Abstract
The Day 14 bone marrow biopsy (D14 BMB) has historically been a key tool for early treatment response assessment in acute myeloid leukemia (AML), particularly following intensive chemotherapy. Traditionally, a blast count of <5% at D14 is associated with higher complete remission (CR) rates and improved overall survival (OS), while persistent disease often prompts re-induction therapy. However, emerging evidence suggests that while re-induction may increase CR rates, it does not consistently improve OS and is associated with significant morbidity. With the emergence of venetoclax-based and other targeted therapies, the traditional role of D14 BMB is being reconsidered, as these agents exhibit slower response kinetics, making later assessments and measurable residual disease (MRD) monitoring more reliable for treatment adaptation. This review critically examines the prognostic utility of D14 BMB in AML, its relevance across different treatment modalities, and its correlation with long-term outcomes. By synthesizing current evidence, we explore whether D14 BMB remains a valuable clinical tool or if a paradigm shift toward later assessments and MRD-guided decision-making is warranted in modern AML therapy.
Keywords
Introduction
Acute Myeloid Leukemia (AML) is a heterogeneous clonal disorder of hematopoietic progenitors characterized by the rapid proliferation of immature myeloid cells. Standard induction chemotherapy, traditionally a “7 + 3” regimen combining cytarabine with an anthracycline, aims to achieve complete remission (CR). 1 Historically, a Day 14 (D14) bone marrow biopsy (BMB) has been utilized as an early predictor of treatment response, assessing residual blasts and bone marrow cellularity. 2
D14 BMB, at this stage, is expected to be hypoplastic, with a blast count below 5%, signaling effective cytoreduction. Patients with a higher percentage of residual blasts or hypercellularity at D14 are at greater risk of treatment failure and relapse. Historically, these findings have informed decisions about re-induction or proceeding with salvage therapies or transplantation. 3
However, the landscape of AML treatment has evolved, particularly with the introduction of targeted therapies, such as venetoclax-based regimens, FLT3 inhibitors (midostaurin, gilteritinib), IDH inhibitors (enasidenib, ivosidenib) and menin inhibitors (Revumenib, Ziftomenib). These novel agents often exhibit delayed bone marrow clearance, raising concerns about the reliability of D14 BMB as a predictive marker in non-intensive treatment settings. Moreover, measurable residual disease (MRD) assessment, utilizing multiparameter flow cytometry (MFC), next-generation sequencing (NGS), and PCR-based methods, is increasingly recognized as a more sensitive tool for predicting relapse risk and long-term outcomes.
As a result, the role of D14 BMB in modern AML therapy is under scrutiny, with questions arising about whether it should remain a standard part of response assessment or if alternative strategies-such as delayed marrow evaluation (Day 21-28), MRD monitoring, and molecular biomarkers-provide greater clinical value. This review critically examines the prognostic utility of D14 BMB, its relevance in the context of evolving AML therapies, and whether a shift toward later response assessments and MRD-driven treatment strategies is warranted. By synthesizing current evidence, we aim to determine whether D14 BMB remains an essential decision-making tool or if its role has become obsolete in the era of personalized AML management.
Clinical Implications of Day 14 Bone Marrow Findings:
Day 14 Blast Clearance and Complete Remission Rates:
Several studies have demonstrated that blast clearance at D14 strongly correlates with achieving CR following induction therapy. In a study
Similarly, a study by
More recent evidence from
In a study,
However, more recent evidence challenges this paradigm. Balev et al (2024) reported that D14 BM status is not an independent predictor of survival in AML, with no significant difference in OS between patients with residual disease (RD) and those with no evidence of disease (NED). While RD patients exhibited lower CR rates, reinduction therapy improved remission but did not enhance OS. Instead, the European LeukemiaNet (ELN) risk classification remained a more reliable prognostic indicator, as D14 disease status did not significantly influence OS within ELN risk groups. 8
These findings call into question the routine use of D14 BM biopsy in reinduction decision-making and highlight the need for prospective studies to refine risk stratification and optimize treatment strategies.
Day 14 Bone Marrow Cellularity
Beyond blast percentage, bone marrow cellularity at D14 serves as a key prognostic marker. Hypocellular marrows (<20% cellularity) are typically associated with favorable outcomes, while hypercellular marrows suggest residual disease or inadequate response to chemotherapy. Griffin et al (2017) found that patients with a hypocellular marrow on D14 had significantly better survival outcomes compared to those with hypercellular or normocellular marrows.
Moreover, combining both blast percentage and cellularity in D14 bone marrow assessments enhances prognostic accuracy. Patients with both <5% blasts and hypocellularity had the highest likelihood of achieving CR, whereas those with persistent blasts and hypercellular marrows had the worst outcomes, including higher rates of relapse and poorer overall survival. 9
A study by Ollila et al (2018) further demonstrated that D14 bone marrow samples with cellularity ≥20% and blasts ≥20% had higher specificity for detecting residual leukemia. CR rates were significantly higher in patients with hypocellular marrow (11 of 16) compared to those with nonhypocellular marrow (4 of 15) (P = .03). Additionally, median OS was longer in hypocellular patients (17 months) than in nonhypocellular patients (8 months) (P = .02). However, the receipt of reinduction therapy did not significantly impact CR rates or OS, suggesting that bone marrow cellularity at D14 may be a more critical determinant of prognosis than the decision to pursue reinduction. 10
Interestingly, Norkin et al (2016) introduced a novel predictive model to enhance the prognostic utility of D14 BMB in assessing remission status in AML patients by incorporating additional clinical and laboratory variables. This model improved predictive accuracy to 88%, with a positive predictive value of 83% and a negative predictive value of 90%. Key factors associated with persistent disease included higher percentages of D14 myeloblasts, a history of prior relapse, and high-risk disease features. In contrast, age, white blood cell count, and D14 BM cellularity did not significantly impact remission outcomes. While these findings suggest a refined approach to risk stratification, further validation in larger, prospective studies is necessary to establish its clinical applicability. 11
Residual Disease at Day 14 and Its Implications for Re-Induction
Patients with residual blasts (>5%) at D14 are at a higher risk of induction failure and often require a second course of chemotherapy or intensified treatment strategies. Studies suggest that early identification of these high-risk patients via D14 BM can guide timely reinduction therapy, potentially improving remission rates.
A study by
Conversely, Campuzano-Zuluaga et al (2016) reported that 79.6% of patients had <5% blasts at D14, with 96.7% of these achieving CR, reinforcing the prognostic value of a favorable D14 BM assessment. Among the 20.4% of patients with >5% blasts, 47.8% received a second chemotherapy course (double induction), while 52.2% were observed (single induction), with no significant difference in CR rates (58.3% vs 45.5%, P = 0.684). These findings suggest that D14 BM evaluation alone may not reliably predict primary induction failure. 13
However, re-induction is associated with significant risks, including increased morbidity and mortality. In a study by
One of the major concerns with re-induction therapy is the increased risk of treatment-related complications. Patients undergoing a second cycle of intensive chemotherapy are more likely to experience prolonged cytopenias, leading to higher rates of infection, bleeding, and organ dysfunction. A study by
In another retrospective study evaluated 50 patients with newly diagnosed AML and indeterminate Day 14 BMB results (≤ 20% cellularity and 5%-20% blasts), comparing those who received reinduction chemotherapy (n = 25) versus observation alone (n = 25). Both groups had similar overall response rates (80% in each) and no statistically significant differences in overall survival (13 months vs 21 months, P = .88) or relapse-free survival (13 months vs 33 months, P = .53). These results suggest that immediate reinduction chemotherapy particularly for patients with hypocellular marrow and low blast percentage may not offer significant benefits and could lead to overtreatment. Larger studies are needed to further assess these findings. 14
Reevaluating the Role of Day 14 Bone Marrow Biopsy in the Era of Novel Therapies
While D14 BMB remains a valuable tool in the era of cytotoxic chemotherapy, the advent of novel therapies such as venetoclax and hypomethylating agents (HMAs) has transformed the treatment landscape for AML. Venetoclax, a selective BCL-2 inhibitor, has revolutionized AML therapy, particularly in older or unfit patients who are ineligible for intensive chemotherapy. When combined with HMAs (eg, azacitidine or decitabine) or low dose cytarabine (LDAC), venetoclax has demonstrated high response rate.15,16 However, its distinct response kinetics, compared to traditional chemotherapy, raise questions about the relevance of D14 BMB in guiding treatment decisions.
Venetoclax induces apoptosis in leukemic cells through inhibition of the BCL-2 pathway, but the pace of blast clearance is slower compared to intensive chemotherapy. As a result, D14 BMB may no longer serve as a reliable predictor of long-term outcomes in patients treated with venetoclax-based regimens.
DiNardo et al (2020) investigated venetoclax combined with hypomethylating agents (HMAs) in elderly AML patients, reporting that the median time to complete remission was approximately one to two treatment cycles-significantly later than the traditional D14 window used for response assessment in standard chemotherapy regimens. Their findings suggest that reliance on D14 BMB for prognostic information in venetoclax-treated patients may lead to premature conclusions about treatment efficacy.
In older, unfit patients receiving venetoclax-based induction, Day 21–28 BPM showed 71% blast reduction (<5%), significantly correlating with improved overall survival (P = 0.0033) and higher overall response rates (ORR) (Baron et al, 2024). Withholding venetoclax to aid count recovery reduced infections and cytopenias, suggesting a strategy to mitigate toxicity. These findings emphasize the importance of early BMBx in guiding venetoclax-based therapy. 17
Similarly,
Since older patients often struggle to tolerate intensive chemotherapy, lower-intensity regimens and targeted therapies such as enasidenib, ivosidenib, glasdegib, venetoclax, midostaurin, and gilteritinib have become promising alternatives, with additional agents in development. Traditional response criteria emphasize rapid morphologic remission, yet increasing evidence indicates that meaningful clinical benefit, including stable disease, reduced transfusion dependence, and improved quality of life, may occur in the absence of early complete remission. Accordingly, long term treatment efficacy is more accurately captured by longitudinal assessment of minimal residual disease and hematologic recovery rather than reliance on day 14 blast morphology alone. 19
In parallel, institutional strategies have evolved to include delayed marrow assessment at day 21 to 28 or after absolute neutrophil count recovery, selective use of re induction based on integrated clinical and biological risk factors, and incorporation of mutation directed therapies. In patients with FLT3 or IDH mutated disease receiving targeted combinations, early marrow evaluation may guide treatment duration or intensity rather than predict definitive remission. 19
Collectively, this heterogeneity in practice reflects a growing consensus that D14 BMB should be interpreted within a comprehensive, risk adapted framework that integrates disease biology, treatment modality, and patient specific factors rather than in isolation.
The Evolving Role of D14 BMB: Not the End of the Story Yet
While D14 BMB remains a valuable tool in AML management-particularly in the context of intensive chemotherapy-its role is becoming less definitive in patients receiving novel agents such as venetoclax, hypomethylating agents, or other targeted therapies. The table below provides an updated summary of recent studies assessing Day 14 bone marrow evaluations in AML, building on the foundational work of Terry et al (2017) 2 (Table 1). It highlights key outcomes related to remission rates, overall survival, and induction failure:
Summary of Key Studies Evaluating Day 14 Bone Marrow Assessments in AML.
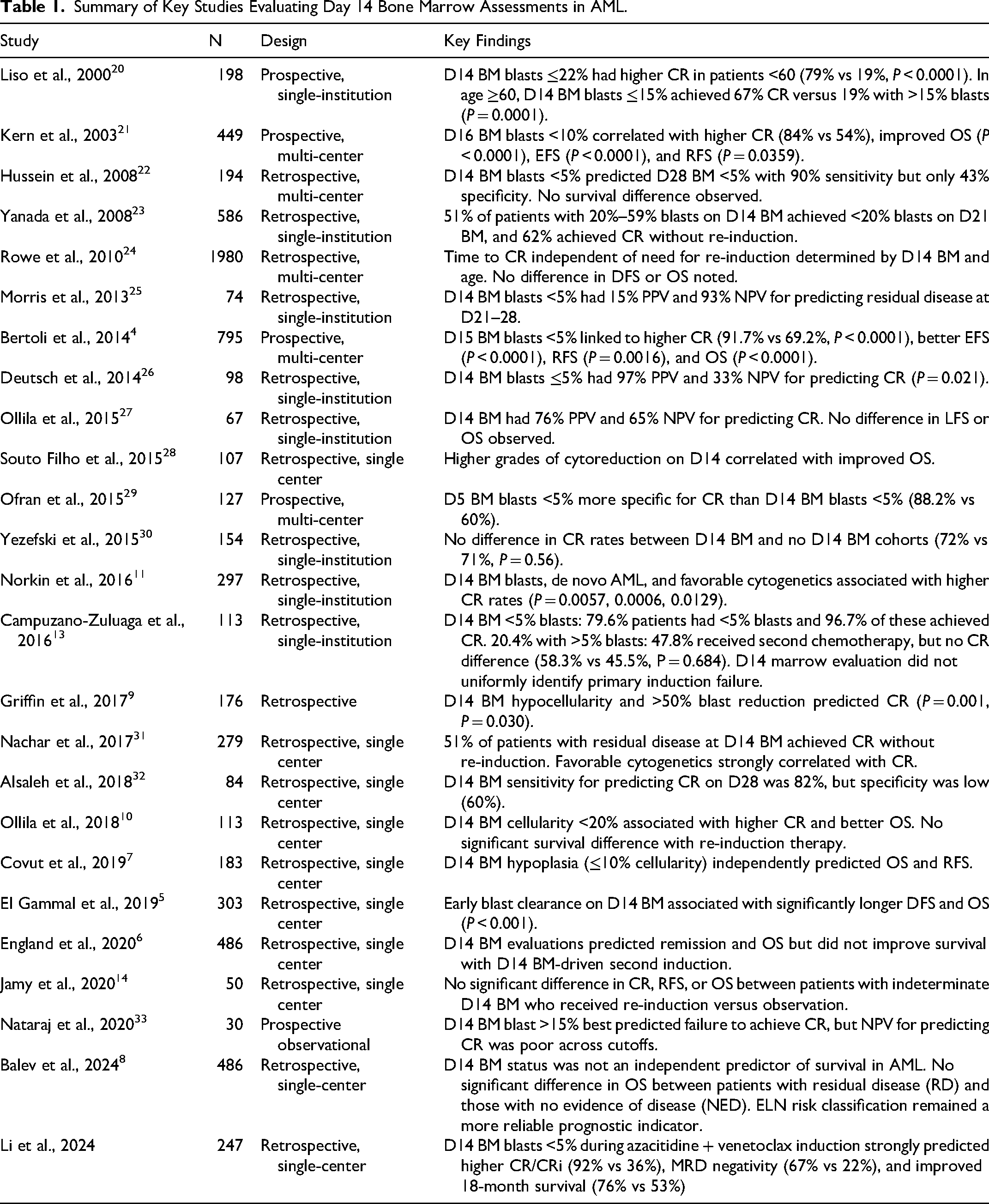
The slower kinetics of modern AML therapies may delay bone marrow blast clearance, making later assessments (eg, post-cycle 1 or cycle 2) more informative. Furthermore, minimal residual disease (MRD) assessment, using flow cytometry or molecular techniques, is emerging as a more precise indicator of long-term outcomes in patients receiving modern therapies.
The 2021 ELN MRD guidelines recommend MRD assessment after two chemotherapy cycles, at consolidation end, and every 3 months for 24 months in high-risk AML. Peripheral blood monitoring may be used in NPM1-mutated and CBF AML, but bone marrow remains the standard. Pre-transplant MRD evaluation aids risk stratification, with extended monitoring individualized based on relapse risk. 34
Given these evolving factors, it is essential for clinicians to interpret D14 BMB findings within the broader therapeutic landscape. For patients undergoing traditional cytotoxic chemotherapy, D14 BMB remains a critical prognostic tool, guiding decisions regarding reinduction and transplantation. However, for those receiving novel agents, a more flexible approach-incorporating later marrow assessments and MRD evaluation-may provide greater predictive value. As treatment paradigms in AML continue to evolve, the D14 BMB remains a key, though increasingly nuanced, tool in early treatment decision-making.
The Role of Measurable Residual Disease (MRD) in the Modern Era
The prognostic significance of MRD has been demonstrated across multiple studies. Pratz et al (2022) showed that MRD response is a strong and independent predictor of prognosis in treatment-naive AML patients receiving venetoclax and azacitidine, with MRD negativity strongly associated with improved outcomes. 35 The 2025 ELN MRD guidelines provide standardized recommendations for MRD assessment timing and methodology, recognizing MRD status as a critical prognostic factor that should guide treatment decisions and risk stratification. 36
MRD assessment is becoming an increasingly important component of AML management. The integration of MRD testing with other prognostic factors, such as cytogenetics, molecular mutations, and clinical response, is the most comprehensive approach to risk stratification and treatment planning. As assays become more standardized and the clinical utility of MRD-guided therapy is further established through prospective studies, MRD monitoring will likely play an even greater role in optimizing AML management in the future.
Conclusion
The D14 BMB remains a valuable clinical tool in the management of AML, particularly for patients undergoing standard induction chemotherapy. However, with the advent of novel agents such as venetoclax and hypomethylating agents, its prognostic utility is becoming less definitive in certain patient populations due to delayed response kinetics. As treatment paradigms evolve, D14 BMB may no longer be the sole determinant of early treatment response but rather one component of a broader, more refined assessment strategy.
While D14 BMB is not yet obsolete, its role is shifting, and it may soon be supplemented-or in some cases replaced-by more advanced methods such as MRD testing, later bone marrow evaluations, and emerging biomarkers that provide a more precise assessment of treatment efficacy. Moving forward, ongoing research and prospective studies will be essential in refining response assessment strategies to optimize AML management in the era of targeted and lower-intensity therapies.
Footnotes
Author Contribution
All authors contributed equally to writing and editing the manuscript and approved submission of final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
