Abstract
Introduction
Tissue-equivalent boluses are used to increase the skin surface dose in patients with breast cancer undergoing postmastectomy radiotherapy. Boluses made from various materials have been developed and applied in clinical practice. However, there is currently no international standardization for their use. This study aimed to analyze the advantages and disadvantages of using compensatory membranes (boluses), identify the optimal patient population for their postoperative application, and determine the most suitable types of compensatory membranes for use in postoperative radiotherapy for breast cancer.
Methods
This study includes a systematic literature review of sources such as websites, books, and articles from January 2013 to December 2023. We analyzed and compared studies on the use of compensatory membranes in postoperative radiotherapy for breast cancer, evaluating differences among various bolus materials as well as the impact of using no bolus. Outcomes such as patient responses and prognosis were also assessed. After excluding abstracts, reviews, and other non-research articles, a total of 32 studies were included in the analysis.
Results
3D-printed boluses and brass mesh boluses show promise as alternatives to traditional bolus materials, with potential for broader clinical application. The use of a bolus can increase the incidence of acute radiation-induced toxicities, without significant improvements in long-term patient prognosis. For patients receiving chest wall radiotherapy after breast cancer surgery, bolus use may be considered selectively, especially in those with high-risk factors.
Conclusion
Currently, there is no unified standard for bolus use in breast cancer patients undergoing chest wall radiotherapy following modified radical mastectomy. This review provides a critical evaluation of existing literature, summarizing the available bolus materials and their influence on radiotherapy outcomes in postoperative breast cancer treatment.
Introduction
The most frequent malignancy in women and the second leading cause of cancer-related mortality is breast cancer. 1 In 2020, approximately 2.3 million women were diagnosed with breast cancer worldwide, and 685 000 died from this disease. 2 Post-mastectomy radiation therapy (PMRT) is used to reduce the local recurrence rate (LRR) and prolong overall survival. 3 Megavoltage (MV) photon beams are commonly used in clinical radiotherapy; however, owing to the lack of electron equilibrium, the surface dosage is lowered. This phenomenon can be called the “dose-build-up effect” or “skin-sparing effect.” 4
Tissue-equivalent boluses can cause the 95%-100% isodose line to shift in the direction of the skin and subcutaneous tissue and have been widely used in postmastectomy radiation therapy.5,6 Boluses made from various materials, such as Superflab™, Vaseline®, brass mesh, and wax have been developed and applied. The use of bolus remains a subject of considerable controversy. By now, there is no consensus on the optimal thickness and timing of using boluses. Moreover, the LRR has continuously decreased as comprehensive therapies have been developed, calling into question the need for boluses. Some researchers have argued that bolus use increases acute and late skin toxicity, even raise the treatment interruption time.7–10 These aforementioned risks may compromise the efficacy of the bolus in reducing the LRR.8,9,11–13 Traditional bolus, due to its uniform one-piece design, often exhibits poor conformity to the chest wall. The resulting incomplete contact can lead to air gaps, which can reduce the surface radiation dose and lead to differences between the actual dose and planning doses. Three-dimensional-printed boluses have been developed to overcome these disadvantages, as they are made to fit each individual patient. However, as this technology is still in its infancy, 3D-printed boluses are associated with their own set of disadvantages, including a long production time, high costs, and the hardness of the material. It also remains unclear whether all patients with breast cancer undergoing postmastectomy radiation therapy require a bolus. Some researchers have noted that boluses can lead to increased skin toxicity and treatment interruptions, resulting in higher rates of local recurrence and breast cancer mortality.7–15 Therefore, using boluses in postmastectomy radiation therapy has been widely questioned, with some researchers proposing their use only in patients at a high risk of local recurrence. 5 The high-risk factors include T3-4 cancer, positive margins, or extensive lymph node metastasis. This review aims to summarize the main types of boluses, analyze the controversy surrounding their use by presenting the evidence supporting the different viewpoints, and discuss future directions.
Method
The methodology of this review was pre-specified according to the recommendations outlined in the latest 2020 update of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 16 A systematic search for articles on breast cancer and bolus materials was conducted in the PubMed and Web of Science databases in December 2023. The publication date is limited from January 2013 to December 2023. No restrictions are placed on countries/regions. The search terms used the following keyword combination: [((breast cancer) OR (breast carcinoma)) AND (bolus) AND ((radical surgery) OR (postmastectomy) OR (radiotherapy) OR (mastectomy))]. The inclusion criteria are as follows: a. Peer-reviewed original articles; b. Studies involving patients diagnosed with breast cancer undergoing postmastectomy radiotherapy; c. Model studies measuring radiation dose; d. Inclusion of all types of bolus materials; e. Inclusion of all types of irradiation beams.
In the initial selection of 395 articles, 363 were excluded after reviewing the titles and abstracts. Articles were excluded if they were abstracts, meeting reports, review articles, case reports, editorials, short communications, meta-analyses, lacked sufficient information, or were non-English language studies. This left 32 articles with adequate details for inclusion in the final review Figure 1.

Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Flowchart.
Result
Types of Bolus Materials
In the 32 articles, conventional bolus like Superflab™ and Vaseline® is the most commonly referenced bolus type, with 13 articles8,11–15,17–23; followed by 3D-Printed Bolus, which appears in 8 articles24–30; and the brass mesh is less common with only 5 articles.31–35 Other types are mentioned in 6 articles, including thermoplastic elastomer (TPE) bolus, 36 custom-made step bolus, 37 thermoplastic bolus, 38 Clearfit bolus, 35 Play-dough material bolus, 39 shape rubber-containing tungsten (STR) bolus. 40 These numbers reflect the varying prevalence of these bolus types in radiotherapy literature. The Table 1 shows the different types of boluses used in radiotherapy based on the number of articles cited.
Types of Boluses.

Regarding bolus thickness, the most commonly used bolus thickness was 5 mm, which appeared in 16 studies.8,11–14,17,18,20,28,30,34–36,38,40 Additionally, 5 studies used a 3 mm bolus thickness,14,17,21,32,35 while 4 studies used a 10 mm bolus.20,21,26,34 A 2 mm bolus thickness was used in 4 studies,19,33–35 and 3 studies used a 1 mm bolus.40–42 One study employed a 1.5 mm bolus thickness, 37 and 9 studies did not specify the thickness of the bolus.22–25,27,29,31,39,43 These findings highlight the variation in bolus thickness used in PMRT planning, with 5 mm being the most frequently chosen thickness.
PMRT Planning
The use of bolus depends on the technique used for radiotherapy. Different radiotherapy techniques require consideration of different factors. For example, the considerations for using a bolus in Intensity-Modulated Radiation Therapy (IMRT) or Volumetric Modulated Arc Therapy (VMAT) are different. Specifically, the number of irradiation fields and the radiation quality will influence the selection and use of the bolus.
In this study, a total of 33 articles were reviewed regarding PMRT planning. Among them, 24 studies used photon beams,8,12,13,17,19–22,25,26,28–36,39–42 accounting for 72.7% of the total; 4 studies used electron beams,24,27,37,38 representing 12.1%; and 5 studies did not specify the type of radiation beam used,11,14,18,23,43 making up 15.2%. These results indicate that photon beams are the most commonly used radiation type in PMRT planning, while the use of electron beams is relatively less common, with a notable proportion of studies not reporting the specific radiation beam type.
Thirteen studies used only 6MV photon beams, making up a significant portion of the total12,13,20,22,29–31,33,34,39–42; 1 study used only a 10MV photon beam 17 ; 2 studies employed a combination of 6MV and 10MV photon beams.26,28 Additionally, 1 study used either a 9 or 12 MeV electron beam, 37 while another study combined 6MV, 10MV, and 15MV tangential photon beams. 19 One study used both 12 MeV and 15 MeV electron beams, 38 and 1 study used a combination of 6MV and 4MV photon beams. 35 Furthermore, 2 studies combined 6MV and 15MV photon beams,21,35 and 1 study used a range of 6-18MV photon beams. 8 Finally, 10 studies did not specify the energy levels or types of radiation used.11,14,18,23–25,27,32,36,43 This variation in radiation beam energies reflects the diversity in treatment planning strategies employed in PMRT. Table 2 shows the detailed types of bolus.
Different Kinds of Bolus.

Dosimetric Effects of Bolus
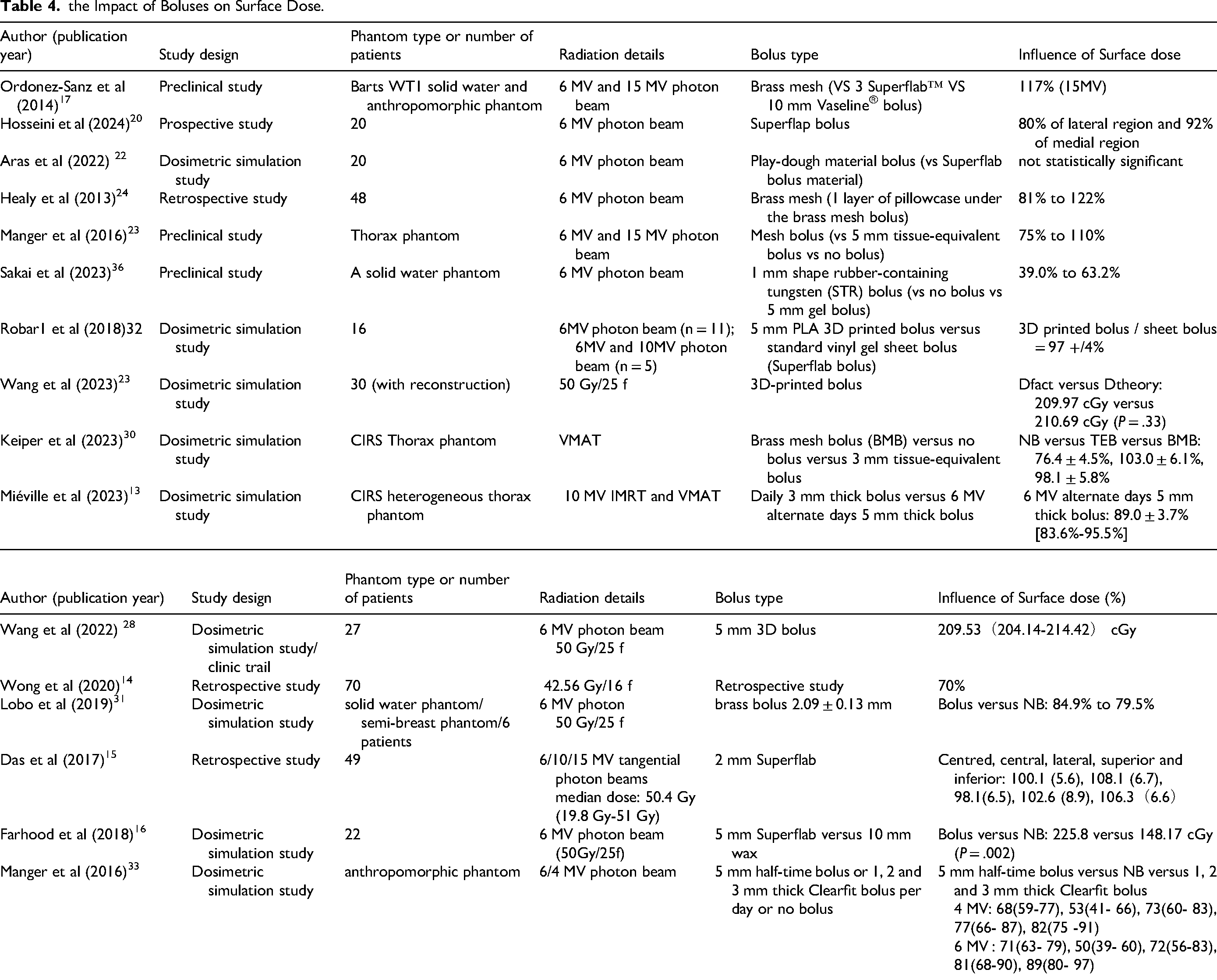
Regarding the dosimetric effects of bolus, 16 articles mentioned surface dose/skin dose.17–22,25,28,30–33,35,39,40 We can find the informaions in Table 3. Among these articles, 1 study reported no significant statistical difference in surface dose when using bolus. 39 8 studies indicated that the surface dose was higher when the bolus was used compared to no bolus.18,20,31–33,35,40,44 Additionally, 7 studies found that the use of a bolus provided adequate surface dose/skin dose.17,19,21,22,25,28,30 These findings suggest that, in general, the application of a bolus increases the surface dose and ensures sufficient dose delivery to the skin area.
the Impact of Boluses on Organs at Risk (OAR).

CB: conventional bolus.
PLA: polylactic acid.
WTESH: water tissue equivalent silicone hydrogel.
A total of 7 articles reported on target volume homogeneity index (HI) and conformity index (CI) after the use of bolus.24–27,29,30,36 Among these, 2 studies found that the homogeneity index (HI) for the target volume was better in the 3D-printed bolus group compared to the traditional bolus group,27,29 with 1 of these studies showing statistical significance (P < .001). 29 1 study reported that the HI with the 3D-printed bolus was significantly better than with no bolus (P < .001). 24 Additionally, 3 studies compared the CI between the 3D-printed and traditional bolus groups,26,27,29 all showing that the CI was superior in the 3D-printed bolus group, with 2 studies demonstrating statistical significance (P < .0001).26,29 And 1 research reported that 3D-printed bolus significantly improved the CI than no bolus. 24 These results suggest that the use of 3D-printed bolus improves both target homogeneity and conformity compared to traditional bolus and no bolus.
Table 4 shows the dosimetric effects of bolus on organs at risk (OAR), 8 articles discussed the impact of bolus on OAR dose.24–27,29,30,36,41 Among these, 3 studies reported that compared to traditional bolus,26,27,29 3D-printed bolus could reduce the dose to OARs, with 2 of these studies showing statistically significant differences between the bolus and no bolus groups (P < .001).26,29 2 studies also indicated that bolus use reduced the dose to OARs when compared to no bolus.24,41 Additionally, 3 studies found that the use of a bolus ensured that the dose to OARs met the required dose limits.25,30,36 These findings suggest that 3D-printed bolus can be more effective than traditional bolus in reducing dose to critical organs and ensuring dose constraints are met.
the Impact of Boluses on Surface Dose.
Impact of Bolus on Radiation Toxicity
As Table 5 shows, a total of 13 articles reported on acute radiation toxicity during patient treatment.8,11–14,19,23,30,36,37,42,43 One study showed that the use of a bolus significantly increased the incidence of grade 2/3 skin reactions compared to no bolus (P = .0039). 8 Three studies indicated that bolus use led to an increase in grade 1/2 skin reactions, with one of these studies showing statistical significance (P = .01).11–13 Additionally, 1 studies reported that the use of a bolus increased the occurrence of grade 3-4 skin reactions. 12 Furthermore, Boulle et al found that patients treated with postmastectomy highly conformal electron radiation therapy with bolus had a lower incidence of grade 1-2 acute skin reactions compared to patients treated with highly conformal photon radiation therapy without bolus, but significantly higher rates of grade 3-4 acute skin reactions (P < .001). 23 These findings suggest that while bolus use can exacerbate acute skin reactions, certain types of bolus, may help mitigate these effects, particularly for moderate skin reactions.
the Effects of Bolus use in PMRT.

SR: patients without skin involvement.
HR: patients with skin involvement.
PMERT (with bolus): patients treated with postmastectomy highly conformal electron radiation therapy.
PMPhRT (without bolus): patients treated by highly conformal photon radiation therapy.
Discussion
Routine Application of Bolus in PMRT
Numerous studies have demonstrated that the use of a bolus can effectively increase the surface or skin dose in the irradiated area; among the 32 articles included in this review, 15 studies confirmed this effect. In addition, the bolus also contributes to improved dose homogeneity and conformity within the target volume. However, there is a huge variability in terms of protocols for bolus use, including differences in bolus thickness, material, and application schedule (daily or on alternating days). Although the American College of Radiology recommends that photons with an energy of 6 MV or higher be routinely used in PMRT, bolus thickness can range from 2 to 10 mm. These changes of bolus thickness may hinder the establishment of consistent treatment quality and increase the risk of treatment errors. He et al 45 founded that 5 mm boluses were associated with fewer inter-fractional setup errors than 10 mm boluses, with superior organ-at-risk (OAR) protection capability, as well as acceptable target coverage and dose homogeneity. A study by Das et al 19 included 49 patients who received radiotherapy after postmastectomy with a daily 2 mm boluses, reporting acceptable dose build-up and skin toxicity.
A study using the Delphi method recommended, from clinical experience, the use of a 3-5 mm bolus to shape around and maintain close contact with the skin, avoiding air gaps. As the thickness of the bolus increased to ≥10 mm, skin toxicity also increased.5,46 In the photon beam radiotherapy, the dose typically increases sharply in the first 10-15 mm below the skin and then decreases in a typical tangential treatment plan. 5 Therefore, 10 mm boluses may lead to high skin toxicity and 2 mm boluses may not meet dose requirements; 5 mm boluses may be the most suitable choice. Especially, In radiotherapy, the distribution of skin dose is significantly influenced by the beam angle and the size of the irradiation field. Especially when using bolus, these factors need to be given special consideration. The role of the bolus is to compensate for the dose deficiency caused by deep tumors by increasing the dose at the skin surface. However, when the treatment beam angle is tilted, the maximum dose of the beam shifts from the tumor target to the skin surface, resulting in an increased dose to the superficial area. In this case, the traditional maximum dose prediction based on 5-10 mm may no longer be applicable. Therefore, when designing the treatment plan, especially in the case of large-angle tilts, the placement of the bolus should be adjusted according to the specific beam angles to ensure precise control of the dose distribution in both the skin and deeper tissues during the treatment process, maximizing the treatment efficacy and minimizing unnecessary side effects.
Controversy also surrounds the issue of bolus schedule. Some researchers recommend using a bolus on alternating days to reduce radiation exposure 5 ; however, others suggest using a bolus daily to ensure accurate calculations and at least 95% coverage. The European Society for Radiotherapy & Oncology (ESTRO) states that although using boluses everyday was found to be related to an increase in skin toxicity, the 5 mm bolus should only be used in patients with high risks of skin involvement, such as T4b, c, d, or any ypT4, and the full dose should only cover the high-risk regions. 5 In general, bolus use on alternating days can effectively protect the skin while minimizing skin toxicity; however, it may reduce target dose accuracy. Another disadvantage of alternating days bolus use is that it required two treatment plans. One of the plans with a bolus and one without a bolus. In comparison, a daily bolus ensured dose accuracy; however, it increased skin toxicity and was recommended only using for patients who have high risks of skin involvement.
Main Bolus Types
Clinically, boluses should be nearly tissue-equivalent, elastic enough to conform to the chest wall, non-toxic, affordable, and durable. But in addition to this some organizations also use brass mesh bolus as an alternative to tissue equivalent bolus. Various materials have been tested for this application. In clinical radiotherapy, tissue equivalent boluses such as Superflab™ and Vaseline® are widely used. Materials with high density or high atomic number, like brass mesh, have been utilized in tangent photon radiotherapy. 47 Thermoplastic sheets and materials, such as wax, paraffin, and cellophane-wrapped wet gauze, have also been used.48–50 Recently, following advances in 3D-printed technology, the use of 3D-printed boluses has become widespread. Research indicated that using 3D printing improves dose uniformity, conforms to the chest wall, and spares healthy organs. 27
Commercial Bolus
Traditional bolus, such as Superflab™ and Vaseline® bolus, is the main component of commercial bolus and is widely used in PMRT. According to statistics, among the 32 studies included in this review, 13 employed traditional bolus materials, accounting for 40.6%. The thickness of this bolus is typically 10 or 5 mm, according to the build-up region of the 6 MV photon beam, which ranges from 1 to 1.6 cm. 22 The density of Superflab is 1.02 g/cm3, giving it a similar tissue equivalence to water. Research conducted by Solanki et al 51 on 50 breast cancer patients after postmastectomy demonstrated that the use of 1 cm Superflab can decrease radiation exposure to normal tissues and enhance radiation delivery to the skin.
Although conventional bolus material is commonly used in clinical practice, there are still some unresolved concerns. Conventional boluses have a consistent flaky structure, which may not conform well to the chest wall, especially in patients who have had a postmastectomy. 39 This results in air gaps between the bolus and skin, causing inaccuracies in the radiation dose at the planned target volume (PTV). 35
3D printing technology has been extensively utilized in producing personalized intricate structures, such as a variety of medical gadgets, in recent years.52,53 3D-printed polylactic acid (PLA) bolus material can fit the chest wall of patients.28,54–57 According to the above statistics, articles related to 3D-printed bolus account for 25% of the total, making it the second most commonly used type of bolus. This indicates that 3D-printed bolus now has being used in clinical applications on a first-class scale and are expected to replace traditional conventional bolus, as commercial bolus.
Studies have been performed using homogenous phantoms such as blue water phantoms, and anthropomorphic phantoms such as RANDO phantoms and other female phantoms with breast attachments. Kim et al 56 utilized both types of phantoms to compare the effectiveness of 3D-printed bolus material against conventional bolus material through the measurement of dosimetric parameters. Using the water phantom, the 3D-printed bolus achieved equivalent dosage coverage compared to the conventional bolus. Using RANDO phantoms, the 3D-printed bolus significantly decreased the air gap between the bolus and the surface as compared to conventional bolus material, thus improving the efficiency and accuracy of radiotherapy. Park et al 58 conducted a study using a female mannequin with breast attachments of varying volumes: 200, 300, 400, 500, and 650 cc. The conventional bolus exhibited notable discrepancies in skin surface doses compared to the estimated values for volumes of 200 and 300 cc. The disparities were −5.1% and −3.2% for 3 mm and −6.3% and −4.2% for 5 mm, respectively. The 3D-printed bolus exhibited minimal variations in volume, with variances of −0.7% and −0.6% for 3 mm, and −1.1% and −1.1% for 5 mm, respectively.
The study demonstrated that the 3D-printed bolus was a viable substitute for the conventional bolus in phantom models.
Robar et al 28 conducted a study comparing a conventional sheet bolus with a 3D-printed bolus in breast cancer patients receiving post-mastectomy radiation therapy, using air gaps and surface doses as outcome measures. The 3D-printed bolus had significantly better fitting accuracy to the chest wall than the standard sheet bolus, reducing the maximum air gap size from 0.5 ± 0.3 mm to 0.3 ± 0.3 mm. Additionally, the implementation of a 3D-printed bolus decreased the setup time from 104 to 76 s. This study shows that using a 3D-printed bolus resulted in better fit and shorter implantation time for patients receiving post-mastectomy radiation therapy compared to the traditional sheet bolus. Zhang et al 29 conducted a study comparing the dosimetry, application, and acute toxicity of 3D-printed and conventional boluses for post-mastectomy radiation therapy with volumetric modulated arc therapy (VMAT). The 3D-printed bolus reduced the average cardiac dosage to the left breast cancer and the average lung dose to the afflicted side by 0.8 Gy, compared to the traditional bolus (P = .035). The value for the conformity index of the PTV was also improved from 0.80 ± 0.03 to 0.03 ± 0.02 (P < .001). We can conclude that a 3D-printed bolus could reduce cardiopulmonary exposure and improve conformity in PMRT.
However, the time-intensive fabrication workflow of 3D-printed boluses (typically >4 h from CT-to-delivery) remains a critical barrier to clinical adoption, particularly for adaptive radiotherapy requiring rapid plan modification. 59 Furthermore, the hardness of PLA, which is not fully conformable to slight deformations or anatomical changes despite personalization. Therefore, further research is needed to develop flexible materials. The ideal material should have customizable 3D-printing and radiotherapy-assisted properties, as well as being tissue equivalent, biocompatible, and antimicrobial. 54
Brass Mesh Bolus
Brass mesh is considered an acceptable replacement for traditional tissue-equivalent bolus materials; however, it is relatively rarely used in PMRT. In this review, articles focusing on brass mesh bolus accounted for 15.6% of the total, ranking third in prevalence. Some studies have shown that brass mesh bolus can be effective in increasing the dose of skin. A retrospective study by Healy et al 31 involving 16 female patients demonstrated that a brass mesh bolus used in conjunction with a 6 MV photon beam can maintain a surface dose between 81% and 122% of the prescribed dose, as measured using a thermoluminescent dosimeter. The increase in surface dose is mainly because the mesh can produce secondary charged particles. Manger et al 35 conducted an experiment using a thorax phantom to assess the superficial dose with several types of boluses: no bolus, a brass mesh bolus, and a tissue-equivalent bolus. The results showed surface doses ranging from 40% to 72%, 75% to 110%, and 85% to 109% of the prescription dose, respectively. The brass mesh bolus offers better conformance to the chest wall than the tissue-equivalent bolus; thus, the surface dose is more accurate. An additional benefit of brass mesh is that it has a minimal effect on the depth–dose curves. Because of this feature, the bolus can be removed at any point without creating a new treatment strategy and therefore significantly reducing the workload of doctors. 21 However, the activation products of photoneutrons should be considered. Neutrons in the brass mesh may result in a neutron dose that is potentially harmful to staff and patients. 35
Other Boluses
In addition to the commonly used boluses described above, other bolus materials have also been assessed. Of the 32 studies included in this review, 6 reported on the use of different types of bolus materials, encompassing a total of six distinct types. Play dough material (PDM) is a solid-liquid mixture consisting of water, a starch-based binder, salt, oil, preservatives, hardeners, humectants, flavors, and food colorants with a density of approximately 1.0 g/cm3. A previous study reported similar radiation doses when using PDM and conventional bolus materials. 60 However, a separate study found that PDM was not suitable for radiation therapy. 39
Tungsten-based materials such tungsten-infused rubber, tungsten-functional paper, and real-time variable-shape rubber with tungsten (STR) are utilized as boluses in photon irradiation. STR is non-toxic and can be easily sliced, allowing it to be applied directly on the skin surface to conform to lesions with uneven surfaces and distorted shapes. The STR bolus is advantageous because it enhances the surface dosage by utilizing the build-up effect and backscatter as the beam penetrates and exits the bolus, hence proving to be an efficient method for photon irradiation of superficial malignancies. 40
Another material, thermoplastic elastomer (TPE), has also been used as a bolus in radiotherapy. TPEs are soft and have self-adhesive properties that mold to the patient more accurately than conventional boluses. A study of 20 patients by Gong et al 36 shown that TPEs resulted in low skin toxicity and pain, did not add to patient suffering or clinical workload, and were considered a better option than 3D-printed bolus materials for patients at high risks of PMRT skin recurrence.
Initially, various materials were tested as boluses. In 2004, a study used perforated Aquaplast and a nearly tissue-equivalent wax. This combination resulted in a bolus with thermoplastic properties. Upon heating, the wax becomes pliable and can be placed on the Aquaplast and the chest wall of the patient to accommodate its irregular surface. Upon cooling, the wax hardens and retains its molded shape. 61 However, this bolus material is not widely used in clinical practice because of its complexity, long fabrication time, and uneven thickness.
Controversy Surrounding Bolus Use
It is widely agreed that a tissue-equivalent bolus should be utilized in post-mastectomy radiation therapy when there is skin involvement to guarantee a sufficient skin dosage.27,30,36,40,54,55,57,61–67 There is disagreement on using a bolus during post-mastectomy radiation therapy in individuals without skin involvement due to varying definitions of target areas, leading to regional differences in guidelines. The RTOG includes skin in its CTV, but the ESTRO protocol does not include the ventral 5 mm of skin and subcutaneous tissue in the chest wall Clinical Target Volume (CTV). 14 The ASCO PMRT guidelines indicate inadequate evidence to support bolus use, the current NCCN Breast Guidelines state that “special consideration should be given to the use of bolus materials to ensure adequate skin dose.”
Among the 32 studies included in this review, five reported that the use of bolus was associated with an increased incidence of radiation-induced skin reactions. This suggests that using a bolus may not be completely advantageous for breast cancer patients, especially those with a low risk of recurrence. In recent years, retrospective studies have shown that a bolus does not significantly decrease the LRR and even leads to an increased LRR due to treatment interruptions caused by the skin toxicity of the boluses.5,7–15
Abel et al 8 did a retrospective study between 106 patients who were receiving chest wall radiation and concluded that administering a bolus during therapy led to a comparable rate of chest wall recurrence compared to treatment without a bolus, but resulted in more intense acute skin reactions. The average length of treatment interruption was likewise greater in the bolus group. Moreover, treatment interruptions were more likely to lead to local control failure (17.4% vs 3.6%, P = .0322).
Another retrospective study by Nichol et al 14 involved 1887 women with breast cancer that did not involve the skin (pT1-4a, any-N, M0) receiving photon 3D conformal radiation therapy from 2007 to 2011, of whom 1569 received a bolus and 318 did not. A multivariable analysis was conducted on cancer stage, subtype, lymph-vascular space invasion, grade, and margin status to assess their impact on locoregional recurrence (LRR) and breast cancer mortality, revealing no significant association between bolus use and the LRR or breast cancer mortality.
Turner et al 13 conducted a retrospective cohort research that involved patients who got chest wall radiation between 2005 and 2010. Out of these patients, 101 were given a bolus whereas 213 were not. Acute skin poisoning occurred more frequently in the bolus group compared to the no bolus group, with a statistically significant difference (P = .002).
Based on the above studies, there are some suggestions that patients without skin invasion after mastectomy should be exempt from the use of a bolus during radiotherapy, thereby reducing skin toxicity and the risk of radiotherapy interruption.
Significant differences in LRR between no bolus and bolus groups may be due to differences in patient selection. According to certain research, people who have a high chance of recurrence are more likely to be given boluses. Research by Yap et al 11 involved 314 patients with invasive breast cancer treated between 2004 and 2009 with adjuvant chest wall radiation. Only after univariate analysis did the four-year LRR differ statistically significantly between the bolus (14%) and non-bolus (3.5%) groups (hazard ratio 3.09; 1.15e8.33; P < .025). The substantial difference vanished when margin status was considered (hazard ratio: 2.45; 95% confidence interval: 0.80e7.50; P = .12) Therefore, the use of a bolus does not raise the LRR in patients with breast cancer, as this retrospective investigation was unable to show.
Prospect of Development
In order to reduce the dosage build-up effect on the target area and increase the skin dose to decrease local recurrence, boluses are now frequently employed in clinical settings. However, there are no uniform standards for the use of boluses, which are mostly applied according to varying guidelines or clinicians’ personal experiences. Increasing evidence indicates that boluses do not have a significant effect on the LRR and have even been shown to increase skin toxicity and radiotherapy interruption time, increasing the LRR. This has led some to question whether all breast cancer patients receiving PMRT need a bolus. Nichol et al 14 reported that boluses can be reserved for individuals who have a high risk of recurrence and excluded from treatment for those who have a low risk.
However, some multivariable analyses have suggested that the high LRR in the bolus group may be related to the fact that patients in the bolus group had more risk factors for recurrence, including margin status, skin involvement, or inflammatory tumor stage. 11 According to some research, patients who have had mastectomy surgery still have a significant chance of recurrence in their skin and superficial tissues. According to a systematic review by Kaidar-Person et al, 68 the mean LRR of 6901 patients who had mastectomy was 3.5%, and 81.8% of recurrences happened in the skin and subcutaneous tissue and 16% in the pectoral muscle. The omission of boluses inevitably reduces the irradiation dose to areas at a high risk of recurrence, including the skin and superficial tissues. It is not consistent with the pattern of local recurrence following mastectomy to reduce the irradiation dose to high-recurrence areas while maintaining an adequate irradiation dose to low-recurrence areas. Based on the above evidence, while using boluses to maintain a high dose in places at a high risk of local recurrence, we can attempt to lower the dose in all target locations, such as the skin and superficial tissues. A different strategy entails improving the PMRT eligibility criteria so that therapy is not required for patients who have little chance of benefiting. A study performed using the Delphi method and presenting international consensus recommendations initiated by the ESTRO stated that only for the treatment of local breast cancer recurrences, ductal carcinoma in situ or invasive breast cancer with superficial margins, inoperable or fungating masses, cutaneous tumor involvement, or inflammatory tumor stages (T4b, c, d, or any ypT4) the boluses need to be apply. 5 Based on ref,5,8,13,14 it should be considered that, when identifying high-risk individuals for whom the higher toxicity associated with bolus administration may be justified, clinical and biological criteria—such as tumor grade, T-stage, molecular profile, response to systemic therapy, and lymphovascular space invasion—should also be taken into account. In addition, the design of the bolus needs to be precisely adjusted according to the shape and location of the tumor, while also paying particular attention to its extension at the edges of the radiation field. This is based on two physical considerations: first, secondary electrons generate dose perturbation effects at the interface between the bolus and air, and extending the bolus beyond the field edges can effectively eliminate the dose cold spot at the interface 69 ; second, considering the risks of treatment positioning errors and tumor motion due to breathing, an appropriate edge buffer ensures that the dose fall-off zone remains outside the target area. 70 Therefore, evaluating whether the bolus extends appropriately to the field edges, as well as how it interacts with other treatment parameters (such as irradiation angles and dose distribution), is a crucial component of optimizing treatment plans and requires further research.
When using bolus, it is also important to consider that different radiotherapy techniques have significantly different requirements for the bolus. In this review, most of the included studies employed IMRT or VMAT techniques. Specifically, among the 32 articles analyzed, 24 (72.7%) utilized photon beams. Regarding beam energy, most studies (13 articles) used 6 MV photon beams, while some adopted a combination of beam energies, ranging from 6 MV to 15 MV. Overall, compared to traditional simple tangential fields, techniques like IMRT and VMAT involve more complex radiation dose distributions. 71 In these cases, the role of the bolus is not only to enhance the dose coverage of the target area but also to consider how it interacts with multiple irradiation fields and different radiation qualities (such as the combination of 6MV and 18MV radiation). Therefore, the use of bolus for these techniques requires systematic research. 72 Through such systematic analysis and optimization, the precision of radiotherapy and the treatment outcomes for patients can be further improved.
The development of 3D printing technology in the medical field has opened new possibilities for the therapeutic usage of boluses. The issues with traditional boluses, such as low conformability and a big air gap between the bolus and the body surface, have mostly been resolved by these 3D-printed boluses. Comparing individually tailored 3D boluses to conventional commercial boluses may increase the precision and effectiveness of the dosage in the intended area. This method of bolus production can also reduce the dose received the OAR, thus reducing the incidence of radiotherapy side effects. In addition, individualized boluses are printed using PLA, a novel renewable and biodegradable material made from plant starch. PLA therefore has the advantages of being environmentally friendly, having good print fluidity, being non-toxic, being less prone to warping, and undergoing minimal shrinkage after printing. Still in its early phases, 3D-printed boluses are being used in clinical settings. Issues such as the long time required to fabricate a 3D-printed bolus, high cost of equipment maintenance, and hardness of PLA need to be resolved. However, there are still fewer clinical studies on 3D printed bolus, and we are conducting a retrospective study on the use of 3D printed bolus in clinic, these study of 3D printed bolus between patients involving OAR, target area dose, acute skin response and other factors.
Overall, the use of boluses after mastectomy is moving towards a more individualized approach and becomes greater precision. At the same time, the specifications for the use of bolus are also being developed in a more standardized way as research is carried out. Further evaluation is required on whether boluses need to be used routinely after mastectomy and more prospective studies on the effect of boluses on the LRR and breast cancer mortality are needed. Moreover, the development of novel materials may address some of the ongoing issues with bolus use.
Conclusion
Bolus devices are widely used as tissue-equivalent compensators in radiotherapy for patients following postmastectomy. They enhance the radiation dose to the skin and subcutaneous tissues by eliminating the photon beam's dose build-up zone. Conventional boluses, typically uniform and reusable medical devices, are commonly used in clinical practice. However, these traditional boluses often do not conform well to the chest wall, leading to air gaps. Such gaps can result in inadequate skin dosing and inaccurate target area coverage, ultimately affecting treatment outcomes. Recently developed 3D-printed boluses can be customized for individual patients, allowing for better conformity to the chest wall and minimizing air gaps, thereby improving dose accuracy. Additionally, the use of biodegradable PLA materials not only enhances patient comfort but also supports environmental sustainability. Ongoing clinical trials on 3D-printed compensatory membranes are expected to provide valuable insights into their practical application and inform future treatment strategies.
Abbreviations
Clinical Target Volume
European Society for Radiotherapy & Oncology
Local Recurrence Rate
Organ-At-Risk
Play Dough Material
Polylactic Acid
Postmastectomy Radiation Therapy
Planning Target Volume
Variable-Shape Rubber-Containing Tungsten
Thermoplastic Elastomer
Three-Dimensional
Volumetric Modulated Arc Therapy.
Footnotes
Acknowledgements
We acknowledge the radiation therapists and the nurses (Ms. Trigo and Ms. Gonçalves) for their participation in the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
