Abstract
Background and Aim
Predictors of neutrophil-to-lymphocyte ratio (NLR) and traditional clinical variables for hepatocellular carcinoma (HCC) prognosis after locoregional therapies were useful while exhibited modest prognostic performances. We dig out the potential of circulating immune features for HCC prognosis prediction.
Methods
244 patients with early-stage HCC who were treated with thermal ablation and performed the peripheral blood mononuclear cells (PBMCs) tests were included. Patients were randomly assigned in 3:1 ratio to discovery (n = 183) and validation (n = 62) sets. Three models, including clinical (Clin-model), NLR-Clin-model and Immune-NLR-Clin-model were constructed using Cox regression model. Concordance index (c-index), integrated discrimination improvement (IDI) and net reclassification improvement (NRI) were used for performance evaluation.
Results
The Immune-NLR-Clin-model exhibited the best performance of 0.706 (95% CI:0.644-0.768) and 0.702 (95% CI:0.566-0.837) in discovery and validation sets, respectively. At 36-month prediction, the IDI and continuous-NRI show trend of improvement, with the IDI was 0.050 (95%CI: −0.5%-12.5%) (
Conclusions
Circulating immune features may be helpful components aiding NLR for HCC predictive models.
Introduction
Primary liver cancer, including hepatocellular carcinoma (HCC), is the fourth most common cause of cancer-related death worldwide. 1 For tumor location and liver function as two important factors on the choice of treatment methods, a large number of HCC patients are not eligible for liver resection and transplantation, various locoregional treatments, like imaging-guided thermal ablation, has been widely used in clinics. 2 With the extensively use of thermal ablation, a pressing need to develop refined prognostic and predictive biomarkers to assist in the personalized prognostic indications. 3 Day-to-day clinical decisions are based on traditional clinical parameters, such as AFP, 4 tumor number 5 and tumor diameter, 6 which underlined the morphology information of HCC while may be insufficient in HCC intrinsic maladjustment.
The chronic and enhancing inflammation induced by the maladaptive immune responses are the “chief culprit” for HCC progression. 7 Myeloid-derived suppressor cells (MDSCs), like neutrophils, 8 modulated tumor microenvironment via multiple direct and indirect manners, including promoting cellular transformation and tumor metastasis, releasing immunomodulatory cytokines to recruit immunosuppressive cells such as regulatory T-cells (Tregs) 9 and macrophages (Mφ). 10 Consistent with this, the circulating neutrophil count and NLR,11,12 indicators of cancer-related inflammation, has long been observed and suggested as both a potential prognostic and therapeutic response markers in tumor patients. 13 Indeed, the circulating NLR is a simple and easy-accessible prognosis index, while the stratification capabilities by NLR alone or combined with some traditional clinical parameters were still insufficiently sensitive.14,15 Other immune indicators which directly modulation of tumor microenvironment may boost enhanced prognosis stratification.
Apart from neutrophils, CD4+CD25+Tregs as an immunosuppressive subset of CD4+ T cells, impair the tumor immunosurveillance in HCC patients.16,17 It is thought that Treg cells may decrease the proliferation of effector CD4+ and CD8+ T lymphocytes by contact inhibition and TGF-β secretion,18,19 subsequently reducing the anti-tumor immune response and resulting in the risk for tumor cells to escape immune surveillance. Therefore, increased infiltration of Tregs into the tumor tissues of patients with HCC has long been observed and suggested as a potential prognostic marker in HCC with multiple causes, like HBV, HCV and non-alcoholic fatty liver disease.20–22 These studies suggested the balance between Tregs and effector T cells as well as neutrophils in tumor microenvironment may be important determinants in HCC progression. However, given the frequent non-invasive diagnosis of HCC 2 and the potential risk of tissue acquisition, 23 histology is not often available before treatment, thus rendering tumor microenvironment evaluation difficult to implement in clinics. In this situation, whether the circulating immune features from PBMCs, incorporated some immune-enhancing and suppressive subsets, could be a reliable and easily-applicable prognostic biomarker is worth exploring.
In this study, we carried out a retrospective analysis identifying potential candidate biomarkers of circulating immune factors for improving prognosis prediction of patients with HCC undergoing curative thermal ablation.
Material and Methods
Patient Cohorts and Baseline Characteristics Collection
We retrospectively searched our department database who performed the PBMCs test 3-5 days prior to thermal ablation consecutively between January 2009 to November 2020. We performed and analyzed this study according to STROBE. 24 Inclusion criteria were as follows: (a) primary HCC which diagnosis confirmed by biopsy or clinical criteria, which was established by one imaging technique (MR or CT) in nodules above 2 cm showing the HCC radiological characteristic and the two coincidental techniques (MRI + CT) with nodules of 1-2 cm in diameter; (b) single nodule ≤5 cm in maximum diameter; (c) ≤3 HCC nodules and each maximum diameter ≤3 cm; (d) ≤2 HCC nodules if each maximum diameter ranging in 3-5 cm;(e) pre-ablation routine blood test results were preserved; (f) Child-Pugh A or B; (g) no portal venous and bile duct tumor embolus and extrahepatic metastasis. Exclusion criteria were listed as: (a) extrahepatic metastasis or main artery/vein invasion; (b) liver function in Child-pugh C class; (c) patients with recurrent HCC; (d) Tumor size >5.0 cm, >3.0 cm and tumor number >3; (e) pre-ablation blood test missing; (f) loss to follow-up; (g) incomplete ablation; (h) receiving liver transplantation after ablation. A total of 244 patients were enrolled and were randomly divided into 3:1 ratio in discovery (n = 183) and validation sets (n = 61) for subsequent analysis (Figure 1).

Study design. PBMCs, peripheral blood mononuclear cells; NLR, neutrophil lymphocyte ratio; TREDN score: Log10(Treg)/Log10(CD8+) and NLR from the immune-related combined model were integrated as a score, reordered as TREND.
We collected details on demographic information, tumor characteristics, liver function parameters prior to thermal ablation from medical records in our institutional database. All the patients were percutaneously treated by a cooled-shaft microwave system (KY-2000, Kangyou Medical, China) or radiofrequency system (WB991029, CelonLab Power, Germany). This study complied with the standards of the Helsinki Declaration and was approved by the hospital Research Ethics Committee, and informed consent was obtained from all patients.
Flow Cytometry and Peripheral Immune Subsets Definition
In order to depict the peripheral immune status in more representative ways, we assessed and integrated 15 classic immune subsets. CD3+ is common T cell markers, 25 CD4+ and CD8+ are used to mark helper T cell and cytotoxic T cell,26,27 respectively. In human, naive cells are held to be CD4+CD45RA+CD45RO−, and memory T cells to be CD4+CD45RA− CD45RO+. 28 The CD28+ was established as a marker of T cell co-stimulation, it was suggested that CD28 engagement by B7-1 could enhance T cell proliferation and interleukin-2 (IL-2) production. 29 CD4+ and CD25+ were Treg cells’ markers. 30 NK were marked as CD3−CD16+CD56+, 31 and NKT were marked as CD3+CD16+CD56+. 32 The flowcytometry strategy (Supplementary Figure S2) and antibodies panels (Supplementary Table S2) used in flow cytometry analysis were described. Samples were obtained using a flow cytometer FC500-MPL (Beckmam Coulter, Inc.), and data analysis was performed using FlowJo software (Tree Star, Inc., Ashland, OR, USA).
Follow-up Protocol
Within one month after thermal ablation, patients underwent contrast-enhanced US or contrast-enhanced MRI/CT to guarantee tumor vitality zone was completely destroyed. During the follow-up period, liver function tests, serum AFP examinations, abdominal ultrasound, contrast-enhanced US/MRI/CT imaging scans were performed once every 3 to 6 months or earlier if clinically indicated. In cases with suspected distant metastasis, chest CT, whole-body bone scans, or positron emission tomography (PET-CT) were performed selectively. The primary endpoint of this study was overall survival (OS), which was defined as the time from day of thermal ablation until death, and patients who were still alive were censored at the date of last contact. The second endpoints were recurrence free survival (RFS) and early recurrence (ER). RFS was time from the date of thermal ablation to either recurrent disease or death from any cause, whichever occurred first. ER was defined as the interval between the date of thermal ablation and the first date of tumor recurrence on imaging within 2 years.
Statistical Analysis
Chi square test for categorical data and the two-sample t-test for continuous variables. Patients were divided into discovery and validation cohorts at 3:1 ratio via random number. For clinical variables, univariable and multivariable analyses were conducted with Cox regression models. In order to evaluate the useful of predictors extensively, we relaxed the inclusion criteria that clinical variables which achieved statistically significant (
Model performances were evaluated by the c-index. We also performed time dependent area under the receiver-operating characteristic curve analyses and reported the corresponding AUC values at 12, 24, 36, 48 and 60 months. IDI, net NRI and R square (Rsq) were used for evaluation of model improvements via reclassification analyses. Two new metrics, the IDI and NRI, have been adopted to quantify the added value of a biomarker to an existing test. These results were validated in validation set. Statistical tests were performed with Empower Statas software (version 4.1), R Foundation for Statistical Computing (R version 4.2.0), SPSS Statistics 25 (IBM) and GraphPad Prism 9.0 (GraphPad Software).
Results
Patient Characteristics
Overall, 183 patients were analyzed in discovery set and 61 were in validation set. Patients in these two sets with balanced clinical parameters (Table 1) and circulating immune characteristics (Supplementary Table S1). In discovery set, 78.69% patients were in BCLC 0-A stage and 95.63% patients were in Child-Pugh class A. The median NLR was1.69 (IQR:1.31-2.19). In validation set, 77.05% patients were in BCLC 0-A stage and the median NLR was1.70 (IQR:1.45-2.28). Other clinical characteristics at baseline were summarized in (Table 1). This study was censored on September 30, 2022, with median duration of estimated follow-up was 45.10 (95%CI: 40.13-50.68) months in the discovery set, and 46.74 (95%CI: 38.10-57.32) months in validation set.
Patient and Tumor Characteristics.

NLR: Neutrophil to lymphocyte ratio; BCLC: Barcelona Clinic Liver Cancer stage.
Traditional Clinical Parameters and the Inflammation index NLR Predict OS in Patients with HCC
Firstly, we evaluated the prognostic performances of clinical and demographic characteristics. Univariate analyses revealed that NLR (≥1.40 vs <1.40), BCLC stage (B vs 0-A) and tumor number (2-3 vs 1) exhibited statistically association with OS (Table 2) in discovery cohort. In multivariate analyses, NLR (≥1.4 vs <1.4), BCLC stage (B vs 0-A) remained independently association with OS. Besides, as tumor number (2-3 vs 1) was a widely accepted prognostic factor, we included it in the final model. Notably, as the HR of NLR was comparably higher than other variables, which indicated the distinct prognosis significance of NLR in HCC patients.
Cox Proportional Hazard Regression Analyses in Discovery Set.
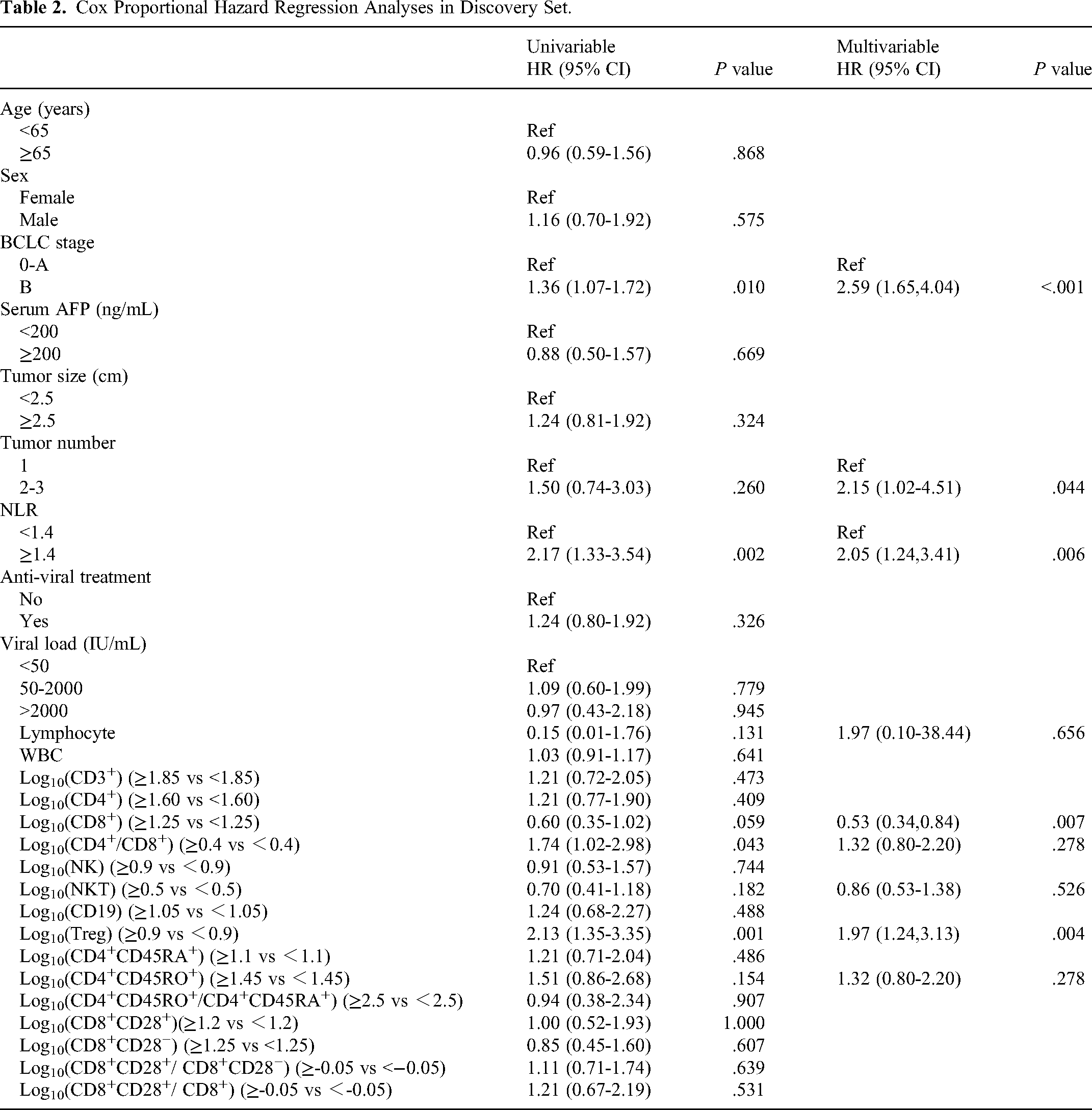
Prognostic value of these traditional factors was evaluated by c-index. Results showed that the c-index of traditional clinical factors (BCLC stage and tumor number) were 0.598 (95%CI:0.543-0.654) and 0.666(95%CI:0.548-0.785) in discovery and validation sets, respectively. When NLR was combined with traditional factors, the c-index were enhanced to 0.649 (95%CI:0.587-0.710) and 0.673(95%CI:0.539-0.801) in discovery and validation sets, respectively.
The Circulating Immune-Indexes Predict OS in Patients with HCC
We next screened and evaluated the potential prognostic values of circulating immune indexes. Firstly, we performed a fitting curve analysis of 15 representative immune subsets using the restrictive cubic spline function. Restricted cubic spline analyses also supported the cut-offs of immune features (Supplementary Figure S1). The discriminatory power of immune features for OS were validated by Cox proportional hazard regression analyses. Among these, the Log10(CD8+) ≥ 1.25 versus <1.25 (
After the collinearity test (Supplementary Table S3), we added immune indexes to the NLR-Clin model, and the c-index increased to 0.706 (95%CI:0.644-0.768) in discovery set and to 0.702 (95%CI:0.566-0.837) in validation set, significantly higher compared with the c-indexes of NLR-Clin model in both discovery and validation sets (Table 3). Using the time-dependent AUCs, discrimination of combined model in the training set were 0.741, 0.706, 0.689, 0.701 and 0.679 at 12, 24, 36, 48 and 60 months, and were 0.712, 0.787, 0.779, 0.749 and 0.788 in validation cohort.
Prognostic Performance of the Models.

Compared the performance between Clin-model and NLR-Clin-model.
Compared the performance between NLR + Clin and Log10Treg/Log10CD8++NLR + Clin.
At 36 month's prediction, the Rsq were 0.208 and 0.130 in Immune-NLR-Clin model and NLR-Clin model respectively in discovery cohort. The IDI and continuous-NRI show trend of improvement, with the IDI was 0.050 (95%CI:-0.005, 0.125) (
Construction of two Immune-Related Indexes as an Easily Applicable TREND Score
As an immune suppressive subset, the Tregs inhibited the effector CD8+ T cells, and the Treg/Teff ratio has been identified as a major contributor to disease progression. Then, we investigated the correlation of Log10(CD8+) and Log10(Treg). Pearson's test indicated that Log10(CD8+) and Log10(Treg) were in negative correlation, with the correlation coefficient = -0.172 (

Construction of TREND score and its prognostic stratification in discovery and validation sets. Construction of TREND score in discovery set and its prognostic stratification in both two sets. (a) Correlation analysis of Log10(Treg) and Log10(CD8+). (b) Combined the Log10(Treg)/Log10(CD8+) and NLR as TREND score and the detailed criterion in TREND score subgroups. (c) Overall survival according to TREND stratification in the discovery set. (d) Overall survival according to TREND score in the validation set.
Apart from the differences of OS, the median RFS of patients with TREND-0/1 and TREND-2 were 36.00 (95%CI 28.00-52.47), 24.40 (95%CI 18.43-33.93) (

Recurrence-free survival (RFS) and early recurrence (ER) rate in different TREND subgroups in discovery and validation sets. (a-b) Patients who were in TREND-high risk subgroup showed decreased RFS compared with patients who were in TREND-low risk subgroup in discovery (P = .019) and validation cohort (P = .095). (c-d) The ER in different subgroups in validation (P = .294) and validation sets (P = .255).
The TREND Score Predicts Outcome in Different Subgroups
To explore the prognostic value of TREND index in different subgroups, we pooled the discovery and validation sets for exploratory analyses. The median OS were different in TREND score strata in subgroups of BCLC stage, treatment history, AFP and age. Details of the median OS were shown (Figure 4).

Hazard ratios (HR) for death of TREND stratifications in different subgroups in the pooled set. OS was compared by Kaplan-Meier method, and HR was analyzed with univariable Cox regression.
Discussion
The key finding from our study is that the levels of pre-existing circulating immune features appear to show association with outcome of HCC patients undergoing thermal ablation. Specifically, when we combined the Log10(Treg)/Log10(CD8+) with clinical variables and NLR, the Immune-NLR-Clin-model reached the c-index of 0.706 in discovery set and 0.702 in validation set, which realized improvement compared with NLR-Clin-model. For characterizing a more reliable circulating immune contexture, we combined the NLR and Log10(Treg)/Log10(CD8+) to construct the easily-applicable TREND score. Accordingly, in contrast to patients with an NLR of 1.4 or higher and a Log10(Treg)/Log10(CD8+) of 0.70 or higher, patients who fulfilled none of these criteria and those who fulfilled only 1 criterion still had a good outcome. These results were confirmed in an independent validation set.
As a mediator of innate inflammatory response, neutrophil makes up a considerable proportion of the immune infiltration in both chronic liver disease and HCC.33,34 High neutrophil counts were independently associated with poor overall survival and more-advanced disease. 35 Therefore, the NLR was a widely-accepted peripheral prognostic index in HCC patients undergoing liver resection or multiple local treatments.35,36 While in previous studies, the NLR alone or combined with other traditional clinical parameters just showed modest prediction accuracy, which partially hampered its clinical application. Further, for those tumor patients which were in early stage, the level of NLR may not as high as those in advanced stage,37‐39 their prognostic indications and may be overlooked. Previous studies have indicated that neutrophils inhibit effector T cells and NK cells proliferation and cytotoxicity through release of multiple metabolites and cytokines.9,40 Besides, neutrophils can stimulate Tregs expansion, which resulted in enhanced tumor immune escape and invasiveness. Therefore, the main mechanistic role of neutrophils in HCC development and progression was the correlation with adaptive immune cells for influencing tumor immune microenvironment.9,41 Treg cells as one of the essential immune homeostasis regulators, dampened the anti-tumor immunity via multiple contact-dependent 42 and contact-independent immune suppressive mechanisms. 16 In addition, previous studies reported the frequency of circulating Tregs was linked to the disease progression and had a potential of serving as a significant biomarker in HCC patients.21,22 Previous studies have reported that the Treg/Teff ratio in situ TME has been identified as a major contributor to disease progression and immunosuppression in HCC and many other tumor types.21,22 In our study, we tried to develop a new index which could supplement the role of NLR ≥ 1.4 as a inclusion criterion for poor prognostic patients, we found that the Treg and CD8+ immune cells could synergize the NLR index and show improved discriminative power. We identified that a dual-positive NLR ≥ 1.4 and Log10(Treg)/Log10(CD8+) ≥ 0.70 represented 28.42% of the cohort with median OS of 62.0 month. The synergizing of Log10(Treg)/Log10(CD8+) not only allow discrimination of more high-risk patients but also indicate that the lower level of NLR were not in absolutely low-risk. Previous studies highlighted NLR ≥ 2.5 or ≥3.0 associating worse OS and RFS,38,39 while in this study we put that the NLR level of the initial HCC patients in early-stage may not as high as those in advanced-stage, and those patients of NLR ≥ 1.4 still have the potential risk of poor survival after thermal ablation, especially when Log10(Treg)/Log10(CD8+) ≥ 0.70 at the seam time. From this point, compared with NLR alone, characterization of NLR as well as Tregs and CD8+ from peripheral blood not only describing a more comprehensive immune contexture in a minimally invasive method, but also realizing an enhanced prediction accuracy.
In the past 20 years, image-guided thermal ablation technique has evolved considerably and been established as a reliable treatment option for HCC patients. 43 For initial HCC in early stage, despite multiple curative treatment techniques have been widely accepted, distinct prognosis still been a great challenge. As for the locoregional therapy, which has been shown to induce immune responses in patients with HCC, exploring the immune-related features for post-ablation prognosis prediction may be more characterized and valuable. On the one hand, it was found that a higher number of antigen specific CD8+ T cells after radiofrequency ablation correlated significantly with the length of HCC recurrence free survival.44,45 It was also suggested that ablation therapy alters the T-cell balance by increasing the systemic CTL/Treg ratio. 46 Additionally, some studies indicated the local ablation may trigger some immune-suppressive subsets, like myeloid-derived suppressor cells (PMN-MDSCs), negatively affected CD8+ T cell infiltration.47,48 Therefore, the ablation-induced immune responses were complex for patients’ prognosis. On the other hand, some studies indicated the pre-ablation inflammatory and immune status influenced the efficacy of thermal ablation. 49 Recently, some studies also suggested that the thermal ablation may not be the optimal treatment for some patients and the pre-treatment semantic information could be used for the treatment selection, which emphasize the pre-treatment traits could influence the prognosis of patients.50,51 Therefore, the pre-treatment clinical and immune characteristics could provide more timely and advanced information for patients’ treatment decisions. In our study, patients who were divided in TREND-high risk have poor survival, with median OS was 62.0 month, accordingly the pre-therapy systemic immune contexture indicated the different prognosis of patients, treatment for reversing the imbalanced systemic immune contexture may be needed in their treatment process.
Our studies have some limitations: firstly, we didn’t analyze and compared other circulating inflammation scores, such as PLR, SII. For this point, Young. et al found out for patients with HCC undergoing TACE, NLR has a higher association with OS than either PLR, ALRI, or SII, and the lymphocyte count may be most predictive of OS. 52 These results were consistent with our study and also indicated the importance of systemic lymphocytes and NLR in overall survival prediction of HCC patients; secondly, for the reason that PBMCs test has not been established in mostly ablation treatment centers, we failed to acquire data from external validation cohorts, but to some extent, the present results confirmed the value of this readily-convenient test in an internal validation cohort, further prospective studies were needed to validated the results in different cohorts to confirm its applicability. Thirdly, undoubtedly, the whole distribution of immune cell subsets, to some extent, was of differences between PBMCs and liver, while both PBMCs and liver tissue contain similar types of immune cells and many immune cells retain their functionality in both locations. Our study aimed to evaluate the main distribution of immune cell subsets, thus could be monitored via the blood tests mini-invasively. If we focused on liver's distinct immunological environment of its composition, activation states, and functional responses, which should be assessed via liver biopsy. Besides, all candidates we included were patients with initial HCC, while they were often treated in real-life practice after the first ablation therapy and it is difficult to assess the potential biases and heterogeneity in the subsequent therapies and the impart on the primary endpoint OS. Finally, in validation cohort the OS and RFS differences did not reach the P value < .05 while approached significant, which may largely be limited by the sample sizes.
Conclusion
We developed a validated score combining systemic NLR and Log10(Treg)/Log10(CD8+) – both known indicating immune suppressive contexture – that predicts outcome of patients undergoing thermal ablation for HCC. Since the TREND score is based on 2 available circulating features, it's easy to perform in clinics. The score may aid in traditional clinical parameters for better prognosis prediction and support decision-making in daily clinical practice. The score needs prospective validation in a large clinical cohort to further test its clinical usefulness.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241309402 - Supplemental material for Circulating Immune Features Synergizing Neutrophil-to-Lymphocyte Ratio in Prediction of Poor Survival of Early-Stage Hepatocellular Carcinoma After Thermal Ablation
Supplemental material, sj-docx-1-tct-10.1177_15330338241309402 for Circulating Immune Features Synergizing Neutrophil-to-Lymphocyte Ratio in Prediction of Poor Survival of Early-Stage Hepatocellular Carcinoma After Thermal Ablation by Fangying Fan, Guoping Dong, Chuanhui Han, Yanchun Luo, Xin Li, Xuanjuan Dong, Zhen Wang, Ping Liang and Jie Yu in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338241309402 - Supplemental material for Circulating Immune Features Synergizing Neutrophil-to-Lymphocyte Ratio in Prediction of Poor Survival of Early-Stage Hepatocellular Carcinoma After Thermal Ablation
Supplemental material, sj-docx-2-tct-10.1177_15330338241309402 for Circulating Immune Features Synergizing Neutrophil-to-Lymphocyte Ratio in Prediction of Poor Survival of Early-Stage Hepatocellular Carcinoma After Thermal Ablation by Fangying Fan, Guoping Dong, Chuanhui Han, Yanchun Luo, Xin Li, Xuanjuan Dong, Zhen Wang, Ping Liang and Jie Yu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgement
We thank the site investigators, study coordinators and patients who participated in the trial.
Author Contributions
Fangying Fan performed conceptualization, data curation, formal analysis, methodology, writing-original draft, writing-review and editing. Guoping Dong performed investigation and methodology. Chuanhui Han performed writing-review and editing. Yanchun Luo performed Methodology. Xin Li, Xuejuan Dong and Zhen Wang were responsible for investigation.Ping Liang was responsible for project administration. Jie Yu performed conceptualization as well as writing-review and editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This research followed the guidelines of the Declaration of Helsinki and was approved by the Chinese PLA General Hospital Research Ethics Committee. The approved protocol number in the Human Subjects section is S2017-045. All patients were required to sign written informed consent before blood specimen collection.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was not supported by any funding.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
