Abstract
Introduction
Uterine sarcoma (US) is an extremely rare and aggressive gynecologic malignancy with a poor overall survival (OS). The efficient prognostic biomarker is currently lacking.
Methods
Utilizing a Sweden microarray dataset from the Gene Expression Omnibus (GEO) (GSE119043, n = 50) and a clinical cohort (n = 31) retrospectively collected from Suining Central Hospital, we analyzed β-catenin expression profiles and corresponding clinicopathological characteristics. Immunohistochemistry (IHC) was used to assess β-catenin expression level. Survival analysis was used to assess the relationship between β-catenin expression and prognosis. Gene set enrichment analysis (GSEA) was performed to characterize the specific pathways involved in β-catenin expression.
Results
Immunohistochemistry indicated that β-catenin expression was significantly upregulated in US group compared to both the normal uterine smooth muscle (UNSM) and uterine leiomyoma (ULM) groups (P < .01). IHC also exhibited a significant difference in β-catenin expression levels in four pathological subtypes. Leiomyosarcoma (LMS) and high-grade endometrial stromal sarcoma (HG-ESS) suggested higher levels of β-catenin expression compared with adenosarcoma (AS) or low-grade endometrial stromal sarcoma (LG-ESS), but no statistically significant difference was found in box plot (P > .05). GSEA indicated that transcriptional dysregulation in cancer, Wnt, AMPK, MAPK, PI3K, p53, Ras, and TNF signaling pathway were positively enriched in β-catenin high-expression group. Though survival analysis showed that β-catenin expression level was not associated with survival, low-β-catenin expression group showed a longer median OS compared to high expression group (56.17 months VS 9.60 months) in Sweden microarray dataset. Similar results were also observed for progression-free survival (PFS) in clinical cohort (not reached VS 45.97 months in high-expression group). Tumor type, lymphadenectomy, family history of malignancy and tumor recurrence remained significant predictors of OS, while only tumor type, stage and tumor recurrence had prognostic significance for PFS. Age, tumor size, menopausal status, CA125, adjuvant chemotherapy, and adjuvant radiotherapy, were not associated with survival (P > .05).
Conclusion
β-catenin was highly expressed in uterine sarcoma and may be promising as a novel potential biomarker for diagnosis and prognosis.
Introduction
Uterine sarcoma (US) is one of the most aggressive gynecologic malignancies with an extremely poor overall prognosis, that accounts for approximately 1% of female genital tract malignancies and up to 7% of uterine malignancies. 1 Unfortunately, the mortality rate associated with this disease is quite high at approximately 30%. 2 Different from endometrial cancer, US derives from uterine mesenchymal cells and usually occurs after menopause. 3 Uterine sarcomas consist of several histological types, such as leiomyosarcoma (LMS), undifferentiated uterine sarcoma (UUS), adenosarcoma (AS), and endometrial stromal sarcoma (ESS), which including low-grade endometrial stromal sarcoma (LG-ESS) and high-grade endometrial stromal sarcoma (HG-ESS).4–6 Notably, carcinosarcoma is categorized as a subtype of endometrial cancer according to the International Federation of Gynecology and Obstetrics (FIGO) system staging and classification in 2023. 7 Currently, the prognosis of US is still extremely poor due to the lack of standard therapeutic options. The 5-year survival rate ranges from 50–55% for early-stage uterine sarcomas to 8–12% for advanced cases. 8 Therefore, further insights are desperately needed to predict the outcome of uterine sarcomas to improve the prognosis. However, the discovery of new biomarkers and specific targeted therapies is still challenging owing to the low incidence of the disease.
The mechanism underlying UCS development remains unsettled. β-catenin, which is encoded by CTNNB1 (ENSG00000168036), serves as an intracellular signal transducer in the canonical Wnt signaling pathway. 9 Numerous studies have shown that the abnormal regulation of Wnt/β-catenin signaling is involved in the occurrence and development of various cancers.10–12 As a key transduction intermediate, beta-catenin plays a crucial regulatory role in these cancers. Ovarian cancer cells have been shown to grow and metastasize by activating the Wnt signaling pathway. 13 The expression and membrane localization of β-catenin are associated with METTL3-promoted cell migration, invasion, and epithelial-to-mesenchymal transition (EMT) in cervical cancer. 14 Mutation of β-catenin exon 3 (87.0%; 47/54) was the main cause in young patients with low-grade and low-stage endometrioid endometrial carcinoma. 9 However, the expression of β-catenin in US remains unclear. In this study, to clarify the role of β-catenin in US, we explored the relationship between β-catenin expression and the prognosis of US.
Materials and Methods
Data Resource
This study included two cohorts in total. The gene expression profiles and corresponding clinicopathological information were downloaded from the open databases, including a Sweden microarray dataset (GSE119043) from the Gene Expression Omnibus (GEO) (https://www.ncbi.nlm.nih.gov/geo/), which contained data from 50 UUS patients. The above data are publicly available. A total of 31 diagnosed patients and clinicopathological information was retrospectively collected and analyzed from the department of pathology from May 2014 to January 2022. The study received approval from the Review Board of Suining Central Hospital (No. LLSLH20220051; August, 31th, 2022) and the Ethics Committee of Zunyi Medical University (No. 2020-1-013; March, 30th, 2020). The written informed consent was obtained from each participant, allowing the use of their clinical data, archived paraffin-embedded specimens, imaging, and pathological images for research and publication purposes. This research report conforms to REMARK guidelines. 15 All patient details have been de-identified.
The primary endpoint was overall survival (OS), defined as days from initial diagnosis to death or last follow-up. Progression-free survival (PFS) was defined as days from initial diagnosis to disease progression or death, whichever occurred first (or the date of last follow-up if progression or death had not yet occurred). The last follow-up was in September 2023. The median follow-up time was 67.97 months (range 0.77-105.90 months). Staging was assessed according to the International Federation of Gynecology and Obstetrics (FIGO) stage system. 16
Pathological Inclusion Criteria
According to the National Comprehensive Cancer Network (NCCN) guidelines, the most common pathological types are leiomyosarcoma (LMS), endometrial stromal sarcoma (ESS), undifferentiated uterine sarcoma (UUS), and adenosarcoma (AS). 17 Notably, carcinosarcoma is categorized as a subtype of endometrial cancer according to the common newest classification. Therefore, we included patients with definitely pathological confirmation in this study, based on the fifth edition of the World Health Organization (WHO) Classification system of Tumors of Female Reproductive Organs in 2020. 18 Patients with metastatic sarcoma from other gynecological sides or without complete information (without a definitive pathological diagnosis, clinical findings, and loss of follow-up data, etc) were excluded.
Immunohistochemistry (IHC)
In this study, we thoroughly reviewed the medical records of 31 patients with pathologically confirmed US, including 16 samples of LMS, 14 of ESS and 1 of AS, in the department of pathology of Suining Central Hospital. 19 Smooth muscle tissue of the uterine wall and leiomyoma tissue were also obtained if present.
The expression of β-catenin was assessed by immunohistochemical method using a mouse-anti-human monoclonal antibody (MX043, MAIXIN, Fuzhou, Fujian). Tissue specimens were fixed with 10% formalin and preserved in paraffin. The paraffin-embedded blocks were sectioned continuously to a thickness of 4 µm and fixed on slides for immunohistochemical staining. Paraffin-embedded sections were dewaxed and then repaired using ethylenediaminetetraacetic acid (EDTA) antigen retrieval buffers. All IHC staining were performed using an automated immunostainer (VENTAN Bench Mark XT, Roche, Swiss). Human colorectal adenocarcinoma tissue was used as a positive control. The higher the expression content of β-catenin was, the greater was the distribution density and the stronger the positive result would be.
β-catenin expression was evaluated using the H score, which is defined as the product of the staining intensity and the percentage of positively stained cells.20–23 The scoring system for staining intensity was as follows: 0 points for no staining, 1 point for light yellow, 2 points for brown‒yellow, and 3 points for brown. The percentage of positive cells was also considered, and the scores were as follows: 0 points, positive cells 5% or less; 1 point, positive cells between 6% and 25%; 2 points, positive cells between 26% and 50%; 3 points, positive cells between 51%-75%; and 4 points, positive cells greater than 75%. The resulting total scores were categorized as negative (0 points), weakly positive (1-4 points), moderately positive (5-8 points), and strongly positive (9-12 points). The scoring process was performed independently by two gynecologic pathologists using optical microscopy. The decision was made after discussion within the group if inconsistent.
Gene Set Enrichment Analysis
Differential expressed genes (DEGs) were analyzed between β-catenin-low- and high-expression group in patients of Sweden microarray cohort using the R package DESeq2 (v1.44.0). Gene set enrichment analysis (GSEA) was then conducted using the R package clusterProfiler (v4.12.0) to explore the potential molecular mechanisms underlying the distinct gene expression. 24 Normalized enrichment score (NES) was calculated with gene set permutations set as 1000 times. Gene sets with |NES| > 1, adjusted P < .05, q < 0.05 were considered as significant enrichment.
Statistical Analysis
The statistical analysis was performed using R (version 4.4.0). The distributions of categorical variables and continuous variables between cases and controls were performed using Pearson χ2 tests and Student's t tests, respectively. Categorical data are presented as counts and percentages and were analyzed using the Chi-square test and Fisher's exact test. One-way analysis of variance (ANOVA) was applied for univariate analysis. The expression levels of β-catenin in patients were determined using the ggplot2 package. Paired t-test was used to compare the differences between tumor tissues and paired normal samples.
Based on the cut-off value of β-catenin expression, the patients were divided into high and low-β-catenin expression groups, Kaplan-Meier analysis was used to evaluate the relationship between overall survival and β-catenin expression level and pathological subtypes in Sweden microarray dataset (GSE119043) from GEO 25 and the clinical cohort from Suining Central Hospital. Univariate and multivariate cox proportional hazard regression analyses were used to explore β-catenin expression as an independent prognostic factor of US. In the multivariate analysis, adjustments were made for potential confounding factors, including β-catenin expression levels, age, menopausal status, family history of malignancy, tumor stage, serum CA125 levels, tumor size, lymphadenectomy, tumor recurrence, adjuvant chemotherapy, and adjuvant radiotherapy. These factors were selected based on their clinical relevance and support from existing literature. P < .05 was considered statistically significant. Hazard ratios (HRs) and 95% confidence intervals (95% CIs) were calculated.
Results
Baseline Characteristics
According to the 2023 FIGO guidelines of the uterine sarcoma, we analyzed the clinical data collected from the patients’ medical records, including age, histology, clinical and pathologic stage, and adjuvant chemotherapy (CT) or radiotherapy (RT).
From an initial cohort of 56 patients, 31 patients were diagnosed with a definite pathological subtype of uterine sarcoma from the department of pathology of Suining Central Hospital. 19 The baseline characteristics of these patients from the clinical cohort were presented in Table 1. All patients involved in the study were female and categorized according to pathological type into groups of adenosarcoma (AS, n = 1), endometrial stromal sarcoma (ESS, n = 14) (including 12 low-grade ESS (LG-ESS) and 2 high-grade ESS (HG-ESS)), and leiomyosarcoma (LMS, n = 16). Nearly a half of them were over age 50. Most of them (n = 20, 64.52%) were premenopausal. Surgical approaches included total hysterectomy + bilateral salpingo-oophorectomy (TH + BSO), TH + BSO + pelvic lymphadenectomy (TH + BSO + PLA), TH + BSO + PLA + omentectomy, and TH + BSO + PLA + omentectomy + appendectomy. A total of 48.39% (n = 15) of all patients underwent lymphadenectomy. In addition, for adjuvant treatment, less than half of the patients (n = 13, 41.94%) received chemotherapy (CT) with doxorubicin and ifosfamide as the primary protocol, while the rest did not. Only six patients received radiotherapy (RT), while most of patients (n = 25, 80.65%) did not.
Baseline Characteristics of 31 US Patients from the Clinical Cohort.
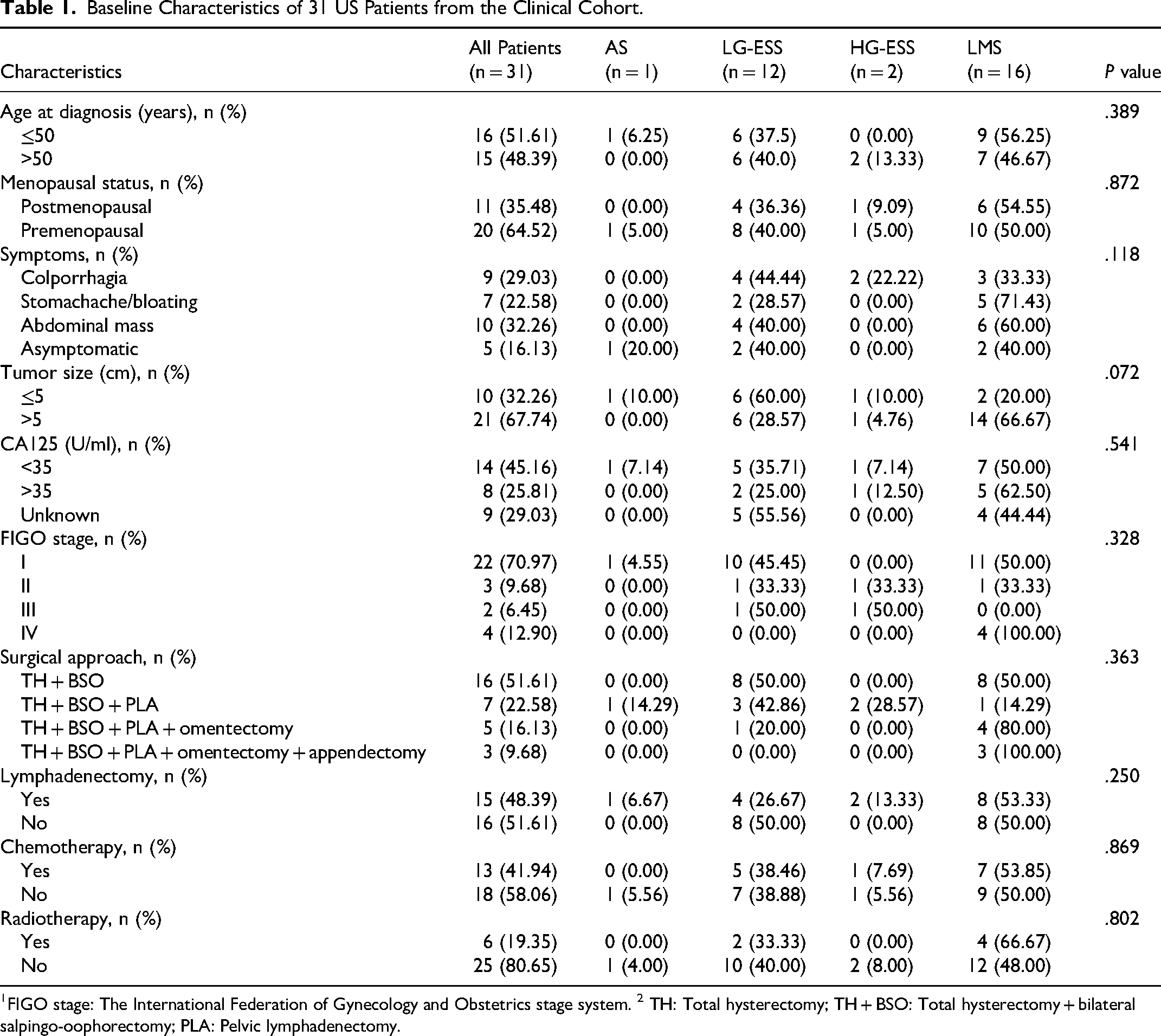
FIGO stage: The International Federation of Gynecology and Obstetrics stage system. 2 TH: Total hysterectomy; TH + BSO: Total hysterectomy + bilateral salpingo-oophorectomy; PLA: Pelvic lymphadenectomy.
IHC Evaluation of β-catenin
Immunohistochemistry was conducted to examine the expression of β-catenin in 31 full-face tissue sections obtained from US patients, including 24 paired samples of normal uterine smooth muscle (UNSM) and 5 paired samples of uterine leiomyoma (ULM). Positive expression of β-catenin was detected in the cell membrane and cytoplasm, appearing as light yellow, brownish yellow or dark brown particles. All specimens from US patients showed varying levels of positivity, ranging from moderate to strong.
As shown in Figure 1, among 5 paired samples, the expression level of β-catenin exhibited a significant increase in the US group compared to both the UNSM and ULM groups (P < .01, both) and in the ULM group compared to the UNSM group (P < .01, Figure 1b). Furthermore, among 24 paired samples, similar results were obtained both in the tumor tissues from US group compared to the normal tissues from UNSM group (P < .001, Figure 1c).

(a) IHC Indicated the Expression Levels of β-Catenin in the UNSM, ULM and US Groups. IHC: Immunohistochemistry; HE: Hematoxylin-Eosin Staining. (b) the box Plot Showed β-Catenin Expression in US, ULM, and UNSM among 5 Paired Samples. (c) the box Plot Showed β-Catenin Expression in and UNSM (Normal) and US (Tumor) among 24 Paired Samples. UNSM: Normal Uterine Smooth Muscle; ULM: Uterine Leiomyoma; US: Uterine Sarcoma.
In different pathological types, immunohistochemistry revealed a significant difference in β-catenin protein expression levels among AS, LMS, LG-ESS and HG-ESS (Figure 2a). The expression of β-catenin in AS showed moderate positivity and was significantly lower than that in LMS. Furthermore, the expression of β-catenin in HG-ESS was substantially up-regulated compared with LG-ESS, although there were only two patients. Obviously, the findings indicated that LMS and HG-ESS group exhibited higher levels of β-catenin expression when compared with other types. However, the box plot showed there was no statistically significant difference in β-catenin expression among AS, LMS, LG-ESS and HG-ESS (Figure 2b, P > .05).
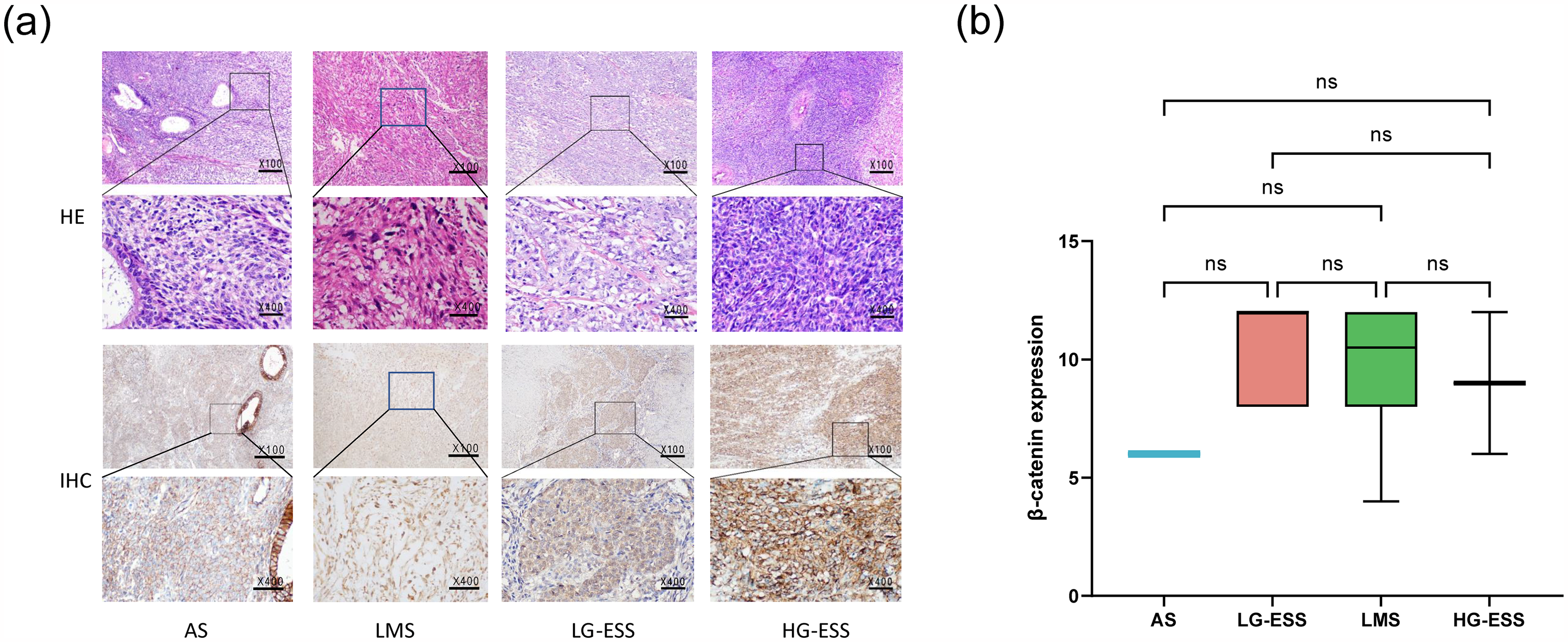
(a) IHC Indicated the Expression Levels of β-Catenin in Different Pathological Types of US. IHC: Immunohistochemistry; HE: Hematoxylin-eosin Staining. (b) The Box Plot Displayed β-Catenin Protein Expression in Different Pathological Subtypes of US. AS: Adenosarcoma; LMS: Leiomyosarcoma; LG-ESS: Low-grade Endometrial Stromal Sarcoma; HG-ESS: High-grade Endometrial Stromal Sarcoma.
Survival Analysis
Survival analysis indicated that high β-catenin expression level was not associated with poor OS (HR, 2.05; 95%CI: 0.43-9.74; P = .36; Figure 3a) and PFS (HR, 0.53; 95%CI: 0.15-1.82; P = .30; Figure 3b) in the clinical cohort. The median OS in the high- or low-β-catenin expression group were not reached (NR). The median PFS in the low-β-catenin expression group was not reached (NR), while that in the high expression group was 45.97 months. The similar results of OS were obtained in 50 UUS patients from Sweden microarray cohort (HR, 0.57; 95%CI: 0.27-1.22; P = .14; Figure 3c), while low-β-catenin expression group also showed a longer but not statistically significant median OS compared to the high expression group (56.17 months VS 9.60 months).

(a) K-M Analysis Suggested OS of β-Catenin High or Low Expression in the Clinical Cohort with 31 Patients. (b) K-M Analysis Suggested PFS of β-Catenin High or Low Expression in the Clinical Cohort with 31 Patients. (c) K-M Analysis Revealed the OS of β-Catenin High or Low Expression in 50 UUS Patients from Sweden Microarray Cohort. (β-Catenin was Coded by CTNNB1 Gene). K-M Analysis: Kaplan-Meier Survival Analysis; OS: Overall Survival; PFS: Progression-Free Survival. UUS: Undifferentiated Uterine Sarcoma.
Univariate and Multivariate cox Regression Analysis
Univariate and multivariate cox proportional hazard regression analyses were carried out with US patients from the Sweden microarray cohort. The univariate analysis indicated that copy number variation (CNV) group (HR = 2.04; 95% CI: 1.05-3.96; P = .034), hormone receptor expression (HR, 0.21; 95%CI: 0.09-0.48; P < .001), mitotic index group (HR, 2.33; 95% CI: 1.21-4.50; P = .012) and nuclear atypia (HR, 1.96; 95% CI: 1.01-3.80; P = .046) were significantly associated with OS in US patients. Multivariate analysis showed hormone receptor expression (HR, 0.24; 95% CI: 0.10-0.60; P = .002), and mitotic index group (HR, 2.70; 95% CI: 1.21-6.05; P = .016) still remained the same results (Supplementary Table S1). However, we noted no significant correlation between CTNNB1 expression and OS (HR, 1.75; 95% CI: 0.82-3.72; P = .148). Additional clinical indicators, including cell density, did not correlate with OS (P > .05). The findings were presented in Table 2.
Univariate cox Analysis of OS in US Patients from the Sweden Microarray Cohort.

OS: overall survival; US: uterine sarcoma. 2 CNV: copy number variation.
Furthermore, we conducted univariate and multivariate cox proportional hazard regression analyses of OS and PFS in US patients from the clinical cohort. Our analyses indicated that tumor type, lymphadenectomy, family history of malignancy and tumor recurrence remained significant predictors of OS, while only tumor type, stage and tumor recurrence were associated with PFS (P < .05, Tables 3 and 4). Univariate analyses demonstrated that only tumor recurrence (OS: HR, 12.46; 95% CI: 3.09-50.27; P < .001; PFS: HR, 16.89; 95% CI: 4.30-66.32; P < .001) and tumor type (OS: HR, 8.94; 95% CI: 1.12-71.02; P = .038; PFS: HR, 9.89; 95% CI: 1.26-77.92; P = .030) were significantly associated with OS and PFS in US patients. Nevertheless, the results indicated β-catenin expression was not associated with OS (HR, 1.53; 95% CI: 0.40-5.93; P = .538) or PFS (HR,1.72; 95% CI: 0.46-6.52; P = .423) in US patients, consistent with prior study from the Sweden microarray cohort. Moreover, other clinical variables did not influence OS and PFS (P > 0.05, Supplementary Table S2-S3).
Univariate cox Analysis of PFS in US Patients from the Clinical Cohort.
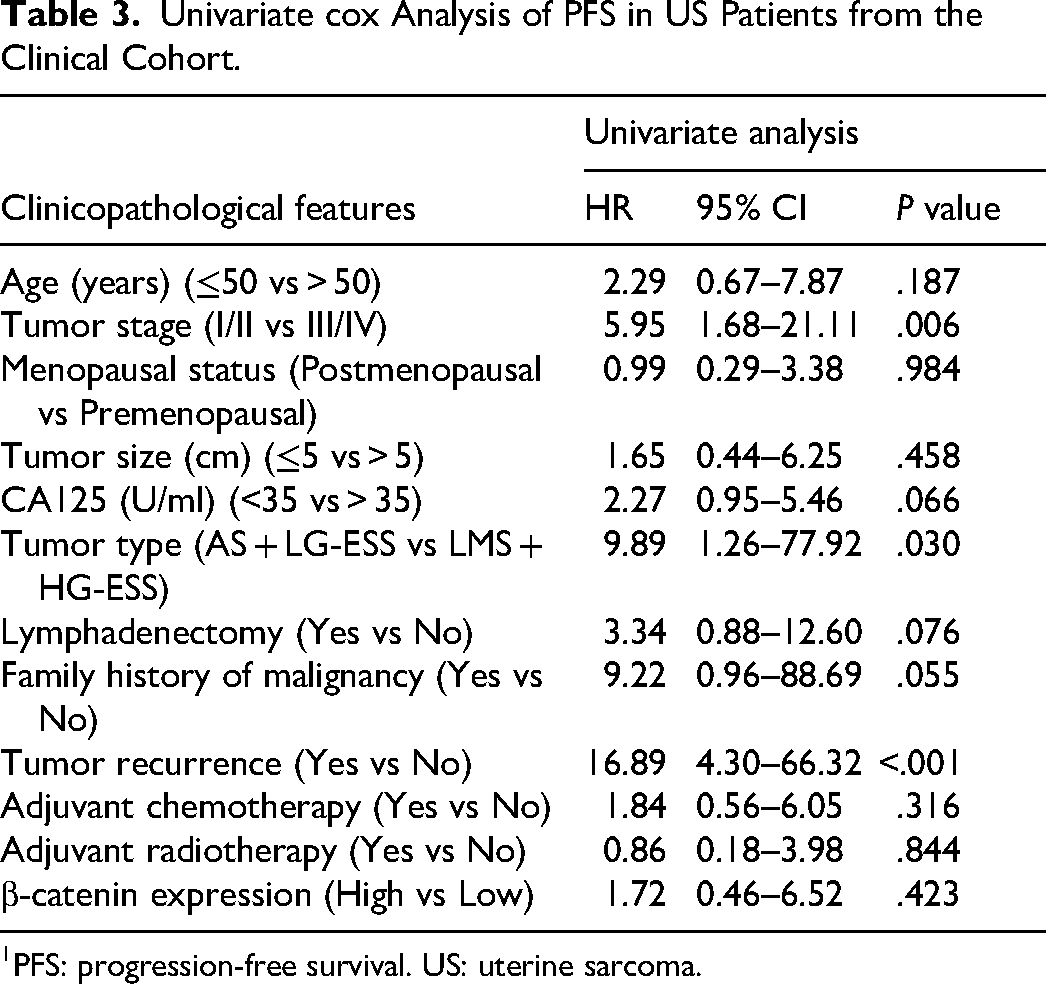
PFS: progression-free survival. US: uterine sarcoma.
Univariate cox Analysis of OS in US Patients from the Clinical Cohort.

OS: overall survival. US: uterine sarcoma.
GSEA to Get First Hints About the Specific Pathways Involved in β-catenin
Based on the potential prognostic significance of β-catenin expression level in uterine sarcoma, we investigated the underlying biological mechanisms. Differential expressed genes (DEGs) were analyzed between β-catenin high- and low-expression group in patients from Sweden microarray cohort, and gene set enrichment analysis (GSEA) was then performed to characterize the specific pathways that involved in the β-catenin expression. The positively enriched gene sets in the β-catenin-high expression group included AMP-activated protein kinase (AMPK) signaling pathway, endometrial cancer, mitogen-activated protein kinase (MAPK) signaling pathway, protein 53 (p53) signaling pathway, phosphatidylinositol-3-kinase/protein kinase B (PI3K-Akt) signaling pathway, rat sarcoma (Ras) signaling pathway, tumor necrosis factor (TNF) signaling pathway, Wnt signaling pathway, and transcriptional dysregulation in cancer. On the other hand, ascorbate and aldarate metabolism, cytokine-cytokine receptor interaction, pentose and glucuronate interconventions, retinol metabolism, steriod hormone biosynthesis, taurine and hypotaurine metabolism, were negatively enriched in the β-catenin-high-expression group (Figure 4, Supplementary Table S4).

(A) Result of GSEA Showed the Positively Enriched Gene Sets in the β-Catenin-High Expression Group. GSEA: Gene Set Enrichment Analysis. (B) Result of GSEA Displayed the Negatively Enriched Gene Sets in the β-Catenin-Low Expression Group. AMPK: AMP-Activated Protein Kinase; MAPK: Mitogen-Activated Protein Kinase; p53: Protein 53; PI3K-Akt: Phosphatidylinositol-3-kinase/Protein Kinase B; Ras: Rat Sarcoma; TNF: Tumor Necrosis Factor.
Discussion
Uterine sarcomas are rare and aggressive gynecologic malignancies, characterized by a relatively high recurrence rate. The prognosis of uterine sarcoma remains poor, with a mortality rate of up to 30% due to its highly aggressive nature. 26 There are few effective prognostic biomarkers or models for improving the clinical outcomes of US patients. Generally, patients with US are diagnosed after menopause. 3 Interestingly, a significant number of women in our study remained in the premenopausal phase. This finding hints at an evolving trend towards rejuvenation worth further monitoring.
Previous research proposed that specific fusion proteins in LG-ESS contribute to overexpression of Wnt ligands with subsequent activation of Wnt signaling pathway and formation of an active β-catenin/Lef1 transcriptional complex. 27 In this study, we confirmed that the expression level of β-catenin significantly upregulated in the US group compared to both the UNSM and ULM groups. Furthermore, immunohistochemistry exhibited a significant difference in β-catenin expression levels in four pathological subtypes. LMS and HG-ESS exhibited higher levels of β-catenin expression compared with AS or LG-ESS, but no statistically significant difference was displayed. Though survival analyses showed that β-catenin expression level was not associated with survival, low-β-catenin expression group showed a longer median OS compared to high expression group in the Sweden microarray dataset. Similar results were also observed for PFS in the clinical cohort. The late endpoint events of patients (reaching beyond follow-up cut-off), may affect the results. And this trend warrants further validation in prospective studies. As indicated by the above results, β-catenin was closely correlated with uterine sarcoma.
Based on our study, we discovered only tumor recurrence and type were significantly correlated with poor OS and PFS. Tumor type, lymphadenectomy, family history of malignancy and tumor recurrence remained significant predictors of OS, while only tumor type, stage and tumor recurrence had prognostic significance for PFS. We respectfully diverged from Wang et al's claim on the correlation between tumor size and survival (OS and PFS). 28 Other clinical variables, such as age, menopausal status, CA125, adjuvant chemotherapy, and adjuvant radiotherapy, did not influence survival. Despite lacking survival correlations, elevated circulating CA125 levels were noted in nearly a half of patients. Focusing on these patients may prompt potential benefits for diagnosis.
Currently, there was no standard therapeutic options for uterine sarcoma, with varying perspectives on surgical method. The cornerstone of the approach to US is hysterectomy and bilateral salpingo-oophorectomy (BSO). 29 It was reported that incomplete surgery was associated with poor prognosis 30 and hysterectomy was an independent risk factor for recurrence. 31 The prognosis of uterine sarcoma to different surgical approaches remains complex. Two studies reported that patients who underwent total hysterectomy without spreading cancer cells had better survival compared with other surgical procedures involving tumor injury.32,33 Further comparison in laparotomy and laparoscopy, laparoscopy was associated with more favorable overall survival than patients undergoing the laparotomy in women with unsuspected uterine malignancy diagnosed after hysterectomy for benign diseases. 34 Our research suggested that lymphadenectomy influenced OS in uterine sarcoma patients, aligning well with Machida's work. 35 On the other hands, differential diagnosis of uterine fibroid and sarcoma was crucial due to non-specific early symptoms similar with uterine myoma. Surgeon's experience, the suitability of the patient, and the size, location, and number of uterine fibroids are critical factors for the choice of surgical approach in uterine leiomyoma management. 36 Reasonable surgical approaches for the patients diagnosed with uterine sarcoma after myomectomy surgery for presumed benign uterine fibroid disease were significant. 29 Additionally, we found that adjuvant chemotherapy or radiotherapy was not associated with survival, consistent with prior research results.37,38 Whether radiotherapy or chemotherapy should be given after surgery is still debated. In summary, the treatment and management in uterine sarcoma remains controversial.
The rarity, heterogeneity, and complexity of US pose enormous challenges in identifying effective biomarkers and treatments, inhibiting progress in survival rates over the past few decades. In this study, we first explored β-catenin expression level and its potential role as an effective predictive biomarker for prognosis prediction of uterine sarcoma. We found that β-catenin was highly expressed in US, compared with UNSM and ULM, while survival analyses and cox regression analyses in the Sweden and the clinical cohorts showed that there was no significant correlation between β-catenin expression level and the prognosis of US patients. However, in the Sweden cohort, low-β-catenin expression group showed a longer median OS compared to high expression group in the Sweden cohort. Similarly, the median PFS showed the same trend in the clinical cohort. These findings suggested that β-catenin was highly expressed in uterine sarcoma and may be promising as a novel potential biomarker for the diagnosis and prognosis of uterine sarcoma. The novel biomarker would contribute to early diagnosis, risk stratification, and prognostic prediction in uterine sarcoma, enhancing clinical decision-making.
Wnt/β-catenin aberrant activation is linked with increased cancer occurrence, tumor progression, adverse prognosis development, and cancer-related mortality risk in human cancers. Altered β-catenin is thought to drive tumorigenesis in multiple cancers, 39 notably colorectal cancer 40 and endometrial carcinoma (EC). 41 Somatic mutation in coded gene hotspots or mutational inactivation of Adenomatous polyposis coli (APC) are two main mechanisms existed in endometrial carcinoma for increasing β-catenin levels. Tumors with β-catenin coded gene hotspot mutations exhibited higher Wnt signaling pathway activity, characterized by increased expression of the key protein β-catenin within the pathway. β-catenin, APC, Axin (axin inhibitor), CK-1α protein (casein kinase 1α protein) and GSK-3β protein (glycogen synthase kinase 3β protein) formed the destruction complex that captured β-catenin by phosphorylating CK1 and GSK3, thus activating the process of β-catenin degradation. The mutational inactivation of APC leaded to the accumulation of β-catenin.12,42 In our study, the positive enrichment of Wnt signaling pathway in the β-catenin high-expression group supported that β-catenin would be a satisfactory tool to predict prognosis in uterine sarcoma patients.
Several tumor progression related gene sets, including Wnt, TNF, AMPK, MAPK, p53, PI3K-Akt, Ras signaling pathway, endometrial cancer, and transcriptional dysregulation in cancer, were the potential predominant molecular pathways implicated in the uterine sarcoma development. For example, YWHAE-NUTM2 regulated cyclin D1 expression and cell proliferation by dysregulating RAF/MEK/MAPK and Hippo/YAP-TAZ signaling pathways in HG-ESS. 43 Intriguingly, autophagy inhibitor 3-Methyladenine (3-MA) potentiated the antitumor efficacy of apatinib in uterine sarcoma by stimulating PI3K/Akt/mTOR pathway. 44 On the other hand, the results suggested that several negatively enriched pathways in β-catenin low-expression group mainly involved in metabolism and hormone biosynthesis. Taurine not only inhibited cancer cell proliferation but also induced apoptosis in certain cancers by differential regulating proapoptotic and antiapoptotic proteins. 30 Taurine and hypotaurine metabolism pathway was continually disturbed during the progression of gastric carcinogenesis (GCG), potentially due to abnormal energy supply for tumor cell proliferation and growth. 31 In endometrial cancer (EC), Kaempferol affected multiple estrogen metabolism pathways by regulating HSD17B1 and HSD17B1-associated genes, which were involved in steroid hormone biosynthesis and regulation of hormone levels. 32 The above pathways were the potential crucial mechanisms to explore the role of β-catenin in uterine sarcoma genesis and progression.
Certainly, our study has limitations. Given a total of only 50 patients for Sweden microarray cohort and 31 patients for the clinical cohort, it is worth noting that the sample size might limit our ability to draw significant conclusions, and additional studies involving a large number of samples and multi-center are necessary. The heterogeneity in each individual would potentially affect the survival of patients, particularly tumor stage, histopathological types. Different surgical approaches and adjuvant treatments for each patient also significantly affected their disease progression and prognosis. In this study, the diverse treatment methods received by each patient may influence the assessment of OS and PFS. Although we conducted the univariate and multivariate cox regression analysis to control the bias, the small size of the cohort amplifies the bias. Besides, due to the sample limitations, with only one patient with adenosarcoma in the clinical cohort, the results for adenosarcoma in our study may not be fully comprehensive. Consequently, the results require validation with a larger sample to obtain a more precise conclusion. Additionally, the median OS and PFS in the low-expression group were not reached in the clinical cohort, suggesting that that the follow-up duration might be insufficient. The relatively long follow-up duration is likely to facilitate more accurate results of long-term survival analysis. Going forward, we plan to keep long-term follow-up for the patients and further explore the mechanism of β-catenin in the development of uterine sarcoma.
Conclusions
β-catenin was highly expressed in uterine sarcoma and may be promising as a novel potential biomarker for the diagnosis and prognosis of uterine sarcoma.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251345208 - Supplemental material for β-catenin is a Potential Prognostic Biomarker in Uterine Sarcoma
Supplemental material, sj-docx-1-tct-10.1177_15330338251345208 for β-catenin is a Potential Prognostic Biomarker in Uterine Sarcoma by Ying Cai, MS, Yunjia Wang, MS, Ling Yang, MS, Yue Huang, MS, Min-Jun Chen, MS, Chi Zhang, MS, Su-Han Jin, MD, PhD, Benjamin Frey, PhD, Udo S. Gaipl, PhD, Hu Ma, MD, PhD, and Jian-Guo Zhou, MD, PhD in Technology in Cancer Research & Treatment
Footnotes
Author Contributions
Conceptualization, Jian-Guo Zhou; Data curation, Yunjia Wang, Ling Yang and Yue Huang; Formal analysis, Min-Jun Chen and Chi Zhang; Funding acquisition, Hu Ma and Jian-Guo Zhou; Resources and methodology, project administration, Supervision, Jian-Guo Zhou; Visualization, Ying Cai; Writing – original draft, Ying Cai; Writing – review & editing, Su-Han Jin, Benjamin Frey, Udo Gaipl, Hu Ma and Jian-Guo Zhou. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Noncommunicable Chronic Diseases-National Science and Technology Major Project (Grant No. 2023ZD0502105), the National Natural Science Foundation of China (Grant No. 82060475), Chunhui program of the MOE (Ministry of Education in China) (Grant No. HZKY20220231), MOE Liberal arts and Social Sciences Foundation (Grant No. 24YJCZH462), Youth Science and Technology Elite Talent Project of Guizhou Provincial Department of Education (Grant No.QJJ-2024-333), Excellent Young Talent Cultivation Project of Zunyi City (Zunshi Kehe HZ (2023) 142), Future Science and Technology Elite Talent Cultivation Project of Zunyi Medical University (ZYSE 2023-02), the Key Program of the Education Sciences Planning of Guizhou Province (Grant No.7), Collaborative Innovation Center of Chinese Ministry of Education (Grant No. 2020-39).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data for uterine sarcoma in GSE119043 are available at the Gene Expression Omnibus (GEO) (![]() ) portal. Analysis tools are listed in Methods. The data for Suining cohort presented in this article are not readily available because the data are part of an ongoing study, due to necessary secrecy. Requests to access the datasets should be directed to the corresponding author.
) portal. Analysis tools are listed in Methods. The data for Suining cohort presented in this article are not readily available because the data are part of an ongoing study, due to necessary secrecy. Requests to access the datasets should be directed to the corresponding author.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Review Board of Suining Central Hospital (No. LLSLH20220051; August, 31th, 2022) and the Ethics Committee of Zunyi Medical University (No. 2020-1-013; March, 30th, 2020). The written informed consent was obtained from each participant, allowing the use of their clinical data, archived paraffin-embedded specimens, imaging, and pathological images for research and publication purposes. This research report conforms to REMARK guidelines. All patient details have been de-identified.
Informed Consent Statement
Written informed consent was obtained from all subjects involved in the study.
Supplemental Materials
Supplemental material for this article is available online. Table S1: Multivariate cox analysis of OS in US patients from the Sweden microarray cohort; Table S2: Multivariate cox analysis of PFS in US patients from the clinical cohort; Table S3: Multivariate cox analysis of OS in US patients from the clinical cohort. ![]() : Results of GSEA in US patients from the Sweden microarray cohort.
: Results of GSEA in US patients from the Sweden microarray cohort.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
