Abstract
Prostate cancer remains one of the most common malignancies in men, with its progression strongly influenced by androgen signaling. While genetic alterations are well-documented in prostate cancer, growing evidence highlights the contribution of environmental factors, particularly diet and the gut microbiome, in modulating disease risk and therapy response. The gut microbiota plays a crucial role in regulating host metabolism, immune responses, and hormone activity. Recent findings suggest that specific microbial communities influence androgen biosynthesis and metabolism through enzymes such as β-glucuronidase, altering systemic androgen availability and imp acting tumor progression. Additionally, microbial metabolites, including short-chain fatty acids, secondary bile acids, and bacterial genotoxins, can affect inflammatory pathways and cellular signaling relevant to prostate tumorigenesis. Experimental studies also indicate that modifying the gut microbiota through dietary interventions, probiotics, or fecal microbiota transplantation can influence tumor growth and improve responses to immunotherapy and hormone-based treatments. In this review we present the current knowledge on gut–prostate axis, examine the mechanistic links between microbial activity and prostate cancer biology, and discuss emerging microbiome-based strategies as potential therapies. A deeper understanding of this bidirectional crosstalk could pave the way for microbiome-informed approaches to prevention, diagnosis, and personalized treatment of prostate cancer
Keywords
Introduction
The human gastrointestinal tract is home to trillions of microorganisms, including bacteria, fungi and viruses. Advances in high-throughput sequencing and multi-omics technologies have shed light on the significant roles these gut microbes play in regulating host hormonal, metabolic, and inflammatory pathways. Disruption of this microbiome balance, called dysbiosis, has been implicated with the onset and progression of various diseases, including neurodegenerative diseases, autoimmune disorders and various forms of cancer. 1
Prostate cancer (PCa), the most commonly diagnosed cancer in men, is generally a slow-progressing disease that predominantly affects individuals over the age of 50. While its genetic underpinnings are well characterized and remain a major area of research, 2 PCa is also shaped by a myriad of lifestyle factors (Figure 1). Incidence rates show wide geographic and demographic variation, influenced by age, ethnicity, socioeconomic status, and behavioral factors such as diet and smoking.3,4 Notably, healthy lifestyle is associated with lower risk of the disease in genetically pre-disposed individuals, 5 underscoring the importance of non-genetic lifestyle factors in PCa susceptibility.
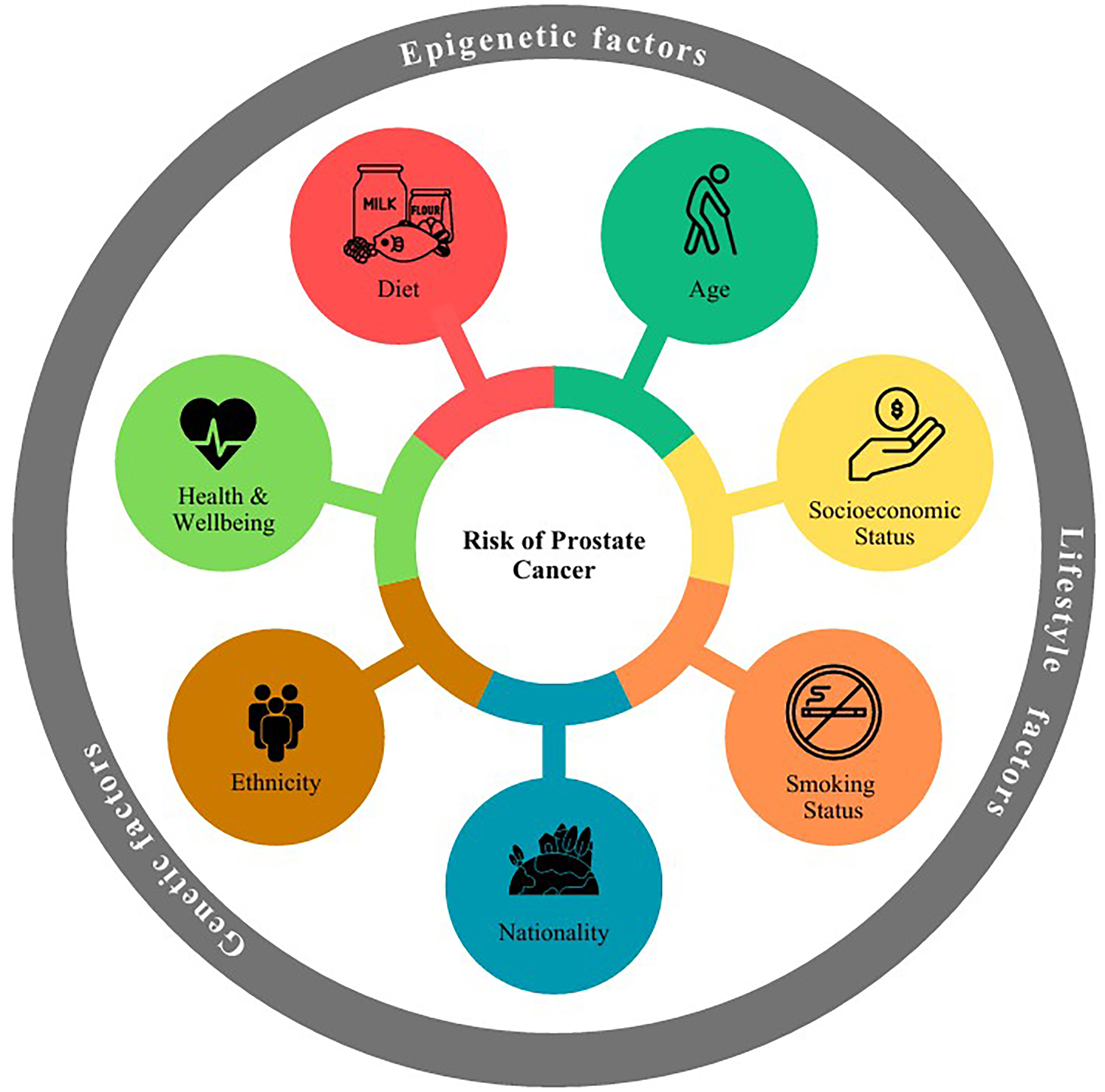
Factors contributing to increased risk of prostate cancer. A range of factors including genetic predisposition, epigenetic modifications and lifestyle factors interact to increase the risk of prostate cancer development and progression.
Early clues linking gut microbiota to PCa emerged through studies associating dietary patterns with disease risk. For instance, several case-control studies have linked high fat diets (HFD) to increased PCa risk. 6 On the contrary, serum from patients with low fat diet was shown to inhibit proliferation in PCa cell lines.7,8 Since gut microbial composition is known to shift in response to diet, this suggests that gut microbes may mediate the diet-associated modulation of PCa risk. Furthermore, in breast cancer, gut microbiota has been shown to influence hormone metabolism, particularly estrogen, 9 raising the possibility of a similar gut–androgen–prostate axis. Indeed, recent studies report distinct differences in gut microbial profiles among healthy individuals, those with benign prostate conditions, and PCa patients. 1
Diet, Gut microbiota and Prostate Cancer
Diet plays a pivotal role in shaping the composition and function of gut microbiota, which in turn influences digestion, nutrient absorption, and overall metabolism. These microorganisms interact with dietary components to produce metabolites which are crucial for maintaining gut health and metabolic balance. Emerging evidence suggests that dietary patterns can also influence genomic stability and gene expression in host tissues via hormonal changes and epigenetic modifications, 10 thereby positioning diet and gut microbiota as potential contributors to cancer risk and progression including in PCa.
PCa is characterized by complex genomic alterations leading to substantial inter- and intra-tumoral heterogeneity. These alterations involve a wide range of molecular pathways, including kinases/phosphatases and other signaling components which regulate processes like cell growth, apoptosis, androgen receptor (AR) signaling, and DNA repair mechanisms. 11 Interestingly, recent studies have revealed a significant gut-prostate axis, highlighting how dietary patterns and gut microbial composition can modulate PCa development and progression. For instance, dietary modulation, particularly the supplementation of polyunsaturated fatty acids (PUFA), can modulate the gut microbiota and influence tumor growth. 12 Experiments involving C57BL/6N, PTEN PE−/− (PE, prostate epithelium) mice fed with purified monoglyceride (MAG) forms of different PUFAs, specifically omega-6 (MAG-AA), omega-3 (MAG-DHA, MAG-EPA), and omega-9 (HOSO) fatty acids rich diet, and subcutaneously injected with TRAMP-C2 PCa cells to assess tumor development showed significant reduction in tumor growth in the omega-3 supplemented group compared to the control (HOSO-treated group), indicating a strong anti-tumor effect of docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA). 13 Additionally, DHA and EPA supplementation led to 1.5-fold reduction in fecal butyric acid levels, displaying reduced PCa growth, slower histopathological progression and increased survival. In comparison, mice fed on an omega-6 FA-rich diet exhibited pro-tumorigenic effects. 13 Similar results were also reported by Gevariya et al and Bilodeau et al, both of whom confirmed that omega-3 fatty acids were associated with reduction in prostate tumor growth, possibly through induction of anti-tumor inflammatory pathways.14,15 Interestingly, some studies have also associated higher levels of EPA and DHA in blood with increased PCa risk, 16 hinting at a bidirectional regulatory effect of PUFAs on tumor growth.
Distinct gut microbial signatures have also been observed between patients with PCa and those with benign prostatic conditions. Studies by Golombos et al and Matsushita et al demonstrated that patients with high-risk PCa exhibit increased abundance of microbial taxa such as Bacteroides massiliensis, Alistipes, Lachnospira, Subdoligranulum, and Eggerthella compared to individuals with benign prostatic conditions.17,18 Lachance et al further demonstrated increased abundance of Bacteroides, Parabacteroides, and Ruminococcaceae in fecal DNA sequences of Pten−/− and Rb−/− PCa mouse models, reinforcing the link between microbial shifts and PCa pathogenesis. 13 These findings suggest that specific microbial communities may be associated with PCa progression. Fecal microbiota transplantation (FMT) further substantiates the causal relationship between microbiota composition and PCa. In syngeneic TRAMP-C2 models, 16S rRNA metataxonomic analysis revealed that FMT from PCa-bearing donors altered microbial profiles, reducing Actinobacteria while increasing Firmicutes, Bacteroides, and Parabacteroides. KEGG analyses revealed significant enrichment of metabolic pathways involving enzymes like aldehyde dehydrogenase, 3-hydroxyacyl-CoA dehydrogenase, and enoyl-CoA hydratase, implicating microbial fatty acid metabolism in tumor progression.13,18
The association between meat consumption and PCa risk remains a subject of ongoing scientific investigation, yielding mixed results and opinions across various studies. While some epidemiological analyses suggest a modest correlation between high intake of red or processed meats and increased PCa risk, 19 other comprehensive meta-analyses have found no significant association. 20 These variabilities may be attributed to variations in study design, population demographics, and potential confounding factors. However, there is a growing consensus that the method of meat preparation plays a crucial role in modulating cancer risk. For instance, heterocyclic amines, such as 2-amino-1-methyl-6-phenylimidazo(4,5-b) pyridine (PhIP), that are generated during high-temperature cooking methods represent a major source of concern. In the liver PhIP is metabolized by the enzyme cytochrome P450 1A2 (CYP1A2) into its bioactive form N-hydroxy-PhIP (N-OH-PhIP). 21 This intermediate is further processed through acetylation or sulfonation into highly reactive electrophilic species capable of forming DNA adducts and promoting mutagenesis. Hence, dietary patterns, fatty acid composition, and gut microbial interactions collectively play a critical role in modulating PCa risk and progression. Targeting this axis through nutritional interventions and microbiome modulation presents a promising avenue for PCa prevention and therapeutic optimization.
Gut Microbiota Associated Metabolites and Their Influence on Prostate Cancer
Short Chain Fatty Acids
Short chain fatty acids (SCFAs) are small metabolites generated when gut microbes ferment undigested or partially digested polysaccharides in the small intestine. Generally consisting of less than six carbons in their aliphatic tails, SCFAs can be categorized as butyrate (C4), propionate (C3)and acetate (C2). Among these, acetate is the most abundant (approximately 60%), followed by propionate and butyrate (each ∼20%). 22 These SCFAs are primarily synthesized by intestinal microorganisms such as Alistipes, and Lachnospira.. Interestingly, studies have shown that microorganisms can regulate PCa through SCFA, with their concentration significantly increased in the intestinal microflora of patients with high-grade PCa, suggesting pro-tumorigenic role.23–25 Matsushita et al demonstrated that SCFA supplementation using HFD in Pten−/− mice models elevates systemic and local levels of insulin-like growth factor 1 (IGF-1), which in turn activates MAPK and PI3 K pathways which frequently crosstalk with the androgen receptor to promote ligand-independent AR activation, a hallmark of CRPC. Furthermore, removing HFD and depleting SCFA producing bacteria using antibiotics was found to reduce systemic and prostate levels of IGF1 as well tumor size (Figure 2). 26 Thus, SCFA-mediated IGF-1 elevation may serve as a critical mechanism for maintaining AR activity even under androgen-deprived conditions. In subsequent studies, HFD was also been found to promote PCa growth via mast cells dependent histamine signaling pathway and zonula occludens-1(ZO-1), highlighting another mechanistic layer by which diet-modulated gut microbiota influences tumor progression. 27

Diet–microbiota–prostate axis in cancer progression. Dietary components modulate the composition and activity of the gut microbiota, which in turn produces bioactive metabolites such as SCFAs, secondary bile acids, and bacterial toxins. These metabolites influence inflammation and oncogenic signaling pathways, ultimately affecting prostate cancer development and progression.
Bacterial Genotoxins and Prostate Cancer
Bacterial genotoxins such as cytotoxic necrotizing factor 1 (CNF1), 28 cytolethal distending toxin (CDT) 28 and colibactin29,30 are known to induce DNA damage in prostatic cells. These toxins cause single- and double-stranded DNA breaks, promote chromosomal rearrangements, and form DNA adducts, which can trigger oncogenic mutations (Figure 2). In animal models, CNF1 promotes metastasis and castration resistance via Cdc42-PAK1 signaling, highlighting its role in tumor progression. 31 The bacterial toxin CDT is primarily composed of three subunits, CdtA, CdtB and CdtC. Of these, CdtB acts as a DNase I like enzyme, generating DNA double stranded breaks (DSB) and leading to genomic instability and cell cycle arrest.32,33 Recent advances have also hinted at a CDT-p53-proteasome-LC3 regulatory axis which suppresses autophagy in cells with sublethal DNA damage, hence retaining mutations and subsequently leading to cancer. 34 Similarly, colibactin, a toxin produced by members of family Enterobacteriaceae containing the pathogenic island pks, is known to cause DNA alkylation and crosslinking, eventually leading to formation of DNA adducts and DSBs.29,35,36 In the context of PCa, colibactin induced DSBs were found to be associated with increase in TMPRSS2:ERG gene fusion, a major driver of PCa initiation. 37 The ERG oncogene in this fusion is placed under the control of androgen-responsive promoters, allowing these microbial toxins directly hijack the AR signaling machinery to initiate and drive PCa progression
Aside from these, several minor genotoxins have also been studied for their role in tumorigenesis. For instance, indolamine, a genotoxin produced by Morganella morganii, is known to induce gut permeability and is associated with increased colon tumor load in mice models for colorectal cancer.38–40 Interestingly, M. morganii was recently also identified in chronic prostatitis and prostatic abscesses. 41 This hints at a possible role of indolamine in PCa and highlights bacterial toxins as a yet underexplored but novel paradigm to study microbial contribution towards cancer development and growth.
Bile Acids and Antitumor Activity
Gut microbial metabolism of bile acids plays an important role in modulating PCa progression. 42 Primary bile acids, such as cholic acid (CA) and chenodeoxycholic acid (CDCA) are synthesized in the liver and secreted into bile. Upon entering the intestinal tract, these primary bile acids are metabolized by the gut microbiota into secondary bile acids like lithocholic acid (LCA), deoxycholic acid (DCA), and ursodeoxycholic acid (UDCA). Several of these secondary bile acids have demonstrated anticancer properties in PCa models (Figure 2). 43 For instance, LCA has been shown to inhibit the proliferation and induce cell death in PCa cells by cleaving Bid and Bax, reducing Bcl-2 levels and triggering permeabilization of mitochondrial outer-membrane, ultimately leading to the activation of caspase-9 and apoptosis. 44 Similarly, UDCA has been reported to suppress the growth of PCa cells by activating the tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) signaling pathway, mediating release of mitochondrial cytochrome c which ultimately leads to apoptosis. 45
Moreover, CDCA and DCA have been implicated in promoting antitumor phenotypes through the destabilization of hypoxia-inducible factor-1 alpha (HIF-1α) subunits. This destabilization impairs hypoxia-driven signaling pathways, thereby inhibiting key processes associated with tumor progression, such as clonogenic growth, cellular invasion, and migration.. 46 Collectively, these findings suggest that gut microbiota-derived bile acids contribute to PCa suppression through diverse molecular mechanisms, highlighting the significance of host–microbe metabolic interactions in cancer biology.
Phthalate-Induced Microbial Dysbiosis
Phthalates, such as dibutyl phthalate (DBP), butyl benzyl phthalate (BBP), and di(2-ethylhexyl) phthalate (DEHP), are high molecular weight compounds that are widely used as plasticizers and are known to cause various developmental and neuroendocrine disorders. 47 Studies have established phthalates as major disruptors of gut microbiota homeostasis, with emerging evidence showing that exposure to DEHP during early stages of development leads to reduced gut microbial diversity and altered levels of microbial metabolites like SCFAs, branched chain amino-acids and simple sugars. 48 Similarly, Zhang et al demonstrated that DEHP exposure led to gut dysbiosis in mice models, increasing abundance of pathogenic bacteria like Desulfovibrio and Muribaculum while decreasing beneficial bacteria like Lactobacillus. 49 Such shifts toward pro-inflammatory microbial communities may create a systemic environment that favors tumour-promoting processes.50–52
In PCa studies, phthalates have been shown to have direct oncogenic effects on prostate cells. DEHP, BBP and DBP promote cell proliferation through upregulation of cyclin D1 and PCNA, downregulation of p21, and activation of the ERK5/p38–AP-1 signaling cascade, suggesting a synergism with AR signaling to drive PCa progression. 53 Supporting these mechanistic findings, multiple population-based analyses identified substantial associations between phthalate exposure and prostate cancer occurrence in the US 54 and Taiwanian 55 populations. Collectively, these studies indicate that phthalates may influence prostate cancer development through dual mechanisms, first by disrupting gut microbial homeostasis, and second by directly activating pro-proliferative signaling pathways in prostate cells. Aside from phthalates, other important xenobiotics like bisphenols, polychlorinated biphenyls, persistent organic pollutants (POP), heavy metals and pesticides have also been found to induce gut dysbiosis and have been associated with various adverse clinical outcomes.56–60 Hence these could also emerge as important risk factors in prostate carcinogenesis.
Antibiotics and Prostate Cancer Risk
Antibiotics are traditionally employed to combat bacterial infections and certain classes of antibiotics, known as antitumor antibiotics, have been repurposed to treat cancer. However, recent studies suggest antibiotic therapies can have adverse effects in PCa via changes in the gut microbiome. Population-based studies revealed that prolonged exposure to antibiotics such as penicillins, tetracyclines, sulfonamides, and quinolones is associated with modestly increased PCa risk.61,62 Similarly, oral administration of broad spectrum antibiotics like ampicillin, neomycin, vancomycin and colistin was found to promote tumor progression and docetaxel resistance in mice models.63,64 Zhong et al demonstrated that this was achieved due to increased intestinal permeability caused by gut dysbiosis in antibiotic-exposed mice, enabling greater translocation of lipopolysaccharides (LPS) into tumor tissues, which activates the NF-κB-IL6-STAT3 axis, driving tumor growth and chemoresistance. 64 These findings suggest that gut dysbiosis may impair immune regulation and influence tumorigenesis, thereby underscoring the potential impact of antibiotics in PCa outcomes.
Gut Microbiota in Androgen Dysregulation and Tumorigenesis
Aside from the gut-prostate axis, recent advances in imaging and microbial research have also illuminated a bidirectional gut–brain axis, wherein the nervous system modulates the gut microbiota via neural, endocrine, and immune routes, 65 while gut microbes, in turn, influence host neurophysiology and behavior (Figure 3). 66 A significant outcome of this crosstalk is the modulation of host hormone levels. Gut microbiota can influence the synthesis, metabolism, and systemic levels of several hormones including sex hormones like estrogen, progesterone, and androgens.67,68 This interaction has been implicated in hormone-driven diseases such as endometriosis and breast and ovarian cancers.69–71

Role of gut microbiota in systemic androgen regulation. The gut-brain axis represents a bidirectional communication between central nervous system and the gastrointestinal tract and its microbiota. A major consequence of this axis is its effect on hormone regulation. Androgen is produced in the testis as testosterone. This is transported to the prostate where most, but not all, testosterone is converted to the more potent DHT by action of enzyme 5α-reductase. These testosterone and DHT molecules can then bind to AR to carry out their biological function. Upon dissociation from AR, androgen molecules freely diffuse into blood vessels where they are carried by plasma proteins. In the liver, the androgen molecules are acted upon by UDP-glucuronosyltransferase (UGT) which converts free androgen to glucuronide conjugates. Conjugated androgens is then excreted into the bile and reach the gastrointestinal tract. Bacterial β-glucuronidase enzymes in the gut deconjugate these metabolites, liberating free androgens. These reactivated androgens can then be reabsorbed through the intestinal epithelium into systemic circulation, increasing circulating androgen levels and potentially enhancing androgen-driven tumorigenesis in the prostate.
Androgen, a steroid sex hormone like estrogen and progesterone, plays an important role in male sexual development and reproduction. Primarily produced in the testes as testosterone, it travels to the prostate via the bloodstream, where it is often converted to the more potent dihydrotestosterone (DHT) and binds to the AR, a ligand-activated transcription factor which promotes cell growth and differentiation. 72 In healthy males, this androgen-AR signaling cascade is associated with the regulation of genes associated with spermatogenesis as well as proper growth, differentiation and homeostasis of male sex organs. 73 However, dysregulation of AR signaling is one of the central features of PCa. 74 Increased AR signaling, either due to high serum androgen levels or ligand-independent activation, has been directly associated with tumor progression and androgen deprivation remains the cornerstone for treatment. 75
There has been emerging evidence hinting at a complex interplay between gut microbiota and androgen where each modulates the expression of the other. Findings suggest that gut microbes play a role in both androgenesis as well as androgen metabolism, with critical implications in health and disease. Recent studies have also demonstrated that certain microbes play important roles in regulating endogenous androgen levels. For instance, higher abundance of gram positive bacteria from phylum Firmicutes (Clostridiales, Turicibacter, and Gemella) has been directly associated with increased serum testosterone levels in elderly Japanese males. 76 In another study, testosterone levels were positively correlated with higher microbial diversity and increased abundance of bacteria like Acinetobacter, Dorea, Ruminococcus, and Megamonas. 77 This was further corroborated by Pernigoni et al who showed that administration of Ruminococcus gnavus in germ free mice increases blood testosterone levels and tumor growth compared to untreated controls. 78 Furthermore, introduction of Lactobacillus reuteri in aging mice increases testosterone to youthful levels. 79 Similarly, colonisation of germ free mice or female mice with conventional male microbiota leads to significantly heightened serum testosterone levels.80,81 Together, these findings indicate a strong association between gut microbiota and androgen levels in the host body.
Having established that microbial alteration can alter host androgen profile, the next step is elucidating the mechanism by which this is achieved. Certain bacterial species can directly regulate serum androgen level by metabolizing or catabolising androgen and its precursors. For instance, bacterial β-glucuronidase, primarily produced by members of phylums Fermicutes and Bacteriodota, plays a pivotal role in androgen reabsorption through enterohepatic recirculation (Figure 3). They achieve this by cleaving glucuronide from androgen conjugates and converting it to its active form. 82 This was further validated by Colldén et al, who demonstrated increased levels of glucuronidated testosterone and DHT in the small intestine (having low bacterial density) and increased levels of free DHT in the cecum and colon (having high bacterial density). The same study also showed that germ-free mice had significantly higher levels of glucuronidated androgens and lower levels of free androgen in distal intenstine as compared to normal counterparts.. 83 Thus, deglucuronidation provides a central mechanism through which gut microbes modulate androgen availability.
Apart from modulating existing androgens, some microbes also possess specific steroid-processing enzymes capable of driving androgen synthesis. For instance, Clostridium scindens mediates formation of androgenic metabolites like androstenedione from glucocorticoids through anaerobic side-chain cleavage.84,85 Other bacteria like Clostridium cadaveris, Clostridium innocuum, and Butyricicoccus desmolans, also express enzymes capable of directly metabolising steroid hormones, thus contributing to androgen regulation 86 Consequently, germ-free mice have generally been found to have lower testosterone levels, lower sperm mobility and decreased weight of reproductive organ. 86 However, microbes are not just involved in androgen production but also in some cases lead to decreased serum androgen. For instance, the betaproteobacterium GD1 catabolises androgen and reduces recirculation, thus lowering serum androgen levels.87,88 Taken together, these results hint at the existence of a strong microbiota-gut-testis axis that significantly modulates androgen levels inside the body.
It is not only the gut microbiome that influences hormone levels, the hormones also influence gut microbiota in return. It is widely established that the composition of gut microbes varies considerably with sex, with specific microbes showing higher abundance in particular sexes. 89 This difference is, at least partly, regulated by differential levels of sex hormones. Study by Yurkovetskiy et al established that the difference in composition of gut microbiota was much more significant in post-pubescent male and female mice as compared to pre-pubescent counterparts. Furthermore, this difference was almost eliminated upon castration, suggesting an important role of androgen in influencing microbial populations. 81 Org et al further substantiated this correlation by using gonadectomy and hormone replacement to establish a direct relationship between microbial diversity and testosterone.90,91 These findings together indicate a defining role of hormonal regulation in controlling the growth of particular microbial populations. Thus, there exists a strong and bidirectional interaction between gut microbiota and host androgen levels, underscoring the importance of microbial regulation for androgen homeostasis.
Altering gut Microbes as Therapeutic Strategy Against Cancer
With growing evidence suggesting the influence of gut microbiota on cancer initiation, progression, and treatment responsethere has been increasing interest in leveraging microbial interventions as adjunct therapeutic strategies in oncology. Among these, FMT has gained immense traction. Historically used to treat gastrointestinal disorders such as Clostridium difficile infections, FMT is now being evaluated in cancer therapy. 92 Recent studies have highlighted the potential of FMT in restoring responsiveness to immune checkpoint inhibitors (CPIs) in patients with advanced melanoma. Baruch et al demonstrated that FMT from CPI-responsive donors induced partial or complete tumor regression in 3 of 10 CPI-refractory patients. 93 In a similar study, Davar et al reported clinical benefit in 6 of 15 patients who had previously been categorized as CPI-refractive. The study found that FMT from CPI responders modified recipient gut microbiomes and altered their proteomic and metabolomic signatures, subsequently leading to decreased IL-8–associated immunosuppression and increased intratumoral CD8+ T-cell activation. 94 While both studies only showed success in a subset of patients, likely due to variable donor compatibility and patient specific immune heterogeneity, they provide strong proof-of-concept evidence for the efficacy of FMT to supplement immunotherapy. In yet another study on melanoma and colon cancer models, oral administration of the probiotic Lactobacillus rhamnosus GG (LGG) was found to improve response to CPI in mice models by activating dendritic cells (DC) and T-cells. Mechanistically, LGG mediated stimulation of DCs leads to increased production of IFN-β via the cGAS/STING pathway, promoting cross-priming of CD8+ T-cells and improving CPI efficacy. .. 95 Similarly, depletion of gut microbes with vancomycin has also been shown to reduce tumor growth and increase efficacy of adoptive T-cell therapy (ACT) in tumor bearing mice. This was linked to increase in CD8α+ DCs which produced higher levels of IL-12, thereby promoting expansion and tumor infiltration of transferred T-cells. Furthermore, this increased ACT efficacy could be transferred between mice by fecal microbiota transplant, further cementing the contribution of microbes. 96
Building upon these observations, microbial manipulation is now being evaluated as a therapeutic strategy in PCa. In murine CRPC models, FMT from healthy donors significantly delayed tumor progression, potentially via downregulation of the TNF-α/NF-κB axis. 97 Conversely, FMT from PCa patients with high tumor burden induced neoplastic features in healthy mice.13Together these results establish FMT as a powerful tool to modulate disease trajectory. Similar findings were associated with microbial derived metabolites, with long chain fatty acids inhibiting tumor growth while butyrate was found to promote metastasis. 98 These studies further reinforce the causal link between gut microbiota composition and PCa development. Given the limited efficacy of CPI therapies in PCa, 99 microbial modulation offers a promising strategy to enhance immunotherapeutic responsiveness. Collectively, these findings advocate for deeper exploration of the gut–tumor axis in PCa, with the goal of developing novel microbiome-targeted interventions.
Current Clinical Trials
Several ongoing clinical studies are actively investigating how modulation of the gut microbiota may improve therapeutic outcomes or serve as a predictive biomarker in PCa management (Table 1). A phase I clinical trial (NCT04116775) is evaluating FMT as a strategy to overcome resistance to immune checkpoint inhibitors in patients with metastatic castration-resistant PCa (mCRPC). This trial involves transferring fecal material from immunotherapy-responsive donors to non-responding recipients, assessing whether gut microbial composition can modulate anti-tumor immune response. 100 Another mechanistic trial (NCT06616597) is investigating the role of gut bacteria in intratumoral androgen production, hypothesizing that specific microbial taxa may contribute to resistance against abiraterone. The study is further exploring whether the administration of broad-spectrum antibiotics and corticosteroids can disrupt this androgenic microbial activity and restore drug sensitivity. 101 In the UK, the microbiome molecular characterization (MMC) study (NCT06423976) is analyzing patient samples to understand how the gut microbiota influences treatment response and side effects across different stages of PCa. 102 Complementing this, the PROMIC study in Finland (NCT06116851) is examining how the composition of gut bacteria may relate to PCa risk and metabolism. Diet and lifestyle are also under investigation with a neoadjuvant trial (NCT04985565) testing the feasibility and biological impact of a plant- and fish-based diet in patients undergoing radical prostatectomy 103 while the microstyle study (NCT05155618) is looking at how a six-month lifestyle intervention during radiotherapy affects gut microbes, inflammation, and treatment-related side effects. 104 Together, these studies suggest the gut microbiome plays a key role in improving prevention, treatment, and understanding of PCa.
Key Clinical Trials Investigating Gut Microbiota in PCa.

Conclusion and Future Perspectives
Recent advances have highlighted the intricate and multifaceted relationship between gut microbiota, diet, and PCa. Compelling evidence now supports the existence of a gut–prostate axis wherein microbial composition and metabolites significantly influence androgen signaling, inflammation, genomic alterations, and ultimately tumor progression. Microbial-derived metabolites such asSCFA, bile acids, and bacterial genotoxins have been shown to modulate oncogenic pathways and hormonal axes. Furthermore, lifestyle choices, particularly diet, play a critical role in shaping the gut microbiota and hence PCa risk. HFD, red meat, and processed foods promote pro-tumorigenic microbial profiles, while omega-3-rich diets and specific bacterial strains show protective effects. Recent studies also underscore the reciprocal interaction between sex hormones and gut microbial communities, hinting at the existence of a microbiota-gut-androgen axis that plays a role in disease development and therapeutic response.
These findings open exciting avenues for future research and therapeutic intervention. Modulation of gut microbiota through dietary interventions, probiotics, antibiotics, or FMT has shown early promise in altering disease trajectory in PCa and other cancers. However, key questions remain regarding the stability, safety, and long-term efficacy of these approaches. Future efforts must focus on defining patient-specific microbial signatures predictive of treatment response and/or disease risk and establishing causal relationships through mechanistic studies and well-controlled clinical trials. Integration of multi-omics data, including metagenomics, metabolomics, and transcriptomics, will be vital in unraveling the precise molecular mechanisms by which microbes influence PCa biology. Finally, combining microbiome-targeted strategies with current standards of care, including androgen deprivation therapy and immune checkpoint inhibitors, may offer synergistic benefits. As research progresses, the gut microbiome holds promise not only as a biomarker but also as a modifiable target for precision prevention and personalized therapy in PCa.
Footnotes
Abbreviations
Acknowledgements
A.M. acknowledges Anusandhan National Research Foundation (CRG/2022/008672) and the Indian Council of Medical Research (NCD/Ad-hoc/224/2022-23) for funding support. G.S. and V.N. are recipients of MoE-JRF fellowship. S.P. acknowledges fellowship support from DBT, GoI.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
No new data was created or analyzed in this study. Data sharing is not applicable to this article.
