Abstract
Background:
The outcomes of patients with postmenopausal breast cancer(PBC) can be improved through the early detection of intraocular metastases(IOMs). In this study, we investigated patients with PBC, and compared those with IOMs with those with non-intraocular metastases(NIOMs) in terms of blood lipid levels, and then differentiated the risk factors associated with IOMs.
Methods:
Student’s t-test and a chi-square test were used to discriminate between the IOMs and NIOMs groups. After establishing a Poisson regression model to analyze risk factors, we plotted receiver operating characteristic curves(ROC) to assess the quality of risk factors predicting IOMs.
Results:
The incidence of IOMs in PBC was 1.16%. There was no significant difference in terms of histopathology between the 2 groups. The levels of total cholesterol (TC), apolipoprotein A1(APOA1) and low-density lipoprotein(LDL) in IOMs were significantly lower than in NIOMs groups. Poisson regression suggested that low levels of APOA1 and LDL were risk factors for IOMs (P = 0.002 and P < 0.001, respectively). ROC curve analysis demonstrated that the cut-off values of APOA1 and LDL were 1.025 g/L and 2.415 mmol/L. The highest prediction accuracy for IOMs involved the combination of APOA1 and LDL (AUC = 0.881, P < 0.001).
Conclusion:
Our research demonstrates that low levels of APOA1 and LDL efficiently predict IOMs in PBC as risk factors, and the combination of APOA1 and LDL was more predictive than single factors.
Keywords
Introduction
Breast cancer (BC) is primary cancer and results in the highest rates of cancer-related deaths in females across the globe. 1 Most cancer-related deaths are a result of metastases. The management and eradication of BC are difficult because of the high rate of metastases within a few decades after BC being diagnosed, particularly in postmenopausal women. 2 However, metabolic syndrome(MS) contributes to the risk of postmenopausal breast cancer (PBC). 3 Additionally, dyslipidemia plays an essential role in MS. Wang M et al reported that dyslipidemia has a substantial adverse effect in PBC. 4 However, the role of dyslipidemia in intraocular metastases (IOMs) in patients with PBC remains elusive.
The uveal membrane is the most common site of the IOMs of blood-borne tumors because of its abundant blood flow. The general clinical symptoms are blurred vision and sight loss. 5 A cohort study of 1,111 patients with uveal metastases performed by Carol L. Shields et al revealed that primary BC lesions accounted for the highest proportion of IOMs, at 37%, with an average patient age of 57. 6 In short, although BC does not commonly metastasize to the eye, it is the most common primary lesion in IOMs. The long-term prognosis and the quality of life of BC patients are poor, because of severe clinical symptoms as well as the late stage of metastases, which mostly occur in the postmenopausal state.
Currently, ocular tumor diagnosis relies heavily on imaging techniques, which are regarded as indispensable, including computed tomography (CT), high-frequency ultrasound, optical coherence tomography, magnetic resonance imaging (MRI) and fluorescein angiography. 7 Despite their remarkable clinical diagnostic value, the high cost and high-radiation doses associated with these tests are problematic during long follow-up periods. Moreover, the incidence of IOMs is somewhat low. Consequently, in terms of patients with PBC, these tests are not suitable for global-scale routine detection during post-treatment follow-up. Serological monitoring may be a preferable option for routine testing during long-term follow-up in PBC, because of its low cost, reproducibility, and non-invasiveness. Recently, it was reported that serological monitoring is useful in BC diagnosis and prognosis; for instance, assessments of programmed cell death protein 1 as well as the systemic immunoinflammatory index. 8,9 Therefore, to facilitate early detection of IOMs in PBC, identifying serum lipid risk factors is important and is becoming more feasible.
With the aim of improving PBC patient prognosis, we carried out a retrospective study to identify the risk factors for IOMs in PBC by assessing the clinical relevance of serum lipids and IOMs.
Materials and Methods
Ethics Statement
The Medical Research Ethics Committee of the First Affiliated Hospital of Nanchang University approved this study(CDYFY-LL2006023). Because of the retrospective nature of the study, informed consent of participants was waived. The methodology applied in this study is based on approved guidelines and relevant regulations.
Study Design
This retrospective study included patients who presented with BC from September 1990 to July 2017 at our hospital, and who were diagnosed using histopathological sections obtained from surgical resection or biopsy. IOMs was confirmed through local CT and MRI.
Inclusion criteria: 1) patients with BC diagnosed as the primary lesion; 2) postmenopausal status
Exclusion criteria: 1) patients with primary ocular malignancies, benign ocular tumors; 2) patients with other malignant tumors; 3) patients with loss of follow-up and incomplete medical data.
Data Collection
Data from retrospective studies were obtained from patient medical records. Once BC was diagnosed, demographics and clinical parameters were recorded before anti-tumor treatment (e.g., radiotherapy, chemotherapy, or surgery) was applied. These mainly included: the age of the tumor being diagnosed, histopathology and the condition of metastases, laboratory tests of lipid metabolism such as total cholesterol (TC), triglyceride (TG), high density lipoprotein (HDL), LDL, APOA1, apolipoprotein B (ApoB), and lipid-protein a (Lp(a)). After the correlation between different clinical-pathological parameters and IOMs was analyzed, we assessed the risk factors for IOMs in PBC and then estimated the predictive value of the risk factors.
Statistical Analyses
Statistical analysis was performed using SPSS software (IBM SPSS Statistics 22.0) and GraphPad Prism (8.0.1.244), and data collation were conducted using Excel software (Microsoft Excel 2016). A comparison between IOMs and NIOMs in terms of clinical factors was performed using Student’s t-test and the chi-square test. Quantitative variables are reported as mean ± standard deviation. Since the occurrence of IOMs in PBC is a rare event (10/865; ≈0.0116), after selecting significant factors, a multivariate Poisson regression analysis model for rare event analysis was established. For the purpose of identifying crucial independent risk factors, the odds ratio (OR) was calculated to estimate their relevance along with the 95% confidence interval (95% CI). Finally, receiver operating characteristic (ROC) curves related to the risk factors were plotted. After the area under the curve (AUC) of the different factors was calculated, the sensitivity and specificity of the optimal cut-off point were assessed to evaluate the ability of the risk factors to predict IOMs. Statistical significance was indicated by a P value of less than 0.05; all reported P values are bilateral. To avoid low effect variables being ignored, P < 0.20 was set as the standard when adding independent variables into the multivariate Poisson regression model. Moreover, the test level (α) was set to 0.05.
Results
Demographics and Clinical Characteristics
A total of 2,373 patients participate in the study, and 1,508 were excluded based on the inclusion and exclusion criteria. Table 1 lists the baseline demographic and clinical data from 10 patients in the IOMs group and 855 patients in the NIOMs group. Median age of IOMs was 64 y(range,54-68); Median age of NIOMs was 57 y (range,35-88)(P = 0.038). The histopathology of the 2 groups was not statistically significant (P = 0.406). The tumor size and stage of the 2 groups are statistically significant (P = 0.020, P = 0.001, respectively). The detailed clinical data for all patients participating in the study are listed in Table 1.
The Clinical Characteristics of Patients With PBC.
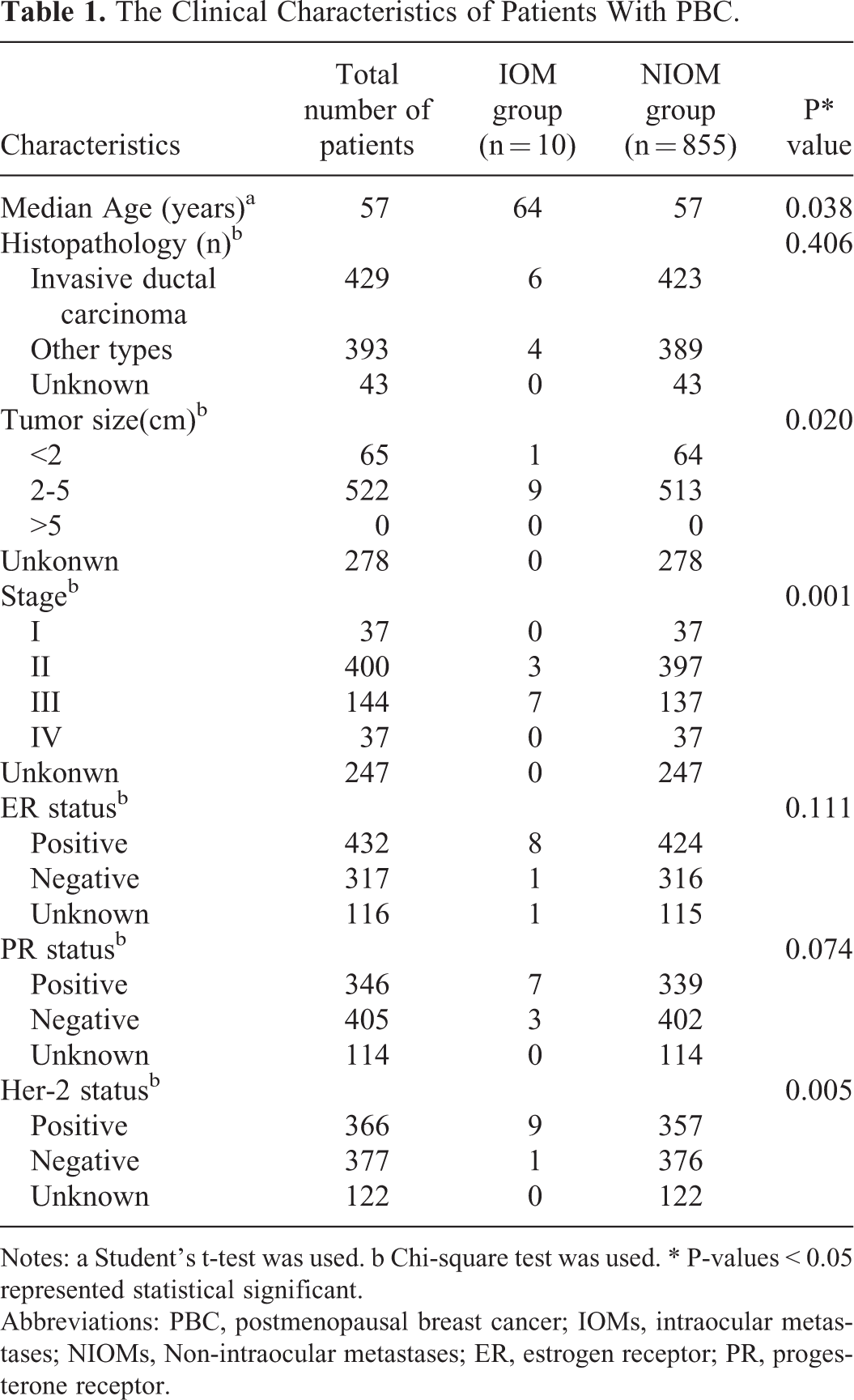
Notes: a Student’s t-test was used. b Chi-square test was used. * P-values < 0.05 represented statistical significant.
Abbreviations: PBC, postmenopausal breast cancer; IOMs, intraocular metastases; NIOMs, Non-intraocular metastases; ER, estrogen receptor; PR, progesterone receptor.
Differences in the IOMs Clinical Features and the Risk Factors for IOMs
The levels of TG, HDL, ApoB, and Lp(a) were not significantly different between the IOMs and NIOMs group (P > 0.05). However, the levels of TC, LDL, and APOA1 were lower in the IOMs group than in the NIOMs group (P = 0.041, P < 0.001 and P = 0.001, respectively) (Table 2). More details are shown in Table 2. A Poisson regression model was constructed to determine potential independent risk factors. It showed that low levels of LDL as well as APOA1 were independent risk factors, contributing to IOMs in PBC (OR = 0.021, 95% CI: 0.002-0.252; OR = 0.313, 95% CI: 0.176-0.557, respectively) (Table 3).
The Differences in Clinical Characteristics Between IOMs and NIOMs.

Note: *: P value < 0.05 represent statistical significance.
a: The SD values are too high in Lp(a). The median value and range are supplied. The median value and range of Lp(a) in IOMs group are 178.00, 124.00-463.00 respectively. The median value and range of Lp(a) in NIOMs group are 127, 0.96-1261.00 respectively.
Abbreviations: TC, total cholesterol; TG, triglyceride; HDL, high density lipoprotein; LDL, low density lipoprotein; APOA1, apolipoprotein A1; ApoB, apolipoprotein B; Lp(a), lipoprotein a; IOMs, intraocular metastases; NIOMs, non-intraocular metastases.
The Poisson Regression Results.

Note: *: P value < 0.05 represent statistical significance.
Abbreviations: TC, total cholesterol; TG, triglyceride; LDL, low density lipoprotein; APOA1, apolipoprotein A1.
The Cut-Off Value, AUC, Sensitivity, and Specificity of LDL and APOA1 in Diagnosing IOMs
On the basis of the ROC curve, it can be observed that APOA1 had the highest sensitivity to IOMs among these factors, with a sensitivity of 97.9% (Table 4). The cut-off values for LDL and APOA1 used for prediction were <2.415 mmol/L and <1.025 g/L, respectively. Therefore, patients with LDL <2.415 mmol/L and APOA1 <1.025 g/L are at higher risk of IOMs in PBC. In terms of the combined risk factors, the combination of APOA1 and LDL had the highest diagnostic accuracy for IOMs in PBC (AUC = 0.881, P < 0.001) (Table 4 and Figure 1).
The ROC Results of Risk Factors for IOMs in PBC.

Abbreviations: AUC, area under the curve; APOA1, apolipoprotein A1; LDL, low density lipoprotein; IOMs, intraocular metastases; ROC, receiver operating characteristics.
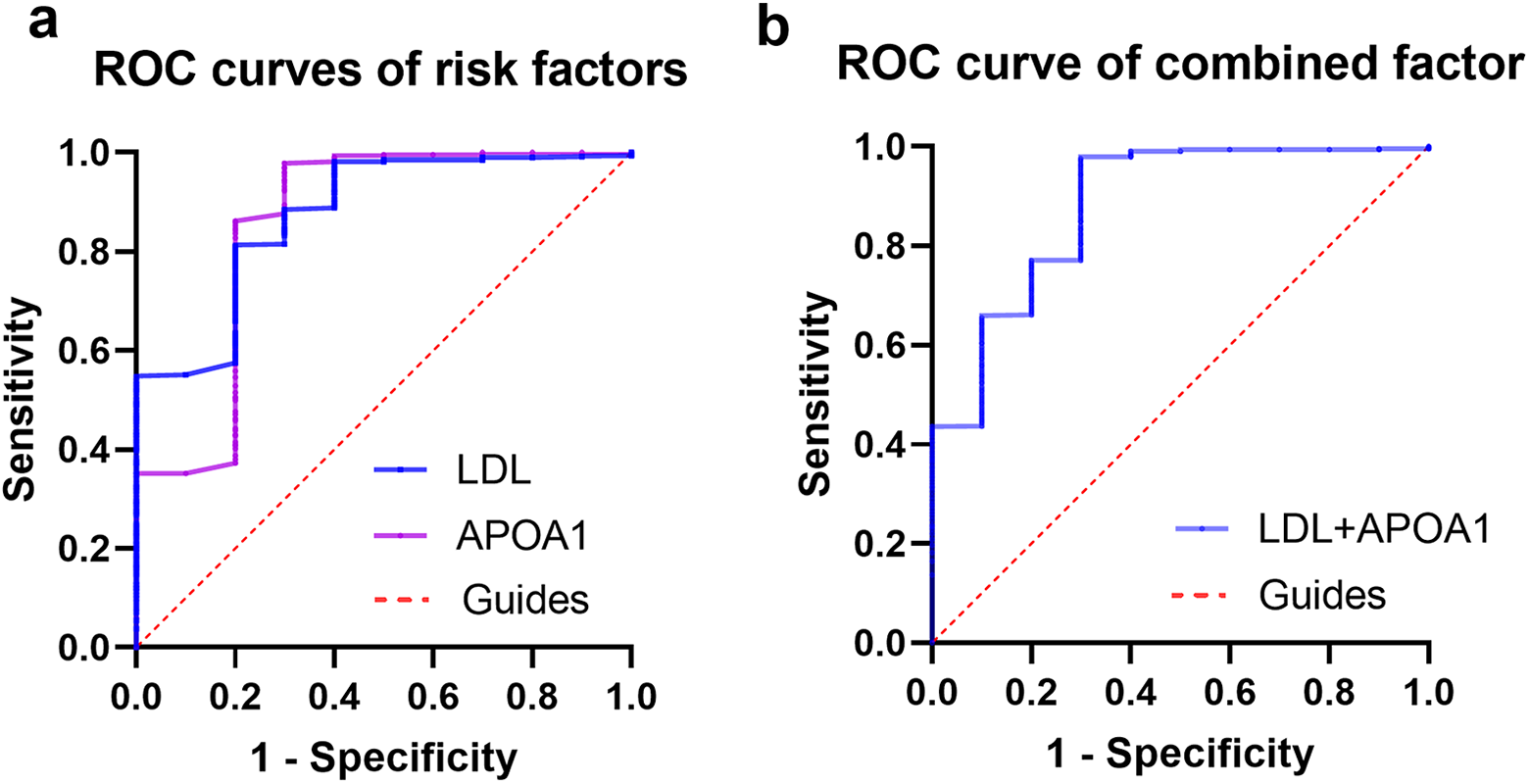
The ROC curves of risk factors for IOMs in breast cancer. Notes: The ROC curve of APOA1. The AUC was 0. 855 (P-value<0.001; 95% CI: 0.699–1.000) (IOMs<NIOMs). The ROC curve of LDL. The AUC was 0.874 (P-value<0.001; 95% CI: 0.769−0.979) (IOMs < NIOMs). The ROC curve of a combination of APOA1 and LDL. The AUC was 0.881 (P-value < 0.001; 95% CI: 0.765–0.997). Abbreviations: ROC, receiver operating characteristic; AUC, area under the curve; CI, confidence interval; IOMs, intraocular metastases; NIOMs, non-intraocular metastases.
Discussion
In the current study, the incidence of IOMs in PBC was 1.16%, which is lower than that reported in other studies. 10,11 This may have resulted from the low detection rate of IOMs in PBC, which could relate to patient data collection over an extended period, subtle initial symptoms, unsatisfactory performance of examination methods, and patient lack of health awareness. The life expectancy of patients with IOMs is usually reduced, and if IOMs are not treated in time, patients are prone to irreversible sight loss. 5 Severe ophthalmic symptoms have an enormously adverse effect on the quality of life in PBC. This type of patient is less compliant than other types, and overall compliance decreases with increasing age. 12 In fact, the imaging screening methods (Figure 2) for conventional intraocular metastatic tumors are not highly accepted by patients. On the contrary, it has been reported by many studies that serum lipid levels are associated with many cancer prognosis, suggesting that these makers are potential screening tests (Table 5). Therefore, it is necessary and feasible to perform more convenient and economical serological monitoring methods to screen IOMs in PBC.

Examples of IOMs. (a): B-ultrasound of an eye with IOMs (right eye): A solid echo of a strong echo is visible in the posterior pole, and the mass grows along the wall of the ball. (b): Fundus photography an eye with IOMs (right eye): A yellow-white, nodular flat bulge can be seen under the retina of the posterior pole. (c): Fundus angiography of an eye with IOMs (right eye): Fluorescence leakage lesion, high fluorescence, and angiography in the lesion are visible. (d): Pathological images of IOMs HE stain×200. The white arrow pointers is to specify the location of the nodular buldge. Abbreviations: IOMs, intraocular metastases; HE, hematoxylin and eosin.
The Serum Lipids as Risk Factors of Different Types of Cancer.

Abbreviations: APOA1, ApolipoproteinA1; APOA-II, ApolipoproteinA-II; PC, phospha-tidylcholine; TG, triacylglycerol; DG, diacylglycerol; PE, phosphatidylethanolamine; LDL, low-density lipoprotein; HDL, high-density lipoprotein.
MS was reported as a significant risk factor for PBC in 22,494 case-cohort study performed by Agnoli C et al. 22 Moreover, a study involving 3,460 subjects demonstrated that a high level of body fat contributed to an increased risk of PBC at a normal BMI. 23 A similar result was obtained in cohort studies including 50,884 participants performed by Park YM et al. 24 It was reported by Wang M et al that dyslipidemia has a substantial adverse effect in PBC. 4 Abnormal lipid metabolism has been demonstrated to be a critical metabolic reprogramming process for tumor cells. 25 However, the role of dyslipidemia in IOMs in PBC remains unclear.
APOA1 is the main component of HDL, at approximately 70%, in terms of structure and function. It is regarded as a crucial mediator in cholesterol transport, is involved in the excretion of cholesterol to the liver from other tissues, and has vital anti-inflammatory, anti-oxidative, anti-apoptotic roles and contributes to innate immunity. 26,27 APOA1 exhibits a role in the onset and progression of cancer. It has been confirmed by numerous studies that decreased levels of APOA1 are linked to pancreatic cancer, 13 gastric cancer, 14 liver cancer, 16 colorectal cancer, 19 renal cancer, 15 and acute lymphoblastic leukemia. 20 Furthermore, it suggested that APOA1 at a low level could be an unfavorable prognostic factor in BC. 28 APOA1 binds and eliminates lysophosphatidic acid which is related to tumor progression and poor prognosis, thereby exerting a negative effect on tumor cell proliferation. 29 Moreover, APOA1 can promote tumor-associated macrophage changes from a tumor-promoting M2 phenotype to an anti-tumor M1 phenotype by regulating the tumor immune environment to delay tumor growth. 30 A cohort study performed by Claudia Agnoli et al showed that low APOA1, as the main component of HDL, increased the risk of BC. His M et al also reported a similar result. 31 In a study involving 38,823 participants, a similar result was found in PBC as well. 32 In recent years, APOA1 has been increasingly connected with ophthalmic diseases. The severity of diabetic retinopathy (DR) is inversely related to APOA1. HDL cholesterol is associated with an increased risk of Age-Related Macular Degeneration. 33 Therefore, we hypothesized that there is a link between low levels of APOA1 and IOMs in PBC. On the basis of the current study, APOA1 is an independent risk factor for IOMs, and APOA1 <1.025 g/L is closely linked to IOMs.
LDL, a cholesterol-rich lipoprotein granule, transports cholesterol to peripheral tissues from the liver. 34 It was reported by Fiorenza A et al that LDL-C at a low level is related to cancer metastases. 35 Additionally, it was reported by Martin LJ et al that LDL-C levels were negatively correlated with BC progression. 36 Knapp ML et al reported low levels of LDL-C in patients with advanced BC. 37 This phenomenon likely results from the excessive consumption of cholesterol as BC cells rapidly proliferate. 38 LDL receptor, which assimilates LDL-C from peripheral blood, is highly expressed in BC tissues. 39 Recently, it has been reported that LDL-C at a high level is linked to BC progression as a risk factor. 21 It was demonstrated by Osae EA et al that LDL is significantly related to Meibomian Gland Dysfunction. 40 Li D et al revealed that high LDL-C is an independent risk factor for age-related cataracts. 41 Consequently, we hypothesized that LDL at a low level is perhaps correlated with IOMs in PBC. Based on our investigation, it can be concluded that LDL contributes to IOMs as an independent risk factor, and LDL <2.415 mmol/L is more likely to be associated with IOMs in patients with PBC.
In the current study, we also assessed the predictive value of a combination of risk factors for IOMs in PBC. Compared with single factor prediction, the combination of APOA1and LDL had higher diagnostic accuracy. Therefore, a combination of risk factors may be beneficial for the prediction of IOMs and provide more critical information for PBC patient follow-up visits.
Although the result of this study is striking, there are some limitations. Firstly, this is a retrospective study. Because of the vast study period, some related data, such as the survival time of IOMs, are not sufficient. Secondly, the sample size may not be large enough to capture the characteristics of the entire BC population, affecting the accuracy of the results to some extent. Moreover, all data for this study come from a single medical institution, which probably adds some confounding bias. Therefore, the results of the current study require validation in a multicenter study.
To sum up, this investigation demonstrates for the first time that low levels of APOA1 and LDL efficiently predict IOMs in PBC as risk factors. APOA1 and LDL are expected to be potential therapeutic targets for IOMs in PBC. These results could motivate novel insights into molecular mechanisms so as to gain a better understanding of the role of LDL and APOA1 for antineoplastic applications.
Footnotes
Authors’ Note
Biao Li, Yao Liu, and Qing Yuan contributed equally to this work. The Medical Research Ethics Committee of the First Affiliated Hospital of Nanchang University approved this study(CDYFY-LL2006023). Because of the retrospective nature of the study, informed consent of participants was waived.
Acknowledgements
The authors will thank Yi Shao for his great help in conceiving and designing the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China (No:81660158); Natural Science Key Project of Jiangxi Province (No: 20161ACB21017); Youth Science Foundation of Jiangxi Province (No: 20161BAB215198); Key Research Foundation of Jiangxi Province (No: 20181BBG70004); Excellent Talents Development Project of jiangxi Province (S2019RCQNB0259); Teaching Reform of Degree and Graduate Education Research Project of Jiangxi Province (No: JXYJG-2018013); Grassroots Health Appropriate Technology Spark Promotion Plan Project of Jiangxi Province (No: 20188003); Health Development Planning Commission Science Foundation of Jiangxi Province (No:20175116); Health Development Planning Commission Science TCM Foundation of Jiangxi Province (No: No:2018A060).
