Abstract
Objective
To validate the diagnostic performance of anti-BNLF2b antibody for detecting nasopharyngeal carcinoma (NPC) compared with healthy controls (HC).
Methods
We conducted a retrospective study including 220 patients with NPC, 61 with tongue cancer (TC), and 88 HC patients. We collected demographic and clinical data, including anti-BNLF2b antibody, EBV DNA, VCA-IgA, EBNA1-IgA, and Rta-IgG. Propensity score matching (PSM) was used to balance baseline characteristics between NPC and comparison groups. Associations between biomarkers and NPC diagnosis were examined using logistic regression. Diagnostic performance was evaluated using receiver operating characteristic (ROC) analysis.
Results
After PSM, 88 patients with NPC were matched to 88 HC with balanced baseline characteristics. Anti-BNLF2b antibody levels were significantly higher in NPC and remained independently associated with NPC diagnosis. For NPC versus HC, anti-BNLF2b antibody showed excellent discrimination (AUC = 0.990; sensitivity 98.9%; specificity 92.0% at a cut-off of 0.210), exceeding the performance of the EBV dual antibody risk probability (AUC: 0.872; P < 0.001). In addition, patients with NPC had higher anti-BNLF2b antibody levels and other EBV-related markers than those with TC.
Conclusion
In this retrospective study, anti-BNLF2b antibody demonstrated excellent discrimination for NPC. It may serve as a complementary serologic marker, pending external validation and prospective assessment of clinically optimized cutoffs.
Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumor arising from the epithelial cells of the nasopharynx, with a notably uneven global distribution. It is especially prevalent in East and Southeast Asia, indicating geographical and ethnic predisposition.1,2 NPC often shows aggressive behavior with early lymphatic spread. Unfortunately, due to its often insidious onset and nonspecific early symptoms, diagnosis frequently occurs at an advanced stage. More than 70% of patients present with late-stage disease at diagnosis, which worsens treatment outcomes and overall prognosis. 3
Epstein-Barr virus (EBV), also known as human herpesvirus 4 (HHV-4), is a member of the Herpesviridae family and is among the most prevalent human viruses. 4 EBV is linked to several malignancies, including lymphomas and epithelial carcinomas; however, its strongest link is with undifferentiated NPC. 5 Extensive research has identified multiple EBV-related serologic markers that aid NPC diagnosis and prognosis. Among conventional serological tests, the combination of viral capsid antigen IgA (VCA-IgA) and Epstein-Barr nuclear antigen 1 (EBNA-1) IgA is widely used for NPC screening, although conventional methods have notable limitations. 6 In a pivotal two-stage study from southern China, Liu et al reported that a dual-marker model combining VCA-IgA and EBNA1-IgA achieved high diagnostic accuracy in a retrospective case-control cohort. However, in a subsequent real-world mass screening of more than 5000 asymptomatic individuals, the same panel showed a lower sensitivity of 75%, which points to a gap between validation and implementation. 7 In high-risk groups such as multiplex families, EBNA1-IgA reaches a sensitivity of approximately 80%, but specificity remains suboptimal, with nearly 40% of non-NPC individuals testing positive, resulting in a high false-positive burden. 8 To address these limitations, new EBV-associated biomarkers have been explored. Recent studies have identified anti-BNLF2b antibody as a promising candidate. It targets the BNLF2b protein, an EBV protein that contributes to immune evasion. 9
Advanced NPC presents with localized pain, mass formation, and cervical lymphadenopathy, which may mimic other malignancies of the head and neck, particularly tongue cancer (TC).10,11 Despite anatomical proximity and overlapping symptoms, NPC and TC have distinct etiologies. TC is largely driven by environmental carcinogens, including tobacco and alcohol.11,12 This divergence between the two diseases underscores the need for highly specific and sensitive diagnostic biomarkers that can distinguish these entities, especially in early-stage presentations where clinical findings are nonspecific.
In this retrospective study, we aimed to validate the clinical utility of anti-BNLF2b antibody for distinguishing NPC from healthy controls, and to benchmark its performance against established EBV-based strategies, including the dual-antibody risk probability and plasma EBV DNA. We also performed an exploratory analysis comparing NPC with TC.
Methods
Study Population
This retrospective study included consecutive individuals who presented with suspected NPC at initial evaluation between January and December 2024. Final diagnoses were established by nasopharyngoscopy and histopathology. Based on the confirmed diagnosis, patients were assigned to the NPC group or the TC group. In parallel, a HC cohort was drawn from individuals who underwent routine health examinations during the same period and met predefined eligibility criteria. Complete clinical and serologic data were available for all participants.
Inclusion criteria for the NPC group were: (1) NPC confirmed by nasopharyngoscopy and histopathology; (2) age over 18 years; (3) no prior antitumor therapy; and (4) complete clinical and laboratory data. Inclusion criteria for TC group were: (1) age over 18 years; (2) no prior antitumor therapy; (3) no symptoms or diagnosis of EBV-associated conditions; and (4) complete clinical and serological data. Inclusion criteria for the HC group were: (1) no history of malignancy or chronic disease; (2) age over 18 years; (3) no symptoms or diagnosis of EBV-associated conditions; and (4) availability of complete clinical and serological data. Individuals with a history of other malignancies, autoimmune diseases, or severe infections were excluded from all groups.
All patient information was fully de-identified before analysis to prevent re-identification. The study was approved by the institutional review board of our hospital, and informed consent was waived by the Medical Ethics Committee due to the retrospective use of de-identified data. HCs were drawn from the institutional health-examination cohort. At the time of examination, all participants signed a general authorization permitting secondary research use of residual specimens and de-identified data. Reporting adheres to the STROBE guidelines.13,14
Data Collection
We collected demographic variables, including age, sex, smoking history, alcohol consumption, and family history of NPC. For patients with NPC or TC, we additionally recorded diagnosis type, clinical stage, and TNM classification.
Anti-BNLF2b antibodies, along with traditional biomarkers such as VCA-IgA, EBV-DNA, EBNA1-IgA, and Rta-IgG, were detected. Anti-BNLF2b antibody levels were measured using the Wan200 + system with a chemiluminescent microparticle immunoassay kit specific for EBV BNLF2b (Xiamen Innodx Biotechnology Co., Ltd, Xiamen, China). 9 EBV DNA was quantified by fluorescence probe-based real-time PCR using the EBV nucleic acid detection kit (Sansure Biotech Inc., Changsha, China). Positivity was defined as greater than 400 copies/mL because values below 400 copies/mL were reported qualitatively as negative without a numeric result. 15 VCA-IgA, EBNA1-IgA and Rta-IgG levels were assessed using enzyme-linked immunosorbent assays (ELISA) with respective antibody detection kits provided by Tracine BioMed Inc. (Beijing, China). 16
The risk probability for NPC based on VCA-IgA and EBNA1-IgA was calculated using the published logistic model: logit(P) = –3.934 + 4.797 × EBNA1-IgA + 2.203 × VCA-IgA(9).
Statistical Analysis
Statistical analyses were performed using SPSS version 26.0 (IBM Corp, Armonk, NY, USA) and R version 4.3.3 (R Foundation for Statistical Computing, Vienna, Austria). Continuous variables were summarized as mean ± standard deviation (SD) for normally distributed data, and as median with interquartile range (IQR) for non-normally distributed data. Between-group comparisons for continuous variables used independent-samples t tests for normal distributions and the Mann-Whitney U test for non-normally distribution. Categorical variables were summarized as counts and percentages and compared using the chi-square test. Two prespecified comparisons were performed: NPC versus HC and NPC versus TC. The primary diagnostic accuracy analysis compared NPC with HC to quantify associations between biomarkers and NPC diagnosis and to estimate discrimination. To reduce confounding, we applied 1:1 propensity score matching (PSM) on age, sex, family history of NPC, and other baseline covariates that differed before matching using R. Balance was assessed using standardized mean differences. The exploratory analysis compared NPC with TC to contextualize the tumor-type specificity of EBV-related markers within head and neck oncology. Associations between biomarker levels and NPC diagnosis were evaluated using logistic regression and reported as odds ratios (ORs) with 95% confidence intervals (CIs). A combined diagnostic model incorporating the anti-BNLF2b antibody and EBV dual antibody risk probability was constructed using multivariate logistic regression. Diagnostic performance of biomarkers and the combined model was evaluated using receiver operating characteristic (ROC) analysis with MedCalc 23.2.6. Sensitivity, specificity, and the area under the ROC curve (AUC) were calculated to compare the diagnostic efficacy, and AUCs were compared using the DeLong test. A p-value < 0.05 was considered statistically significant.
Results
Demographic and Clinical Characteristics
After applying the eligibility criteria, a total of 220 NPC patients and 88 HC subjects were enrolled (Figure 1). Compared with the HC group, the NPC group was older and had a higher prevalence of family history of NPC (P < 0.05). To reduce confounding, 1:1 PSM was conducted for NPC versus HC (n = 88 per group), ensuring comparable baseline characteristics. Demographic and clinical characteristics before and after matching are detailed in Table 1. After PSM, EBV DNA positivity, anti-BNLF2b antibody levels, the EBV dual-antibody risk probability, and Rta-IgG levels were significantly higher in the NPC group than in the HC group (P < 0.001) (Table 1).
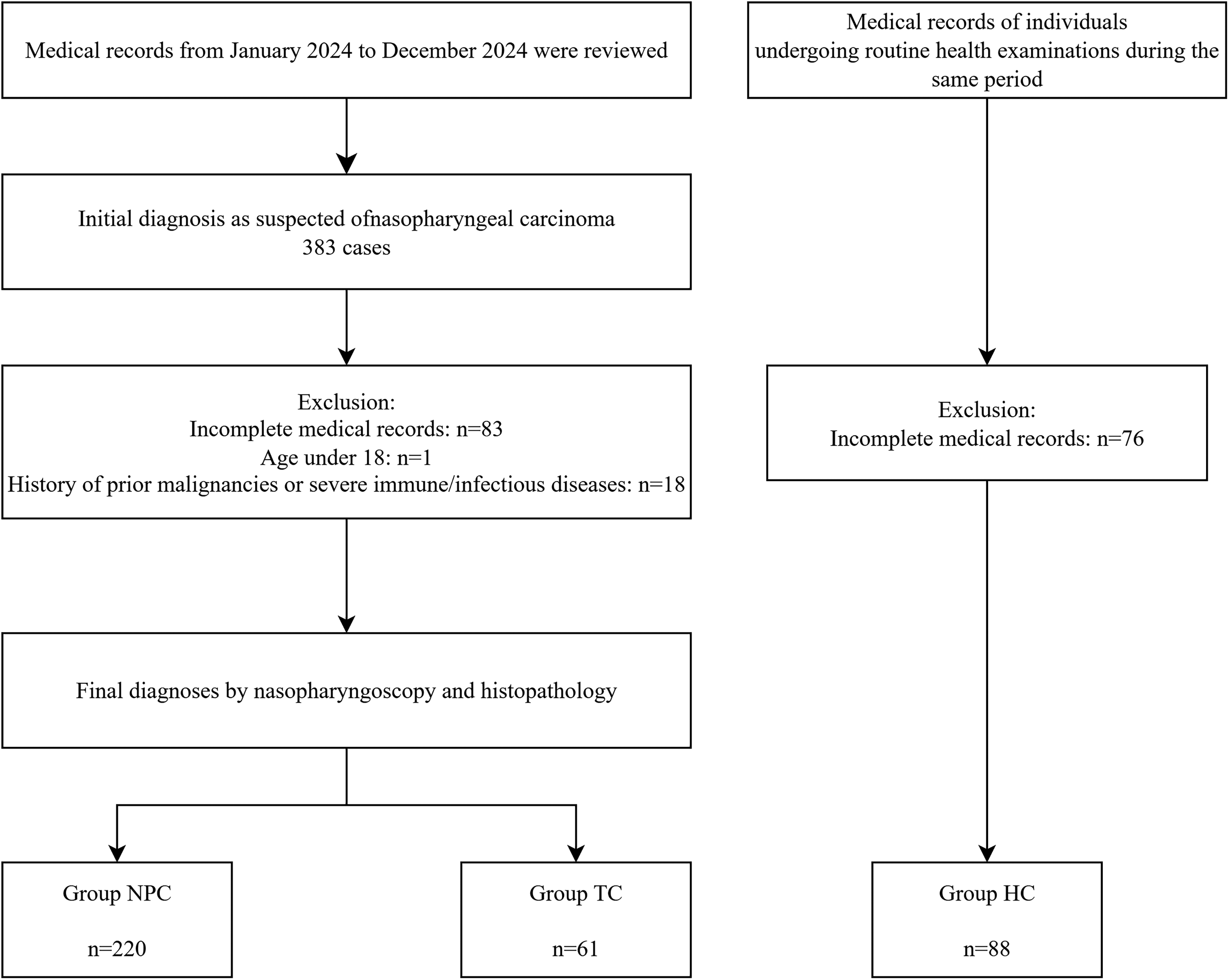
Patient selection flowchart.
Comparison of Clinical and Demographic Characteristics Between NPC and HC Groups.
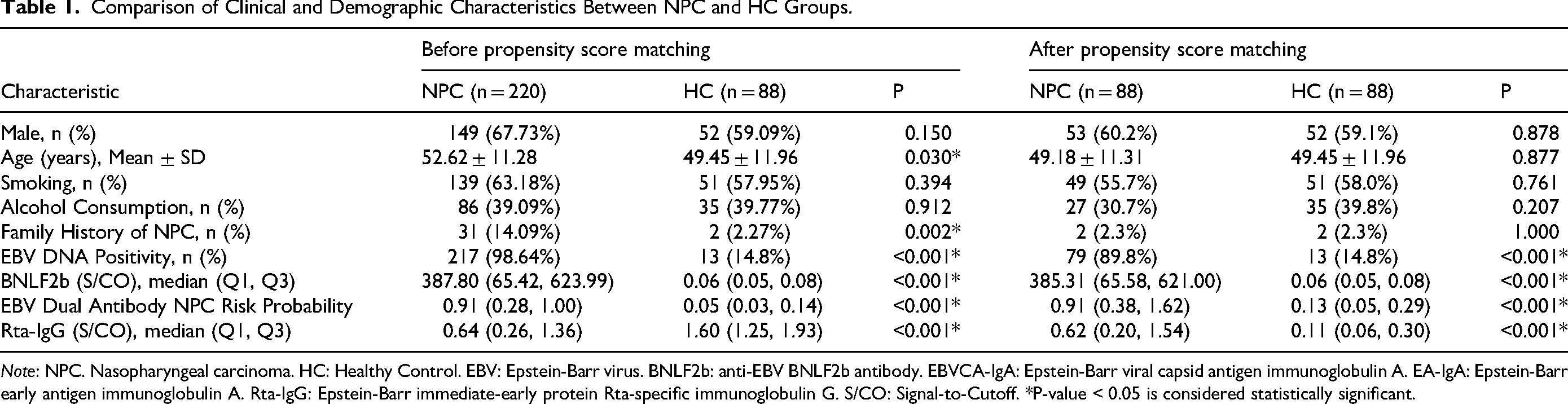
Note: NPC. Nasopharyngeal carcinoma. HC: Healthy Control. EBV: Epstein-Barr virus. BNLF2b: anti-EBV BNLF2b antibody. EBVCA-IgA: Epstein-Barr viral capsid antigen immunoglobulin A. EA-IgA: Epstein-Barr early antigen immunoglobulin A. Rta-IgG: Epstein-Barr immediate-early protein Rta-specific immunoglobulin G. S/CO: Signal-to-Cutoff. *P-value < 0.05 is considered statistically significant.
Association of Biomarkers with NPC Diagnosis
Multivariate logistic regression was performed to evaluate association between biomarkers and NPC diagnosis (Table 2). In the comparison between NPC and HC, high anti-BNLF2b antibody levels (OR = 72.093, 95% CI: 6.749-770.059, P < 0.001) and high EBV dual antibody risk probability (OR = 20.575, 95% CI: 2.331-181.621, P = 0.006) were strongly associated with NPC diagnosis. EBV DNA positivity and Rta-IgG levels were not significantly associated with NPC diagnosis. (P > 0.05).
Multivariate Logistic Regression.

Note: NPC. Nasopharyngeal carcinoma. EBV: Epstein-Barr virus. BNLF2b: anti-EBV BNLF2b antibody. Rta-IgG: Epstein-Barr immediate-early protein Rta-specific immunoglobulin G. *P-value < 0.05 is considered statistically significant.
Diagnostic Performance of Biomarkers
We evaluated the diagnostic performance of prespecified biomarkers using ROC analysis (Table 3; Figure 2). In distinguishing NPC from HC, the anti-BNLF2b antibody alone demonstrated excellent diagnostic accuracy, with an AUC of 0.990 (95% CI: 0.961-0.999), which was significantly higher than that of the EBV dual antibody risk probability (AUC = 0.872, 95% CI: 0.814-0.918; P < 0.001). A combined model incorporating the anti-BNLF2b antibody and the EBV dual antibody demonstrated superior discrimination, with an AUC of 0.991 (95% CI: 0.964-0.999). However, this was not significantly different from the anti-BNLF2b antibody alone (P = 0.578).
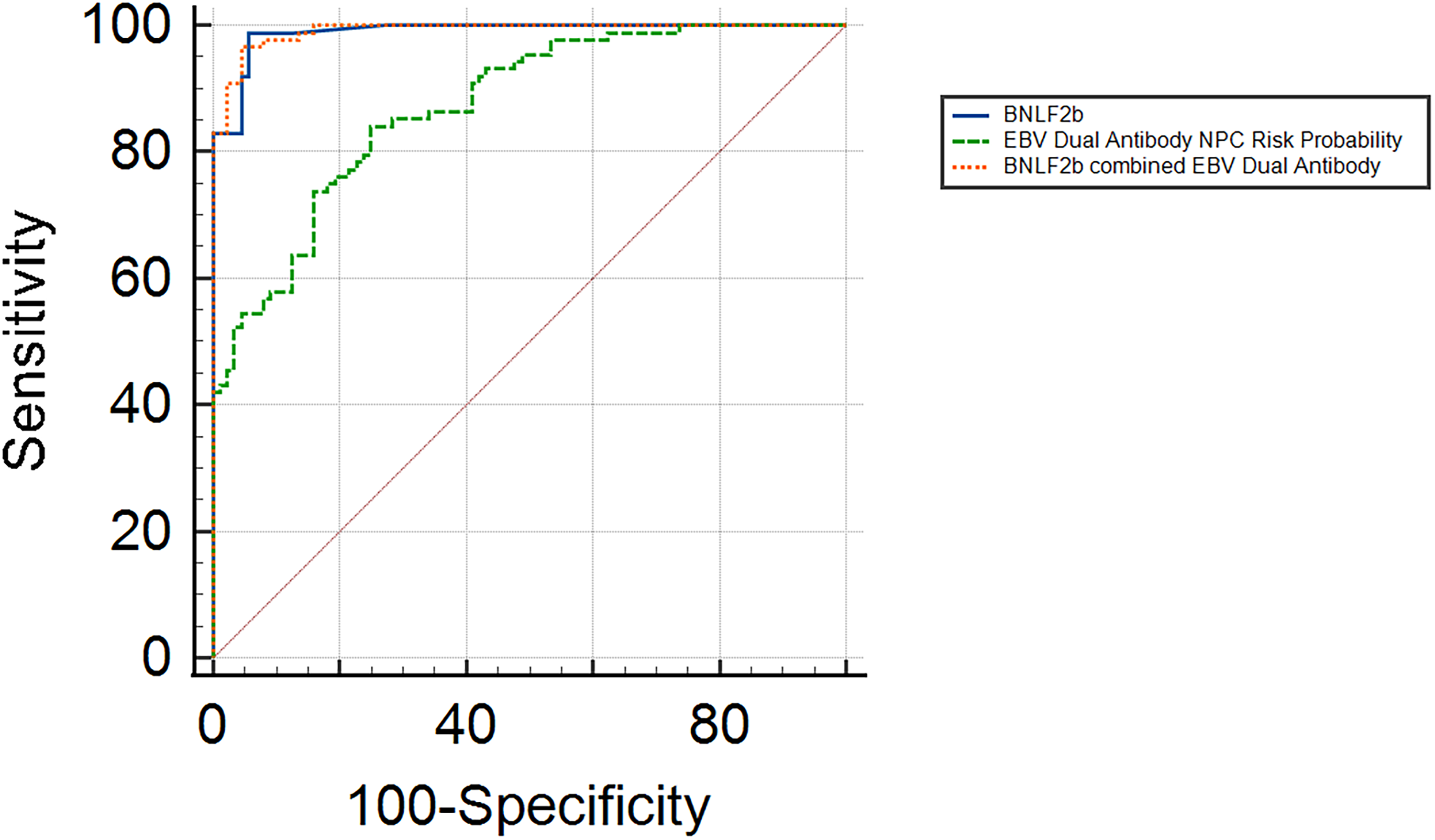
Comparison of ROC curves for differentiating nasopharyngeal carcinoma from healthy controls.
Comparison of Biomarker Diagnostic Accuracy in NPC Versus HC.

Note: NPC. Nasopharyngeal carcinoma. HC: Healthy Control. EBV: Epstein-Barr virus. BNLF2b: anti-EBV BNLF2b antibody.
Anti-BNLF2b Antibody Distributions Across Clinical Stage and Metastasis
Among patients with available anti-BNLF2b antibody and staging information (n = 220 for tumor stage; n = 234 for nodal/metastatic status), anti-BNLF2b antibody levels did not differ significantly across tumor stages (1-4) or nodal stages (0-3) (Kruskal–Wallis H = 4.734, df = 3, p = 0.192; H = 1.738, df = 3, p = 0.628), and no monotonic trends were detected (Jonckheere–Terpstra Z = −1.385, p = 0.166; Z = −0.185, p = 0.854). By metastatic status, anti-BNLF2b antibody distributions differed significantly (Mann–Whitney U = 4860, Z = −2.119, p = 0.034), although the effect size was small (r = 0.139).
Exploratory Tongue Cancer Analysis
61 TC patients were included in this study. Compared with TC, the majority of NPC patients had non-keratinizing carcinoma, while all TC patients had squamous carcinoma (P < 0.001). NPC cases also presented with more advanced clinical stage (P = 0.001), greater nodal involvement (P < 0.001), and a higher proportion with distant metastasis (P < 0.001). EBV-related markers were consistently higher in NPC than in TC, including EBV DNA positivity, anti-BNLF2b antibody, and Rta-IgG (P < 0.001). Likewise, the EBV dual antibody risk probability was higher in NPC patients (P < 0.001) (Table 4). A family history of NPC was also more common in NPC than in TC (P < 0.05).
Comparison of Clinical and Demographic Characteristics Between NPC and TC Groups.

Note: NPC. Nasopharyngeal carcinoma. TC: Tongue cancer. EBV: Epstein-Barr virus. BNLF2b: anti-EBV BNLF2b antibody. EBVCA-IgA: Epstein-Barr viral capsid antigen immunoglobulin A. EA-IgA: Epstein-Barr early antigen immunoglobulin A. Rta-IgG: Epstein-Barr immediate-early protein Rta-specific immunoglobulin G. S/CO: Signal-to-Cutoff. *P-value < 0.05 is considered statistically significant.
Discussion
In this retrospective, propensity score-matched analysis, anti-BNLF2b antibody showed excellent diagnostic performance for NPC diagnosis and remained independently associated with NPC after multivariable adjustment. These results support anti-BNLF2b antibody as a strong complement to existing EBV-based approaches for screening and diagnosis.
In our PSM analysis, anti-BNLF2b antibody remained independently associated with NPC in multivariable models, and showed excellent diagnostic performance. These findings are consistent with recent large-scale evidence showing that anti-BNLF2b antibody provides high diagnostic accuracy and improves positive predictive value in NPC screening workflows. 9 In a prospective cohort, anti-BNLF2b antibody achieved 97.9% sensitivity and 98.3% specificity with a positive predictive value of 10.0%, outperforming legacy dual-antibody algorithms. 9 Another study reported an AUC of 0.951, closely aligning with our ROC estimates. 17 For the EBV dual-antibody-based probability metric, our cohort showed moderate discrimination, a pattern consistent with population-dependent specificity fluctuations reported previously.6,18 Although dual-antibody testing (VCA-IgA and EBNA1-IgA) is widely used in current practice, our results indicated that anti-BNLF2b antibody achieved higher overall accuracy in this setting, supporting its role as a strong complementary marker within EBV-based strategies. By contrast, plasma EBV DNA positivity differed markedly between groups after matching, but was not independently associated with NPC in multivariable models. This aligns with prior prospective work indicating that EBV DNA is best used for longitudinal risk tracking or early event prediction rather than as a universal standalone serologic screen.19,20
Prior studies have suggested that patients with NPC who undergo radiotherapy may develop radiation-induced TC as a secondary malignancy.21,22 Because our enrollment mirrored the real-world diagnostic pathway for suspected NPC, a subset of participants received a final diagnosis of TC, providing a practical disease-control group for biomarker comparisons. In our cohort, several factors significantly differed between NPC and TC. A positive family history of NPC was more frequent among NPC patients, whereas the TC group showed no such connection. Consistent with our observation, large epidemiologic studies have shown that a first-degree family history confers a substantially elevated risk of NPC, exceeding fourfold in a population-based case-control study from southern China. 23 Evidence from multiplex-family cohorts further supports familial co-aggregation, and contemporary reviews estimate an approximately four- to twentyfold increase among first-degree relatives.24,25 Pathologically, NPC is predominantly a non-keratinizing carcinoma, whereas TC is exclusively a squamous cell carcinoma strongly associated with smoking, alcohol consumption, and human papillomavirus infection.26,27 In terms of clinical progression, NPC is often diagnosed at a more advanced stage than TC, with a higher likelihood of lymph node involvement and distant metastasis at diagnosis. This pattern aligns with NPC's strong propensity for early lymphatic spread and distant dissemination. 28 Consequently, earlier and more precise diagnostic approaches are crucial for improving survival rates and treatment outcomes in NPC patients. However, since most patients in our cohort were diagnosed at later stages, we were unable to fully evaluate the dynamic changes in anti-BNLF2b antibody levels across different NPC stages. From a biomarker perspective, compared with TC, NPC showed markedly higher EBV DNA positivity, elevated anti-BNLF2b antibody levels, higher dual-antibody NPC risk probability, and increased Rta-IgG. Taken together, these findings indicate that EBV-linked serology, particularly anti-BNLF2b antibody, reflects EBV-driven NPC biology and is not elevated in TC. Although TC and NPC are usually distinguishable in routine practice, scenarios such as cervical nodal metastasis with an unknown primary or overlapping histomorphology can occur, in which EBV-related serologic markers may provide supportive clues to the primary site. However, our dataset did not include sufficient cases to evaluate this application, and no further analyses were performed. NPC is often diagnosed at a late stage due to its insidious onset and non-specific symptoms. 29 Given that the majority of our cohort consisted of patients with advanced-stage disease, we further investigated whether circulating anti-BNLF2b antibodies could serve as a proxy for anatomical tumor burden. Stratified analyses revealed no significant differences or ordinal trends in anti-BNLF2b levels across T1-T4 or N0-N3 stages. A small difference was observed by metastatic status, there was substantial overlap in antibody levels across groups, but distributions overlapped substantially, suggesting that anti-BNLF2b antibody may reflect host-tumor immune interactions driven by EBV, rather than providing a linear correlate of anatomical tumor volume. However, due to the imbalance in clinical staging and considerable within-group variability, these findings are exploratory and likely underpowered.
Despite the promising findings, this study had certain limitations. First, as a retrospective observational study, potential selection bias and confounding factors could not be eliminated. Second, although 1:1 matching was used for NPC versus HC to balance baseline characteristics, independent prospective validation in external cohorts is still needed. Third, spectrum effects may be present because most NPC cases were treated at a tertiary cancer center with stage distribution skewed toward advanced disease, whereas healthy controls were drawn from a health-examination cohort; these features may overestimate diagnostic separability compared with real-world triage populations. Moreover, the overall sample size, particularly within subgroup analyses, limited statistical power to detect small to moderate effects; consequently, non-significant results should be interpreted with caution, as suggested by the wide 95% confidence intervals. Another limitation is that EBV DNA values below 400 copies/mL were reported only as negative due to assay limitations, which prevented finer analysis at low viral loads. Additionally, longitudinal follow-up was not available, so we could not assess whether anti-BNLF2b antibody levels correlate with disease progression, recurrence, or treatment response. Future studies should be prospective and multicenter with larger cohorts, use higher-sensitivity assay platforms, and standardize prospective sample handling to enable fine-grained analyses of low-level viremia and to establish the long-term clinical utility of this biomarker.
Conclusion
Anti-BNLF2b antibody showed high discriminatory performance for NPC in this retrospective cohort. The EBV dual-antibody NPC risk probability, though associated, had lower accuracy and specificity. Exploratory comparisons with TC indicated that EBV-linked serology, particularly anti-BNLF2b antibody, is not elevated in TC, supporting its role as a high-specificity complement to existing EBV-based strategies. However, these findings should be interpreted with caution given the study's observational design and modest sample size, and they require confirmation in prospective, multicenter cohorts with external validation and standardized assay procedures.
Supplemental Material
sj-pdf-1-tct-10.1177_15330338251412015 - Supplemental material for Evaluation of Anti-BNLF2b Antibody and Epstein–Barr Virus Biomarkers for the Diagnosis of Nasopharyngeal Carcinoma: A Retrospective Study
Supplemental material, sj-pdf-1-tct-10.1177_15330338251412015 for Evaluation of Anti-BNLF2b Antibody and Epstein–Barr Virus Biomarkers for the Diagnosis of Nasopharyngeal Carcinoma: A Retrospective Study by Hongyu Deng, Qinglin Liu, Haoming Shen and Ping Xiao in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338251412015 - Supplemental material for Evaluation of Anti-BNLF2b Antibody and Epstein–Barr Virus Biomarkers for the Diagnosis of Nasopharyngeal Carcinoma: A Retrospective Study
Supplemental material, sj-docx-2-tct-10.1177_15330338251412015 for Evaluation of Anti-BNLF2b Antibody and Epstein–Barr Virus Biomarkers for the Diagnosis of Nasopharyngeal Carcinoma: A Retrospective Study by Hongyu Deng, Qinglin Liu, Haoming Shen and Ping Xiao in Technology in Cancer Research & Treatment
Footnotes
Acknowledgements
We thank Xiamen Innodx Biotechnology Co., Ltd for providing the BNLF2b antibody detection kit.
Ethics Approval
This study was approved by the Medical Ethics Committee of Hunan Cancer Hospital (KY2025582).
Consent to Participate
Due to the retrospective nature of the study, consent to participate was waived by the Medical Ethics Committee of Hunan Cancer Hospital.
Author Contributions
H.D. conceived the study and acquired funding. Q.L. curated the data. Q.L., H.S., and P.X. performed the formal analyses. H.S. prepared the visualizations. H.D. drafted the original manuscript. Q.L., H.S., and P.X. critically reviewed and edited the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Hunan Province of China (Grant numbers [S2024JJQYLH0226]).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
