Abstract
Background
The prognostic value of the Geriatric 8 (G8) screening score in metastatic renal cell carcinoma (mRCC) patients receiving first-line immunotherapy remains unclear. This study aimed to evaluate the prognostic role of G8 within the context of the Meet-URO classification in mRCC patients treated with first-line ipilimumab-nivolumab.
Methods
This retrospective multicentre study analysed 106 mRCC patients treated with first-line ipilimumab-nivolumab. G8 and Meet-URO scores were calculated before treatment initiation. Primary endpoint was overall survival (OS), defined as duration from first administration of Nivolumab to death. OS was analysed in relation to age groups, G8 scores, and Meet-URO score categories, with data censored for patients still alive at the last follow-up. The secondary endpoint, progression-free survival (PFS), was measured from initiating Nivolumab to the earliest instance of disease progression or death. OS and PFS were assessed using Kaplan-Meier methods and Cox regression analyses. The reporting of this study conforms to the REMARK guidelines.
Results
Patients with G8 > 14 had more favorable IMDC and Meet-URO risk classifications and lower neutrophil-to-lymphocyte ratios. While PFS did not differ significantly between G8 ≤ 14 and >14 groups (1-year 29.3% vs 46.2%, p = 0.2), OS was significantly longer in G8 > 14 group (1-year 76.1% vs 58.6%, p = 0.006). In multivariable analysis, G8 ≤ 14 was independently associated with worse OS (HR 2.36, 95% CI 1.06-5.08, p = 0.03) but not PFS. The Meet-URO score was prognostic for both PFS and OS. In patients ≥70 years, G8 lost its prognostic value, while Meet-URO remained prognostic for OS.
Conclusions
The G8 score is an independent prognostic factor for OS but not PFS in mRCC patients receiving first-line ipilimumab-nivolumab. The Meet-URO score shows consistent prognostic ability for PFS and OS across age groups. These findings suggest that while G8 may be useful for individual patient-level OS prediction, the Meet-URO score may be superior for guiding treatment decisions in clinical practice.
Keywords
Introduction/Background
With ever growing life expectancy, physicians should expect to be faced with the challenges of managing a frailer oncological patient cohort. Given the recent advancements in immunotherapy for metastatic renal cell carcinoma (mRCC), it is vital to equip prognostic tools that risk stratify patient groups and help to determine survival outcomes by taking into consideration variables such as age, comorbidities, performance status, time between diagnosis and initiation of therapy as well as haematological and biochemical parameters.
The European Society for Medical Oncology (ESMO) clinical practice guideline advocates for the use of prognostic models in RCC such as the TNM classification for operable malignancies and the International Metastatic Renal Cancer Database Consortium (IMDC) score for advanced RCC. It is however important to note that the IMDC score was developed and validated at a time when tyrosine kinase inhibitors (TKIs) were more widely used as the first-line treatment option rather than immune checkpoint inhibitor (ICI) therapy where its prognostic value is now ambiguous. 1
The G8 geriatric screening tool was designed to identify older cancer patients and has been shown to amplify the predictive value of the ECOG performance status scale. 2 It has also been validated as a screening test for patients aged above 70 with cancer before initiation of treatment. Patients that have an abnormal G8 score undergo a geriatric assessment followed by relevant geriatric interventions and potentially adapted treatment measures such as best supportive care instead of immunotherapy that may have toxic inflammatory side effects.3,4
The Meet-URO score, a novel and cost-effective composite clinical prognostic score, incorporates further biomarkers, the pre-treatment neutrophil-to-lymphocyte ratio (NLR) which is a more accurate indicator of inflammation and bone metastases, into the IMDC model to formulate a risk stratification tool that holds prognostic value for predicting both OS and PFS and hence can be utilized to guide key immunotherapy decisions. 5
The aim of this research study is to evaluate the prognostic role of the G8 geriatric screening tool within the context of the Meet-URO classification against the backdrop of the evolving mRCC treatment landscape.
Methods
Study Design and Data Collection
This retrospective multicentre study evaluated clinical outcomes and prognostic factors in 318 patients with metastatic renal cell carcinoma (mRCC) treated with first-line Nivolumab and Ipilimumab. Data was collected from the Italian Expanded Access Program (EAP) across 85 centers between April and October 2019. The study adhered to ethical standards, including good clinical practice, and received Regional Ethical Committee approval. All participants, aged 18 and older with stage-IV mRCC, provided informed consent and had completed at least one cycle of nivolumab. All patient details have been de-identified. The reporting of this study conforms to the REMARK guidelines. 6
In the first-line setting, Nivolumab (3 mg/kg) and Ipilimumab (1 mg/kg) were administered intravenously every three weeks for four cycles, followed by maintenance therapy with Nivolumab at either 240 mg every two weeks or 480 mg every four weeks. For second-line or later therapy, Nivolumab was initially given at 3 mg/kg every two weeks. From May 2018 onward, the dosing regimen was adjusted to 240 mg biweekly or 480 mg every four weeks. The study timeline spanned from April 2019, when the first patient with mRCC commenced immunotherapy, to March 2021, which marked the last immunotherapy session included in the study. Treatment continued until disease progression, unacceptable toxicity, death, or patient withdrawal, with continuation beyond progression allowed if clinical benefit was evident. This study assessed the utility of the G8 score in predicting immunotherapy outcomes in elderly patients, comparing outcomes for scores above and below 14, with lower scores linked to worse prognoses according to existing literature. The primary endpoint was OS, defined as the duration from the first administration of Nivolumab to death. OS was analysed in relation to age groups, G8 scores, and Meet-URO score categories, with data censored for patients who were still alive at the last follow-up. The secondary endpoint, PFS, was measured from the initiation of Nivolumab to the earliest instance of disease progression or death. PFS data were also censored for patients who remained alive and free of progression at the last follow-up. CT scans of the chest, abdomen, pelvis, and head (when indicated) were used for radiological assessments, performed at baseline, every 2–4 months during treatment, or when progression was suspected. Tumor response was evaluated using RECIST version 1.1, with outcomes classified as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD).
Statistical Analysis
Patient characteristics were summarized as counts and percentages for categorical variables and as medians with ranges for continuous variables. Categorical variable associations were assessed using Fisher's exact test or Chi-square test, while the Kruskal-Wallis test was used to compare medians of continuous variables across groups. The Meet-URO score was calculated from baseline data, including IMDC risk stratification, bone metastases, and the NLR. The G8 score was also calculated before the first cycle of immunotherapy. Kaplan-Meier curves estimated OSand PFS. Median OS and PFS, along with follow-up durations, were computed using product-limit and reverse Kaplan-Meier methods. Differences in OS and PFS related to the G8 score, and other prognostic categories were evaluated with the log-rank test. Cox proportional hazards regression was used for both univariable and multivariable analyses to assess the influence of inflammatory markers, treatment lines, and other clinical factors on the endpoints, providing hazard ratios (HRs) and 95% confidence intervals (CIs). Factors with p-values <0.10 in univariable analyses were included in the multivariable models.
Statistical analyses were conducted using R Statistical Software (version 4.2.3; R Core Team, 2023), with significance set at p < 0.05.
Patient Selection and Flow
A total of 318 consecutive patients with mRCC were initially evaluated for this study. However, not all patients had complete clinical information available to calculate the G8 score. Patients were not excluded based on any criteria; rather, the analysis was limited to those with sufficient data to apply the G8 score. The patient flow is summarized in the flow diagram (Figure 1).

Diagram depicting patient flow through the study, highlighting both primary and secondary endpoints.
Results
Patients’ Characteristics
Our study analysed data from 106 patients with metastatic renal cell carcinoma (mRCC) treated with first-line ipilimumab and nivolumab combination immunotherapy.
Table 1 shows the patients’ characteristics stratified by G8 score with a cut-off of 14. Significant differences between the G8 populations were observed in the IMDC classification, with an association of a better G8 (i.e., > 14) with more intermediate and less poor IMDC risk groups (p = 0.007). Patients with G8 > 14 had a higher proportion in the intermediate risk group (83.3% vs 56.3%) and a lower proportion in the poor risk group (16.7% vs 43.8%) compared to those with G8 ≤ 14. Similarly, although with borderline statistical significance, the Meet-URO score risk groups were different, with better G8 scores associated with lower risk categories (p = 0.05). There was a significant difference in NLR values between the G8 groups (p = 0.04). Higher NLR values were associated with worse G8 scores (≤14) (60.9% with NLR ≥ 3.2 vs 38.1% in the G8 ≤ 14 vs G8 > 14, respectively).
Patients’ Characteristics.

Abbreviations: IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; NLR, Neutrophil-to-Lymphocyte Ratio.
In bold statistical significance.
The results of Table 2 compare the characteristics of patients over 70 years old, stratified by G8 score (≤14 vs > 14). Significant differences were confirmed in the IMDC score (p = 0.03), with a higher proportion of patients with G8 > 14 in the intermediate risk group (83.3% vs 50.0%) and fewer in the poor risk group (16.7% vs 50.9%). Additionally, there was a significant difference in NLR (p = 0.009), with a lower proportion of patients with G8 > 14 having an NLR ≥3.2 (50.0% vs 57.1%).
Over 70 Patients’ Characteristics.
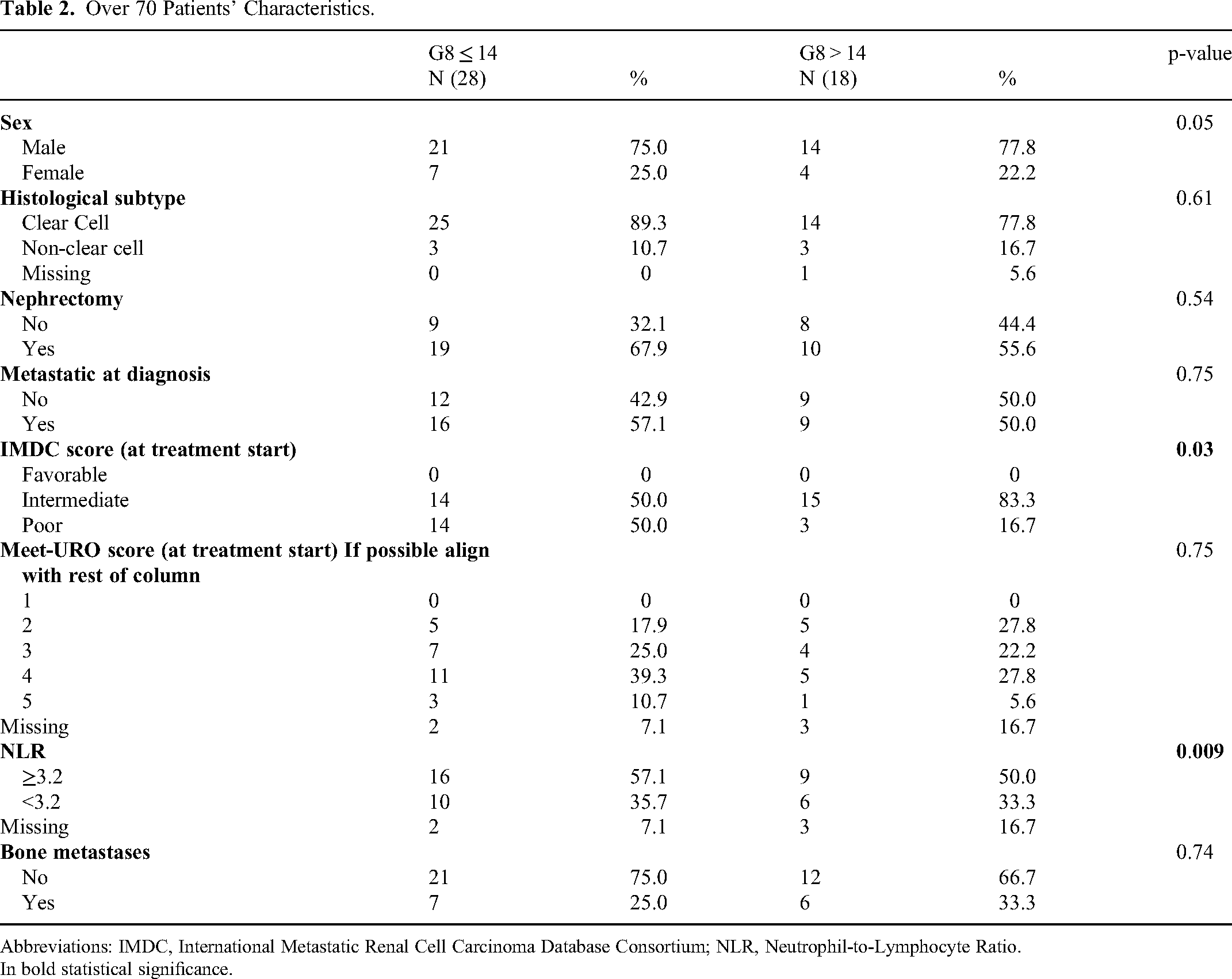
Abbreviations: IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; NLR, Neutrophil-to-Lymphocyte Ratio.
In bold statistical significance.
These findings suggest that patients with better G8 scores tend to have more favorable prognostic factors according to established risk classification systems and inflammatory markers.
Progression-Free Survival (PFS) and Overall Survival (OS)
The median follow-up was 16.8 months (95% CI 16.1-17.5) for the overall population, with no significant difference between the G8 ≤ 14 and >14 groups (p = 0.1).

^Statistical comparison by Log-rank between under and over G8 of 14.

Abbreviations: CI, confidence interval; H, high; L, low; OS, overall survival; PFS, Progression-Free Survival; yr, year.
In bold statistical significance.
For the entire cohort, the 1-year PFS was 34.8% (95% CI 29.4-41.1), and the 1-year OS was 63.3% (95% CI 57.8-69.4) (Figure 2A and 2B).

Progression-free and Overall Survival of the overall population (panels A and B) and according to G8 ≤ 14 (C and D).
When stratified by G8 score, there was no significant difference in PFS between patients with G8 ≤ 14 and >14 (1-year 29.3% vs 46.2%, p = 0.2) (Figure 2C). However, OS was significantly different, with patients having G8 ≤ 14 showing shorter survival compared to those with G8 > 14 (1-year 58.6% vs 76.1%, p = 0.006) (Figure 2D).
In the subgroup of patients aged ≥70 years, neither PFS nor OS showed significant differences according to the G8 score (p = 0.3 for both, data in figure 2 legend). Notably, the Meet-URO score classification was significantly prognostic for both PFS (p = 0.001) and OS (p = 0.001) in the overall population and for OS only (p = 0.02) in the over 70 group.
Multivariable Analysis
Table 3 presents the results of the univariable and multivariable analysis. For PFS, the independent prognostic factors were Meet-URO score overall, and groups 3, 4, and 5 (p < 0.001, and p = 0.003, p < 0.001 and p < 0.001, respectively) with a progressive hazard ratio (HR) from 1.86 for group 3 to 4.96 for group 5. For OS, alongside Meet-URO score overall, and groups 4 and 5 (p < 0.001, and p = 0.02 and p = 0.001, with HR of 3.80 and 8.24, respectively), the G8 score was confirmed as an independent prognostic factor (HR 2.36, 95% CI 1.06-5.08, p = 0.03).
Univariable and Multivariable Analysis of Progression-Free and Overall Survival.

Abbreviations: CI, confidence interval; H, high; L, low; OS, Overall Survival; PFS, Progression-Free Survival.
In bold statistical significance.
In the population over 70 years old (Table 4), the multivariable analysis revealed that non-clear cell histology (p = 0.003, HR 2.85) and Meet-URO score overall, and group 4 (p = 0.008, and p = 0.01, HR 2.82) were independent factors for PFS. For OS in this subgroup, nephrectomy (p = 0.02, HR 0.40) and Meet-URO score overall, and groups 4 and 5 (p = 0.005, and p = 0.03 and 0.04, HR 3.57 and 4.28, respectively) emerged as independent prognostic factors.
Univariable and Multivariable Analysis of Progression-Free and Overall Survival in Patients age ≥ 70.

Abbreviations: CI, confidence interval; H, high; L, low; OS, Overall Survival; PFS, Progression-Free Survival.
In bold statistical significance.
These results demonstrate the prognostic value of the G8 score for OS in the overall mRCC population receiving first-line ipilimumab-nivolumab, while the Meet-URO score shows consistent prognostic ability for both PFS and OS across different age groups.
Analysis on the Association Between Meet-URO Score and G8 Score
The Chi-squared test was conducted to assess the association between the Meet-URO score and the G8 score (graphically displayed as Sankey diagram in Figure 3). The test yielded a Chi-squared statistic of 7.69 with 3 degrees of freedom and a p-value of 0.05 suggesting a marginal association between the Meet-URO score and the G8 score.
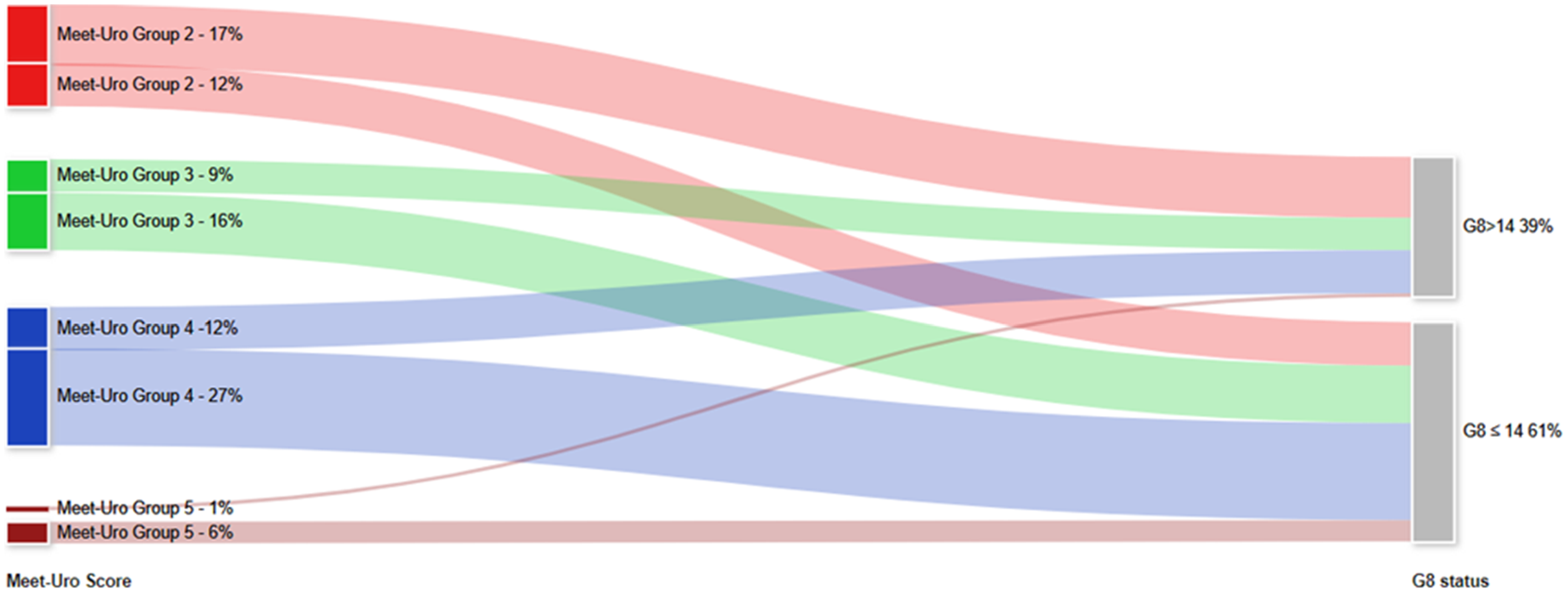
Sankey diagram demonstrating association between the Meet-URO and the G8 score.
The univariate Cox model for PFS, was not significant (likelihood ratio test p = 0.1), additionally no significant interaction was observed between Meet-URO Score and G8 Score suggesting no synergistic effect in PFS.
Regarding OS, the model was statistically significant as indicated by the likelihood ratio test p = 0.001. Within this model the interaction between Meet-URO and G8 score was not significant (Supplementary Table 1). However, a high Meet-URO score of 5 was strongly associated with an increased risk of death (HR = 31.50, p < 0.01), suggesting its importance as a prognostic factor for OS, with G8 Score having a limited role.
Variable Aggregation and Outcome Comparison in Patients with High G8 and High Meet-URO Score Versus Those with Low G8 and High Meet-URO Score
The Meet-URO score was aggregated into two levels: Low: Combines Groups 2 and 3; High: Combines Groups 4 and 5. Only patients in high level were selected for this analysis.
The high group was further stratified based on G8 scores (≤14 indicates frailty): Group > 14 (Meet-URO High, G8 > 14): n = 16; Group ≤ 14 (Meet-URO High, G8 ≤ 14): n = 36.
Patients with a G8 score ≤ 14 within the high Meet-URO score groups (4 and 5) exhibited the poorest outcomes in both PFS and OS although these differences were not statistically significant in PFS (p = 0.1) and of borderline significance in OS (p = 0.05) (Supplementary Figure 1 and Supplementary Table 2).
Discussion
Over the last four decades, the cornerstone of mRCC treatment has shifted progressively from cytokine therapy to targeted VEGF receptor TKIs and mTOR inhibitors and subsequently on to immune-based therapies. We have witnessed how ongoing advancements have increased survival and expanded the toolkit of the uro-oncologist. 7 As the treatment landscape continues to evolve, it is important to have prognostic tools that can help us effectively apply these treatment options either as monotherapy or as combination therapies in the clinical setting across varying demographics.
The goal of this analysis was to investigate the prognostic role of the G8 geriatric screening score within the context of the Meet-URO classification in mRCC patients receiving first-line ipilimumab and nivolumab. The multivariable analysis confirms that the G8 score is an independent prognostic factor for OS, though not for PFS in our study population. As has been previously demonstrated, the Meet-URO score appears to be predictive for both OS and PFS.
To our knowledge, this is the first study to evaluate the G8 geriatric screening score as a predictor of immunotherapy outcomes in older mRCC patients receiving the nivolumab-ipilimumab combination as first-line treatment. It also provides a head-to-head comparison of the G8 score and the Meet-URO score as prognostic factors in this disease and treatment setting. We chose to evaluate the G8 screening tool as there is a strong clinical need for prognostic models in the frail and older mRCC patient cohort.
In 2021, as part of the multicentre Meet-URO 15 study, the Meet-URO score was validated to carry prognostic value in the mRCC cohort receiving second and later lines of nivolumab. By incorporating bone metastases and NLR, the Meet-URO score further boosted the accuracy of the IMDC score. 8 Three years on, we compare the Meet-URO score to the G8 geriatric screening score in a similar disease context, but in mRCC patients receiving the first-line ICI combination nivolumab and ipilimumab.
Our results extend the work of Rebuzzi SE et al by demonstrating that G8 is an independent prognostic factor for OS and confirming our findings that the Meet-URO score has prognostic value for both OS and PFS in mRCC patients treated with first-line combination ICI therapies. 5 Furthermore, in mRCC patients receiving nivolumab, the Meet-URO score was far superior to the IMDC score as a predictor of both time to strategy failure and PFS. 9
The analyses performed by Araujo DV et al 10 established that there is no difference in OS, time to treatment failure or time to next treatment between the older or younger mRCC cohort treated with ICI. While the study by Domański et al 11 did not show any correlation between the G8 tool and OS or PFS in TKI-treated mRCC patients, our analysis identified the G8 geriatric screening tool as a prognostic factor for OS but not PFS in the ICI-treated cohort. It is important to note that Domański et al's study 11 investigated a geriatric mRCC cohort over 65 years of age whereas we evaluated patients 18 and above. Importantly, we also found that the G8 tool loses its prognostic value in the over 70 group in our sub-analysis. This may indeed be explained by the fact that 70 may not be an accurate threshold. Domański et al's study 11 also interestingly highlighted that lower pan-immune-inflammation (PIV) values and obesity are associated with longer OS in geriatric TKI-treated mRCC patients. Sustaining a geriatric perspective, a study by Damassi A et al in 2024 validated the Meet-URO score in older adults (aged 70 and above), receiving Cabozantinib. 12
Our findings support the use of G8, a screening score used to identify which patients require a comprehensive geriatric assessment (CGA), as a predictor of OS in mRCC patients treated with combination ICI treatment. Pierantoni et al validated the use of CGA to inform the clinician about OS and PFS in older mRCC patients above the age of 70 treated with first-line TKIs namely Sunitinib and Pazopanib. 13 Similarly, the Meet-URO prognostic score was validated to have superior accuracy to the IMDC score as part of a multicenter retrospective analysis evaluating mRCC patients treated with second-line Cabozantinib and beyond. 14
It is vital to continue our pursuit of clinically relevant biomarkers that can improve patient selection and hence inform the clinician of likely survival outcomes post initiation of immunotherapy. We would hence like to highlight other clinical characteristics that have been investigated in recent years. For instance, the work of Maruzzo M et al identified baseline thyroid hormone impairment, specifically a low fT3/fT4 ratio, as an independent prognostic factor for mRCC patients treated with systemic treatment. 15 Other significant blood markers include sodium levels and RBC-based scores (comprising haemoglobin, MCV and red cell distribution width).16,17 From a pathological standpoint, various immunohistochemical pattern differences have been identified between nivolumab-responsive and non-responsive patients. 18
The limitation of our study is that it is retrospective in nature however the strength lies in its multicentric design. We acknowledge a potential selection bias related to the availability of clinical information to calculate G8 scores. However, based on a comparative analysis of PFS and OS between the formally analysed cohort of patients where G8 scores were available and the excluded patients (without G8 scores), we did not find a significant difference in survival outcomes.
The lack of interaction between the Meet-URO and G8 scores supports their complementary roles in assessing different aspects of prognosis rather than overlapping measures and the primary prognostic role of the Meet-URO score. The Meet-URO score appears to contribute more to the risk prediction for PFS, where the G8 score contributes little to none. The Meet-URO score, particularly for groups 4 and 5, is a strong predictor of mortality, where G8 may complement the Meet-URO score by adding prognostic value related to patients’ frailty.
The Meet-URO score should be prioritized for risk stratification of mRCC patients receiving immunotherapy. The G8 score retains utility as a complementary tool for tailoring treatment in frail patients, providing insights into patient vulnerability that may influence clinical management.
Therefore, our study validates the Meet-URO score as an important prognostic factor for both OS and PFS in the mRCC cohort receiving ICI combination nivolumab-ipilimumab. These results hence cement the foundation for future studies to validate the G8 and Meet-URO scores in a larger patient cohort, other demographic groups, and nascent immune-based therapies. A meta-analysis can then potentially further boost the validation of the Meet-URO score in mRCC patients treated with the first-line nivolumab-ipilimumab combination.
The Meet-URO 33 study is ongoing and will help to further refine our conclusions, as analyses on the G8 score will be conducted on a larger scale and other prognostic factors for mRCC will also be explored as part of the study. It aims to provide further survival and toxicity data for the various different immuno-combinations. 19
Conclusion
Our analysis confirms that the Meet-URO classification can be used as a key prognostic tool to guide treatment decisions as it informs the clinician about PFS, demonstrating its superiority to the G8 geriatric screening score. However, the G8 score is useful at an individual patient level as a predictor of OS but not useful to guide patient or treatment selection in a clinical setting.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251316626 - Supplemental material for Prognostic Value of G8 Geriatric Screening and Meet-URO Scores in Metastatic Renal Cell Carcinoma Patients Receiving First-Line Ipilimumab-Nivolumab Combination Immunotherapy
Supplemental material, sj-docx-1-tct-10.1177_15330338251316626 for Prognostic Value of G8 Geriatric Screening and Meet-URO Scores in Metastatic Renal Cell Carcinoma Patients Receiving First-Line Ipilimumab-Nivolumab Combination Immunotherapy by Ria Nagpal, MB, BCh, BAO, Marina Campione, PhD, Sara Elena Rebuzzi, MD, Lucia Fratino, MD, Pasquale Rescigno, MD, Sergio Bracarda, MD, Davide Bimbatti, MD, Ugo De Giorgi, MD, Matteo Santoni, MD, Fabio Calabrò, MD, Mimma Rizzo, MD, Alessio Signori, PhD, Diana Giannarelli, PhD, Giuseppe Fornarini, MD, Umberto Basso, MD, and Giuseppe Luigi Banna, MD in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
SER and GF would like to thank the Italian Ministry of Health (Ricerca Corrente 2018-2021 grants) that financially support their current research focused on identifying prognostic and predictive markers for patients with genitourinary tumors. All authors would like to thank the Italian Network for Research in Urologic-Oncology (Meet-URO).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SER received honoraria as a speaker at scientific events and travel accommodation from Amgen, GSK, BMS, MSD. DB received honoraria as speaker or advisory/board member from Ipsen, Astellas, Janssen, Novartis, MSD, Pfizer, Merck, Gentili and travel/accommodations/expenses from Ipsen, Janssen, MSD, Merck, Astrazeneca, Novartis. GLB reported personal fees from Astellas, Astrazeneca, Amgen, Bayer, Merck, Pfizer. UDG, financial interests, personal, advisory board: Pfizer, BMS, MSD, PharmaMar, Astellas, Bayer, Ipsen, Novartis, EISAI, Janssen; financial interests, personal, invited speaker: Roche, BMS, Clovis Oncology, AstraZeneca; financial interests, institutional, research grant: AstraZeneca, Sanofi, Roche. FC has received consulting fees from Merck Sharp & Dohme Oncology and Pfizer. MR received honoraria as a speaker/consultant by MSD, Bristol-Myers Squibb, Astrazeneca, Johnson & Johnson, Eisai, Gilead, Ipsen, Merck Serono and congress travel accommodation from Merck Serono, MSD, Johnson & Johnson, Ipsen. PR served as an advisory board for MSD and Astra Zeneca Italy. GF served as an advisory board member for Astellas, Janssen, Pfizer, Bayer, MSD, Merck and received travel accommodation from Astellas, Janssen, Bayer. DG served as an advisory board for Sanofi and received consulting fee from Amgen. All other authors have declared no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
The EAP (Expanded Access Program) involved 86 centees in Italy between April and October 2019. The study was carried out under the Declaration of Helsinki, Good Clinical Practice and local ethical and legal regulations. The Regional Ethical Committee approved the analysis
Ethical Approval statement
The original study is reported here: Basso, U.; Paolieri, F.; Rizzo, M.; De Giorgi, U.; Bracarda, S.; Antonuzzo, L.; Atzori, F.; Cartenì, G.; Procopio, G.; Fratino, L.; et al Compassionate Use Program of Ipilimumab and Nivolumab in Intermediate or Poor Risk Metastatic Renal Cell Carcinoma: A Large Multicenter Italian Study. Cancers 2022, 14, 2293. https://doi.org/10.3390/cancers14092293. This was a retrospective, non-interventional study evaluating the clinical outcomes of ipiliumab-nivolumab treatment in patients with metastatic renal cell carcinoma (mRCC). Ipilimumab and Nivolumab were provided by Bristol-Myers Squibb (BMS) upon physician request, following approval by local Ethics Committees and in accordance with Italian compassionate use regulations (Ministry of Health Decree, 7 October 2017). All patients provided informed consent and signed privacy management forms prior to starting treatment. The retrospective multicenter analysis received approval from the
Consent Statement
Patients consented to the Italian EAP, which included first-line nivolumab plus ipilimumab and for the related analyses.
Data Availability Statement
Data used for this analysis are available upon justified request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
