Abstract
Introduction
Peripherally inserted central catheter (PICC)-related bloodstream infections (BSIs) are severe complications in breast cancer patients undergoing chemotherapy. This study evaluated the diagnostic potential of calcitonin gene-related peptide (CGRP), programmed cell death protein-1 (PD-1), and its ligand (PD-L1) as biomarkers for PICC-related BSIs.
Methods
A total of 384 breast cancer patients with PICC placement were retrospectively identified from medical records, of these, 78 developed BSIs and 306 did not. Serum levels of CGRP, PD-1, and PD-L1 were measured using enzyme-linked immunosorbent assay (ELISA) and quantitative polymerase chain reaction (qPCR), respectively, to evaluate their potential as diagnostic biomarkers for BSIs. Blood cultures were performed to confirm infections and identify pathogens.
Results
The BSIs group showed significantly lower CGRP and PD-L1 levels, and higher PD-1 expression and PD-1/PD-L1 ratios compared to the non-BSIs group (all P < 0.001). Receiver operating characteristic (ROC) curve analysis showed area under the curve (AUC) values of 0.84 for CGRP, 0.77 for PD-1, 0.70 for PD-L1, and 0.86 for the PD-1/PD-L1 ratio. Combined detection achieved an AUC of 0.96, with 88% sensitivity and 92% specificity. Gram-negative bacteria (59.8%) were the predominant pathogens, with Escherichia coli (29.3%) being the most common.
Conclusion
CGRP alone showed strong diagnostic utility, but combining CGRP, PD-1, and PD-L1 markedly enhanced accuracy. ELISA and qPCR detection of these markers provides results within hours, enabling earlier diagnosis than conventional blood cultures.
Keywords
Introduction
Breast cancer is a major global health concern for women, with the highest incidence and mortality rates among female cancers. 1 Chemotherapy is a cornerstone of comprehensive breast cancer treatment, and peripherally inserted central catheters (PICC) are widely used as a safe and convenient route for the administration of chemotherapeutic agents. 2 However, PICC-related bloodstream infections (BSIs) are a frequent complication, often causing treatment disruptions, extended hospitalizations, higher medical expenses, and severe, sometimes fatal, outcomes. 3 The reported incidence of PICC-related BSIs ranges from 2% to 20%, with breast cancer patients being particularly vulnerable due to chemotherapy-induced neutropenia and impaired immune surveillance. 4 Prompt and accurate diagnosis is therefore vital to mitigate adverse outcomes and prevent treatment delays.
Currently, the diagnosis of PICC-related BSIs primarily relies on clinical symptoms (eg, fever, chills) and microbiological culture results. 5 However, clinical symptoms are often nonspecific, and microbiological cultures are time-consuming and lack sufficient sensitivity, making early diagnosis challenging. 6 These limitations frequently delay diagnosis and treatment initiation, highlighting the critical need to identify reliable, rapid, and accessible diagnostic tools. Therefore, identifying reliable, rapid, and minimally invasive biomarkers is crucial for the timely recognition and management of high-risk patients.
Biomarkers are increasingly important in infectious diseases. Calcitonin gene-related peptide (CGRP), linked to immunity, plays a key role in bacterial infections and inflammation.7,8 Changes in CGRP levels may reflect the activation or suppression of the immune response, making it a potential biomarker for infectious diseases. 9 Additionally, programmed cell death protein-1 (PD-1) and its ligand (PD-L1), known for their role in immune checkpoint regulation and tumor immune evasion, have yet to be fully elucidated in the context of infections. 10 Emerging evidence suggests that the PD-1/PD-L1 pathway may promote pathogen immune escape by inducing an immunosuppressive state, thereby exacerbating infections. 11
In the clinical setting of PICC-related BSIs in breast cancer patients, the potential of CGRP, PD-1, and PD-L1 as effective biomarkers for early diagnosis and prognosis assessment remains to be validated through clinical and experimental data. This study aims to evaluate the predictive value of CGRP, PD-1, PD-L1, and their ratios in PICC-related BSIs among breast cancer patients. Furthermore, we explore whether the combined application of these biomarkers can enhance the sensitivity and specificity of early diagnosis, with a focus on reducing the time to detection compared to conventional culture-based methods, thereby providing a more precise diagnostic tool for clinical practice.
Methods
Study Design and Participants
The reporting of this study conforms to the STARD 2015 guidelines for reporting diagnostic accuracy studies. 12 A retrospective cohort of 503 breast cancer patients who underwent PICC placement at a single tertiary care center between October 2023 and September 2024 was initially identified. After applying inclusion and exclusion criteria, 384 patients were included in the final analysis (see Figure 1). Patients were identified through electronic medical record (EMR) using diagnostic codes for breast cancer and PICC placement for chemotherapy. A convenience sampling approach, commonly used in retrospective observational studies, 13 was used to include patients meeting the following criteria: histologically confirmed breast cancer, age ≥18 years, and absence of active infection at PICC insertion (temperature <38 °C within 24 h, no systemic antibiotics within 7 days, and negative blood cultures within 72 h if available). Exclusion criteria were active infection within 30 days before PICC placement, immunodeficiency (eg, HIV/AIDS, immunosuppressive therapy, stem cell transplantation), incomplete medical records, or concurrent enrollment in infection-related clinical trials. The study was approved by the local Institutional Review Board, and written informed consent was obtained for use of anonymized clinical data and samples.

Flow Diagram of Study Design.
Data Collection
Patient information, encompassing demographics, cancer stage, treatment specifics, and infection-related results, was retrieved from the EMR system. PICC-related BSIs were diagnosed according to the Infectious Diseases Society of America criteria and the Centers for Disease Control and Prevention guidelines.14,15 A confirmed BSI required systemic infection signs (eg, fever ≥38 °C, chills, or hypotension) with no alternative infection source, and microbiological evidence based on: (1) concordant positive cultures from PICC and peripheral blood samples (PICC positive ≥2 h earlier), (2) ≥ 15 CFU/plate from catheter tip cultures using the semi-quantitative roll-plate method, or (3) matching pathogens isolated from catheter tip and blood cultures. Recurrent BSIs were categorized as relapse (same pathogen ≤72 h post-treatment) or new infection (different pathogen). Colonization was defined as ≥15 CFU on catheter tip culture without systemic signs of infection. 16 Two independent reviewers verified all BSI cases, with discrepancies adjudicated by a third expert.
Blood samples were collected from both the PICC line and peripheral veins using BACTEC Plus Aerobic/F and Anaerobic/F bottles (BD Diagnostics, USA). Cultures were incubated in the BACTEC FX system at 37 °C for up to five days. Positive results were automatically flagged, and pathogens were identified via Gram staining, colony morphology, biochemical profiling, and the VITEK 2 automated identification system (bioMérieux, France). Catheter tip cultures were grown on Columbia Blood Agar and MacConkey Agar at 37 °C for 48 h. All specimens were processed individually (not pooled) in the hospital's certified microbiology laboratory by trained technicians.
Biomarker Measurement
Peripheral blood samples were collected at the onset of clinical symptoms suggestive of BSI (eg, fever ≥38 °C) and prior to antibiotic administration. Blood was drawn into serum-separating tubes (SST; BD Vacutainer®, Franklin Lakes, NJ, USA), allowed to clot for 30 min at room temperature, and centrifuged at 3000 rpm for 10 min. The resulting serum was aliquoted and stored at −80 °C until analysis
Serum CGRP concentrations were measured using a commercial enzyme-linked immunosorbent assay (ELISA) kit (Human CGRP ELISA Kit, Cat. No. YJ57929, Shanghai Yaji Biological Technology Co., Ltd, China), following the manufacturer's protocol.
PD-1 and PD-L1 expression levels were assessed using quantitative real-time polymerase chain reaction (qPCR). Total RNA was extracted from whole blood using TRIzol reagent (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA). Reverse transcription was performed with the PrimeScript™ RT reagent kit (Takara Bio, Dalian, China), and qPCR was carried out using SYBR Green PCR Master Mix on a QuantStudio™ 5 Real-Time PCR System (Thermo Fisher Scientific, Waltham, MA, USA).
Gene-specific primers for PD-1, PD-L1, and the reference gene GAPDH were designed and synthesized by Sangon Biotech (Shanghai, China). The sequences and amplicon lengths are provided in Table 1.
Primer Sequences Used for Quantitative Real-Time PCR.

Abbreviations: F, forward primer; R, reverse primer; bp, base pairs.
Statistical Analysis
Continuous variables were expressed as mean ± SD, and categorical variables as frequencies (%). Group differences were analyzed using the t-test, Mann-Whitney U test, or chi-square test. Receiver operating characteristic (ROC) curve analysis was used to was conducted to assess the diagnostic performance of each biomarker, and the area under the curve (AUC), sensitivity, and specificity were calculated using the Youden index. A P-value <0.05 was considered statistically significant. Analyses were performed using SPSS software version 22.0 (IBM Corp., Armonk, NY, USA).
Results
Study Population and Baseline Characteristics
A total of 384 breast cancer patients were included in the final analysis, among whom 78 (20.3%) developed PICC-related BSIs and 306 (79.7%) did not. Baseline clinical and biomarker characteristics are summarized in Table 2. No significant differences were noted between the groups in age, BMI, clinical T/N stage, pathology type, hormone status, HER2 status, or Ki-67 status (all P > 0.05). However, significant differences were noted in biomarker levels: patients with PICC-related BSIs had lower CGRP levels (37.62 ± 10.43 pg/mL vs 58.68 ± 16.72 pg/mL, P < 0.001), higher PD-1 levels (2.63 ± 0.76 vs 1.83 ± 0.59, P < 0.001), lower PD-L1 levels (1.19 ± 0.38 vs 1.52 ± 0.48, P < 0.001), and higher PD-1/PD-L1 ratios (2.11 ± 0.67 vs 1.20 ± 0.39, P < 0.001). Although numeric differences varied among biomarkers, the highly significant P-values (P < 0.001) were consistent across appropriate statistical methods and reflect data variability.
Clinicodemographic and Biomarker Characteristics between Patients with and Without PICC-Related BSIs.
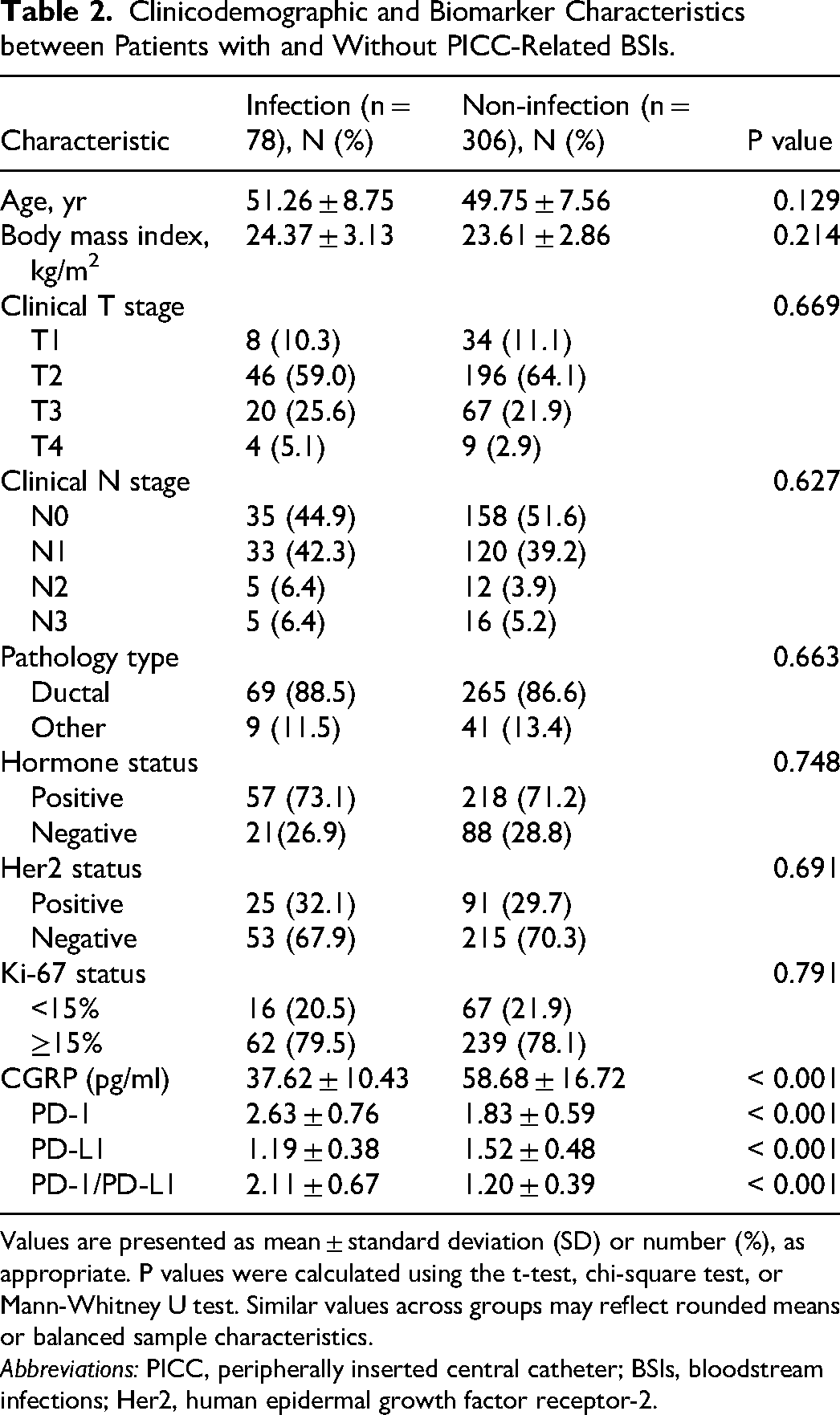
Values are presented as mean ± standard deviation (SD) or number (%), as appropriate. P values were calculated using the t-test, chi-square test, or Mann-Whitney U test. Similar values across groups may reflect rounded means or balanced sample characteristics.
Pathogen Distribution
The pathogens associated with PICC-related BSIs are detailed in Table 3. A total of 102 pathogens were isolated from the 78 BSI patients, as some patients presented with polymicrobial infections. Gram-negative bacteria accounted for 59.8% (61/102) of the isolates, with Escherichia coli (29.3%) and Klebsiella pneumoniae (15.9%) being the most common. Gram-positive bacteria constituted 40.2% (41/102) of the isolates, predominantly Staphylococcus epidermidis (29.4%) and Staphylococcus aureus (6.9%).
Distribution of Pathogens Isolated from PICC-Related Bloodstream Infections (n = 102 Strains).
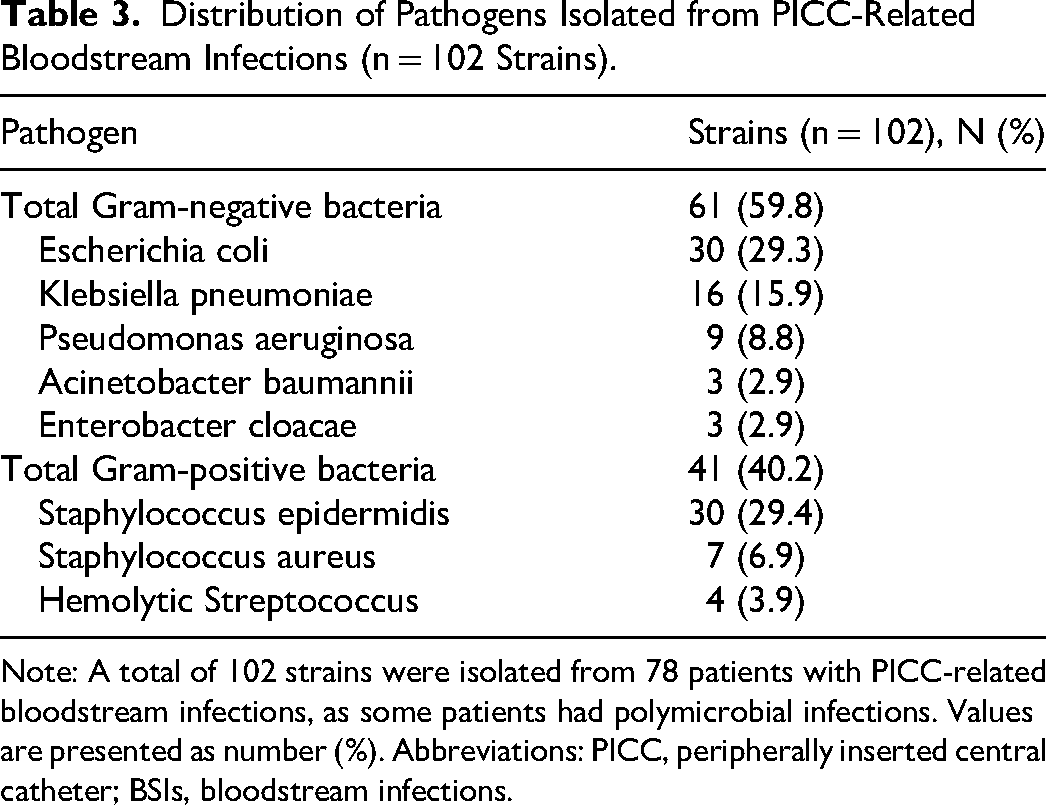
Note: A total of 102 strains were isolated from 78 patients with PICC-related bloodstream infections, as some patients had polymicrobial infections. Values are presented as number (%). Abbreviations: PICC, peripherally inserted central catheter; BSIs, bloodstream infections.
Predictive Performance of Biomarkers
The predictive value of CGRP, PD-1, PD-L1, and PD-1/PD-L1 for PICC-related BSIs was evaluated using ROC curve analysis (Table 4). CGRP demonstrated an AUC of 0.84, with a sensitivity of 60% and specificity of 65% at a cutoff of 42.817 pg/mL. PD-1 showed an AUC of 0.77, with a sensitivity of 82% and specificity of 65% at a cutoff of 2.301. PD-L1 had an AUC of 0.70, with a sensitivity of 52% and specificity of 55% at a cutoff of 1.613. The PD-1/PD-L1 ratio exhibited the highest individual AUC of 0.86, with a sensitivity of 86% and specificity of 77% at a cutoff of 1.634. When these biomarkers were combined, the predictive performance improved significantly, achieving an AUC of 0.96, with a sensitivity of 88% and specificity of 92% at a cutoff of 0.135.
Predictive Value of CGRP, PD-1, PD-L1 and PD-1/PD-L1 for PICC-Related BSIs.
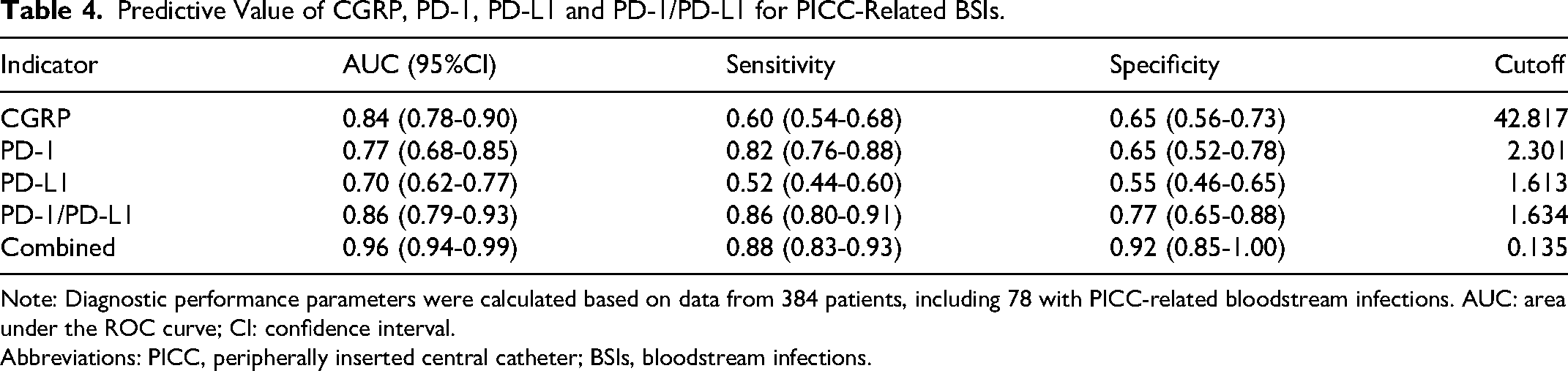
Note: Diagnostic performance parameters were calculated based on data from 384 patients, including 78 with PICC-related bloodstream infections. AUC: area under the ROC curve; CI: confidence interval.
Abbreviations: PICC, peripherally inserted central catheter; BSIs, bloodstream infections.
The ROC curves for each biomarker and the combined model are presented in Figure 2. Visual inspection of the curves revealed that the combined model consistently outperformed all individual biomarkers, with a steeper curve and a higher AUC value (Figure 2E). Among the individual biomarkers, the PD-1/PD-L1 ratio exhibited the best performance (Figure 2D), followed by CGRP (Figure 2A), PD-1 (Figure 2B), and PD-L1(Figure 2C). These findings underscore the importance of combining multiple biomarkers to enhance predictive accuracy

ROC Curves for Individual Biomarkers Based on ELISA and qPCR Data Collected at the Onset of Suspected BSI: A: CGRP, B: PD-1, C: PD-L1, D: PD-1/PD-L1 Ratio. E: Combined Model Based on Logistic Regression of All Biomarkers. F: Calibration Curve Demonstrating the Agreement Between Predicted Probabilities and Observed Outcomes for the Combined Model. ROC Analysis was Performed Using SPSS 22.0, and AUC, Sensitivity, and Specificity were Determined via the Youden Index.
To further evaluate the predictive combined models, calibration curves were constructed to assess the agreement between predicted probabilities and observed outcomes. The calibration curves for the combined model are shown in Figure 2F. The results indicated that the combined model exhibited excellent calibration, with predicted probabilities closely aligning with actual outcomes.
Discussion
This study demonstrates that serum levels of CGRP, PD-1, and PD-L1 are significantly associated with PICC-related BSIs in breast cancer patients. Our findings reveal that patients with PICC-related BSIs exhibit lower levels of CGRP and PD-L1, higher levels of PD-1, and an elevated PD-1/PD-L1 ratio compared to non-infected patients. These biomarkers, particularly when combined, show high diagnostic accuracy, with an AUC of 0.96, sensitivity of 88%, and specificity of 92%. These findings suggest that CGRP, PD-1, and PD-L1 may serve as valuable biomarkers to support earlier diagnosis of PICC-related BSIs, as they can be detected rapidly through ELISA and qPCR assays, well before blood culture results become available. This advantage may facilitate prompt clinical decision-making and infection control.
CGRP is a neuropeptide with dual roles in vasodilation and immune regulation.17-19 Its presence in circulating serum and high sensitivity to detection via ELISA makes it a practical candidate for BSI biomarker development. In this study, we observed significantly reduced CGRP levels in patients with PICC-related BSIs, suggesting a potential association with impaired immune response. Mechanistically, CGRP has been shown to inhibit the release of pro-inflammatory cytokines (eg, TNF-α, IL-1β) and enhance endothelial barrier integrity, thereby mitigating tissue damage during bacterial infections and sepsis.20,21 Therefore, lower CGRP levels may reflect dysregulated neuro-immune signaling, leading to excessive inflammation and increased vascular permeability that facilitates microbial dissemination. 22 This interpretation is consistent with previous evidence showing that CGRP-deficient mice are more susceptible to severe bacterial infections. 23 Owing to its robust diagnostic performance (AUC = 0.84), CGRP represents a promising surrogate marker of immune dysregulation in catheter-related infections.
The PD-1/PD-L1 axis, a well-established immune checkpoint in cancer, is increasingly recognized for its role in infectious diseases. 24 Elevated PD-1 levels and reduced PD-L1 expression in our cohort suggest a state of T-cell exhaustion, wherein persistent antigen exposure (eg, from catheter biofilm colonization) leads to diminished effector T-cell function. 25 PD-1 upregulation inhibits T-cell activation and cytokine production, impairing pathogen clearance, while reduced PD-L1 may disrupt feedback mechanisms that temper excessive immune responses. 26 The elevated PD-1/PD-L1 ratio (2.11 vs 1.20) further emphasizes this imbalance, mirroring findings in chronic viral infections such as HIV and hepatitis C. 24 These results imply that targeting the PD-1/PD-L1 pathway—either diagnostically or therapeutically—could restore immune competence in PICC-related BSIs. For example, checkpoint inhibitors (eg, anti-PD-1 antibodies), though primarily used in oncology, might hold promise for reversing immune exhaustion in refractory infections. 27
Given the distinct biological characteristics and detection platforms of CGRP (a circulating peptide detected by ELISA) and PD-1/PD-L1 (membrane-bound immune checkpoint molecules detected by qPCR), we propose a combined diagnostic approach that leverages the complementary strengths of both methods. Such a strategy—implemented either sequentially or in parallel depending on clinical laboratory resources—could provide a more comprehensive immune and inflammatory profile, enabling faster and more accurate identification of PICC-related BSIs in breast cancer patients.
The predominance of Gram-negative bacteria (59.8%), particularly Escherichia coli, aligns with global reports of rising Gram-negative infections in catheter-associated BSIs. 28 E. coli's propensity for biofilm formation on catheter surfaces likely contributes to its prevalence, as biofilms shield bacteria from host defenses and antibiotics. 29 Notably, the high resistance rates to first-line antibiotics (eg, amoxicillin, cefazolin) underscore the urgency of antimicrobial stewardship. Our findings advocate for empirical therapy targeting Gram-negative pathogens in breast cancer patients with PICC-related BSIs, guided by local resistance patterns. Furthermore, the integration of biomarker-driven diagnostics could facilitate earlier pathogen-specific interventions, reducing reliance on broad-spectrum antibiotics and mitigating resistance development.
Several limitations must be acknowledged. The retrospective, single-center design may introduce selection bias and limit generalizability. Biomarker measurements were taken at a single time point, preventing dynamic assessment over the course of infection. Furthermore, this study did not include a sample size calculation or a breast cancer control group without PICC placement, limiting our ability to fully delineate infection-specific biomarker changes. Additional confounders, including comorbidities and treatment regimens, may have influenced biomarker expression. Future multicenter, prospective studies with appropriate controls are needed to validate our findings and assess their broader clinical applicability.
To translate this biomarker panel into clinical practice, several research directions are warranted. These include: (1) mechanistic studies exploring the roles of CGRP and PD-1/PD-L1 in host-pathogen interactions; (2) validation of diagnostic cutoffs in diverse patient populations; (3) development of point-of-care platforms integrating.
Conclusion
In summary, this study identifies CGRP, PD-1, and PD-L1 as promising biomarkers for the early diagnosis of PICC-related BSIs in breast cancer patients. The combined detection of serum CGRP by ELISA and PD-1/PD-L1 mRNA expression by qPCR enables rapid, complementary immune profiling and delivers results within hours—well before conventional blood culture results become available. This advantage may facilitate earlier clinical decision-making and timely intervention. Future validation and mechanistic studies are warranted to refine this biomarker panel and explore its role in personalized infection management.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to all the patients who participated in this study for their invaluable contribution
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The raw data supporting this study's findings can be provided by the authors upon request.
Informed Consent
Written informed consent was obtained from all participants for the use of their anonymized clinical data and biological samples in this study, as approved by the IRB of the Nanchang People’s Hospital.
Institutional Review Board Statement
The study protocol was reviewed and approved by the IRB of Nanchang People’s Hospital (Ethics Committee reference number: k-lw2023002).
