Abstract
Introduction
To investigate the efficacy of a knowledge-based planning (KBP) model in optimizing dose distribution, and identify the inter-institutional variation in radiotherapy of recurrent nasopharyngeal carcinoma (rNPC).
Methods
A total of 70 rNPC patients treated with intensity-modulated radiotherapy (IMRT) were recruited to build a KBP model. Following model refinement, 36 patients were retrospectively enrolled for dosimetric comparison between manually optimized and KBP-generated plans. Ten experienced physicists from six different institutions were engaged to independently design manual plan for a representative case, to assess inter-institutional variations, and differences between KBP and manual plans.
Results
The refined KBP model provided significant reduced dose in brainstem D1cc (the dose received by the “hottest”1 cm3 volume, 41.14 ± 8.51 Gy vs 38.48 ± 8.60 Gy, P < 0.001) and spinal cord D1cc (17.48 ± 9.38Gy vs 12.23 ± 6.56Gy, P < 0.001). In addition, The mean dose (Dmean) of brainstem, spinal cord, mandible, parotid, temporomandibular joint and inner ear were statistically decreased (P < 0.05). In validation cohort, KBP model eliminated the hotspot (0.57 ± 0.01% vs 0.00 ± 0.00%, P < 0.001), improved target homogeneity (HI: 0.06 ± 0.00 vs 0.08 ± 0.00, P < 0.001), and performed superior to the manual plans in sparing organs. While all institutions achieved comparable target coverage, manual plans exhibited substantial variability in sparing brainstem. KBP implementation reduced inter-institutional dose disparities for brainstem (46.30 ± 10.08 Gy vs 41.80 ± 5.80 Gy, P = 0.041) and spinal cord (26.08 ± 7.06 Gy vs 18.19 ± 1.98 Gy, P = 0.002). Additionally, planning efficiency increased by 48.7% (39 vs 76 min).
Conclusions
This KBP framework optimized rNPC reirradiation from three dimensions: 1) Enhanced OARs’ protection; 2) Improved target homogeneity; 3) Improved the multi-institutional consistency and efficiency of planning. These advancements established a clinically actionable paradigm for precision reirradiation.
Keywords
Introduction
In recent years, radiotherapy has emerged as the cornerstone of nasopharyngeal carcinoma (NPC) management, due to the tumor's inherent radiosensitivity and deep-seated anatomical constraints. Conventional radiation therapy has demonstrated favorable long-term survival outcomes, though often at the expense of acute and late complications.1,2 The introduction of advanced techniques such as intensity-modulated radiation therapy (IMRT) has markedly enhanced local control rates.3,4 Nevertheless, 10%-15% of patients still experienced local recurrence. 5
Salvage therapy for locally recurrent NPC (rNPC) remains clinically demanding. 6 Reirradiation carries substantial risks of cumulative radiation-induced morbidity. 7 While IMRT is the preferred option, it is not devoid of acute and late complications. 8 Current evidence supports a prescription dose ≥60 Gy during reirradiation to optimize overall survival.9,10 Recent work by You et al. highlighted that hyperfractionated IMRT may reduce severe late toxicity rates and improve survival in locally recurrent cases. 11 However, cumulative dose constraints to previously irradiated organs at risk (OARs) from the initial treatment course critically limit dose escalation. Consequently, achieving optimal therapeutic balance—maximizing tumoricidal efficacy through minimal effective tumor dose while strategically redistributing radiation burden away from critical OARs—may represent a promising paradigm.
Current clinical practice in rNPC reirradiation planning predominantly relies on manual optimization, which narrowly focuses on trade-offs between target coverage maximization and OAR sparing. However, inherent limitations of treatment planning systems (TPS), dosimetrist expertise variability, and time-intensive iterative optimization processes render the manual identification of globally optimal dose distributions clinically impractical. Recent advancements in automated planning algorithms have introduced transformative tools for radiation oncology. 12 Among these, knowledge-based planning (KBP) constitutes a data-driven methodology that employs historical dose distribution databases to predict patient-specific optimal dosimetry.13,14 By integrating anatomical data from computed tomography (CT) and delineated structures, KBP facilitates individualized, protocol-consistent dose predictions.
To rigorously evaluate KBP's clinical utility, we curated an IMRT plan dataset from a cohort of rNPC patients. We conducted a granular comparative analysis between KBP-generated and manual optimized plans, followed by cross-institutional validation for a representative sample. This study aims to to determine whether KBP can consistently achieve dosimetrically superior plans and quantify inter-institutional variations in planning practices.
Methods and Materials
Model Building and Refinement
A cohort of 70 rNPC patients treated with IMRT between 2017 to 2022 was retrospectively analyzed for model development. Target volumes and OARs were contoured by radiation oncologists in accordance with Radiation Therapy Oncology Group protocols, with clinical judgment applied to customize planning target volume (PTV) expansions while sparing critical OARs.15,16 Planning risk volumes (PRVs) were exclusively assigned to brainstem and spinal cord, employing uniform 3-mm expansion margins across all cases. The PTV hierarchy comprised a primary gross tumor volume (PGTV; prescription dose: 64.8 Gy), and a prophylactic clinical target volume (PCTV; prescription dose: 54 Gy), delivered in 54 fractions as per established protocols. 11 All the planning CT datasets (3-mm slice thickness), structure sets, radiotherapy plans and dose matrices were imported into the Eclipse TPS (version 15.6, Varian Medical Systems, Palo Alto, CA, USA) for analysis using the Rapidplan knowledge-based planning module. Each plan was re-optimized using the KBP model to achieve dosimetrically favorable distributions. If the re-optimized plan demonstrated dosimetric superiority over the initial treatment plan, it was selected as the modeling plan; otherwise, the original treatment plan was retained for model construction.
Model Evaluation
Between January 2022 and June 2024, 36 consecutive non-metastatic rNPC patients were retrospectively enrolled (information detailed in Table 1). Treatment plans were evaluated by a experienced physicist team, and manually re-optimized as perfect as possible. Following KBP implementation, dosimetric parameters of automatic plans were compared against manually optimized counterparts.
Patients’ Characteristics.

*The eighth edition of the American Joint Committee on Cancer staging system.
To assess inter-institutional variability, ten experienced radiotherapy physicists from six institutions using Varian TPS were engaged to independently design a plan for a representative patient under identical protocol constraints, described in Table 2.7,17 These manual plans were compared to identify the inter-institutional variations in dose distribution. In addition, the KBP-generated plan for this representative patient was generated by Eclipse and divered the ten physicists to re-optimize. The dose of brainstem and spinal cord in re-optimized KBP plan were compared to their manual plan.
Protocol Constraints of Target Volumes and Critical OARs for rNPC Patients.

*If the dose constraints could not be satisfied, the dose to the structure was minimized to the greatest extent feasible.
Model Application
The KBP implementation workflow comprised two stages (Figure 1): Stage 1, manually imported the CT images and structure sets of a new patient into Eclipse to generate an OAR-specific ‘line’ constraints through the KBP model. Stage 2, Eclipse systems can produce a automated clinical plans based to the aforementioned ‘line’ constraints. For institutions utilizing non-Varian TPS, the KBP model exported patient-specific extensible markup language (XML) file containing optimization objectives, serving as reference templates for manual plan optimization.
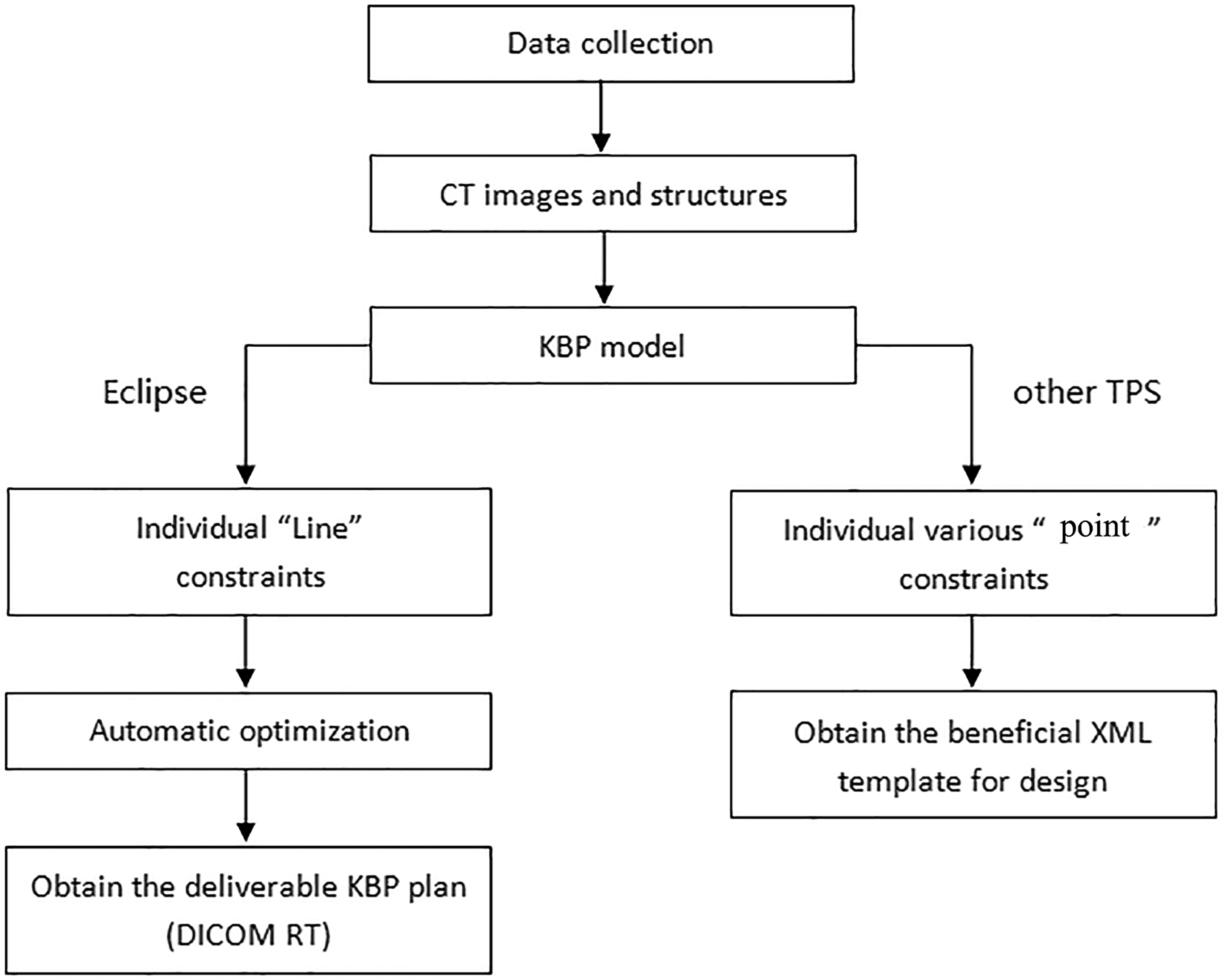
The Workflow of KBP Model Application for Eclipse and Other TPS.
Dosimetric Analysis
For parallel organs (eg, parotid), we typically employed the mean dose (Dmean) for evaluation. In contrast, for serial organs (eg, spinal cord), the maximum dose (Dmax) has traditionally been used. However, Dmax is highly sensitive to the accuracy of organ delineation. Additionally, as highlighted in ICRU Report 83, a meaningful hot spot dose assessment requires a clinically relevant volume. For this reason, our study adopted D1cc (the minimum dose to the “hottest” 1 cm³ volume) to evaluate serial organs such as the brainstem and spinal cord. Nevertheless, for small-volume structures (eg, lens, optic nerve, optic chiasm) with a total volume less than 1 cm³, D1% (the minimum dose to the “hottest” 1% volume) may serve as a more reasonable metric. The Conformity Index (CI) was calculated using the following equation
18
: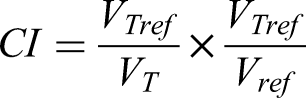
Where VTref was the volume of the target tissue receiving at least the reference prescription dose. VT denoted the total volume of the delineated target structure and Vref indicated the total volume enclosed by the reference isodose.
The homogeneity index (HI) was defined as
19

Where Dx% was the absorbed dose received by x% of the target volume.
Statistical Analysis
Analyses were performed using SPSS Statistics (v23.0, IBM Corp., Armonk, NY). Normality was assessed via Kolmogorov-Smirnov testing. Parametric data underwent paired t-tests; non-parametric comparisons used Wilcoxon signed-rank tests. All stests were two-sided with a P value <0.05 was considered significant.
Results
Dosimetric Data of KBP Model Refinement
Following model refinement, as summarized in Table 3, no statistically significant differences in target coverage were observed between the initial and KBP plans for 70 patients. However, significant reductions were achieved in brainstem D1cc from 41.14 ± 8.51 Gy to 38.48 ± 8.60 Gy (P < 0.001), and spinal cord D1cc from 17.48 ± 9.38 Gy to 12.23 ± 6.56 Gy (P < 0.001). Furthermore, KBP optimization demonstrated significantly greater reductions in Dmean of brainstem, spinal cord, mandible, parotid, temporomandibular joint (TMJ), inner ear (P < 0.05).
Comparison of the Dosimetric Parameters Between the Initial and KBP Plans.

‘Line’ Constraints Optimization of KBP Model
Figure 2 illustrated the KBP-derived dose-volume histogram (DVH) prediction framework. The green band represents the model-generated DVH probability distribution for brainstem dose based on the anatomical features extracted from newly acquired CT datasets. The optimized dose-volume constraint line (dotted line) was algorithmically derived from the lower boundary of this predictive band (left) (Figure 2). The green solid line exhibited the final DVH optimized by the KBP model for the brainstem (right) (Figure 2b).
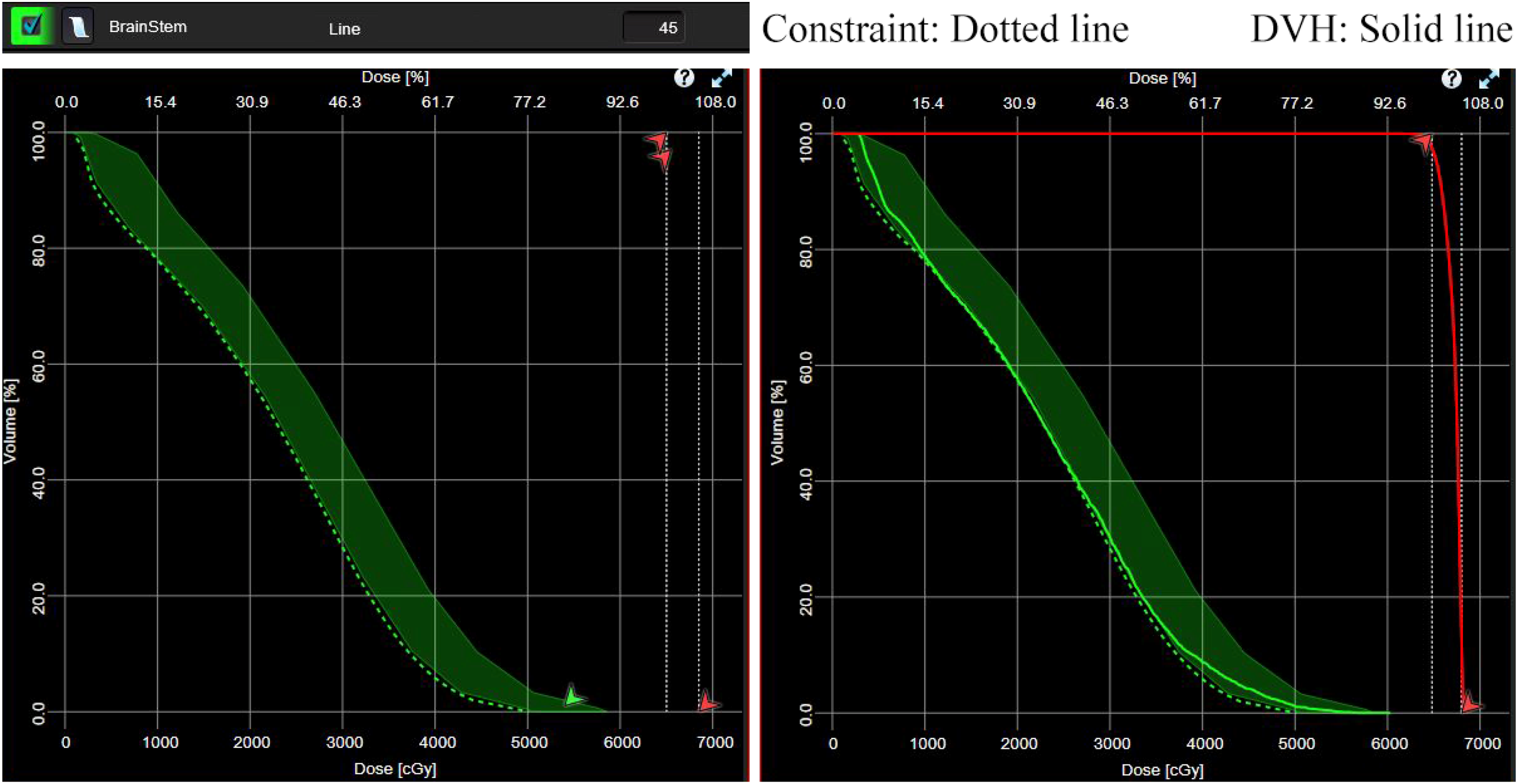
‘Line’ Constraints Optimization of Brainstem Generated by KBP Model.
Dosimetric Data of KBP Model Evaluation
Under normalized target coverage conditions (V98% = 95% for PGTV), KBP plans exhibited superior dosimetric performance compared to manual plans (Table 4). Significant improvements were observed in PGTV hotspot (0.00 ± 0.00 vs 0.57% ± 0.01%, P < 0.001) and HI (0.06 ± 0.00 vs 0.08 ± 0.00, P < 0.001). Enhanced OAR sparing was consistently achieved for brainstem, spinal cord, temporal lobe, mandible, parotid, TMJ, inner ear and larynx (P < 0.05).
Comparison of the Dosimetric Parameters Between the Manual and KBP Plans for 36 Patients.

Dosimetric Data Across Institutions
The DVHs of ten manual plans from six institutions for the representative patient were shown in Figure 3, indicating that all ten manually optimized plans met PGTV coverage requirements, but considerable inter-institutional variation existed in brainstem DVH.
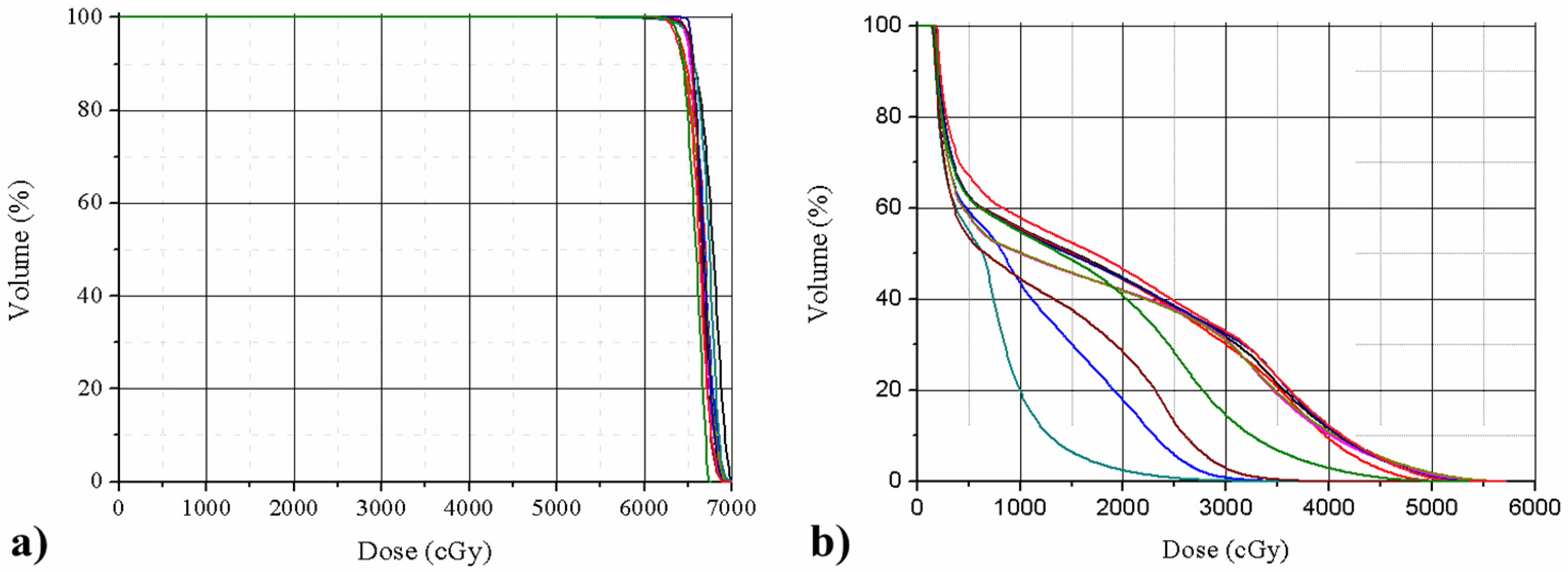
The DVHs of ten Manual Plans from six Institutions for the Representative Patient (a: PGTV; b: Brainstem).
As shown in Figure 4, implementation of KBP modeling yielded substantial dose reduction for neurological structures: Brainstem D1cc from 46.30 ± 10.08 Gy (manual) to 41.80 Gy ± 5.80 Gy (KBP), 9.72% reduction (P = 0.041); Spinal cord D1cc from 26.08 Gy ±7.06 Gy (manual) to 18.19 ± 1.98 Gy (KBP), 30.25% reduction (P = 0.002), respectively. Additionally, Planning efficiency analysis revealed KBP required 39 min per case versus 76 min (range: 39-169 min) for manual optimization, representing a 48.68% reduction in planning time.
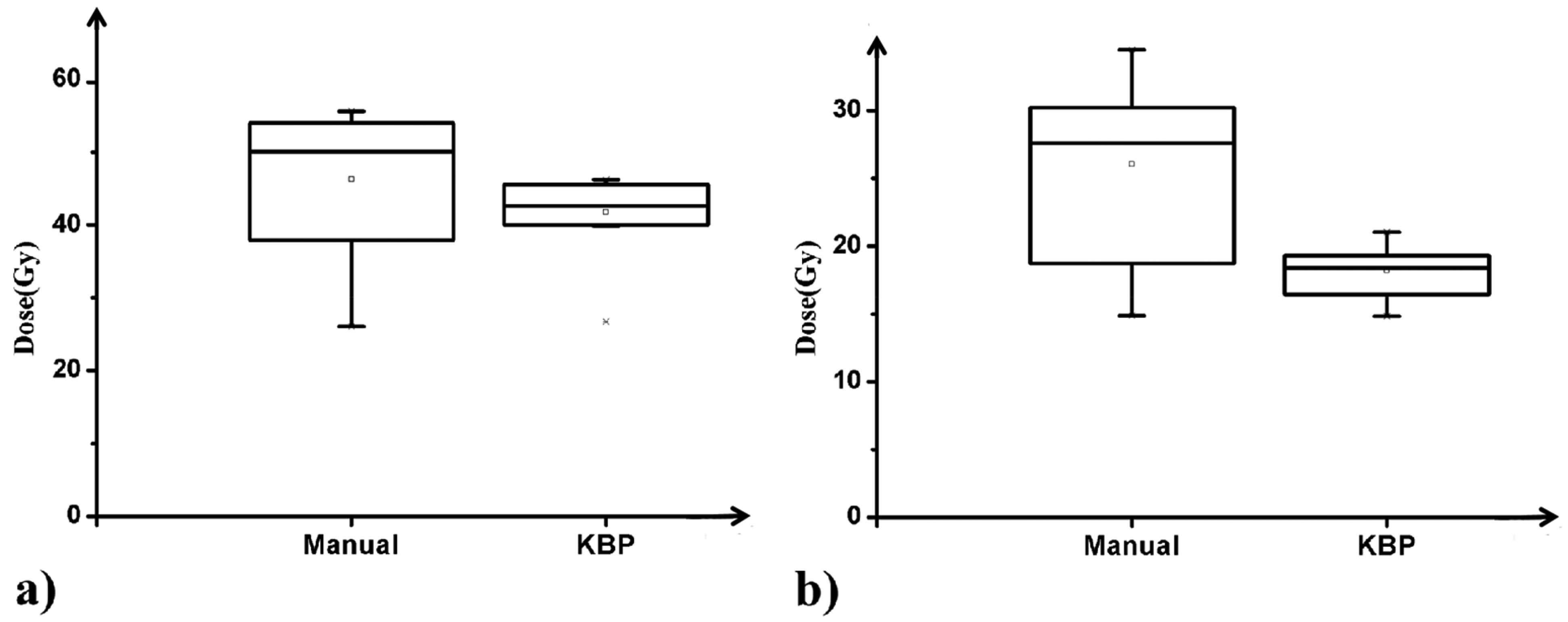
The Inter-Institutional Average D1cc Values of Adjacent Critical Neurological Structures from the Manual and KBP Plan (a: brainstem, b: spinal cord).
Discussion
The dosimetric superiority of KBP over manual optimization has been well-established across multiple malignancies,13,14 while evidence specific to locally rNPC remained rare prior to this investigation. Our study developed the disease-specific KBP framework for rNPC reirradiation using IMRT, demonstrating clinically meaningful improvements in dose HI for PGTV and sparing for some critical OARs. This optimized balance between target volume dose escalation and OAR protection addressed the critical clinical need for minimizing cumulative radiation toxicity in rNPC patients, potentially improving long-term survival and quality of life.
In this study, the KBP approach demonstrated significant improvement in dose homogeneity, reducing the HI from 0.07 in manual plans to 0.06 (P < 0.001). This aligned with prior KBP applications in primary NPC radiotherapy reported by Toi et al. (HI reduction:0.09→0.07) and our earlier research (HI reduction:0.08→0.07).20,21 Given that reirradiation patients have already received high cumulative doses from initial treatment, with reported incidences of nasopharyngeal necrosis (8/38) and catastrophic hemorrhage (10/38) in second-course radiotherapy. 22 Optimizing target volume dose homogeneity becomes paramount for rNPC compared to primary cases. Our findings build upon existing evidence for HI optimization in primary NPC radiotherapy, further advancing homogeneity standards to potentially mitigate reirradiation-related mucosal complications in recurrent disease.
Furthermore, the KBP methodology overcame inherent limitations of conventional manual planning's narrow focus on isolated dose parameters. By implementing DVH-based ‘line’ constraints, KBP induced a systematic left and downward shift of OARs’ DVH, thereby reducing all clinically relevant dose metrics—a critical advancement for reirradiation where cumulative OAR tolerance was profoundly compromised. Prior studies report alarmingly high rates of severe (grade ≥4) late toxicities after reirradiation (actuarial 28% at 5 years), including mucosal necrosis, osteonecrosis, arterialblowout and ohters. 23 Our KBP model achieved comprehensive dose reduction across multiple priority OARs (brainstem, spinal cord, temporal lobe, mandible, parotid, TMJ, inner ear, and larynx), with particularly pronounced sparing of the inner ear Dmean and spinal cord D1cc over > 6 Gy (Table 4). These magnitude of dose reductions may substantially lower risks of severe radiation-induced complications.
These dosimetric advantages of KBP approach for rNPC may be derived from model refinement and the the suboptimal dose constraint settings used by planner. 24 However, limitations still exist in conforming the optimal DVH during KBP model building process. As Tambe et al. demonstrated in lung and Scaggion et al. validated in prostate cancer,25,26 strategic model re-optimization enhanced plan quality—a principle we operationalized through rigorous case curation (78 initial samples refined to 70 after physics-led exclusion of anatomically aberrant cases). As shown in Table 3, KBP plans provided the superior brainstem D1cc (P < 0.001) and spinal cord D1cc (P < 0.001) compared to the initial sample plans. Moreover, the Dmean of mandible, parotid, TMJ, inner ear were significantly reduced. Hence, model refinement was indispensable. Nevertheless, the limited sample size and current commercial TPS architectures may not obtain the optimal DVH, needing future large-scale validation.
The another advantage of KBP model was that its automation leverages patient-specific anatomical features derived from CT-based contours to generate individualized OAR dose-volume constraints, effectively decoupling plan quality from planner experience and institutional resource disparities. 27 This capability holds particular promise for standardizing rNPC reirradiation across multi-center networks, which has been reported to be an effective instrument in multi-center clinical trials in terms of head and neck tumors. 28 Our six-institution analysis for representative case revealed consistent PGTV coverage (V98% = 95%) but substantial variability in brainstem dose sculpting (Figure 3), mirroring patterns observed in prostate cancer by Bossart et al. and head-and-neck cohorts by Sean et al.29,30 Implementation of our KBP framework reduced inter-institutional variability in serial OAR doses (brainstem D1cc: 9.7% reduction, P = 0.041; spinal cord D1cc: 30.3% reduction, P = 0.002), while maintaining target coverage (Figure 4).
At present, this KBP model has been validated in our single-center cohort, we proposed a multi-center implementation workflow: 1) Directly generated automated IMRT plan via inputting CT/structure sets into Eclipse RapidPlan; 2) XML-based constraint templates for non-Varian TPS users KBP increased clinical efficiency by significantly reducing planning time. 31 In recent years, there have also been relevant research reports on dose prediction using KBP combined with deep learning.32,33 Consequently, it is expected to utilize the Internet to achieve multi-center sharing of the KBP model resource in future, and generate clinical treatment plan within half a day. We hope that more institutions join us to enhance the accuracy of the multi-institution KBP model for rNPC. Moreover, future research may further explore the application of KBP in other high-risk cases.
Conclusion
The KBP paradigm demonstrated its potential in rNPC reirradiation from three dimensions: 1) Enhanced homogeneity of PGTV and reduced the hotspot; 2) Dose reduction of critical OARs potentially decreased the severe radiation toxitciy; 3) Enhanced multi-center consistency with improved efficiency. This methodology established a foundational framework for high-precision, resource-efficient rNPC radiotherapy.
Footnotes
Ethical Considerations
The study protocol was approved by Institutional Review Board of Sun Yat-Sen University Cancer Center with the approval number B 2019-131-01, where the need for written informed consent was waived due to the retrospective nature of data analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China NO.82373211, and Research funding project of Sun Yat-Sen University Cancer Center NO.qnrc-2024-07.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
