Abstract
Objective
To systematically investigate the impact of adjusting the relative weight of the built-in Stereotactic Radiosurgery Normal Tissue Objective (SRS-NTO) on dosimetric quality, plan complexity, and delivery efficiency in HyperArc™ stereotactic radiosurgery (SRS) for brain oligometastases.
Methods
In this retrospective planning study, a cohort of 20 patients with 1-3 brain oligometastases was analyzed. For each case, six distinct HyperArc plans were designed and optimized using the Varian Eclipse™ Treatment Planning System. To precisely isolate its impact, the relative weight of the SRS-NTO to the PTV objective was systematically varied across six levels—50%, 75%, 100% (default), 125%, 150%, and 200%—while all other planning parameters were held constant. A comprehensive comparative evaluation was then performed to assess the plans across four key domains: (i) dosimetric quality, evaluated by metrics including the Paddick Conformity Index (CI), Gradient Index (GI), and dose to Organs at Risk (OARs); (ii) plan complexity, characterized by various modulation and aperture-based indices; (iii) delivery efficiency, primarily quantified by the total Monitor Units (MUs); and (iv) physical deliverability, verified via Gamma analysis.
Results
Increasing NTO weight did not significantly alter dosimetric quality; key metrics for CI, GI, and OAR sparing remained statistically equivalent (p > .05). Conversely, higher NTO weights prompted a significant reduction in total MUs (p < .001) that reached an optimum at the 150% setting, and enhanced plan deliverability, evidenced by significantly higher Gamma passing rates under stricter verification criteria. An inflection point was observed beyond the 150% setting, with higher weights leading to degraded plan complexity and efficiency. Strategies within the 125% to 150% range demonstrated a superior balance, achieving optimal dosimetric trends while maximizing gains in efficiency and precision.
Conclusion
In HyperArc SRS for brain oligometastases, moderately increasing the SRS-NTO weight from the default 100% into the 125% to 150% range is a superior clinical strategy. This adjustment significantly enhances treatment efficiency and delivery precision by reducing plan complexity, without compromising dosimetric quality, thereby achieving a superior overall performance.
Introduction
In Stereotactic Radiosurgery (SRS) for brain metastases, achieving an optimal balance between the delivery of an ablative dose to the target and the protection of surrounding normal brain tissue is paramount for determining clinical efficacy and safety.1–3 Specifically, the creation of a steep dose fall-off gradient at the target edge is crucial for mitigating the risk of severe complications such as Radiation Necrosis (RN).4–6 The Varian HyperArc™ technology, a highly integrated SRS solution, has significantly improved the quality and efficiency of treatment planning through its automated non-coplanar trajectory design and built-in algorithms.7–9
Among the various optimization tools, the Normal Tissue Objective (NTO) is a key function for actively shaping the dose gradient.10,11 However, the application of existing NTO strategies is constrained by notable limitations. While manual NTO (mNTO) offers high flexibility, its optimization process heavily relies on the dosimetrist's experience and is difficult to standardize due to its vast parameter space, leading to uncertainty in plan quality. Conversely, conventional auto-NTO, though efficient, often demonstrates dosimetrically inferior performance compared to meticulously tuned mNTO in complex clinical scenarios.10,12
The Stereotactic Radiosurgery-specific NTO (SRS-NTO) integrated into the HyperArc technology is a more advanced automated tool. It applies spatially-weighted constraints based on target geometry to efficiently suppress dose bridging between multiple targets and enhance the dose gradient.7,9 Although SRS-NTO largely improves plan quality and efficiency, its optimization weight—its priority relative to other objectives like target coverage—is typically set to the system-recommended default.7,10 While robust, this default value may not represent the optimal solution for achieving the best balance between “efficacy” and “safety” in all situations. Recent studies have shown that non-coplanar RapidArc plans, utilizing more flexible beam paths and optimized mNTO, can even outperform HyperArc plans in certain key normal tissue-sparing metrics, indirectly suggesting that optimization strategies within the HyperArc framework warrant further exploration and enhancement.13–15 An aggressive NTO weight might compromise target dose homogeneity and increase plan modulation complexity and monitor units (MUs), whereas a conservative weight might fail to achieve ideal normal tissue sparing. Therefore, the SRS-NTO weight setting itself represents a critical yet under-investigated trade-off point.
This study aims to systematically investigate the quantitative impact of the SRS-NTO's relative weight on the dosimetric quality, plan complexity, and delivery efficiency of SRS plans for brain oligometastases within the Varian HyperArc framework. We hypothesize that an optimal weight range exists, within which the dose gradient can be further improved and normal brain tissue dose can be reduced without unduly sacrificing target coverage quality or significantly increasing treatment delivery complexity. The goal of this research is to provide evidence-based guidance for the refined adjustment of SRS-NTO parameters in clinical practice, ultimately aiming to maximize the therapeutic ratio of HyperArc for brain metastases.
Materials and Methods
Patient Cohort and Ethical Statement
This retrospective analysis was approved by the local ethics committee, and all procedures adhered to the ethical principles of the Declaration of Helsinki. Anonymized data from 20 patients with brain oligometastases who had previously undergone HyperArc™ SRS were included. Inclusion criteria were: (1) diagnosis of solid tumor brain metastasis confirmed by histology or definitive imaging, with completed radiotherapy; (2) 1 to 3 intracranial metastases confirmed by pre-treatment magnetic resonance imaging; (3) availability of complete planning CT and MRI data for plan redesign; and (4) a maximum diameter of any single lesion not exceeding 4 cm, and a planning target volume (PTV) not exceeding 25 cm³; (5) a Karnofsky Performance Status (KPS) score of ≥70. Exclusion criteria included: (1) severe artifacts in planning CT images caused by metallic implants (eg, dental fillings) that hindered accurate delineation of organs at risk (OARs); and (2) any single metastasis with a volume exceeding 25 cm³. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 16 All patient details were de-identified to protect patient privacy. A summary of the patient and lesion characteristics is provided in Table 1.
Baseline Patient and Tumor Characteristics (n = 20).

*A total of 37 lesions were analyzed across the 20 patients. Percentages for location are calculated based on the total number of lesions. Lesion dimensions were measured based on the maximal 3D diameter of the Gross Tumor Volume (GTV) on post-contrast T1-weighted MRI.
Radiotherapy Equipment and Software
Patient positioning and CT simulation were performed using a Philips Brilliance™ Big Bore 16-slice CT scanner (Philips Healthcare, Amsterdam, Netherlands). All treatment planning and optimization were conducted on the Varian Eclipse™ Treatment Planning System (TPS) v15.6 (Varian Medical Systems, Palo Alto, CA, USA). The HyperArc™ planning module (v15.5) was utilized for the automated generation of non-coplanar treatment plans. All plans were designed for delivery via a Varian TrueBeam™ 2.7 linear accelerator. Plan-specific quality assurance was performed using the accelerator's integrated aSi-1000 amorphous silicon electronic portal imaging device (EPID), with Gamma analysis conducted via Portal Dosimetry™ software (v15.6, Varian Medical Systems, Palo Alto, CA, USA).
Treatment Planning
For each patient, six new HyperArc plans were generated, maintaining the original target delineations (GTV, PTV) and OAR constraints. The sole variable among these plans was the relative weight of the built-in SRS-NTO compared to the PTV optimization objective, which was set to 50%, 75%, 100% (system default), 125%, 150%, and 200%. The resulting plans were named NTO_50%, NTO_75%, NTO_100%, NTO_125%, NTO_150%, and NTO_200%, respectively.
To ensure a fair comparison, all plans were generated in a single optimization run using a standardized template and were normalized to a prescription dose of 27 Gy delivered in 3 fractions (27 Gy / 3f), such that 98% of the Planning Target Volume (PTV) (D98%) received 27 Gy. This hypofractionated regimen is a common clinical approach for brain metastases of the size range included in our cohort. All other optimization parameters were kept strictly identical. Key technical parameters were set as follows: a dose calculation and optimization grid resolution of 1.25 mm; the Acuros™ XB (AXB) algorithm v15.6 for dose calculation; a 6 MV flattening filter-free (FFF) beam energy; and a dose rate of 1400 MU/min. The HyperArc module automatically generated four standard non-coplanar arcs with system-optimized collimator angles. Convergence Mode was enabled during optimization to ensure adequate target coverage under varying NTO constraints.
Dosimetric Evaluation
PTV Dosimetric Indices
Based on RTOG guidelines and relevant literature,17–19 the following indices were evaluated:
Gradient Index (GI): GI = V0.5RX/VRX, where V0.5Rx is the volume covered by 50% of the prescription isodose. It quantifies the dose fall-off steepness outside the target, with lower values being better.
D2cm: The maximum point dose in normal brain tissue at a 2 cm distance from the PTV edge, assessing high-dose exposure in the near-target region.
Additionally, the mean dose to the PTV (Dmean),D99.9 (the minimum dose received by 99.9% of the PTV, assessing cold spots), and D0.1 (the minimum dose received by 0.1% of the PTV, assessing hot spots) were assessed.
OAR Dosimetric Indices
The maximum point dose (D0.03cc) to critical OARs—including the brainstem, optic nerves, optic chiasm, lenses, and globes—and the V12Gy to the whole brain were recorded.
Isodose Distributions and Dose-Volume Histograms
A qualitative evaluation of each plan was performed through visual inspection. Isodose distributions were reviewed on representative axial, sagittal, and coronal slices, with a focus on the conformality of the high-dose (eg, 100% prescription isodose line) and low-dose (eg, 50% prescription isodose line) regions, as well as the efficacy of OAR sparing. The characteristics of the PTV's Dose-Volume Histogram (DVH) curve were also assessed.
High Dose Spillage and Delivery Efficiency
To specifically address high dose spillage, three additional metrics were evaluated: (1) the volume of the prescription isodose (27 Gy) outside the PTV (V27Gy_external); (2) the maximum dose within a 1-3 mm concentric ring structure created around the PTV (Dmax Ring_1-3mm); and (3) the maximum dose within a 3-5 mm ring (Dmax Ring_3-5mm). Delivery efficiency was quantified by total Monitor Units (MUs) and the total beam on time reported by the planning system.
All dosimetric parameters for PTV and OARs were batch-extracted using an in-house Python-based platform (Radiotherapy Dosimetry Omics Platform, RadDOP).
Plan Complexity and Treatment Efficiency Assessment
To quantify plan complexity and potential treatment efficiency, the following parameters were evaluated:

Calculated per arc as where is the

Calculated per control point as where


All complexity indices were calculated using an in-house C# script via the Eclipse Scripting API (ESAPI).
Plan Verification
All plans underwent delivery verification using the EPID. Gamma analysis24,25 was performed to assess the agreement between calculated and delivered doses, with passing rates evaluated at 3%/2 mm, 2%/2 mm, and 1%/1 mm criteria. It should be noted that the EPID-based verification utilizes a “Perpendicular Composite” methodology. While effective for the relative performance comparison in this study, we acknowledge the potential limitations of this method in detecting certain delivery errors, as highlighted by guidelines such as AAPM TG-218. 26
Statistical Analysis
Statistical analysis was performed using SPSS v26.0. Levene's test was used to assess the homogeneity of variances. One-way analysis of variance (ANOVA) was used for data with equal variances, while Welch's ANOVA was used for data with unequal variances. The Tukey's HSD (Honestly Significant Difference) test was used for post-hoc multiple comparisons. A two-tailed p-value <.05 was considered statistically significant.
Results
PTV Dosimetric Indices
The NTO weight strategies had a minimal impact on the core dosimetric quality of the PTV. As shown in the new Figure 1, no statistically significant differences were observed across the six NTO strategies for any of the PTV dosimetric indices, including Paddick CI, GI, D2cm, Dmean, D99.9, and D0.1 (F-values ranging from 0.043 to 1.619, all p > .05). However, despite the lack of statistical significance, a trend was observed where NTO_125% group exhibited the most favorable values for CI, GI, and D2cm (Figure 1).

Comparison of PTV dosimetric parameters (Paddick CI, GI, D2cm, Dmean, D1 and D0.1 ) for HyperArc plans under different NTO strategies.
High Dose Spillage
The evaluation of high dose spillage metrics (Figure 2) also revealed no significant differences among the groups. The external prescription dose volume (V27Gy_external) and the maximum doses in the 1-3 mm and 3-5 mm peritargetal rings were statistically equivalent across all plans (F-values ranging from 0.417 to 0.827, all p > .05)

Comparison of PTV High Dose Spillage for HyperArc plans under different NTO strategies.
OAR Dosimetric Parameters
Increasing the NTO weight did not systematically compromise the sparing of OARs. The D0.03cc for all evaluated OARs (brainstem, optic pathway, lenses, globes) and the critical parameter of whole brain V12Gy was also assessed (Figure 3). No significant differences were found for any OARs (F-values ranging from 0.001 to 0.074, all p > .05).
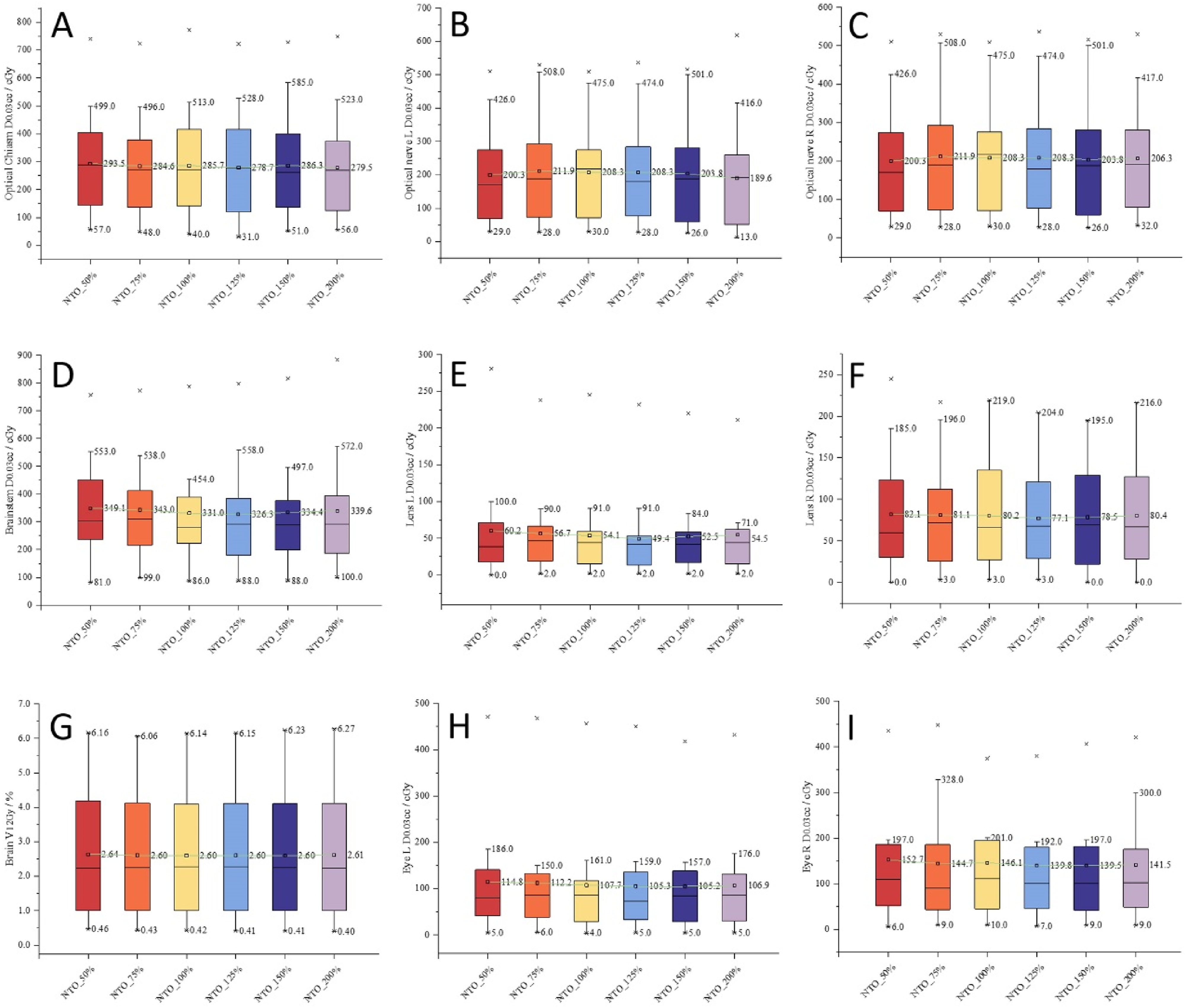
Comparison of OAR dosimetric parameters for HyperArc plans under different NTO strategies.
Plan Complexity and Delivery Efficiency
Modulation Complexity and Segment Number
Plan complexity indices, with data aggregated from all four arcs, showed varied results (Figure 4). While SN (F = 1.086, p > .05) and ALT (F = 0.357, p > .05) showed no significant differences (p > .05), a statistically significant difference was found for MCS (F = 8.232, p < .05). Post-hoc tests indicated that the NTO_150% group yielded the simplest plans. A clear trend was observed where complexity indices initially improved with increasing NTO weight, reached an optimum around the 150% setting, and then deteriorated in the 200% group. Furthermore, the Aperture Irregularity (AI) for a representative case is displayed in Figure 5. These plots illustrate a trend toward more regular aperture shapes (lower AI values) for the NTO_125% and NTO_150% strategies compared to the other groups.

Comparison of plan complexity parameters (SN, MCS, ALT) for HyperArc plans under different NTO strategies.
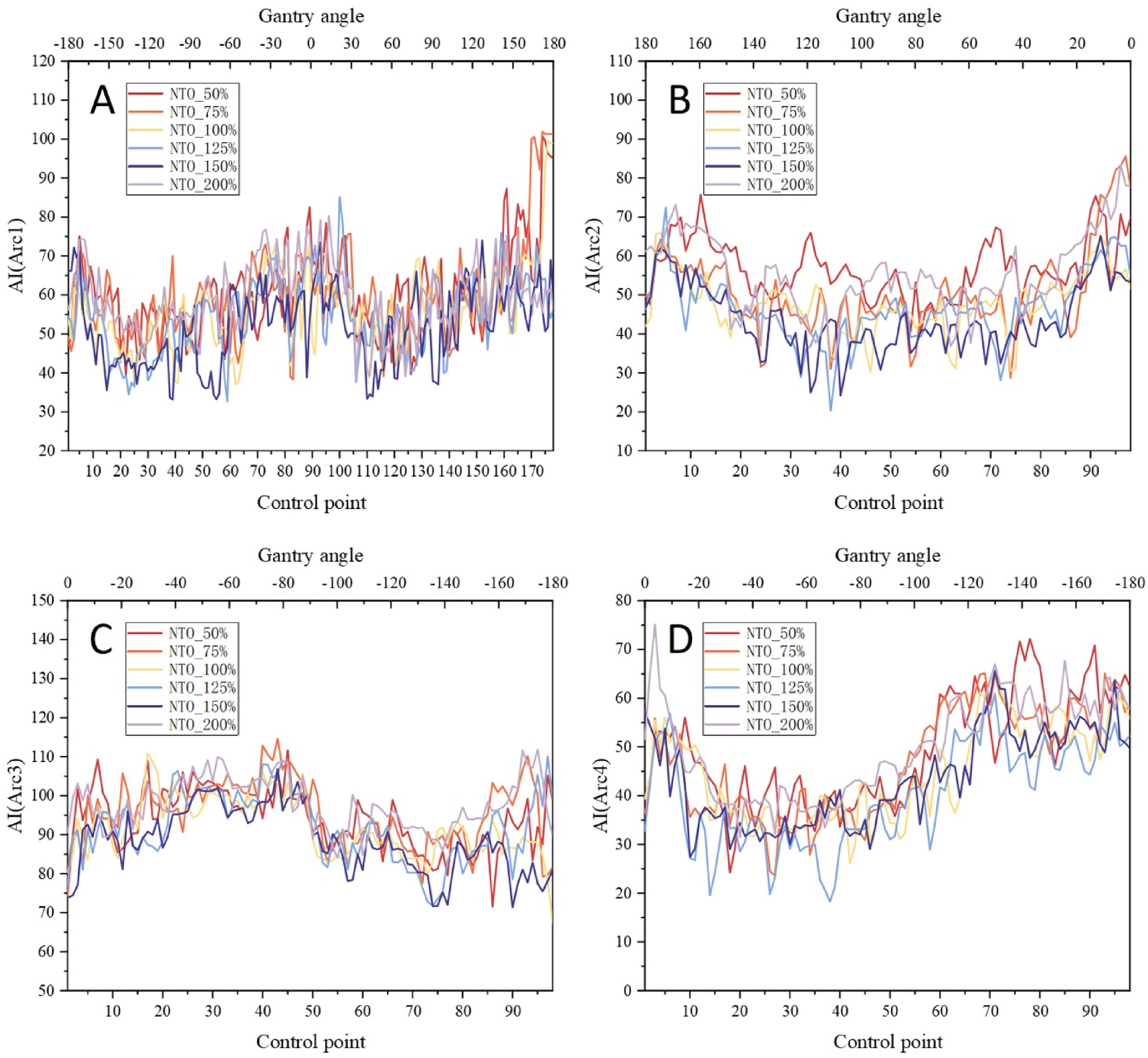
Aperture Irregularity (AI) for a representative case under different NTO strategies in HyperArc plans.
Delivery Efficiency
Delivery efficiency metrics demonstrated a strong and significant trend (Figure 6). Increasing the NTO weight led to a significant reduction in both total MUs (F = 18.385, p < .001) and beam on time (F = 19.688, p < .001) that reached a minimum at the 150% setting, which had the lowest mean values. Notably, the NTO_200% group exhibited higher MUs and longer beam-on times than the NTO_150% group, indicating a reversal of the efficiency gains. Post-hoc multiple comparisons using Tukey's HSD further elucidated the specific inter-group differences (Table 2 and Table 3). The NTO_150% group had the lowest MUs (2516.5 ± 309.4), representing an approximate 24% reduction compared to the NTO_50% group (3311.7 ± 353.3) (Figure 6).

Comparison of total Monitor Units (MUs) and Beam on time for HyperArc plans under different NTO strategies.
Post-Hoc Multiple Comparisons of Total MUs (Tukey's HSD Test).
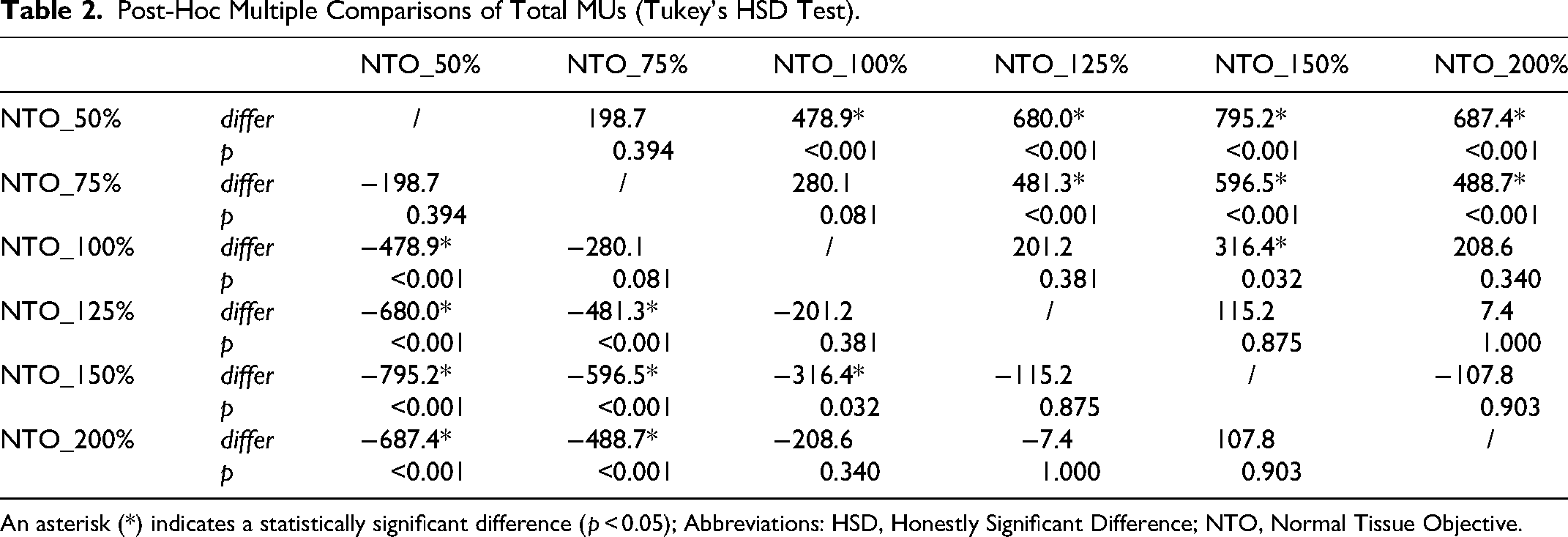
An asterisk (*) indicates a statistically significant difference (p < 0.05); Abbreviations: HSD, Honestly Significant Difference; NTO, Normal Tissue Objective.
Post-Hoc Multiple Comparisons of Beam on Time (Tukey's HSD Test).

An asterisk (*) indicates a statistically significant difference (p < 0.05); Abbreviations: HSD, Honestly Significant Difference; NTO, Normal Tissue Objective.
Plan Dose Verification
Gamma passing rates, with data aggregated from all arcs, showed a significant improvement with increased NTO weight (Figure 7). The passing rates at 2%/2 mm and 1%/1 mm criteria were significantly higher for the NTO_125% and NTO_150% groups compared to the default setting (F-values ranging from 2.198 to 36.949, p < .05). The improvement trend appeared to peak within the 125% and 150% settings, with a subsequent decline in performance for the NTO_200% group, which showed lower passing rates.

Comparison of Gamma passing rates for each treatment arc under different NTO strategies.
Isodose Line Distribution and DVH
Visual inspection of the isodose distributions for all plans confirmed that all strategies met clinical requirements for PTV coverage and OAR sparing, with highly comparable overall dose distributions. Only minor variations were observed in the high-dose region contours within the target and the precise location of the maximum dose point, none of which resulted in clinically unacceptable hot or cold spots. Furthermore,the DVHs also showed that the PTV mean dose peaked at the 150% NTO weight setting (Figure 8).
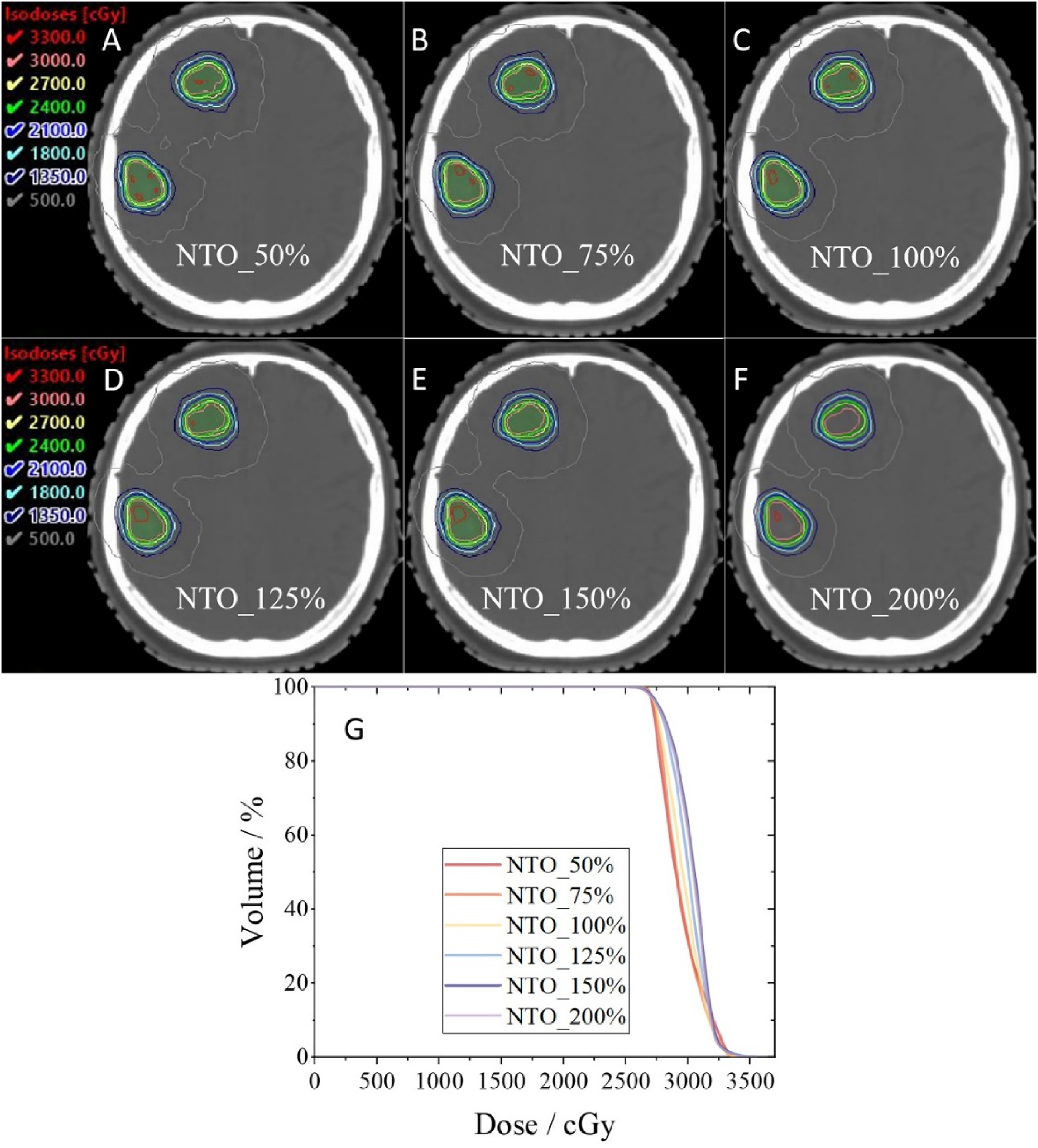
Example of isodose distributions for a representative case under different NTO strategies (axial view).
Discussion
This study is the first to systematically investigate the comprehensive impact of adjusting the SRS-NTO's relative weight as a single variable on the dosimetric quality, plan complexity, and treatment efficiency of SRS plans for brain oligometastases within the Varian HyperArc framework. Our central finding is that a moderate increase in the NTO weight to 125% from the system default of 100% into an optimal range of 125% to 150% can significantly enhance plan efficiency and delivery precision without compromising, and even slightly improving, dosimetric quality. This result provides crucial evidence-based guidance for the clinical fine-tuning of HyperArc plans and offers new insights into the behavior of NTOs in highly automated radiotherapy workflows. To ensure a controlled and unbiased comparison for this planning study, we utilized a standardized prescription of 27 Gy delivered in 3 fractions (27 Gy / 3f). It is important to clarify that this was not a single-fraction dose but a clinically relevant hypofractionated regimen, appropriate for the lesion sizes within our cohort, which addresses concerns regarding clinical applicability. Furthermore, it is also important to contextualize the NTO weight optimization against the backdrop of the actual treatments. In the clinical plans delivered to this cohort, prescription doses were determined individually, and the SRS-NTO weight was selected by the planner based on clinical judgment, typically falling within a range of 75% to 125%. This existing variability in clinical practice underscores the importance of our systematic analysis. Our finding that selecting an NTO weight within the 125% to 150% range consistently provides a superior balance of plan quality and efficiency offers clear, evidence-based guidance to standardize and improve upon this practice, moving from empirical selection to an optimized, data-driven strategy.
An important finding, contrary to conventional wisdom, is that a more aggressive NTO weight did not come at the cost of PTV dosimetric quality. Our data show no statistically significant differences in Paddick CI, GI, D2cm, or OAR doses across all strategies. To address concerns about both cold and hot spots in ablative therapy, we evaluated target dose coverage at the periphery (D99.9%) and within the high-dose core (D0.1%). Preventing cold spots is paramount in SRS to ensure complete tumor ablation. Our data confirmed that even at the highest NTO weights, D99.9% remained statistically unchanged, indicating that target coverage integrity was not compromised. The evaluation of hot spots, which is also important for characterizing dose heterogeneity, showed these also remained stable. This contrasts with some mNTO studies where achieving ultimate normal tissue sparing in non-coplanar RapidArc plans, as demonstrated by Muthu S. et al (2025), 10 necessitates complex multi-parameter trade-offs. Our study suggests that within the automated HyperArc framework, merely strengthening the NTO constraint allows the optimizer to find a superior solution without “eroding” the target dose. We postulate this is primarily due to HyperArc's highly optimized non-coplanar beam geometry, which intrinsically facilitates steep dose gradients. On this foundation, a stronger and clearer NTO objective may provide better “navigation” for the optimizer, allowing it to converge faster to a more globally optimal dose distribution rather than settling for a “compromise” on the PTV.
The findings regarding treatment efficiency and plan complexity offer a new perspective on the role of the NTO. We observed a statistically significant decrease in total MUs and beam on time with increasing NTO weight up to the 150% setting. This is in stark contrast to some mNTO optimization studies, which often increase MUs to achieve extreme gradients.13,14 It also offers a different viewpoint from the work of Muthu S. et al (2025), 10 who found that flexible beam selection in non-coplanar RapidArc could sometimes yield lower MUs than HyperArc. Furthermore, our results may seem to differ from those of Oh SA. et al (2025), 15 who found that the HyperArc SRS-NTO algorithm increased MUs compared to a generic auto-NTO.
Our study's unique contribution, distinct from prior work comparing different platforms,12–15 is its focus on optimizing within the HyperArc framework. Our findings reveal that a higher NTO priority guides the optimizer toward inherently more efficient solutions characterized by lower modulation and simpler aperture shapes—a conclusion supported by the trends in MCS and AI in our data. This “simpler is better” approach yields a direct cascade of benefits: reduced MUs, shorter treatment times, and—most critically—enhanced plan deliverability. The strong correlation between increased NTO weight and improved Gamma passing rates, especially under stringent 2%/2 mm and 1%/1 mm criteria, robustly validates this conclusion. It demonstrates that simpler plans achieve a higher fidelity between the intended and the delivered dose, thereby ensuring greater treatment precision.
Considering all evaluated dimensions, this study provides conclusive evidence that an optimal therapeutic window for the NTO weight exists between 125% and 150%. Our inclusion of a 200% NTO group was decisive in defining the upper limit of this window and refuting the notion that a higher NTO weight is always better. Compared to the 100% default, both the 125% and 150% strategies delivered significant reductions in MUs and higher Gamma passing rates. While the most favorable dosimetric trends (for CI and GI) were noted at 125%, the absolute peak in efficiency (lowest MU/BOT) and plan simplicity (best MCS) occurred at 150%. Critically, beyond this range, at 200%, we observed a clear deterioration in performance, with worsening complexity (lower MCS), declining Gamma rates, and a reversal of efficiency gains. This empirically demonstrates that while the entire 125%-150% range is superior, arbitrarily increasing NTO priority further is detrimental. Therefore, selecting a weight within this 125%-150% “sweet spot” strikes the most desirable Pareto-optimal balance, aggressively enhancing plan quality and efficiency without approaching the zone of negative trade-offs. This provides a simple and effective optimization pathway for clinicians, resonating with previous research,27–29 and aligning with HyperArc's core philosophy of automation.
Nevertheless, this study has several limitations. First, as a retrospective, single-center planning study, its conclusions are based on dosimetric and physical parameters and lack a direct correlation with long-term clinical outcomes like radiation necrosis or local control. Second, the cohort was limited to brain oligometastases (1-3 lesions), and the generalizability of these findings to more complex cases (eg, > 3 metastases or larger total volumes) needs further validation. Therefore, the clinical applicability of applying this optimal NTO range to cases with more than 3 lesions or large cumulative tumor volumes remains unknown and warrants caution. Finally, this study did not directly compare the optimized SRS-NTO strategy with potentially superior non-coplanar RapidArc plans using mNTO, which limits our exploration of the deeper interplay between HyperArc's inherent beam geometry and NTO optimization. Future research should build on these findings by initiating prospective studies to validate the clinical benefits of the NTO 125%-150% setting, extending this paradigm to more numerous metastases, and exploring the integration of optimized NTO strategies with advanced tools like multi-criteria optimization (MCO) 30 and knowledge-based planning (KBP) 31 to achieve individualized and intelligent plan optimization.
Conclusion
The results of this study demonstrate that for HyperArc™ SRS of brain oligometastases, moderately increasing the relative weight of the SRS-NTO from the system default of 100% into an optimal range of 125% to 150% is a superior clinical strategy. This adjustment significantly reduces total monitor units, thereby enhancing treatment efficiency, without compromising the core dosimetric quality of the plan, including target conformity, dose gradient, and OAR sparing. Importantly, the reduction in plan complexity associated with the higher NTO weight translates into improved plan deliverability, particularly under stricter physical verification standards.
In light of these trade-offs, we recommend the adoption of an SRS-NTO weight within the 125% to 150% range for HyperArc plan optimization in this clinical context. This range strikes the optimal balance among dosimetric quality, plan complexity, and treatment delivery efficiency. Such a simple yet effective parameter adjustment has the potential to shorten treatment times, improve the overall efficiency of the radiotherapy workflow, and ensure a high-quality, high-precision dose distribution—ultimately enhancing both the patient experience and the utilization of clinical resources.
Footnotes
Acknowledgements
We thank Dr Weihua ZHU (Varian Medical Systems Inc.) and Dr Shixiong HUANG (Hunan Cancer Hospital) for their assistance with batch data extraction and processing in this study.
Ethics Approval and Consent to Participate
This retrospective analysis was approved by the Science and Technology Ethics Committee of TFCH (Tianjin First Central Hospital), located in Tianjin, China (IRB Approval No: 20241031-1, approved on 31-October −2024). Informed consent was waived by the IRB for this study. All patient data were de-identified to protect privacy, and all procedures adhered to the ethical principles of the Declaration of the 1964 Helsinki.
Consent for Publication
The need for consent for publication was waived by the Institutional Review Board due to the retrospective nature of the study and the use of anonymized data.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Tianjin Health Research Project (TJWJ2024MS049) and Tianjin Natural Science Foundation (21JCYBJC00930).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
