS (all log-rank

Effects of demographic and clinical variables on the OS and DSS of elderly patients diagnosed with cervical cancer using Kaplan–Meier method and log-rank test in training set.
Univariate and Multivariate Analyses in Training set
Univariate and multivariate Cox PH analyses were conducted to screen for independent prognostic factors associated with OS and DSS, as shown in Table 2. Univariate analysis showed that the absence of any treatment (surgery, RT, or chemotherapy), FIGO stages III and IVa, and aged 76 to 80 and 81 to 98 years were significantly associated with diminished OS and DSS. Thereinto, marital status of widowed/others (vs married, OS: hazard ratio [HR] = 1.334, 95% confidence interval [CI] = 1.057-1.684,
Univariate and Multivariate Analysis of Demographic and Clinical Factors in Patients with Primary Cervical Cancer in Training set.
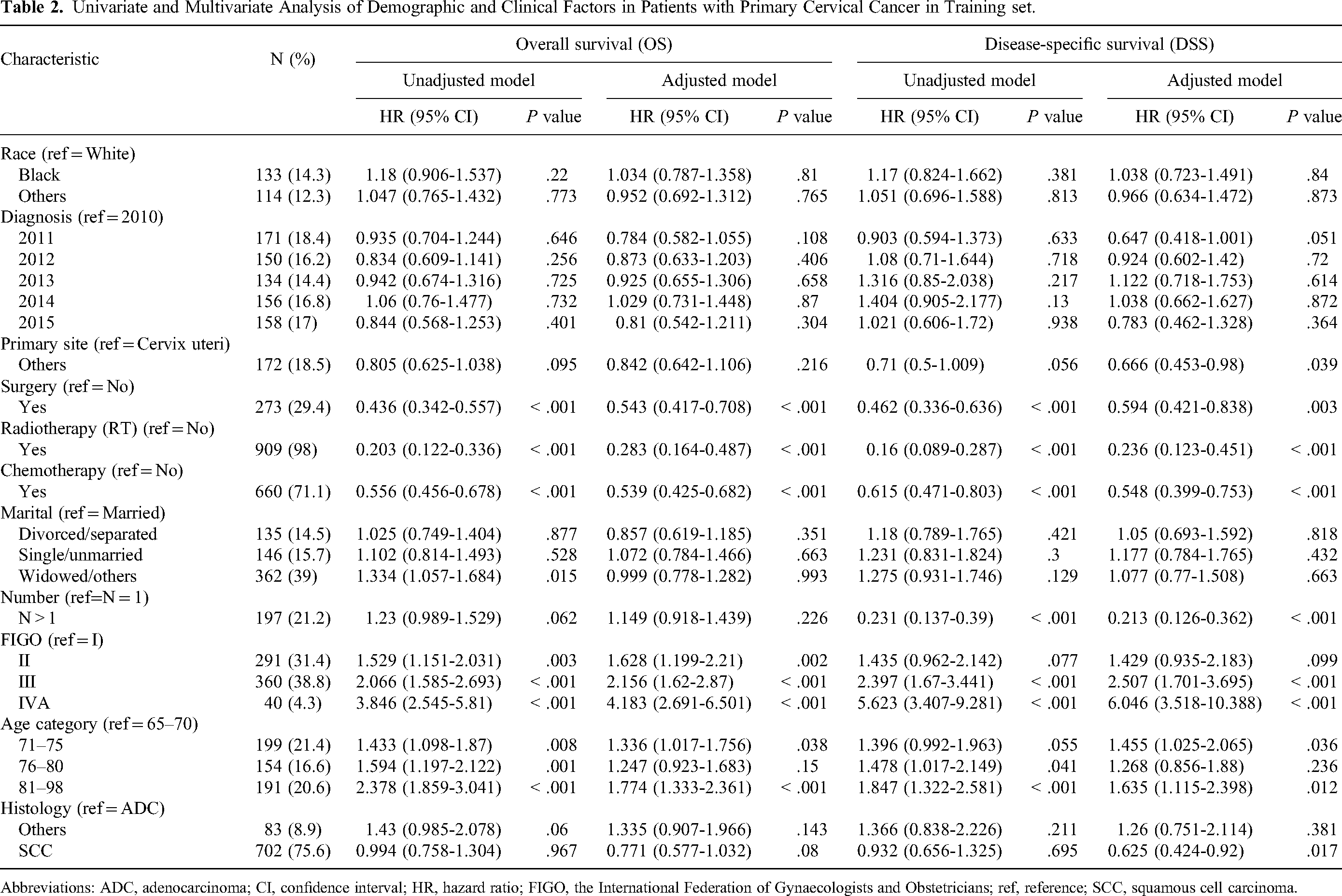
Abbreviations: ADC, adenocarcinoma; CI, confidence interval; HR, hazard ratio; FIGO, the International Federation of Gynaecologists and Obstetricians; ref, reference; SCC, squamous cell carcinoma.
All factors were included in the multivariate analysis. Results suggested that receiving surgery (vs no surgery, OS: HR = 0.436, 95% CI = 0.342-0.557,
Competing Risk Analysis of Demographic and Clinical Factors in Training set
In the training set, 238 (26%) patients died of cervical cancer and 181 (20%) died of other causes out of total 928 patients. The CIF for death from cervical cancer suggested that age, higher FIGO stage, multiple in situ malignancies, and having had no surgery, RT, or chemotherapy significantly increased risk of death (
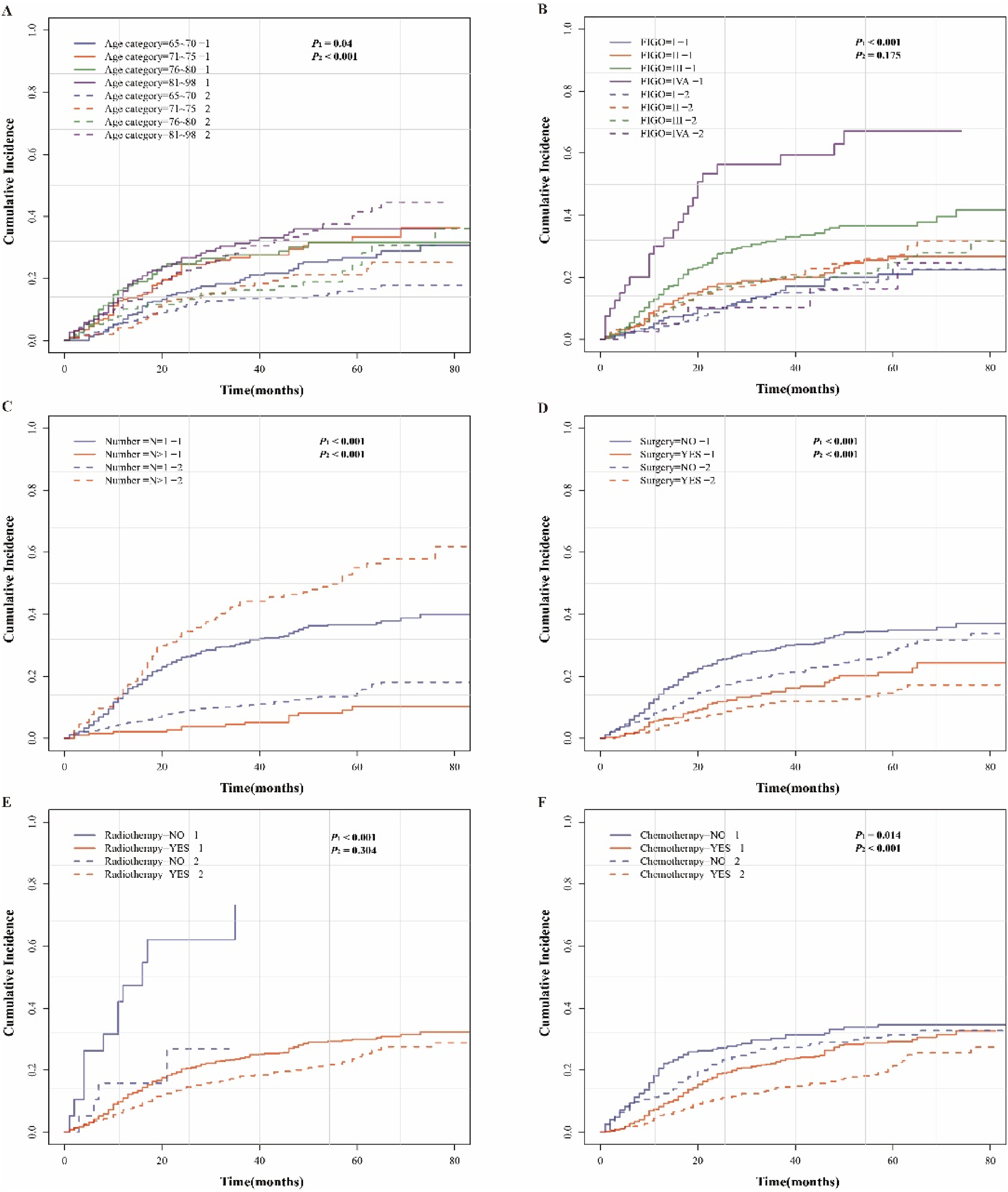
Evaluation of cumulative incidence rate for death of prognostic factors in elderly patients with cervical cancer.
Fine and Gray's analysis was applied for univariate and multivariate analyses of DSS in the CRM, and subdistribution hazard ratios (sHRs) were estimated. Univariate analysis showed consistent results with the Cox model; age, number of in situ malignancies, FIGO stage, surgery, RT, and chemotherapy significantly influenced the prognosis of patients with cervical cancer. The results of multivariate analysis suggested that age was still an independent risk factor, and 71 to 75 years old (sHR = 1.447, 95% CI = 1.019-2.053,

Forest plot illustrating the univariate and multivariate competitive risk models (CRMs) for predicting disease-specific survival (DSS) in the training set (n = 928) of elderly patients with cervical cancer.
Competing Risk Model Nomogram Construction and Evaluation
According to the results of the Fine-Gray analysis, primary site, histology, age, FIGO stage, in situ malignancy number, chemotherapy, RT, and surgery were integrated into the CRM nomogram to predict 1-year, 3-year, and 5-year cumulative survival probabilities (Figure 5A). Each included variable corresponded to a score at the “points” axis. Thereafter, we could get the overall score according to every patient's characteristics, then the predictive cumulative probability of cervical cancer specific survival at 1-year, 3-year, and 5-year could be observed using the bottom axis. All the calibration curves were close to the standard curve, which demonstrated favorable concordance; however, consistency deviated later because the actual survival rates of patients were too low. For example, the actual 1-year survival rate of the included cases was only 25% (Figure 5B and C). The C-indexes of the nomogram in All model and the BIC model were 0.641, 0.647 and Brier scores were 0.094, 0.903 at the 1-year cut-off in the training set, respectively. All C-indices and Brier scores are shown in Supplementary Table S1. The time-AUCs of the CRM nomogram at the 1-year, 3-year, and 5-year intervals in the training set were 77.6%, 77.3%, and 74.5% (Figure 5D), respectively, and the time-AUC of BIC model at the 1-year, 3-year, and 5-year intervals were 77.4%, 77%, and 74.2% (Figure 5E), respectively. These results show that CRM had a better predictive power than the Cox regression model. Moreover, the DCA suggested that the CRM nomogram had good net benefits at different survival outcomes, and the results of All model and the BIC model were consistent, indicating the actual compatibility for clinical decision making (Figure 5F-H). The nomogram was also internally validated (Supplemental Figure 1).
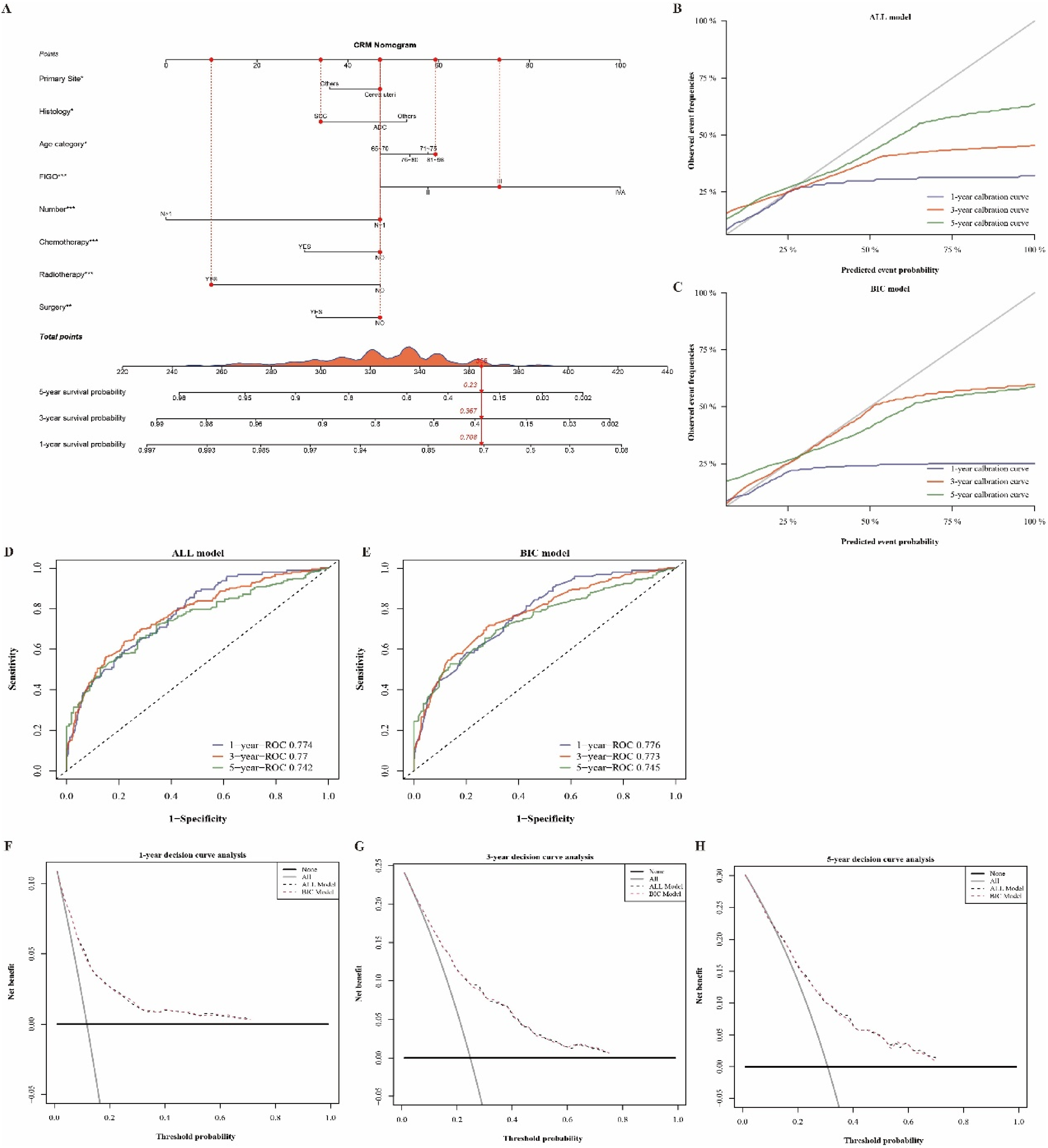
Construction and evaluation of the CRM nomogram for predicting the probability of 1-year, 3-year, and 5-year DSS in elderly patients with cervical cancer in the training set. (A) CRM nomogram for DSS; (B) Calibration curve of CRM nomogram in All model; (C) Calibration curve of CRM nomogram in BIC model; (D) ROC curve of CRM nomogram in All model; (E) ROC curve of CRM nomogram in BIC model; (F) DCA for 1-year DSS; (G) DCA for 3-year DSS; and (H) DCA for 5-year DSS.
The median value was used as the cut-off point recognized by the CRM, and patients in the training set were divided into low-risk and high-risk groups according to the median value. Subgroup analysis demonstrated that the cumulative incidence of cervical cancer-specific mortality was higher in the high-risk group, while other causes of mortality was lower (Figure 6,
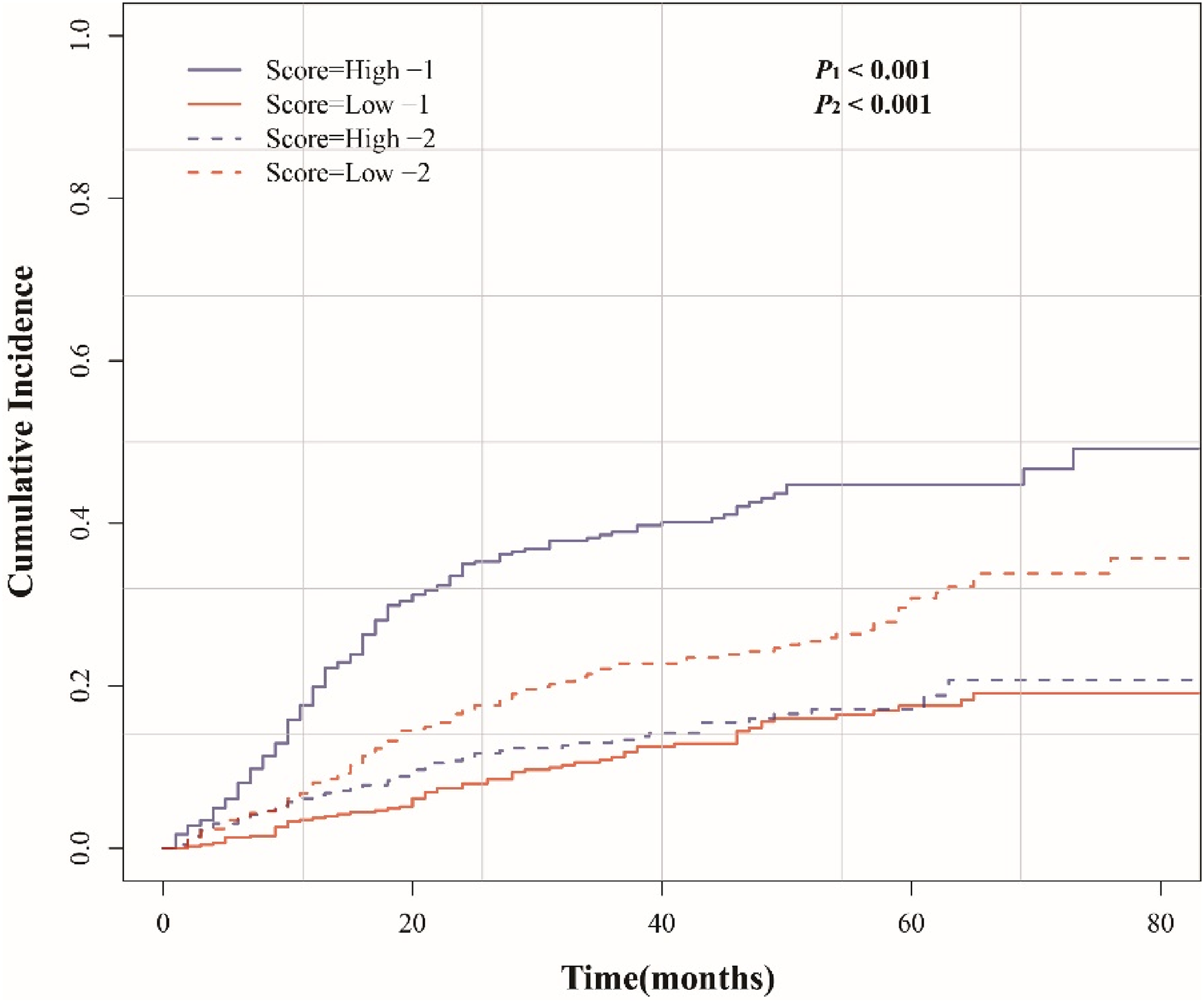
Cumulative incidence of death from cervical cancer and other causes in the low- and high-risk groups of elderly patients with cervical cancer in the training set.
Discussion
Cervical cancer has a high incidence and mortality rate, especially in developing countries. 2 As the population ages, the number of elderly patients is increasing, and nearly 25% of cervical cancer patients are over 65 years old, with a 5-year survival rate of 40.8%.23,24 The prognosis and curative effect in elderly patients are worse than those in young patients because elderly patients might experience severe toxicity and have reduced compliance. Therefore, the prediction of prognosis-related risk factors in elderly patients diagnosed with cervical cancer is particularly important in today's emphasis on precise personalized treatment.
Traditional survival analyses, such as the Kaplan–Meier marginal regression and the Cox PH model, usually focus on events of interest; however, the probability of a targeted event is closely related to competitive events. Therefore, it is easy to defect the accuracy of the prediction, and bias may arise when using classical methods. A competitive risk regression model is more favorable for identifying the impact of competitive events and improving predictions.
In this study, we used a competitive risk regression model to construct a nomogram to identify the risk factors that are important for predicting prognosis in elderly patients with nonmetastatic cervical cancer. In our study, the median age of the included patients was 73 years, 79% of the patients had only 1 in situ tumor, and 41% of the patients were windowed/others. Moreover, 85% of patients’ primary sites were cervix uteri, and 74% of histology types were SCC. Patients who underwent surgery, chemotherapy, and RT had better outcomes; however, RT (98%) and chemotherapy (71%) were the main treatments for elderly patients, and only 29% of patients underwent surgery. We speculated that age and physical conditions are critical in determining whether a patient is eligible for surgery. Primary site, histology, age, FIGO stage, number of in situ malignancies, chemotherapy, RT, and surgery were identified as independent prognostic factors and aggregated into the CRM nomogram to predict 1-year, 3-year, and 5-year cumulative survival probabilities. The CRM results were more accurate than those of the Cox proportional regression analysis, and the bias was less pronounced. The results of the C-index, Brier score, time-AUC, DCA, and calibration curve suggested that the CRM nomogram had a better predictive power.
In previous studies, 25 Black race and unmarried status were associated with elevated death risks; however, there were no significant differences in our analyses. Race and marital status had no effect on the survival of elderly patients with cervical cancer in both the Fine-Gray and Cox models. Racial differences are closely related to access to health care and medical screening efforts, especially when it comes to preventable diseases such as cervical cancer. We hypothesized that the differences in access to medical care might be diminished with age due to mobility. Racial disparities caused relatively low awareness of seeking medical support; the race in the SEER database was predominantly Caucasian (73%). Decreased sexual activity due to aging or being divorced/widowed leads to less prominent symptoms, such as contact bleeding. Marital status in most previous studies was categorized into married and unmarried; we divided it into married, divorced/separated, single/unmarried, and widowed/others, so a more detailed classification might impair the differences between groups (Figure 4). Therefore, more evidence is needed to confirm the impact of racial and marital status in terms of prognosis and to resolve the controversy.
Elderly people are at an elevated risk of developing cancer, accounting for nearly 70% of all cancer deaths.
26
Compared with young and middle-aged cancer patients, elderly patients have other contributing factors, such as aging of various organ functions, hidden onset, and multiple comorbidities. Hypertension and diabetes were both more likely to be reported in the medical histories of women over 65 years of age (
RT is an integral part of the treatment of cervical cancer, either for patients who are at risk of recurrence after surgery or as an initial curative treatment. Chemoradiation is recommended for stages IB3-IVA, and RT or chemoradiation is conditionally recommended for stages IA1 to IB2 if medically inoperable. 29 Many studies have demonstrated that RTis necessary to treat cervical cancer and is beneficial for prognosis in both the young and elderly. 30 Our results showed that 98% of the included patients received RT, which appeared to be a protective factor for elderly patients both in the Fine-Gray and Cox models. However, a recent study revealed that RT has an adverse effect on the prognosis of patients diagnosed with ovarian cancer. 31 We speculated that RT was more commonly administered to ovarian cancer patients with nonepithelial type, distant metastases, and AJCC stages III/IV, so the poor prognosis may be attributed to a more malignant phenotype in the RT group. Thus, further subgroup and stratification analyses are required in any study. A SEER-based study showed that RT leads to a reduced OS in patients with cervical SCC. 32 In comparison, our study included patients with SCC, ADC, and other histological types; therefore, a larger sample size and including several histological types may be behind this disparity. In addition, the long-term adverse side effects of RT did not emerge owing to the short lifespan of elderly patients. Chemotherapy for cervical cancer, especially adjuvant chemotherapy before and after surgery, has received increasing attention. However, in the NCCN guidelines, it is mainly recommended for the treatment of advanced and recurrent metastatic cervical cancer or in combination with RT. According to relevant statistics, the proportion of American cervical cancer patients undergoing concurrent RT and chemotherapy after surgery is relatively stable (41.8%); however, the proportion of patients receiving RT alone (23.6%) decreased, while the proportion of patients receiving chemotherapy (24.8%) increased. 33 In this study, 71% of the included patients received chemotherapy, and it was an independent prognostic factor that could reduce the mortality rate in the Cox and Fine-Gray models, which is consistent with previous studies. Surgery is mainly performed for early stage (Ia-IIa) patients. Indeed, most elderly patients cannot undergo radical surgery due to physical fragility. We found that only 29% of patients aged > 65 years with nonmetastatic cervical cancer underwent surgery. This indicates that age and associated complications limit the possibility of surgery. The Cox and Fine-Gray models suggested that surgery could reduce the risk of death. Nevertheless, RT was superior to surgery and chemotherapy as a protective factor in elderly patients.
Histological diagnosis was an independent prognostic factor in the present study. According to a study on cervical cancer patients aged < 45 years, ADC had better prognosis than SCC. In addition, other histological types, besides SCC and ADC, lead to a reduced OS and DSS.
30
On the other hand, other studies have shown that patients with cervical ADC or adenosquamous cell carcinoma have a worse prognosis than those with SCC.
34
In this study, 74% (1376) of patients had SCC, 17% (311) had ADC, and 9% (169) had other histological types, and the multivariate analysis results showed that SCC was a protective factor compared with ADC (adjusted HR = 0.625, CI = 0.424-0.92,
In this study, the Cox model suggested that FIGO stages II, III, and IVA were risk factors for reduced OS in elderly patients with cervical cancer. However, there was no statistical significance for the impact of FIGO stage II on DSS in the Cox model, and the Fine-Gray model revealed consistent results for DSS. Remarkably, the Cox model results underestimated the risks for all FIGO stages, and the CRM conclusions were still more valid. Moreover, the number of malignant lesions can reflect the severity of cervical cancer to a certain extent. Previous studies have revealed that the depth of invasion is associated with worse prognosis in patients with cervical cancer,35,36 however, very few studies have reported a correlation between the number of in situ lesions and prognosis. We explored the relationship between the number of in situ malignant lesions and prognosis of patients, and the Cox model showed that multiple malignancies may lead to better prognosis compared to 1 in situ tumor when it comes to DSS. The results were more accurate in the CRM. In conclusion, our results suggest that it is not the case that patients with multiple tumor lesions have a worse prognosis, but the depth and site of tumor invasion. We inferred that the number of tumor lesions accounted less weight on prognosis and other risk factors, such as age, FIGO stage, and histology, had a greater effect on prognosis.
Precision medicine involves individualized prevention, diagnosis, and treatment. Thus, it needs to rely on predictive models that are well calibrated. The CRM nomogram is optimized for usability, acceptability and interpretability. To our knowledge, this is the first SEER database linked study to construct a CRM nomogram for predicting prognosis in patients aged > 65 years with nonmetastatic cervical cancer. Mortality rate caused by competitive events cannot be ignored especially in the case of high mortality in elderly patients, and traditional survival analysis method (eg, Cox proportional risk regression model) will overestimate the risk of the disease, resulting in competitive risk bias. The CRM predicted more accurately and represented better predictive power and reliability. Our study was based on the SEER database, which improved the accuracy and reliability of prediction. Nevertheless, our study had some limitations. First, although the SEER is a population-based high-quality database, the data collection is still incomplete. Some data such as chemotherapy regimen, RT dose, whether there are multiple combinations of treatments, and the sequences of treatments are not available; these factors may be confounding variables that affect prognosis. Second, this was a retrospective study and selection bias was unavoidable. Third, the CRM limits the consideration of competitive risk events. In this study, only the binary endpoints in the Cox model were extended to the triple endpoints in CRM, namely the event of interest, the deletion event, and the competitive risk event. Therefore, it should be more fully evaluated when choosing statistical methods, and the influence of confounding factors should be considered as well. As our future direction, we will collect data from multi-clinical center in different regions and at different levels to optimize and validate the model. Some vital genetic or epigenetic signatures associated with these risk indicators, which has been validated by multiomics data and wet experimental assays will be integrated to develop a more rigorous nomogram. Subgroup and stratified analyses should also be applied in future research.
Conclusion
In general, this study is the first to construct a CRM nomogram based on the SEER database to predict DSS in nonmetastatic cervical cancer patients aged > 65 years. The CRM predicted the endpoints more accurately and had a better predictive power and reliability. This model is helpful for clinicians in implementing more accurate personalized diagnosis and treatment modalities for elderly patients with nonmetastatic cervical cancer.
Supplemental Material
sj-docx-1-tct-10.1177_15330338231164191 - Supplemental material for Competitive Risk Model Nomogram to Predict Prognosis in Patients Aged Over 65 Years with nonmetastatic Cervical Cancer: A SEER Population-Based Study
Supplemental material, sj-docx-1-tct-10.1177_15330338231164191 for Competitive Risk Model Nomogram to Predict Prognosis in Patients Aged Over 65 Years with nonmetastatic Cervical Cancer: A SEER Population-Based Study by Shengyuan Jiao, MS, Li Guo, PhD, Fei Da, PhD, Qiaohui Gao, MS, Zhenghua Ren, MS, JianyuWang, MS, Quanwei Fu, PhD, and Junye Liu, PhD in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank Surveillance, Epidemiology, and End Results (SEER) program staff for invaluable providing the free open source from the database.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The SEER data are public and free of charge so no ethical review and individual consent are required.
Funding
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This study was financially supported founded by the grant (grant numbers 31770908 and 81272490) from the National Natural Science Foundation of China.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
