Abstract
Colorectal cancer ranks as one of the most common malignancies globally, with mortality rates second only to lung cancer. Despite improvements in diagnostic and therapeutic approaches, long-term survival rates for colorectal cancer patients remain unsatisfactory. The onset and progression of this disease involve intricate molecular mechanisms, influenced by a range of biological factors. Zinc finger proteins play a critical role in these processes, impacting tumor development and patient prognosis. This review summarizes current research on zinc finger proteins in colorectal cancer, highlighting key findings and advancements in understanding their role in tumor biology.
Colorectal cancer (CRC) is among the most common cancers worldwide, ranking third in incidence and second in cancer-related mortality. Recently, new favorable subsets of cancers of undefined origin (CUP) seem to emerge including CRC CUP. This new clinical entity contributes to the current increased incidence of CRC. 1 In 2020, global cancer statistics reported over 1.9 million new CRC cases and 935 000 deaths. 2 In China, the latest data shows 517 100 new CRC cases and 240 000 deaths, placing CRC as the second most common and fourth most deadly cancer, respectively. Even though older patients are more prone to severe postoperative complications, there is no consensus that age affects survival outcomes. The prognosis of older patients may be confounded by differences in stage at presentation, tumor site, preexisting comorbidities, and type of treatment received. 3 With an aging population, CRC incidence in China is rising annually. The primary treatment approach remains surgery, often combined with radiotherapy, chemotherapy, targeted therapy, and immunotherapy. Preoperative neoadjuvant therapies and postoperative treatments have improved cure rates to some extent, yet overall treatment outcomes for CRC remain unsatisfactory. Recent advances in various omics technologies and deeper CRC research have led to the discovery of new biomarkers for CRC diagnosis and treatment.4,5 As biomarker testing is recommended as a part of the standard investigation in CRC. Among several major genetic mutations in CRC, RAS mutation is correlated with oncological aggressiveness and the pathologic response to chemotherapy. There is also growing evidence that inflammation drives the development of the disease. As a result, many studies have investigated the predictive and prognostic role of various blood based inflammatory markers, including neutrophil–lymphocyte ratio (NLR), lymphocyte–monocyte ratio (LMR), and platelet–lymphocyte ratio (PLR). Finally, miRNAs have roles as tumor suppressor genes and oncogenes, and their diagnostic, prognostic, and predictive implications are now being explored. 6 However, much remains to be understood about the complex mechanisms driving CRC development and progression. Zinc finger proteins play a crucial role in regulating the tumor immune microenvironment. Studies have shown that Zinc Finger Protein 703 (ZNF703) is associated with the infiltration of CD3+ and CD8+ cells in colorectal cancer, and it shows potential as a target for immune checkpoint blockade (ICB) therapy in colorectal cancer. Zinc finger proteins may contribute to further advancements in the treatment of colorectal cancer. 4
Zinc finger proteins (ZFPs) are transcription factors characterized by finger-like domains that regulate human genes (Figure 1). Initially identified as structural elements in transcription factor IIIA in the African clawed frog, “zinc finger” now broadly describes various protein structures found in eukaryotes. ZFPs are encoded by 2% of human genes, forming the largest family of DNA-binding proteins. 7 Many contain cysteine or histidine residues, stabilizing their structure through coordination with zinc ions. Zinc finger motifs, classified into types such as Cys2His2 (C2H2)-like and zinc ribbon, play diverse regulatory roles depending on cellular contexts. 8 These proteins interact with Deoxyribonucleic acid (DNA), Ribonucleic acid (RNA), poly ADP-ribose (PAR), and proteins, influencing transcriptional regulation, protein degradation, signaling, DNA repair, and cell migration. 9 Increasing evidence suggests that abnormal ZFP expression is linked to several malignancies, including liver, 10 gastric, 11 breast, 12 lung, 13 and prostate cancers. 14 Through effects on cell proliferation, differentiation, migration, DNA methylation, signaling, and inflammation, ZFPs can either promote or inhibit CRC progression. 15 This article reviews the current research on ZFPs in CRC.
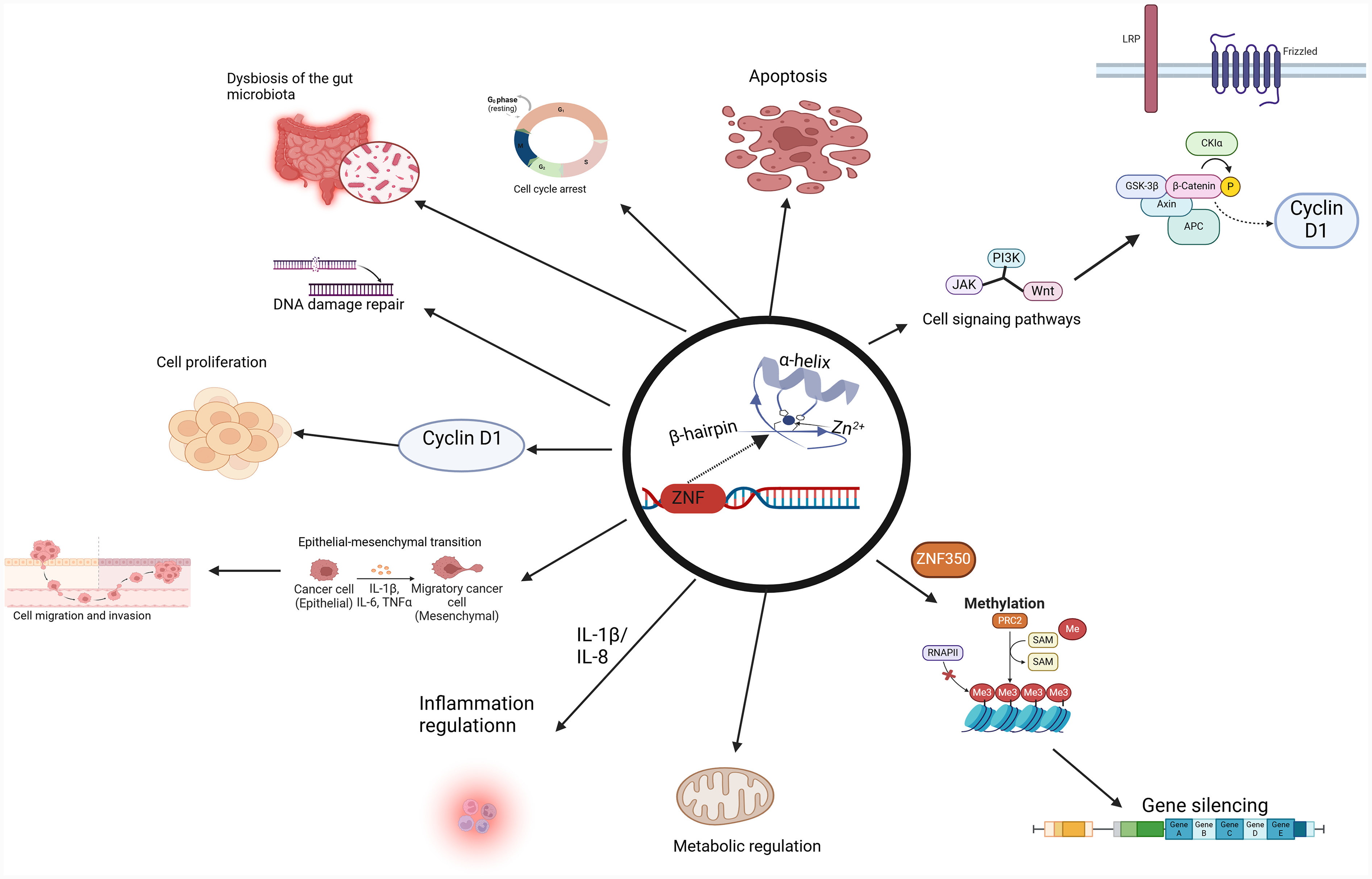
Zinc Finger Proteins Play a Vital Role in Living Organisms. The Figure Illustrates the Structure of Zinc Finger Proteins and the Various Biological Functions: Transcriptional Regulation, DNA Repair, Post-Translational.
Zinc Finger Proteins and Colorectal Cancer Cell Proliferation and Differentiation
Intestinal stem cells (ISCs) and undifferentiated transit-amplifying cells (TACs) maintain the balance of intestinal epithelial cells.16,17 TACs rapidly proliferate to produce various cell types, like goblet cells and functional epithelial cells, playing a key role in cell growth and differentiation. 18 When TACs lose regulation, this balance may disrupt, potentially leading to tumor formation. Zinc Finger Protein 277 (ZNF277) as a critical regulator of TAC proliferation, enhancing tumorigenesis. Additionally, ZNF277 is linked to the cell cycle inhibitor p21WAF1, suppressing cell aging by inhibiting p21WAF1 in colon cancer cells, thus promoting CRC development. 18
Cyclin-Dependent Kinase 1 (CDK1), essential for G1/S and G2/M cell cycle transitions, is regulated by ZNF880, as shown by Dong et al, who found that ZNF880 enhances CRC progression by binding to the CDK1 promoter and increasing transcriptional activity. 19 Suski J.M. et al demonstrated that progression through the G0, G1, and S cell cycle phases requires Cyclin D1-CDK4/CDK6 and Cyclin E/CDK2 complexes.20,21 Zinc Finger Protein 692 (ZNF692) functions as an oncogene by altering Cyclin D1 and CDK2 expression in CRC, while Heat Shock Protein 4 (HSF4), a ZNF692 target, promotes cell proliferation by preventing apoptosis. 22 Zinc finger protein 143 (ZNF143) is involved in cell cycle progression, and small molecules that inhibit the transcriptional activity of E2F or c-Myc can induce cell cycle arrest at the G0/G1 phase. YPC-21661 is a novel inhibitor of ZNF143 that exerts cytotoxic effects on cancer cells by inducing G2/M phase cell cycle arrest and apoptosis. In colorectal cancer cells, YPC-21661 downregulates the expression of ZNF143 target genes by inhibiting the binding of ZNF143 to DNA. Additionally, ZNF143 binds to cisplatin-modified DNA and is involved in cisplatin resistance. 23 YPC-21661 enhances the therapeutic efficacy of cisplatin. This study reveals that YPC-21661 is the first ZNF143 inhibitor that demonstrates anticancer activity both in vitro and in vivo, and it may become a novel anticancer drug for the treatment of colorectal cancer. 24
Hypoxic environments, common in tumors, drive angiogenesis and invasion by stabilizing HIF-1α, a transcription factor central to oxygen regulation.25,26 In CRC, HIF-1α stabilization fosters angiogenesis, invasion, and chemotherapy resistance. 27 Zinc Finger Protein 91 (ZFP91) was shown to increase HIF-1α levels, thereby promoting CRC cell proliferation and tumor progression. 28 Furthermore, ZIC5 enhances tumor progression by upregulating Platelet-Derived Growth Factor D (PDGFD), 29 which activates the FAK/STAT3 pathway. Silencing ZIC5 or PDGFD reduces STAT3 phosphorylation, thereby inducing cell death, suggesting their role in sustaining CRC cell proliferation and migration. 29
Studies also identify Zinc Finger Protein 281 (ZFP281) as crucial for stem cell differentiation, with knockdown of ZFP281 significantly reducing CRC cell proliferation. 30 Clinical studies have identified ZNF281 as a resistance factor against radiation-induced DNA damage, which increases the survival rate of CRC cells exposed to x-ray radiation. ZNF281 knockdown in colorectal cancer cells enhances radiation resistance by promoting Double-Strand Break (DSB) repair in colorectal cancer cells. The upregulation of ZNF281 in colorectal cancer is associated with poor prognosis in locally advanced rectal cancer (LARC) patients who receive radiotherapy, suggesting that ZNF281 could serve as an effective biomarker for predicting the sensitivity to radiotherapy in colorectal cancer and a target for radiosensitization in LARC patients. This could contribute to the development of precision treatment strategies in future clinical practice. 5 Additionally, Zic2, a ZFP with C2H2 zinc finger domains, enhances CRC cell survival under low glucose conditions. 31 Zhao et al found that Zic2 supports CRC cell growth by activating Cyclin D1 transcription. 32 ZNF169, in association with peptide-tRNA hydrolase (ANKZF1), regulates cell cycle, apoptosis, autophagy, and DNA repair, further influencing CRC cell proliferation. Zhao et al found that the Wnt/β-catenin, Zic2, and Zic5 signaling pathways regulate the glucose transporter 1 (GLUT1) gene to modulate glucose metabolism, thereby affecting the development of colorectal cancer.33,34
Ribosome biogenesis is pivotal in cancer cell growth. 35 Wang et al showed that Zinc Finger Protein 545 (ZNF545) inhibits ribosome biogenesis in CRC by binding to the rDNA promoter and recruiting transcriptional repressors. Knockdown of ZNF545 enhances CRC development, suggesting a tumor-suppressive function. 36 These findings collectively indicate that zinc finger proteins regulate CRC progression through control of cell proliferation and differentiation.
Zinc Finger Proteins and Colorectal Cancer Cell Motility and Migration
Metastasis, including cell adhesion, matrix degradation, and migration, is critical to CRC prognosis, with liver metastasis occurring in approximately 50% of CRC cases. Metastasis is a primary cause of poor CRC outcomes. Hao et al reported that Zinc Finger Protein 460 (ZNF460) is highly expressed in CRC, with elevated levels linked to lower survival rates. ZNF460 upregulation was shown to promote CRC metastasis via activation of the JAK/STAT3 pathway. 37 Daisuke Furukawa et al identified Zinc Finger Protein 185 (ZNF185) as an independent predictor of liver metastasis, further associating ZNF185 with poor CRC prognosis. 38
Zinc Finger Protein 350 (ZNF350), which regulates epithelial-mesenchymal transition (EMT), influences CRC cell migration, with methylation of the ZNF350 promoter enhancing metastatic potential. 39 ZNF281, also crucial for EMT, increases cell migration and invasion, as shown by reduced invasive capacity in CRC cells following ZNF281 knockdown. 30 These findings highlight that zinc finger proteins promote CRC metastasis by regulating cell movement and invasiveness, underscoring their potential as prognostic biomarkers in CRC.
Zinc Finger Proteins Influence Colorectal Cancer through DNA Methylation Regulation
In addition to their roles in cell proliferation and migration, zinc finger proteins (ZFPs) also influence colorectal cancer (CRC) progression through the regulation of DNA methylation, a key epigenetic mechanism that modulates gene expression. DNA methylation, a process involving the addition of a methyl group to the fifth carbon of cytosine in CpG dinucleotides, is catalyzed by methyltransferases. 40 This modification affects DNA structure, causing chromatin condensation and altering gene transcription. Hypermethylation in promoter regions of tumor-suppressor genes disrupts critical regulatory pathways, leading to tumor progression. Hiroki Tanaka et al found that promoter methylation of zinc finger protein 350 (ZNF350) enhances CRC cell motility, with hypermethylation of three CpG sites within the ZNF350 promoter reducing gene expression and increasing cell migration. 39 Methylation changes are frequently linked to cancer, with high promoter methylation often correlating with gene silencing. 41 In CRC, tumor-suppressor gene downregulation through hypermethylation can be reversed by demethylation. 42 Bin et al discovered that 29 CpG sites of ZNF334 are highly methylated in CRC, with DNA methylation driving its downregulation. TET1, a methylation regulator, demonstrates anti-tumor effects in CRC cells by modulating ZNF334 methylation. Targeted demethylation of ZNF334 inhibits CRC cell growth. 43 These findings suggest that the methylation status of ZNFs can influence their expression and, consequently, affect CRC progression.
Zinc Finger Proteins Influence Colorectal Cancer through Signal Transduction Regulation
Research indicates that ZFPs affect CRC progression by modulating signal transduction pathways. The Wnt pathway, critical in CRC, includes both canonical and non-canonical pathways, with β-catenin playing a key role in the canonical pathway. 44 β-catenin, through Wnt pathway activation, facilitates tumor progression, promoting uncontrolled cell growth and migration when dysregulated. Emerging evidence indicates that ZNF281 modulates the Wnt/β-catenin signaling pathway by regulating its downstream effector Proto-Oncogene Protein (c-Myc). Studies demonstrate that upregulation of ZNF281 correlates with enhanced activation of the Wnt/β-catenin pathway and concurrent overexpression of c-Myc, suggesting a mechanistic role for ZNF281 in promoting colorectal cancer (CRC) cell proliferation and migration through Wnt/β-catenin signaling activation. These findings collectively underscore the potential of ZNF281 as a critical regulator in CRC progression. 30 Similarly, Zic2 directly represses the transcription of Axin2, a component of the β-catenin destruction complex. By doing so, it prevents the degradation of β-catenin, allowing its accumulation and nuclear translocation, which enhances Wnt signaling. Zic2 also interacts with β-catenin, further boosting Wnt activity in colon cancer cells. This multi-level regulation by Zic2 contributes significantly to CRC cell proliferation and tumorigenesis, making Zic2 a potential therapeutic target (Figure 2). 31

Zic2 Inhibits Axin2 Transcription, Resulting in β-Catenin Accumulation and Heightened Wnt Signaling, Which Drives Downstream Gene Transcription and CRC Cell Proliferation.
The JAK-STAT pathway, essential in cell proliferation, apoptosis, and immune regulation, is another critical target. ZNF460, for instance, promotes CRC invasion and metastasis via JAK-STAT activation, correlating with poor prognosis and increased metastatic potential in CRC. 37 In addition, STAT3, a key component of JAK-STAT, upregulates oncogenes, driving tumor progression. 45 ZNF70 activates the STAT3 pathway to promote IL-1β secretion by macrophages, facilitating CRC cell proliferation (Figure 3). 46 ZNF32 promotes the self-renewal of colorectal cancer stem cells and facilitates the development of colorectal cancer by activating the LEPR-STAT3 pathway. 47

ZNF70 Activates the STAT3 Pathway to Promote IL-1β Secretion by Macrophages, Facilitating CRC Cell Proliferation.
In the PI3K/AKT pathway, which is associated with CRC cell proliferation, ZNF692 promotes cell cycle progression and reduces apoptosis by upregulating cyclin D1 and CDK2 while downregulating p27Kip1 through the PI3K/AKT pathway. This activity increases MMP-9 expression, enhancing CRC progression. 48 Together, these studies reveal that ZFPs support CRC cell proliferation, invasion, and migration by modulating key signaling pathways.
Zinc Finger Proteins Influence Colorectal Cancer through Inflammation Regulation
In the tumor microenvironment, immune cells release factors that foster tumor progression, with inflammation playing a critical role in early tumorigenesis. Immune cell PD-L1 expression is significantly higher in mismatch repair (MMR)-deficient (MSI-H) CRC as compared to MMR-proficient (MSI-L) tumors, with no differences among the different MSI-H molecular subtypes. The recommended screening for defective, DNA MMR includes immunohistochemistry (IHC) and/or MSI test. However, there are challenges in distilling the biological and technical heterogeneity of MSI testing down to usable data. It has been reported in the literature that IHC testing of the mismatch repair machinery may give different results for a given germline mutation and has been suggested that this may be due to somatic mutations. 49 The risk of CRC escalates with chronic inflammation. IL-8, a major inflammatory factor produced by cancer cells, sustains the tumor environment by recruiting myeloid-derived suppressor cells, stimulating myofibroblast growth, and promoting angiogenesis. 50 IL-8 binds to CXCR1/2 receptors, triggering downstream pathways linked to CRC invasion and poor prognosis. ZNF143, when silenced, suppresses IL-8 expression, while activation of the IL-8-CXCR axis drives CRC progression. 51
In colitis-associated CRC, immune cell infiltration and inflammatory factor release activate pro-inflammatory transcription factors.52,53 IL-1β, for example, fosters cancer-associated inflammation. Studies demonstrate that ZNF70 enhances IL-1β secretion by promoting NLRP3 component expression and inflammasome assembly. In LPS/ATP-treated THP-1 cells, ZNF70 activates Src/STAT3, boosting Pro-IL-1β synthesis. ZNF70 further supports IL-1β secretion and CRC cell growth by activating NLRP3 and STAT3 pathways. 46
The Myc-associated zinc finger (MAZ) is highly active in inflammatory sites, playing a role in hypoxia-driven inflammatory responses in CRC. 54 Elevated MAZ expression in colitis intensifies tumorigenesis through both HIF-2α-dependent and independent mechanisms, with studies showing that MAZ regulates CRC cell growth. MAZ advances CRC by promoting the expression of Cxcl1, a potent neutrophil chemoattractant, through HIF-2α. 55 Additionally, dysbiosis in intestinal microbiota activates CRC-related inflammation. 56 Ji et al found that microbiota depletion reduced ZFP90's carcinogenic effects in a mouse model of colitis-associated CRC, with fecal microbiota transplantation studies revealing that ZFP90 accelerates CRC via the TLR4-PIK3-AKT-NF-κB pathway. 57
CD103+ CD8+ T cells, known as tissue-resident memory T (Trm) cells, 58 play a role in suppressing cancer progression and metastasis. Recent studies have shown that ZFPs, such as ZNF683, play a critical role in modulating the tumor microenvironment by regulating the function of tissue-resident memory T cells (Trm cells). These cells are essential for suppressing tumor progression, and their activity is influenced by ZFP-mediated signaling pathways. 59 These findings underscore that ZFPs influence CRC development by regulating inflammation.
Conclusion
Zinc finger proteins play both supportive and inhibitory roles in CRC progression, impacting patient prognosis (Table 1). These proteins are regulated by factors such as post-translational modifications, cell cycle changes, DNA methylation, inflammatory mediators, immune cells, and gut microbiota. The clinical implications of ZFPs in CRC are profound. Their involvement in key signaling pathways and tumor progression makes them promising candidates for early diagnosis and prognosis assessment. Additionally, targeting ZFPs through small-molecule inhibitors or gene-editing technologies could pave the way for novel precision therapies, particularly for patients with advanced or treatment-resistant CRC. Despite significant progress in understanding the role of ZFPs in CRC, several challenges remain. The functional diversity and tissue-specific expression of ZFPs complicate their targeting in therapeutic interventions. Current research on zinc finger proteins (ZFPs) in colorectal cancer (CRC) remains in the exploratory phase, with a notable gap between mechanistic insights and clinical translation. While preclinical studies highlight their regulatory roles in tumorigenesis, there is a lack of robust clinical validation and in-depth exploration of their therapeutic relevance, particularly regarding practical clinical applications and patient-centered outcomes. Future research should focus on several key areas: (1) exploring the potential of ZFPs as therapeutic targets, particularly in the context of drug resistance, metastasis and the need for personalized medicine approaches; (2) investigating the role of ZFPs in the tumor microenvironment, including their interactions with immune cells and stromal components; (3) developing precision therapies that selectively modulate ZFP activity to improve CRC treatment outcomes. Expanding research on the role of ZFPs in CRC will further uncover molecular mechanisms of CRC development, offering a theoretical foundation for exploring new therapeutic strategies.
The Roles of Zinc Finger Protein in Colorectal Cancer.

Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (82003072).
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
