Abstract
Introduction
SCINTIX® therapy, a form of biology-guided radiotherapy using real-time positron emission tomography (PET) imaging on the RefleXion® X1 platform, recently received FDA clearance for FDG-guided treatment of lung and bone tumors. This study evaluates the feasibility and dosimetric quality of Biology-Guided Radiotherapy (BgRT) plans for FDG-avid lesions in anatomical sites beyond current indications using processed diagnostic PET (dPET) images.
Methods
Eleven previously treated FDG-avid lesions in ten patients (sites: liver, head and neck, pancreas, kidney, pelvic/abdominal nodes) were retrospectively analyzed. dPET images were processed to emulate RefleXion X1 PET characteristics. BgRT plans were generated using a non-clinical version of the RefleXion treatment planning system, delivering stereotactic body radiation therapy-level doses (48Gy/3fx for liver, 40Gy/5fx for other sites). Plan deliverability was evaluated based on normalized target signal (NTS >2.7) and activity concentration (AC > 5 kBq/ml). Plans were categorized based on the presence of dose-limiting organs at risk (OAR) constraints. Dosimetric parameters were assessed according to institutional standards. Statistical analyses examined relationships between dPET standardized uptake value (SUV), PET-derived parameters, planning target volume (PTV) characteristics, and plan complexity.
Results
BgRT plans were successfully generated for all lesions. All met the NTS threshold (mean = 9.3); 72.7% met the AC goal (mean = 12.1 kBq/ml). Among cases with dPET SUV > 6, 87.5% met AC criterion, supporting this threshold as a reasonable selection criterion. Plans without dose-limiting OARs (n = 6) achieved higher PTV coverage (V100%=94.4%, conformity index (CI) = 1.08) compared to plans with OAR constraints (n = 5, V100%=78.0%, CI = 1.4). All plans were deemed clinically acceptable despite observed heterogeneity within target regions. MLC modulation intensity correlated strongly with target volume (r = 0.907) and superior-inferior extent (r = 0.896) rather than OAR proximity.
Conclusion
BgRT planning using processed dPET is feasible for non-lung/bone FDG-avid lesions. SUV > 6 may serve as a reasonable threshold, though feasibility is affected by tumor location, FDG background, and nearby critical structures. Findings support clinical potential of SCINTIX expansion into new anatomical sites.
Introduction
External beam radiation therapy has traditionally relied on anatomical information for target delineation and treatment planning. Over recent decades, technological advancements have significantly improved the geometric precision of radiation delivery through techniques such as intensity-modulated radiation therapy (IMRT) and stereotactic body radiation therapy (SBRT).1–3 However, a fundamental limitation persists: the reliance on anatomical rather than biological information for treatment guidance, leading to challenges in addressing tumor heterogeneity and motion management during treatment delivery.4,5
Biology-guided radiotherapy (BgRT) represents a paradigm shift in radiation oncology by directly incorporating biological information into treatment delivery.4–7 Early research in BgRT focused on utilizing positron emission tomography (PET) for improved offline target delineation and adaptive planning.4,5,7 Several studies demonstrated the value of 18F-fluorodeoxyglucose PET (FDG-PET) in identifying metabolically active tumor regions for dose escalation or adaptation.4,7–11 However, these approaches still maintained the conventional workflow separation between imaging and treatment, with PET data incorporated into planning but not available during delivery. Unlike conventional radiotherapy approaches that rely on pre-treatment imaging for planning and intermittent image guidance during delivery, the concept of integrating real-time biological guidance during treatment utilizes real-time biological signals to guide beam delivery continuously. This integration has the potential to overcome key limitations of conventional radiotherapy, including the management of tumor motion, adaptation to metabolic changes, and the targeting of heterogeneous intratumoral regions.12,13 This concept ideally integrates functional imaging with linear accelerator technology to deliver radiation based on the biological characteristics of tumors rather than solely on their anatomical location. However, it remained a significant technical challenge due to hardware limitations and the complex signal processing required.
The development of the RefleXion® X1 system (Hayward, CA), a ring gantry linac that incorporates a PET/CT scanner, represents a meaningful advancement in the field. This is the first commercial system that combines PET/CT and radiotherapy into a single system, allowing molecular signals to be detected and processed in real-time to guide radiotherapy beam delivery, resulting in effective treatment execution and tracking of the dose to the target motion.12–15 The RefleXion X1 system features a rotating ring gantry with a 6MV FFF compact linear accelerator, alongside PET detector arcs positioned orthogonally to the MV beamline. This configuration is complemented by a binary multileaf collimator (MLC) and onboard fan-beam kV computed tomography (kVCT), creating a unique dual-modality treatment platform that offers both biologic and anatomic guidance capabilities.12,13 The RefleXion X1 system offers two distinct treatment modalities: IMRT and BgRT (marketed as SCINTIX® therapy). In the BgRT mode, the system detects emissions from PET tracers within the tumor, processes these signals in real-time, and uses them to guide the delivery of radiation beamlets. By using the radiolabeled tumor as a biological beacon, SCINTIX therapy has the potential to improve radiation delivery by increasing intrafraction treatment accuracy.13–16 Compared with a motion inclusive internal target volume (ITV) approach, a tracked delivery method such as PET-guided therapy can potentially reduce treatment volumes for tumors that move with respiration and therefore reduce dose to critical organs at risk (OARs). This approach is particularly appealing for patients with multiple tumors throughout the body, which is a current clinical challenge that may require multiple treatment modalities.12,13
Initial research on the RefleXion X1 system focused on commissioning, quality assurance, and validation of its capabilities. Simiele et al performed the treatment planning system (TPS) commissioning with specific focus on the IMRT and SBRT treatment modes. 15 Han et al conducted the beam commissioning of the first clinical RefleXion X1 system linac, characterizing mechanical alignment, and beam parameters. 17 Pham et al further verified that clinically acceptable IMRT/SBRT plans could be generated with the X1 TPS for both 10 mm and 20 mm jaw settings across multiple treatment sites. 18 Additionally, Zhuang et al evaluated its kVCT imaging performance. 19 For the BgRT mode specifically, Surucu et al conducted a comprehensive commissioning of the SCINTIX delivery system, evaluating its sensitivity to PET contrast variations and its performance under both static and dynamic conditions. 16 However, the unique BgRT capability still required separate clinical validation due to its novel approach to treatment guidance. The first clinical evaluation of the BgRT mode came through BIOGUIDE-X, an Investigational Device Exemption (IDE) trial (NCT04788147). This prospective feasibility study assessed the imaging and planning capabilities of the SCINTIX system in patients with FDG-avid tumors (SUVmax ≥ 6) in lung and bone. 14 These anatomical sites were strategically selected to evaluate the technology in both moving (lung) and relatively stationary (bone) targets, as well as in settings with different tumor-to-background FDG uptake ratios. The study established patient selection criteria and validated the treatment approach. Based on promising clinical data, the Food and Drug Administration (FDA) granted marketing clearance for SCINTIX biology-guided radiotherapy for FDG-guided treatment of lung and bone tumors in 2023.14,16
Despite this important regulatory milestone, several challenges remain in expanding BgRT to other anatomical sites. Different anatomical regions present varying levels of background FDG uptake, physiological motion, and proximity to critical organs, potentially affecting both signal detection and plan quality. Additionally, the unique delivery characteristics of the RefleXion X1 system, including its rotating gantry and binary MLC design, may interact differently with various anatomical constraints compared to conventional delivery platforms. A significant practical barrier to implementing BgRT across diverse anatomical sites is predicting feasibility before subjecting patients to SCINTIX therapy-specific imaging protocols. Currently, patients must undergo a dedicated PET imaging session, known as functional modeling, on the RefleXion X1 system as part of the treatment planning process, representing a resource-intensive commitment.12,13 The ability to pre-screen patients using existing diagnostic PET (dPET) data would substantially streamline workflow and expand accessibility. However, this requires a method to translate standard dPET signals to the specific characteristics of the RefleXion X1 PET subsystem, which has different detector configurations and resolution properties.
To address these challenges, we conducted a systematic investigation of BgRT planning feasibility for anatomical sites beyond lung and bone using emulated RefleXion X1 PET data derived from standard diagnostic PET scans. The RefleXion X1 PET subsystem is specifically designed to acquire line-of-response (LOR) data for real-time guidance of radiation delivery, rather than producing diagnostic-quality PET images. This fundamental design choice prioritizes rapid signal processing and localization capabilities over the high-resolution image quality typical of diagnostic PET scanners. Our approach simulates the differences in system sensitivity, acquisition time, reconstruction method and geometry between the diagnostic PET system and the RefleXion X1 PET subsystem, allowing for assessment of plan feasibility and quality without requiring patients to undergo actual imaging on the RefleXion platform. This study specifically aims to: (1) evaluate whether processed dPET can effectively assess BgRT plan feasibility across extrapulmonary and extraosseous sites; (2) determine if the SUV > 6 criterion established for lung and bone lesions remains valid for other anatomical locations; (3) assess the dosimetric quality of generated BgRT plans for sites with varying levels of OAR complexity; and (4) characterize plan deliverability parameters across different anatomical regions. By addressing these objectives, we aim to establish a practical methodology for pre-screening patients for BgRT eligibility and provide guidance for the expansion of this technology beyond its current FDA-cleared indications.
Methods and Materials
RefleXion X1 System
The RefleXion X1 system is a highly compact radiotherapy treatment system that includes a 6MV FFF linac mounted on an O-ring gantry rotating at 60 RPM. The system operates with a nominal dose rate of 1000 MU/min at isocenter plane with 85 cm source-to-axis distance. The split-jaw design of the RefleXion X1 system features two clinical jaw settings capable of adjusting to 10 mm or 20 mm in the longitudinal (superior/inferior) direction. In addition, the system is equipped with a multi-leaf collimator (MLC) system, employing a binary leaf design, consisting of 64 leaves with 6.25 mm width. The open and close MLC transition rate is 5-10 ms, enabling the system to deliver modulated beamlets at 50 unique gantry angles. The couch enters the bore in discrete steps of 2.1 mm in the longitudinal (superior/inferior) axis, where the gantry rotates around the patient and delivers the modulated beamlet sequences based on the plan optimization. The system features two 90-degree PET detector arrays orthogonal to the MV beamline with an axial field of view (FOV) of 5.2 cm. The RefleXion X1 system offers both conventional imaging guidance treatment (IGRT mode) and BgRT (SCINTIX delivery mode).12,13,16,17
Patient Data Collections
This retrospective study was approved by the Institutional Review Board (IRB) of the University of Texas Southwestern Medical Center, Dallas, Texas, USA (approval number: STU 082013-008, approval date: December 6, 2021). Due to the retrospective nature of this study utilizing previously acquired clinical data, the requirement for informed consent was waived by the IRB. All patient data were de-identified prior to analysis to ensure patient confidentiality. The study was conducted in accordance with the Declaration of Helsinki and institutional human research ethics guidelines. The clinical sites were selected to investigate the potential expansion of BgRT beyond the lung and bone indications established in the BIOGUIDE-X IDE trial, based on data availability and clinical significance. Ten previously-treated patients who had liver, head-and-neck (HN), pancreas, renal, and pelvic-abdominal lymph node lesions were identified for this study. A total of 11 treatment sites were determined with one patient's treatment plan divided into two separate sites. Patients were simulated with head-first-supine (HFS) on a simulation CT scanner with uniform slice thickness of either 1 mm or 2 mm. These cases were selected to evaluate the feasibility and robustness of the modality across different anatomical regions, specifically the head and neck, abdominal, and pelvic regions. BgRT planning utilized previously acquired simulation CTs, diagnostic PETs, and clinical contours. To ensure planning feasibility, dPET images of the treatment sites were first reviewed and analyzed before treatment planning. The target selection criteria included a minimum SUV of 5 and a target size of less than 10 × 10 cm. Additionally, for patients with multiple targets, the targets were separated from each other by at least 5 cm apart from each other. Table 1 presents the detailed characteristics of the lesions evaluated in this study, including the prescribed radiation dose, the volume and length of the planning target volume (PTV) in the superior-inferior (SUP-INF) direction, and the SUV from the dPET scans.
Lesion Characteristics - Site, Prescribed Dose, PTV Volume (cc), Length in SUP-INF Direction (cm), and SUV on dPET.

Abbreviations: NTS, normalized target signal; dPET, diagnostic positron emission tomography; PTV, planning target volume; SUV, standardized uptake value.
RefleXion BgRT Plan Generation
BgRT plans were generated on a RefleXion non-clinical TPS. The dPET images were first imported into the RefleXion TPS after being checked for fusion accuracy. The non-clinical TPS platform emulated PET images acquired on the RefleXion X1 machine by accounting for differences in system sensitivity, acquisition time, reconstruction method and geometry of the X1 system as follows. For the purposes of generating the emulated X1 PET image, the dPET image is considered ideal (ie, assumed to be noiseless, precise data) since current state-of-the-art diagnostic PET/CT systems provide clinically well-accepted image quality and resolution. To account for the lower sensitivity of the X1 due to its unique geometry (ie, rotating dual 90-degree PET detector arcs and limited axial FOV of 5.2 cm), the dPET image is forward-projected and attenuated based on the patient's CT density map into projection data (sinogram) space. The measured absolute sensitivity map in projection space from an actual X1 system is multiplied with the forward projected data and Poisson statistics, random coincidences, and a simple model of patient scattered photons are all then added in the projection space. The X1 PET geometries are also modeled, including the discrete 2.1 mm steps of the couch and the 4-pass back and forth motion of the couch as relevant for the image being simulated. Finally, the exact filtered-back projection reconstruction method and post-smoothing parameters that are used on the X1 system are employed to reconstruct the emulated X1 PET image from the simulated projection data. While SUV values are reported in this study, they are derived exclusively from the diagnostic PET (dPET) images. The emulated RefleXion X1 PET images, generated within the non-clinical TPS, do not compute or display SUV values. Rather, they simulate the spatial distribution of PET signals for use in BgRT plan optimization. Absolute SUV quantification is not preserved or intended in the emulation process. Instead, internal system parameters such as normalized target signal (NTS) and activity concentration (AC) are used to assess signal adequacy and plan deliverability within the BgRT workflow.
For all cases, gross tumor volumes (GTVs) were delineated based on physician-drawn clinical contours. Subsequently, the BgRT PTV was created with a uniform 5 mm margin expansion around the GTV. BgRT planning uses an additional virtual structure referred to as the biology-tracking zone (BTZ). The BTZ is a conceptual volume encompassing the target's entire range of motion, inclusive of a margin. During treatment delivery, the BTZ obscures all PET signals emanating from beyond its boundaries. In this investigation, the BTZ was established with a 5 mm margin either from the BgRT PTV for static targets or from the previously delineated ITV for moving targets, which accounts for the full range of GTV motion. Subsequently, individual BgRT plans were formulated, delivering 48 Gy in 3 fractions for liver cases and 40 Gy in 5 fractions for all other sites.
The TPS optimization and dose calculation employ accelerated proximal gradient descent, leveraging the Fast Iterative Shrinkage-Thresholding Algorithm (FISTA) and Collapsed-Cone Convolution-Superposition, operating on a fixed 2.1 mm dose calculation grid, respectively.20,21 Regarding the optimization process, the optimizer accounts for user-specified dose constraints and objectives while also integrating the PET signal for fluence generation. The final BgRT plan is the dose distribution ascertained by applying the firing filters to the planning PET images. The firing filter is an optimized operator derived during treatment planning that defines the relationship between PET signal and radiation delivery. When applied to PET signals (planning PET for optimization, and subsequently, real-time PET signals during delivery), the firing filter determines the modulation of radiation beamlets to achieve the desired dose distribution based on the biological signal. The fluence map (F) is mathematically defined by the firing filter (P) acting on the tumor PET projections (X) as F = P⋅X, enabling real-time adaptation during treatment delivery. All plans mandated a minimum of five revolutions per beam station to meet the criteria for plan creation and deliverability. 12 Figure 1 delineates the treatment planning workflow and the primary evaluation criteria utilized in this study.

BgRT Plan Workflow. Diagnostic PET (dPET) is Used to Generate Simulation PET Images on RefleXion. Emulated X1 PET Images Along with Simulation CT and Clinical Contours were Used to Generate a BgRT Plan. Both Plan Quality Metrics and Plan Deliverability Parameters were Derived to Assess the Plan. PET, positron emission tomography.
Plan Evaluation and Data Analysis
Both plan quality and deliverability were evaluated based on dosimetric criteria and PET-derived imaging parameters. The feasibility of planning and delivery for BgRT plans was initially assessed through the evaluation of AC and NTS derived from emulated X1 PET images, with requirements that NTS exceed 2.7 (a hard constraint) and AC surpass 5 kBq/ml (a defined deliverability goal). Plan quality was evaluated following institutional and national guidelines. Key dosimetric metrics included PTV coverage, Organ at Risk (OAR) doses, maximum dose (Dmax), Conformity Index (CI), new Conformity Index (nCI), Heterogeneity Index (HI), among others. CI was calculated as
Additional analysis focused on dose distribution patterns, including hotspot localization, dose fall-off characteristics, and the spatial relationship between isodose contours and adjacent OARs. In addition to dosimetric metrics and dose distribution, plan parameters such as delivery time, MLC modulation (both maximum and average transitions per second), and jaw size were assessed. An analysis of treatment time in relation to the Superior/Inferior length and volume of the GTV/PTV was also undertaken.
The impact of OAR constraints on BgRT plan quality was analyzed by stratifying treatment plans into two groups: (1) sites where target coverage was not significantly compromised by OAR constraints, despite the anatomical location (including liver lesions and specific nodal regions in the head and neck); and (2) sites where dose-limiting OAR constraints necessitated substantial reduction in target coverage to meet clinical OAR dose limits (including mesenteric, pancreatic, and renal regions). Notably, while head and neck regions typically contain numerous critical structures, the specific nodal targets in our study (left supraclavicular and pharyngeal nodes) were anatomically positioned such that critical OARs did not significantly impede target coverage during clinical plan optimization.
Statistical Analysis
Statistical analyses were conducted to compare dosimetric parameters and treatment delivery characteristics between the two groups. Welch's t-test was used to assess differences in PTV volume, PET superior-inferior length, dPET SUV, PTV V100%, and Dmax between the two groups. To determine whether the presence of dose-limiting OAR constraints affected treatment complexity, Welch's t-test was also used to compare treatment time, MLC transitions per second, and average transition rates between constrained and unconstrained plans. Pearson correlation analysis was performed to evaluate the relationships between PET-derived imaging parameters and dosimetric outcomes, as well as treatment delivery metrics and anatomical characteristics. The correlation coefficients between dPET SUV and BgRT-derived NTS and AC were computed to assess their predictive value for plan feasibility. Additionally, correlation analysis was conducted to examine the relationships between BTZ volume, superior-inferior length, and treatment delivery parameters, including treatment time, maximum MLC transitions per second, and average MLC transition rate per second. A significance level of P < .05 was used for all statistical comparisons.
Results
Table 1 presents the detailed characteristics of the 11 BgRT plans evaluated in this study. These 11 BgRT plans from ten patients were successfully derived, accommodating two separate targets within a single patient's treatment plan. The initial phase of this study successfully emulated PET images on the RefleXion X1 system for all ten patients. However, compared to standard dPET images, those acquired using the RefleXion X1 system exhibited a noticeable decline in image quality, characterized by increased noise and blurriness—limitations inherent to the system's imaging technology. Figure 2 illustrates the comparative PET/CT images obtained from the conventional diagnostic device alongside those from the RefleXion X1. This comparison is demonstrated through images from three representative patients, each with different pathologies: Patient A with liver cancer, Patient B with a PET-avid left supraclavicular (SCV) node, and Patient C with a lesion in the superior mesenteric artery.

Comparative PET/CT Imaging Across Systems and Pathologies. the Left Column Displays Diagnostic PET/CT Images, While the Right Column Shows Emulated X1 PET Images Overlaid on the Diagnostic CT. Each row Represents a Different Patient with Distinct Oncological Diagnoses: (A) Liver Cancer, (B) PET-Avid Left Supraclavicular (SCV) Node, and (C) Lesion in the Superior Mesenteric Artery. PET, positron emission tomography.
Quantitative analysis of the PET images yielded an average dPET SUV of 12.6, with derived NTS and AC values on emulated X1 PET images standing at 9.3 and 12.1 kBq/ml, respectively (Table 1). Notably, statistical analysis revealed that dPET SUV exhibited only weak correlations with derived AC (r = 0.170) and NTS (r = 0.393). In contrast, PTV volume, which showed almost no correlation with dPET SUV (r = 0.048), demonstrated a strong correlation with derived AC (r = 0.908) and NTS (r = 0.779). All BgRT plans met the NTS >2.7 requirement, confirming sufficient target signal detection. However, only 8 of 11 plans achieved AC >5 kBq/ml, a key criterion for plan deliverability. The three plans that failed to meet the AC threshold are detailed in Table 1, including, R Renal Bed (dPET SUV = 5.8, AC = 2.4 kBq/ml), Pancreas (dPET SUV = 5.0, AC = 4.51 kBq/ml), and R Ext Iliac LN (dPET SUV = 30.5, AC = 3.34 kBq/ml), leading to insufficient PET signal intensity and BgRT plan in feasibility. However, the third case (R Ext Iliac LN) presented a high SUV (30.5) yet failed to meet AC deliverability, likely because only a small subvolume within the PTV was FDG-avid.
To further assess plan quality and dosimetric performance, all cases were categorized into two groups (shown in Table 2): 1) Sites without dose-limiting OARs near the target (n = 6); 2) Sites with dose-limiting OAR constraints (n = 5). Statistical analysis confirmed no significant differences (P > .05) in PTV volume, PET sup-inf length, or dPET SUV between the two groups. However, PTV V100% and Dmax exhibited significant differences (P < .05) between the two groups. In cases without limiting OAR constraints (n = 6), all plans successfully met dose constraints for critical structures, including the duodenum, small bowel, large bowel, and spinal cord. These cases achieved an average PTV V100% of 94.4%, with a conformity index (CI) of 1.08, heterogeneity index (HI) of 1.3, and maximum dose (Dmax) of 124.0%. Among these, five plans achieved an even higher average PTV V100% of 95.22% and Dmax of 120.8%, with the exception of a single pelvic site (LN Lymphoma Groin, PTV V100% = 90.3%, Dmax = 140%), which exhibited slightly reduced target coverage and an elevated maximum dose. In cases with adjacent dose-limiting OAR constraints (n = 5), target coverage was reduced as expected. These plans achieved an average PTV V100% of 78.0%, with Dmax of 133.2%, CI of 1.4, and HI of 1.3. The observed coverage reduction reflects the necessary dose trade-off to prioritize OAR sparing while maintaining therapeutic dose levels within the target volume.
Dosimetric Outcomes for BgRT Plans Treatment Sites with and Without Limiting OAR Structures.

Abbreviations: PTV, planning target volume; nCI: new Conformity Index.
Figure 3 further illustrates the dose distribution patterns, highlighting the contrast between cases with and without OAR constraints. Patient A (Liver Cancer, PTV V100% = 95.0%, Dmax = 126%, CI = 1.0) and Patient B (Left SCV node, PTV V100% = 94.8%, Dmax = 118%, CI = 1.1), both lacking significant OAR constraints, demonstrated higher dose conformality, with centrally concentrated dose localization within the PTV. Patient C (Superior Mesenteric Artery, PTV V100% = 79.2%, Dmax = 132%, CI = 1.2) exhibited a “butterfly effect”, where larger firing arcs were not utilized due to OAR constraints, limiting dose modulation and leading to compromised dose conformality (Figure 3(c)).

Dose Dstribution and Dose-Volume Histograms (DVH) for BgRT Plans. The Left Panels Depict Axial Slices of PET/CT Images with Overlaid Dose Distribution for (A) a Patient with Liver Cancer, (B) a Patient with a PET-Avid Left SCV Node, and (C) a Patient with a Lesion in the Superior Mesenteric Artery. The Right Panels Present the Corresponding DVHs, Delineating the Dose Received by the Target Volume and Various Organs at Risk (OARs). PET, positron emission tomography.
Table 3 summarizes the treatment delivery parameters and machine dynamics across all plans. The average BgRT treatment time was 1014.6 s, with an average maximum MLC transition rate of 75.9 transitions per second and an average transition rate of 36.6 transitions per second. Correlation analysis revealed a strong positive relationship between BTZ superior-inferior (sup-inf) length and treatment time (r = 0.896), maximum MLC transition rate (r = 0.875), and average transition rate (r = 0.648). Similarly, a strong correlation was observed between BTZ volume and these parameters, with correlation coefficients of 0.907, 0.967, and 0.758, respectively. These results suggest that larger BTZ volumes and increased sup-inf target length contribute to longer treatment times and higher MLC modulation demands. A comparison of treatment time, maximum MLC transitions per second, and average transition rate between plans with and without dose-limiting OAR constraints revealed no significant differences (P > .05). This finding indicates that the presence of OAR constraints did not directly influence MLC modulation or treatment duration. Instead, MLC modulation intensity and time were predominantly driven by the extent of anatomical motion and target size rather than OAR-related dose limitations.
Treatment Delivery Parameters and Machine Dynamics for BgRT Plans.
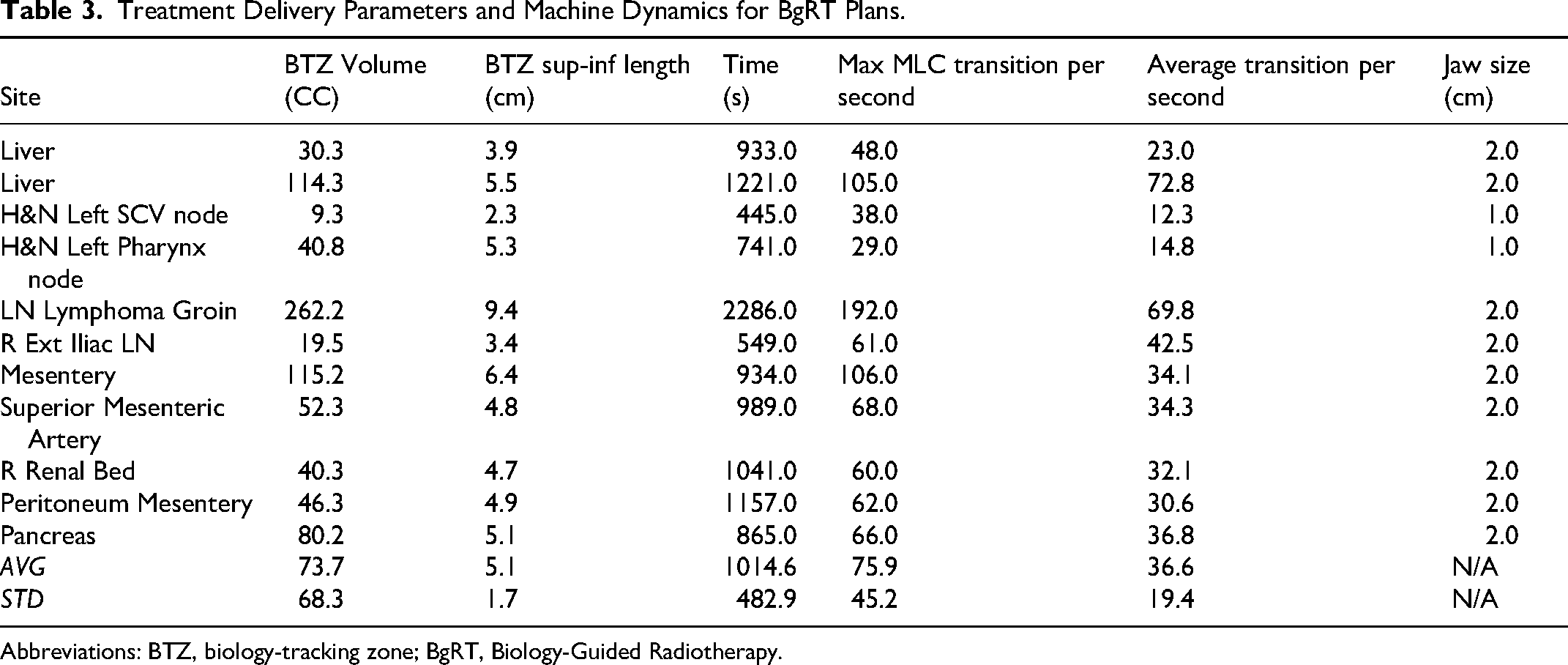
Abbreviations: BTZ, biology-tracking zone; BgRT, Biology-Guided Radiotherapy.
Discussion
This study pioneers the exploration of the RefleXion X1 TPS for BgRT in the treatment of various cancers beyond lung and bone lesions.
Feasibility Assessment Using dPET
Despite the challenge of reduced PET image quality, all eleven plans met the NTS thresholds, confirming the feasibility of SBRT-style BgRT planning. Furthermore, 8 of the 11 plans (72.7%) achieved the AC deliverability (>5 kBq/ml), suggesting a robust signal during BgRT delivery. Consistent with the BIOGUIDE-X trial on lung and bone tumors, where an SUV threshold of >6 was established as an initial criterion for potential BgRT implementation, the present study observed a similar trend. Among the 11 plans, the average dPET SUV was 12.6; and for the 8 cases with dPET SUV >6, 7 (87.5%) successfully met the AC > 5 kBq/ml deliverability threshold. Among the three cases that failed to meet the AC threshold, two cases had a dPET SUV <6 (R Renal Bed, SUV = 5.8, AC = 2.4 kBq/ml; Pancreas, SUV = 5.0, AC = 4.51 kBq/ml). One potential contributor to low AC values is the difference in FDG injection protocols. While clinical BgRT protocols typically use a 15 mCi FDG injection, the dPET scans in this study used 6–10 mCi, which may have resulted in reduced PET signal intensity, particularly in tumors with low SUV or high background FDG uptake. Increasing FDG injection doses to 15 mCi in BgRT as studied in BIOGUIDE-X may enhance AC values and improve deliverability in borderline cases. These findings support the continued use of SUV >6 as a reasonable initial selection criterion for BgRT in other anatomical sites. In general, this study demonstrates the feasibility of using dPET as an emulation tool for evaluating BgRT eligibility before treatment planning. This approach is particularly valuable for assessing patients with borderline FDG uptake tumors or those with dose-limiting organs-at-risk (OARs), enabling for early identification of potential limitations.
Beyond SUV: Multi-Factorial Determinants of BgRT Feasibility
Further statistical analysis revealed that dPET SUV exhibited only a weak correlation with AC (r = 0.170) and NTS (r = 0.393), indicating that SUV alone is insufficient to predict BgRT feasibility across different anatomical sites. Notably, our findings highlight key exceptions. For instance, the R Ext Iliac LN case (dPET SUV = 30.5, AC = 3.34 kBq/ml) failed to meet the AC threshold, despite having a high SUV. This discrepancy is likely attributable to only a small subvolume within the PTV was FDG-avid. A comparative analysis of two cases with identical dPET SUV values (5.8)—H&N Left SCV Node versus Right Renal Bed—further highlights the critical role of PET-avid subvolume within the PTV. The H&N Left SCV Node (PTV = 2.3 cc) met the AC threshold (5.8 kBq/ml), with over 80% of the PTV volume exhibiting PET avidity. In contrast, the Right Renal Bed (PTV = 15.6 cc) failed to meet the threshold (2.4 kBq/ml), likely due to a larger PTV volume that included non-avid regions. These findings underscore that tumor location, the extent of PET-avid subvolume, and background PET signal must all be considered alongside SUV when evaluating BgRT eligibility. This reinforces the need for a comprehensive patient selection framework that integrates multiple parameters—beyond SUV or volume alone—to optimize BgRT outcomes. Future studies with larger sample sizes may enable the development of machine learning-based predictive models (eg, logistic regression, support vector machines) to improve pre-treatment feasibility assessments. By incorporating multivariate analysis, such models might enhance BgRT eligibility predictions by identifying complex interactions between plan characteristics.
Plan Quality Assessment and Impact of OAR Constraints
In this study, treatment sites were categorized into two groups based not simply on anatomical location but on the actual dosimetric impact of adjacent critical structures on plan optimization. This functional classification, determined through the treatment planning process rather than anatomical assumptions, allowed for objective evaluation of the SCINTIX planning system's ability to balance target coverage and OAR sparing across various clinical scenarios. Subsequent dosimetric evaluation of BgRT plans revealed that treatment planning feasibility and target coverage were largely dependent on proximity to dose-limiting OARs. Plans were divided into two groups: those with no adjacent OAR constraints (n = 6) and those with OAR constraints that affected PTV coverage (n = 5). No significant differences were observed between these two groups in terms of PTV volume, PET sup-inf length, or dPET SUV (P > .05), indicating that anatomical target characteristics were comparable across groups. However, significant differences (P < .05) were observed in PTV V100% and Dmax. In the unconstrained group, average PTV V100% was 94.4%, with an average Dmax of 124.0%. Excluding one pelvic site that was a bit of an outlier (LN Lymphoma Groin, PTV V100% = 90.3%, Dmax = 140%), the remaining five cases achieved an even higher mean PTV V100% of 95.22% and a lower mean Dmax of 120.8%. Conversely, for cases with limiting OARs, average PTV V100% was reduced to 78.0%, with an average Dmax of 133.2%, reflecting the necessary trade-off to spare adjacent dose-sensitive structures. This pattern was particularly evident in the Superior Mesenteric Artery case, where OAR constraints resulted in a dose distribution that exhibited a “butterfly effect,” characterized by non-uniform beamlet delivery and limited target conformality.
Notably, despite the general perception that head and neck regions present significant planning challenges due to numerous critical structures, the specific nodal targets in our study (left supraclavicular and pharyngeal nodes) were successfully planned with high target coverage. While previous studies have demonstrated the capability of the RefleXion X1 system's IMRT mode in head and neck planning, showing improved OAR sparing compared to conventional techniques for nasopharyngeal carcinoma, 22 our study represents the first evaluation of BgRT mode in this anatomical region. The anatomical positioning of these targets relative to critical OARs allowed for adequate target coverage without significant dosimetric compromise. The two head-and-neck treatment plans required the use of 1 cm jaws (compared to 2 cm for other sites) to meet the minimum number of revolutions per beam station necessary for BgRT delivery, while simultaneously achieving the finer dose distribution needed for this complex anatomical region. The smaller jaw setting enabled greater beam overlaps, increasing flexibility in beamlet selection and potentially enhancing dose gradient control at OAR interfaces. This technical adaptation demonstrates the system's ability to handle multi-OAR optimization challenges while maintaining clinically acceptable plan quality in the head and neck region—an important finding for future implementation of SCINTIX therapy in this anatomical site.
Treatment Delivery Parameters
Treatment delivery analysis demonstrated that treatment time and MLC modulation rates were primarily influenced by target volume and motion range rather than by OAR constraints. In the RefleXion X1 system, MLC transition rate represents the frequency of leaf movements during treatment, with higher rates indicating more complex beam modulation patterns and increased plan complexity. These transitions reflect the dynamic adaptations of the beam aperture required to conform to the target while sparing surrounding tissues.
The correlation analysis revealed strong positive relationships between BTZ volume and treatment time (r = 0.907), maximum MLC transition rate (r = 0.967), and average transition rate (r = 0.758). Similarly, superior-inferior target length demonstrated strong correlations with treatment time (r = 0.896), maximum MLC transition rate (r = 0.875), and average transition rate (r = 0.648). These strong correlations indicate that larger targets and those with greater superior-inferior extent require more complex modulation and longer treatment times.
Importantly, no significant differences (P > .05) were observed in treatment time, MLC transitions per second, or average transition rates between plans with and without OAR constraints. These findings suggest that MLC modulation intensity is primarily dictated by the extent of anatomical motion and target size rather than by dose-limiting structures. This observation has important implications for clinical workflow and treatment scheduling. While all plans in this study were successfully generated by the non-clinical TPS, the clinical implications of highly modulated plans (such as potential deliverability challenges or QA passing rates) were not evaluated in this preliminary study and would require further investigation in future clinical implementation studies.
Study Limitations and Future Directions
There are several limitations to this study. The emulated PET workflow, while designed to reflect actual RefleXion PET acquisitions, introduces some uncertainty, as real treatment requires a PET functional modeling session on the X1 system, which may yield different PET signal characteristics. The sample size is limited, and patient selection was based on dPET SUV >5 and data availability, which may introduce selection bias. Furthermore, the TPS used in this study is under continuous development, and improvements in both hardware and software may enhance plan optimization and PET signal processing.
The ongoing BIOGUIDE-X2 trial (NCT06634706), which focuses on BgRT imaging, planning, and emulated delivery studies for lesions outside lung and bone, will provide further validation of these findings and guide future clinical implementation. While FDG is currently the only FDA-cleared radiotracer for this application, future research should explore expanded patient cohorts, potential dosimetric benefits of margin reduction, and the integration of tumor-specific tracers to further broaden the therapeutic scope of this promising technology. In particular, prostate-specific membrane antigen (PSMA) PET has shown promise for BgRT applications. Gaudreault et al demonstrated the feasibility of using PSMA-PET for BgRT to boost dominant intraprostatic tumors, with over 50% of analyzed cases exhibiting suitable signal-to-background ratios for BgRT delivery. 23 Such tumor-specific tracers could potentially improve target definition and delivery precision beyond what is currently achievable with FDG-PET for select patient populations.
Conclusion
This preliminary study successfully generated BgRT plans using emulated X1 PET images for 11 treatment sites across head and neck, abdominal, and pelvic regions. All plans met NTS constraints and 72.7% achieved AC deliverability thresholds, with SUV > 6 serving as a reasonable initial selection criterion for evaluating SCINTIX therapy feasibility beyond the FDA-cleared lung and bone indications. Our findings demonstrate that BgRT feasibility is determined by multiple factors beyond SUV alone, including tumor location, background FDG uptake patterns, and proximity to critical structures. The dosimetric quality of all plans was deemed clinically acceptable according to institutional standards with appropriate trade-offs between target coverage and OAR sparing in anatomically challenging regions. MLC modulation intensity correlated strongly with target volume and superior-inferior extent rather than OAR constraints, providing valuable insights for clinical workflow implementation. This study establishes a practical methodology for pre-screening patients for BgRT eligibility using existing diagnostic PET data, potentially expanding the application of SCINTIX therapy to additional anatomical sites.
Footnotes
Ethics Statement (Committee Approval Number,etc)
This retrospective study was approved by the IRB of University of Texas Southwestern Medical Center(Study Number:STU 082013-008). The requirement for informed consent was waived due to the retrospective nature of the study. All patient data were de-identified prior to analysis, and the study was conducted in accordance with the principles outlined in the Declaration of Helsinki and institutional guidelines for human research ethics.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is partially funded by RefleXion Medical (UTSW SRA202302-0018).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.C. receives a research grant from RefleXion Medical that directly supported this study. B.C. also receives trial support and honoraria from RefleXion Medical outside the scope of this work. T.D. receives trial support from RefleXion Medical outside this work. S.N.B. receives grant support from RefleXion Medical and AstraZeneca unrelated to this study. A.G. receives grant support from RefleXion Medical, Teleflex Medical, and Elekta, all outside the submitted work. G.G., A.D.S., and G.K. are employees of RefleXion Medical. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
