Abstract
Background
Gamma Knife radiosurgery has proven to be highly effective for small brain lesions and those with a limited number of metastases. This study aimed to evaluate the treatment outcome of Gamma Knife radiosurgery in Vietnamese breast cancer patients with brain metastasis.
Methods
This retrospective descriptive study included 75 patients treated between January 2019 and December 2023. Eligible patients had 1 to 5 brain lesions ≤ 3 cm, a Karnofsky Performance Status (KPS) score of ≥ 60, and no prior whole-brain radiotherapy or brain tumor resection. Clinical characteristics were documented, and imaging responses were evaluated using RECIST criteria. Overall survival (OS), brain-specific progression-free survival (BSPFS), and overall response rates were analyzed using Kaplan-Meier survival curves and Cox regression.
Results
Seventy-five patients were included in the study, with a median follow-up time of 15.9 months. The median age was 53.0 years (range: 29-73 years), and 39 patients (52%) were HER2-positive. The median total tumor volume per patient was 3.78 cm3. A total of 152 brain tumors were identified, of which 131 (86.2%) were <2 cm in diameter, 36 patients (48.0%) had a single lesion. The intracranial tumor control rates were 96% at 3 months and 92.5% at 6 months. The median overall survival (OS) was 17.2 months (range: 13.7-20.7 months). Multivariate analysis revealed that the total volume of metastatic lesions ≤7.0 cm3, hormone receptor negativity, ≤ 3 lines of systemic treatment before brain metastasis and controlled extracranial metastases were correlated with both BSPFS and OS.
Conclusion
Gamma Knife radiosurgery is an effective treatment for limited brain metastases, demonstrating a high rate of local control over brain lesions, which contributed to the promising survival outcome in Vietnamese breast cancer patients.
Introduction
Brain metastases from breast cancer occur in 5.1% of cases, with a higher prevalence in HER2-positive and triple-negative patients, ranging from 33% to 46%. 1 Metastatic breast cancer is diagnosed at the initial presentation (de novo) in 6% to 10% of cases, while 30% progress from earlier stages. 2 The average survival time for metastatic breast cancer is 2 to 3 years, with brain metastases associated with the worst prognosis. Previously, treatment options for brain metastases were limited, leading to rapid deterioration with neurological symptoms and significantly reduced survival. Currently, localized treatments for brain metastases include surgery, radiosurgery, and whole-brain radiation therapy, all considered fundamental approaches. Surgery can improve symptoms and extend survival but is typically reserved for single lesions with mass effect due to potential complications. 3 For decades, whole-brain radiation therapy has been the standard treatment for patients with brain metastases.
However, whole-brain radiation therapy can damage a significant amount of healthy brain tissue and lead to various neurological side effects. 4 Stereotactic radiosurgery, first proposed by Leksell in the 1950s, 5 has been shown in studies worldwide to offer advantages for treating brain metastases, including high tumor control rates, rapid symptom relief, prolonged survival, and fewer side effects. Vietnamese breast cancer patients often present at later stages, have a younger age at diagnosis and the proportion of HER2-positive subtype is considerably higher compared to Western population.6,7 Consequently, breast cancer patients in Vietnam have a generally poorer prognosis and higher risk of brain metastasis. Additionally, due to limited availability and high cost, access to targeted therapies with recorded intracranial activities such as trastuzumab deruxtecan or tucatinib is limited. Thus, local treatments remain a cornerstone of care. This article aims to evaluate survival outcomes and associated prognostic factors for breast cancer patients with brain metastases treated with Gamma Knife radiosurgery in Vietnam. The reporting of this study conforms to STROBE guidelines. 8
Materials and Methods
Patients and Methods
This retrospective study was conducted at a public tertiary referral center which receives cancer patients across Northern Vietnam, which is one of the three centers in Vietnam equipped with radiosurgery machines. Since 2019, our hospital has utilized the Leksell Gamma Knife ICON unit (Elekta AB) for stereotactic radiosurgery.
The study included 75 patients with either recurrent brain metastases from breast cancer or newly diagnosed stage IV breast cancer with brain metastases January 2019 and December 2023. Diagnosis of brain metastases was confirmed by Magnetic Resonance Imaging (MRI) or biopsy results in cases of which the lesions on MRI were not typical for brain metastasis. Inclusion criteria included (1) good general condition, with an Eastern Cooperative Oncology Group (ECOG) performance status of 0–2 or a Karnofsky Performance Status (KPS) score above 60%; (2) no prior whole-brain radiotherapy or brain tumor resection; (3) five or fewer brain lesions, with no lesion larger than 3 cm in diameter; and (4) availability for follow-up examinations for a minimum of three months. Patients with symptoms of increased intracranial pressure, life-threatening conditions, coma, or stupor were excluded. Informed consent was waived due to the retrospective nature of this study.
Treatment Procedure
All patients were treated with Gamma Knife radiosurgery using the ICON model (Elekta Instruments). A Leksell head frame was applied to the patient's head under intravenous sedation and local anesthesia. MRI scan was used for treatment planning and was conducted on the same day as the treatment. Treatment planning was performed using Leksell GammaPlan software, version 11.0 and was in line with the AAPM Task Group 101. 9 Patients underwent single-session stereotactic radiosurgery (SRS), with dose selection based on the recommendations from the SRS dose escalation trial RTOG 90-05 adjusted according to individual patient needs. 10 Tumor size and location were considered when selecting the prescription dose, which ranged from 18 to 24 Gy at the margin. Prescription isodose lines varied from 50% to 95%, with lower isodoses used for larger tumors. The median prescription isodose was 50%.
Follow-up and Evaluation
Patients were followed up with clinical examinations and MRI of the brain at 90-day intervals. Radiological responses to GKS were categorized as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) according to the RANO-BM (Response Assessment in Neuro-Oncology Brain Metastases), assessed at 3 and 6 months after Gamma Knife radiosurgery (GKRS). CR was defined as the disappearance of the enhanced lesion; PR was a tumor volume reduction of more than 30%; SD was neither sufficient shrinkage to qualify for PR or CR nor sufficient increase to qualify for PD; PD was the appearance of new lesions or an increase in tumor volume greater than 20%. Tumor progression was differentiated with radionecrosis by clinical evaluation and perfusion-weighted imaging or MR spectroscopy when clinically indicated. The intracranial tumor control (ITC) rate is calculated as the proportion of lesions that show a complete response, partial response, or stable disease. Brain-specific progression-free survival (BSPFS) is defined as the time from the start of GKRS until the brain metastatic lesions progress (ie, increase in size or the appearance of new lesions), or until death or loss to follow-up. Overall survival (OS) is defined as the time from the start of GKRS to the time of last follow-up or the patient's death.
Statistical Analysis
The Kaplan-Meier method was used to describe overall survival and time to brain recurrence. The log-rank test was employed for univariate analysis to compare these endpoints. Cox regression analysis was conducted to identify predictors of survival, incorporating eight potential prognostic factors: Karnofsky Performance Status (KPS) (≥80 vs < 80), number of tumors (single vs multiple), total tumor volume (≤7.0 cc vs > 7.0 cc), presence of extracranial metastasis (no vs yes), control of extracranial metastasis (yes vs no), hormone receptor status (negative vs positive), HER2 receptor status (positive vs negative), and the number of treatment lines before brain metastasis (BM) (≤3 vs 3). Factors included in the multivariate analysis were selected based on a literature review.11–15 p-values < .05 were considered statistically significant.
Results
Between January 2019 and December 2023, 75 breast cancer patients with brain metastases were treated with GKRS. The median follow-up duration was 15.9 months, with a range of 4.0 to 40.3 months.
Characteristics of Patients and Tumor Characteristics
Seventy-five patients with a total of 152 brain metastases lesions were enrolled in the study. Patient characteristics are summarized in Table 1. In the overall cohort, the median age at the diagnosis of brain metastases was 53.0 years (range: 29-73 years). Most patients had a good performance status at the time of brain metastasis diagnosis, with 62.7% having a Karnofsky index of ≥80. The interval between the diagnosis of breast cancer and the identification of brain metastases ranged from 1 to 124 months, with a mean of 46.4 months and a median of 36.5 months. The predominant histological type was invasive ductal carcinoma (n = 62, 82.7%), with 46.7% of tumors classified as grade 3. At the time of breast cancer diagnosis, 17.3% of patients had metastases. Regarding breast cancer subtypes, 34 patients (45.3%) were HER2-positive, and 19 patients (25.3%) were triple-negative. The majority of patients (n = 55, 73.3%) had extracranial metastases at the time of brain metastasis diagnosis, most commonly in the lungs (50.7%), bone (49.3%), and liver (42.7%). The brain was the first site of recurrence in 19 patients (25.3%). Brain metastases without active systemic disease were present in 26.7% of patients. Neurological symptoms were documented in 62 patients.
Characteristics of the Patients.
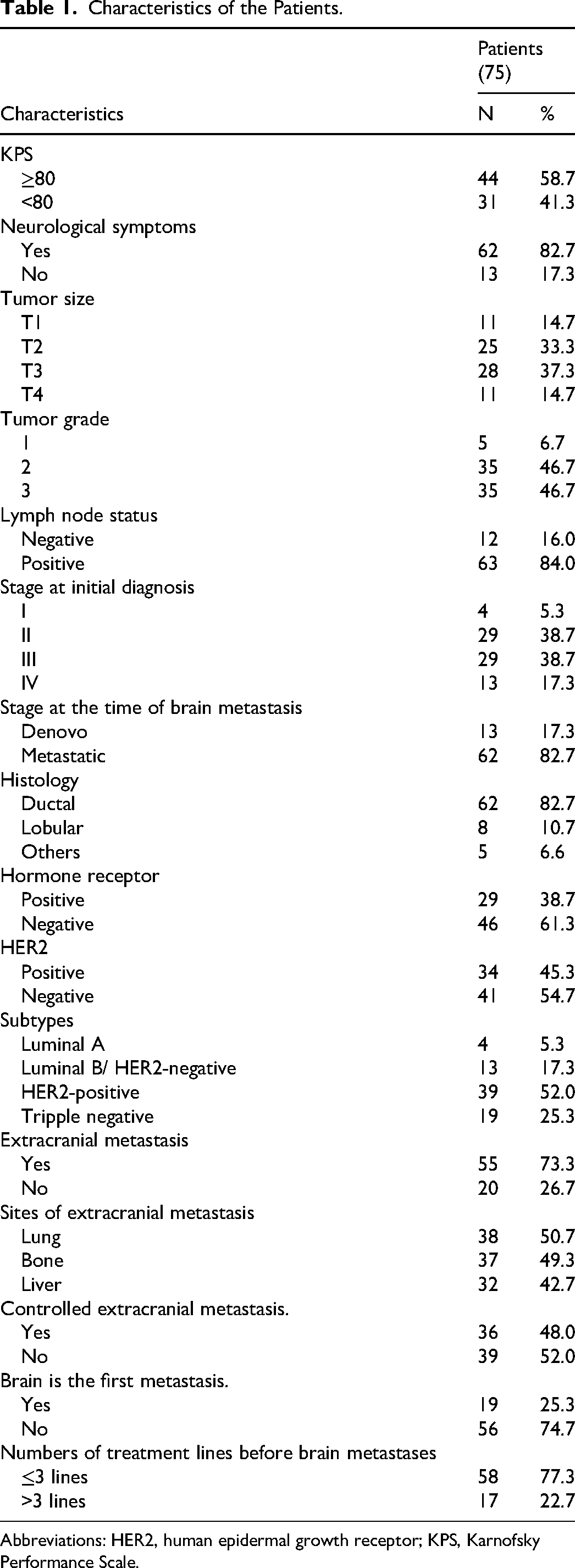
Abbreviations: HER2, human epidermal growth receptor; KPS, Karnofsky Performance Scale.
Of the 75 patients, 48.0% (n = 36) had a single brain lesion. The median total tumor volume per patient was 3.78 cm3, with a range from 0.06 to 34.9 cm3. A total of 152 brain tumors were identified, 86.2% of which were less than 2 cm in diameter. The distribution of brain lesions by location was as follows: frontal lobe (33.5%), parietal lobe (15.8%), occipital lobe (12.5%), cerebellum (22.4%), temporal lobe (13.8%), and other locations (2.0%). The neurologic symptoms included headache (54.6%), motor deficits (25.3%), seizures (12%), and cognitive impairments (9.3%). Tumor characteristics are summarized in Table 2.
Brain Tumor Characteristics.

Intracranial Tumor Control (ITC)
Among the 75 patients treated with GKRS and followed up with systematic imaging at 3 and 6 months, the intracranial tumor control (ITC) rate was 96% at 3 months. This rate included three subsets: complete response (25.3%), partial response (57.4%), and stable disease (13.3%). After GKRS, 10.6% of patients reported transient headaches, and 5% experienced mild nausea. At 6 months, the ITC rate remained high at 92.5%. At follow-up, most patients experienced stabilization or improvement in clinical symptoms, correlating with tumor control. Among five patients who progressed at 6 months after GKRS, only one patient had progression at the previously treated brain lesion. Detailed results are presented in Table 3.
Intracranial Tumor Control After Gamma Knife Therapy.

Brain Specific Progression – Free Survival (BS-PFS)
Among the 75 patients treated with GKRS, the median brain-specific progression-free survival (BS-PFS) was 15.6 months. The BS-PFS rates at 6 and 12 months were 86.0% and 58.6%, respectively. No statistically significant differences in BS-PFS were observed based on Karnofsky Performance Status (KPS), the total number of lesions, the total volume of lesions, HER2 receptor status, or the number of treatment lines before brain metastasis (BM).
However, significantly prolonged median BS-PFS was associated with hormone receptor status (18.6 months for hormone receptor-negative vs 12.2 months for hormone receptor-positive, p = .003) and the absence of extracranial metastases. Patients without extracranial metastases had a median BS-PFS of 29.0 months, compared to 14.4 months for those with extracranial metastases (p = .007). Additionally, patients with controlled extracranial metastatic lesions had a higher median BS-PFS of 22.1 months compared to 12.2 months for those with uncontrolled extracranial metastases (p < .05). Detailed results are presented in Table 4.
Brain Specific Progression-Free Survival.

Abbreviations: BM, brain metastasis; HER2, human epidermal growth receptor; KPS, Karnofsky Performance Scale.
Overall Survival
The median overall survival (OS) for the entire GKRS cohort following the diagnosis of brain metastases (BM) was 17.2 months (95% CI: 13.7-20.7 months). Survival rates at 6 and 12 months after GKRS were 90.6% and 69.3%, respectively. The median survival after BM diagnosis was 20.0 months for patients with a single lesion, compared to 15.9 months for those with multiple lesions (p = .051). For patients with a total metastatic tumor volume of ≤ 7 cm3, the median survival was 19.9 months, whereas it was 14.2 months for those with a tumor volume > 7 cm3 (p = .375).
Factors associated with significantly longer OS included the absence of extracranial metastases, controlled extracranial metastatic lesions, hormone receptor-negative status, and fewer lines of treatment before brain metastases (p < .05). No significant differences in survival were observed based on Karnofsky Performance Status (KPS), HER2 receptor status, stage at initial breast cancer diagnosis, the total number of brain lesions, or the total volume of brain lesions. Detailed OS and prognostic factors are presented in Table 5.
Overall Survival.

Abbreviations: BM, brain metastasis; HER2, human epidermal growth receptor; KPS, Karnofsky Performance Scale.
Predictive Factors of Survival
For brain-specific progression-free survival (BS-PFS), significant prognostic factors included hormone receptor status, control of extracranial metastasis, number of tumors, number of previous lines of treatment and total tumor volume. For overall survival (OS), significant prognostic factors were hormone receptor status, control of extracranial metastasis, total tumor volume, and the number of treatment lines before BM. Detailed results are presented in Table 6.
Prognostic Factors for Survival in Multivariate Analysis.

Abbreviations: HER2, human epidermal growth receptor; KPS, Karnofsky Performance Scale. HR: hazard ratio (lower HR indicated better chance of survival).
Discussion
Intracranial Tumor Control
Stereotactic radiosurgery (SRS) is a technique that delivers high doses of radiation precisely to tumors while minimizing irradiation of surrounding healthy tissue, leading to effective local control. In our study, the intracranial control rates at 3 and 6 months were 96% and 92.5%, respectively. Firlik et al, in a study of 58 breast cancer patients with brain metastases treated with GKRS, reported an intracranial control rate of 93%. 14 Similarly, Kyung et al reported brain control rates of 80.7% and 77.0% at 6 and 12 months, respectively. 16 In comparison, conventional whole-brain radiation therapy (WBRT) typically shows lower response rates, around 40–60%. Nieder et al evaluated WBRT (30 Gy in 10 fractions) in 108 patients with 336 lesions and found an overall response rate of 59% at 3 months, with 24% achieving a complete response and 35% achieving a partial response. 17 Additionally, radiosurgery offers advantages over surgery, particularly for tumors in challenging or deep brain locations, and helps avoid serious postoperative complications such as bleeding and infection.
Although GKRS is highly effective in controlling metastatic brain tumors, the emergence of new lesions remains a possibility due to its localized effect. Distant brain progression was the predominant form of intracranial progression in our series. This finding mirrors studies in high-income countries. For example, studies by Yamamoto et al and Aoyama et al reported that while SRS achieved robust local control, the risk of distant progression remained a significant challenge, especially in patients with active extracranial disease.18,19 McTyre's study of 2657 patients with brain metastases treated with GKRS found a 27.7% rate of new lesions at 6 months. 20 Similarly, Hanssens et al studied 837 patients with brain metastases treated with GKRS, including 81 with primary breast cancer, and reported new tumors in 40% of cases. 21 The relatively high incidence of new lesions can be attributed to the fact that once breast cancer has metastasized to the brain, it indicates that cancer cells have disseminated through the bloodstream and crossed the blood-brain barrier. The observed high rate of distant brain progression in our study underscores the need for comprehensive follow-up strategies, including periodic brain imaging and the importance of systemic therapies to treat extracranial diseases. Currently, there are no effective systemic treatments for micrometastatic brain lesions, which are lesions not detectable by modern imaging techniques. Due to the risk of recurrence in the brain after GKRS, prophylactic whole-brain radiation therapy (WBRT) has been considered. However, a randomized clinical trial by Aoyama et al involving 132 patients with 1–4 brain metastases smaller than 3 cm found no survival benefit from adding WBRT to radiosurgery alone. 19 In our study, among the patients with progressive disease or an increase in lesion size, some underwent re-treatment with stereotactic radiosurgery (SRS), while others with multiple recurrent brain metastases underwent whole-brain radiation therapy.
Survival Outcomes
Despite recent advances in breast cancer treatment and the development of new drugs, breast cancer with brain metastases continues to have a poor prognosis and remain a leading cause of morbidity and mortality. However, with improvements in radiation techniques, the treatment of brain metastases has significantly advanced, with radiosurgery playing a crucial role. In our study, patients with 1–5 brain metastases treated with GKRS had an overall survival (OS) of 17.2 months. This finding is consistent with several previous studies in resource-rich settings such as South Korea or the UK that report similar OS times of 15–18 months for patients with brain metastases from breast cancer.15,22,23 For example, Kondziolka's study reported a median OS of 11.2 months for patients treated with radiosurgery. 24 Norbert et al found that patients treated with radiosurgery alone had a median OS of 17.1 months, compared to 15 months for those who received a combination of radiosurgery and whole-brain radiation therapy. 15 Similarly, Wilson's study of 91 patients with brain metastases from breast cancer reported a median OS of 15.9 months for those treated with radiosurgery. 23
Analyzing the overall survival (OS) of 75 patients treated with radiosurgery revealed no significant difference in OS between those with single versus multiple brain tumors, or between those with total tumor volumes ≤7 cm3 versus >7 cm3. However, multivariate analysis indicated that patients with total tumor volumes ≤7 cm3 had better overall survival (p < .05). Kondziolka et al identified factors associated with longer survival, including controlled extracranial disease, a higher Karnofsky Performance Status (KPS), fewer brain metastases, a smaller total tumor volume per patient, and HER2/neu overexpression. 24 Additionally, fewer brain metastases and a smaller total tumor volume were linked to longer progression-free survival. Our study observed a trend toward increased OS in HER2-positive breast cancer patients compared to HER2-negative patients (19.9 months vs 15.9 months), although this difference was not statistically significant (p = .288). In a retrospective study by Kirch et al, involving 108 breast cancer patients with brain metastases, the HER2-positive group had a significantly better median survival compared to the HER2-negative group (22 months vs 9 months; p = .002). 25 Similarly, Kondziolka et al found that HER2-positive patients had longer survival in their study of 350 breast cancer patients with brain metastases (p < .05). 24 The reason for the longer survival in HER2-positive patients with brain metastases is not fully understood. It has been suggested that HER2-targeted therapy may help control tumor activity at extracranial metastatic sites. Recent studies have also indicated the potential intracranial anti-tumor activity of new drugs such as T-DM1, T-DXd, and tucatinib, offering new hope for treating breast cancer patients with brain metastases.26–28
Additionally, control of extracranial metastatic lesions significantly prolongs overall survival (OS) in breast cancer patients with brain metastases. Kondziolka et al found that controlling extracranial metastatic lesions significantly extended OS (p < .0005, HR = 2.2075). 24 While the absence of extracranial metastasis was associated with longer progression-free survival (PFS), it did not impact OS in our study. Our findings also showed that hormone receptor-negative patients had better survival rates compared to hormone receptor-positive patients, and this difference was also statistically significant in multivariate analysis. This may be attributed to the fact that most hormone receptor-positive patients in our study had extensive extracranial metastases before the development of brain metastases, which likely contributed to their poorer prognosis. Moreover, patients with extracranial metastases who received ≤3 lines of systemic therapy before brain metastasis had significantly longer OS compared to those who received >3 lines of therapy. This aligns with previous research. Kyung et al, in their study of 105 breast cancer patients with brain metastases treated with GKRS, found that a smaller number of metastases (≤3) and the absence of extracranial metastasis significantly affected brain-specific progression-free survival (BSPFS). 16 They also identified that a high Karnofsky Performance Status (KPS ≥70) and the receipt of systemic chemotherapy were associated with more favorable outcomes (p < .001). This suggests that patients who have undergone fewer lines of treatment before the development of brain metastases are more likely to achieve better control of extracranial metastases.
Several potential limitations of our study should be acknowledged. This was a retrospective, single-arm study and the sample size was relatively small, which may limit its statistical power and potentially impact the results. Despite these limitations, our study was not only the first report of treatment outcome of Gamma Knife in Vietnamese breast cancer patients but also provided data to support clinical management of breast cancer patients with brain metastases in similar resource-limited settings where access to new targeted therapies is limited.
Conclusion
Gamma Knife radiosurgery is an effective treatment method with high tumor control rates and safety, with promising overall survival for patients with limited brain metastases. Factors associated with better survival include a smaller total number of metastatic lesions, a smaller total tumor volume, fewer treatment lines before brain metastasis, the absence of extracranial metastases, and controlled extracranial disease at the time of GKRS. Therefore, Gamma Knife is worth pursuing for patients in Vietnam or other resource-limited settings, especially for those with the characteristics associated with improved survival as above. Besides, early detection, timely intervention for brain metastases, and effective control of extracranial metastatic lesions are crucial for enhancing overall survival.
Footnotes
Acknowledgements
None.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical Approval
This study was approved by Hanoi Medical University in Vietnam (approval no. 3214/QÐ-ÐHYHN). Informed consent was waived due to the retrospective nature of this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
