Abstract
Keywords
Introduction
Breast cancer is the most prevalent form of malignant cancer among women globally.1 An estimated 2.3 million new cases were reported worldwide in 2023.1 Advancements in screening technologies and improved accessibility have facilitated the detection of breast cancer at earlier stages. In the United States, approximately 63% of diagnosed patients had tumors smaller than 2 cm.2
Numerous studies are exploring treatments that increase cure rates, reduce toxicity, and are less invasive while maintaining equivalent efficacy. As such, percutaneous ablative procedures have garnered significant interest. Among the primary methods under investigation, ranked by the number of publications, are radiofrequency ablation (RFA), cryoablation, percutaneous interstitial laser ablation (PLA), high-intensity focused ultrasound, and microwave ablation.3 Previous studies on the ablation of early-stage breast cancer have shown promising results, especially with the use of RFA, PLA, and cryoablation. Since these methods can be applied to other types of tumors, equipment with these technologies is available in specialized hospitals around the world. Therefore, refining techniques for using these ablative methods could broaden their indications and allow treatment for a greater number of patients. This review primarily focuses on PLA, as this method has shown promising results in isolated studies, albeit with significant discrepancies and a lack of standardization.4–8
Laser interstitial photocoagulation for solid organ tumors, including hepatic nodules, was initially reported in 1983 by Bown S.G. as a minimally invasive technique providing localized destruction.9 Since the first reports of this method for the treatment of breast cancer in 1989, nearly 310 women have undergone laser ablation. However, the results obtained from these procedures exhibit significant diversity.3,10–12
This review aimed to assess the studies published to date and identify appropriate methodological and patient selection parameters to optimize results with laser ablation. Additionally, we aimed to analyze whether this method is a viable alternative to current surgical treatments.
Methods
As a systematic review article, an ethics committee approval is not required for this research. The study protocol was registered in International Platform of Registered Systematic Review and Meta-Analysis Protocols-INPLASY (registration number: INPLASY2024100045)
A PROSPERO search was performed and no review studies on laser ablation for patients with breast cancer were found. The 2020 PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) method was employed.13 The terms
The inclusion criteria comprised original articles utilizing laser as an ablation method for local breast cancer treatment, irrespective of its association with other methods or whether subsequent surgery was conducted. Review articles, editorials, letters, and non-human studies (eg,
All articles were selected by a single author (DRMM), and the selections were subsequently validated by the second author (GF). Data were collected and tabulated based on several topics of interest, including year of publication, number of patients included, type of device used, and technical parameters such as power and energy. Additionally, the imaging method used to guide ablation and evaluate the area of necrosis and any residual lesions, ablation rates, and the necessity for surgery following ablation were recorded.
The data were compiled and described in terms of proportions or intervals. Whenever feasible, averages were calculated based on descriptions provided in the texts or tables within the articles.
Results
The search yielded 1778 and 739 articles in the Pubmed and Embase platforms, respectively. During the review of the selected articles, 10 studies not previously identified by the search platforms were actively found through references and included for analysis. Among the 2527 total articles, 56 were excluded due to duplication. On reviewing the title and abstract, 2429 studies were excluded because they did not meet the inclusion criteria. Among the 42 articles reviewed, 25 were excluded because they referred only to technical or pathological aspects or image analysis (10 articles); did not correspond to the technique studied (7 articles); or were conducted on cadaver, animal, or
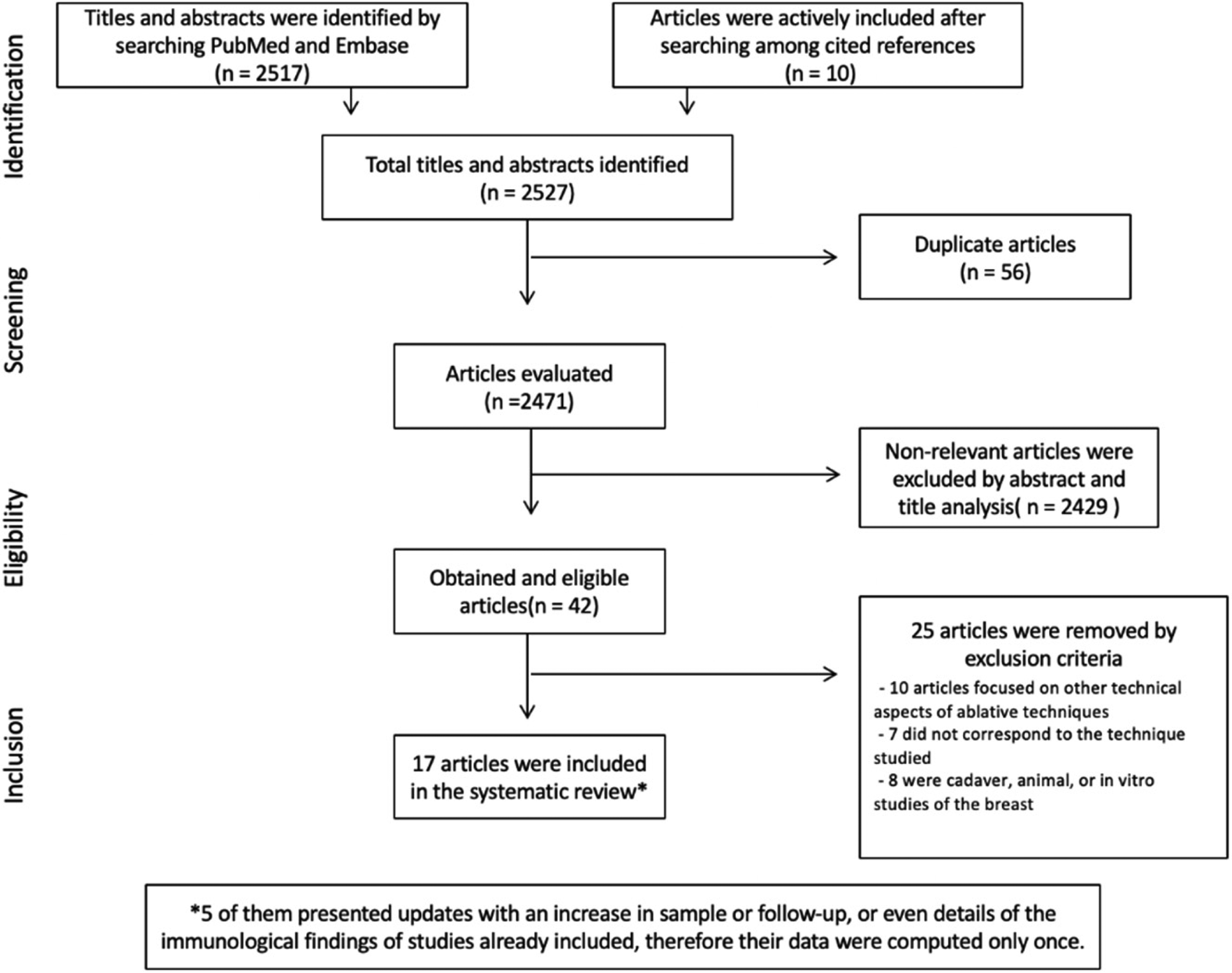
Selection of items included.
Findings of Studies That Used PLA for the Treatment of Breast Cancer
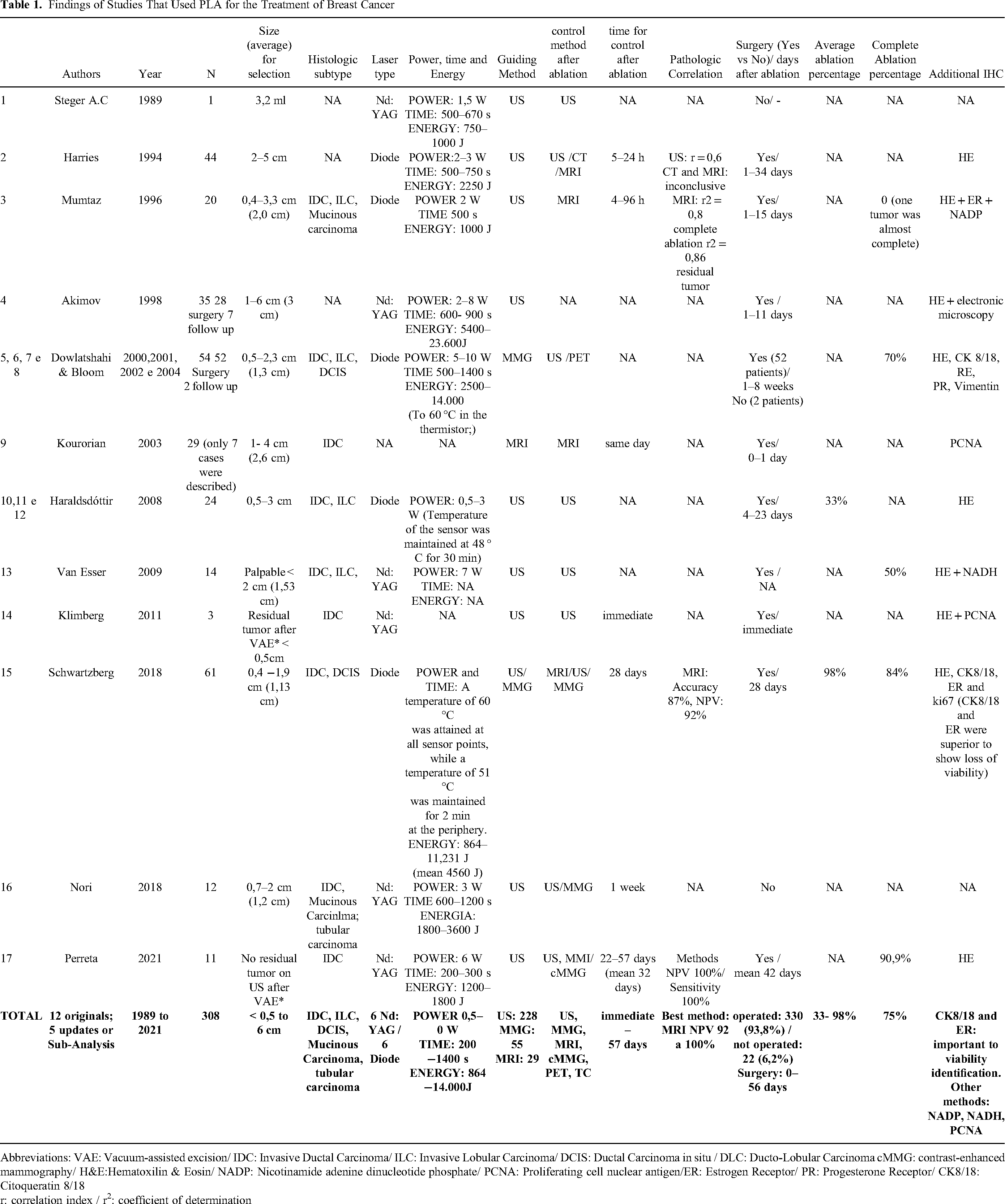
Abbreviations: VAE: Vacuum-assisted excision/ IDC: Invasive Ductal Carcinoma/ ILC: Invasive Lobular Carcinoma/ DCIS: Ductal Carcinoma in situ / DLC: Ducto-Lobular Carcinoma cMMG: contrast-enhanced mammography/ H&E:Hematoxilin & Eosin/ NADP: Nicotinamide adenine dinucleotide phosphate/ PCNA: Proliferating cell nuclear antigen/ER: Estrogen Receptor/ PR: Progesterone Receptor/ CK8/18: Citoqueratin 8/18
r: correlation index / r2: coefficient of determination
The first report of PLA for a patient with breast cancer dates was an anecdotal study by Steger in 1989.14 Despite the lack of efficacy, the study is the first to describe the technique. It also mentions the importance of defining parameters for patient selection and imaging techniques that can predict the response to ablation.14
The first comprehensive study focused on the effects of PLA on breast cancer patients involved 44 women and was conducted in 1994 by Harries et al15 A diode laser was used, with power ranging from 2 to 3 W for 500 to 750 s, resulting in energy levels between 1000 and 2250 J per nodule. Ultrasonography (US) was used as the primary imaging modality for guiding the ablation procedure, with evaluation of the necrotic area conducted via US, computed tomography (CT), and/or magnetic resonance imaging (MRI). The study's key observations pertain to the anatomopathological outcomes of the ablated region and the challenges posed by US in accurately predicting the initial tumor volume. Concerning necrotic findings, the authors observed that central cauterizing of the tumor correlates with a more extensive peripheral necrotic area. To improve the central carbonization rate of the tumor, a “pre-carbonization” of the fiber tip was conducted in 18 patients, resulting in a more predictable and expansive necrotic area. This may be ascribed to the initiation of tumor ablation at higher temperatures.15,16
Considering existing data regarding the inability of US to effectively estimate the extent of the necrosis area, Mumtaz et al published their study using MRI after ablation for improved evaluation of the tumor necrosis area in 1996.17 Twenty patients underwent US-guided PLA using a diode laser device with fibers also subjected to pre-carbonization. Surgical resection occurred between 1 and 15 days following the ablative procedure. Using linear regression analysis (r2), the authors reported that MRI presented an adequate correlation in estimating both the area of necrosis (r2= 0.8) and the residual tumor (r2 = 0.86).17
In 1998, Akimov et al published the data of their study, which was divided into three phases: preclinical phase (
Dowlatshahi and Bloom et al assessed technical and pathological aspects of the application of PLA on 54 patients whose tumor diameters ranged from 5 to 23 mm and reported their results in studies published between 2000 and 2002.6,19 The ablative procedure was conducted with a diode laser coupled to a thermistor, and mammography (MMG) was the guiding method. Surgery was conducted after 1 to 8 weeks, and tumor destruction was complete in 70% of cases. Operator inexperience, improper technique usage, and underestimation of tumor diameter, which exceeded 2 cm according to imaging methods, contributed to the incomplete destruction of the target lesion in 16 patients.6,19
Korourian et al assessed cell viability by Proliferating Cell Nuclear Antigen (PCNA) analysis in 2003.20 MRI-guided PLA was applied to 29 patients with invasive tumors between 1.0 and 4.0 cm. The authors do not mention the technical parameters applied. Surgery was conducted on the day of or the day after the ablation. Of the 29 patients submitted to the protocol, only 7 had their data analyzed because the tumor periphery and the carbonization area were represented on the same slide. On pathological evaluation, PCNA activity increased progressively with each millimeter of distance from the central carbonization area. The seven selected cases exhibited the same pattern of PCNA activity. This allowed the authors to conclude that, in addition to the effects of direct cellular injury by carbonization, ablation reduced the proliferative capacity of adjacent tumor cells, possibly by driving them to apoptosis.20 Clique ou toque aqui para inserir o texto.
In the PLA reported by Haraldsdóttir et al in 2008, diode laser with thermistor was used for ablation under US view of breast cancer in 24 patients.7 The number of fibers was proportional to the lesion volume: 1 fiber for lesions up to 10 ml and 4 for lesions between 10 and 15 ml. The power ranged from 0.5 to 3 W and was adjusted to maintain a temperature of 48 °C for 30 min on the thermistor. Ablation rates ranged from 0% to 100% (mean 33%), and 3 patients had complete tumor necrosis. Part of the incomplete response was attributed to the failure of diagnostic methods to adequately estimate tumor volume or to difficulty in visualizing the lesion. In 2011, the same group published a study describing immune alterations following ablation. CD20+, CD68+, and CD83 + cells increased in the tumor borders, as did intratumoral CD8 + and CD68 + cells. Therefore, PLA may be a potential source of tumor antigens thus eliciting an immune response.7,21
In 2009, Van Esser et al published data on ablation of breast carcinomas in 14 patients using a Nd:YAG laser with 7 W of power.8 Complete ablation was accomplished in 7 patients with a success rate of 50%. Among patients with tumors smaller than 2 cm (8 patients), the ablation rate was notably higher at 88%. The main reasons cited for partial success were the imaging underestimation of tumors and the association with an extensive
After a long pause in publications regarding PLA for breast cancer, Schwartzberg et al reported in 2018 the results of a multicenter investigation wherein laser ablation was applied to 61 patients with invasive carcinoma measuring 20 mm or less.4 The breast lesion was assessed by MMG, US, and MRI before and 28 days after laser ablation. The equipment used was a diode laser with thermistor. The procedure was maintained until a temperature of 60 °C was reached at all points of the sensor or 51 °C for more than two minutes at the peripheral points, which resulted in average energy values of 4560 J. Complete ablation was observed in 84% of cases. MRI had a negative predictive value (NPV) of 92%. To confirm necrosis and assess cell viability, immunohistochemical (IHC) analysis with markers such as cytokeratin (CK) 8/18 and estrogen receptor (ER) are crucial. These markers provided superior information compared to hematoxylin and eosin (HE) staining alone and ki67 staining. Furthermore, given late cellular effects, the group theorizes that the results could have been better if the surgical excision had been conducted with a longer interval after PLA, with longer observation of changes.4
Despite the partial success rates reported in previous studies, Nori et al evaluated PLA as the exclusive method for local treatment in 12 patients with individualized cases and published their results in 2018.5 The patients were over 75 years old with single tumors smaller than 2 cm and could not undergo surgery. An Nd:YAG laser was used with 3 W of power and a target energy of 1800 J per application (with repositioning of the fiber in the nodules between 1 and 2 cm) or until complete vaporization of the nodule upon US. After an average follow-up of 28 months, regional and distant recurrence were not observed. No updates on the longer-term follow-up of these patients have been published.5
Two studies assessed PLA as an additional method after vacuum-assisted excision (VAE). In the study by Klimberg et al, 3 patients with tumors smaller than 1.5 cm underwent ablation immediately following VAE and surgical excision soon after. The area of necrosis and the images after ablation were incompatible on pathological examination, and the authors judged that the behavior of the ablation in the hematoma cavity presented different aspects from those observed in the tumor. They concluded that PLA was unsafe immediately following VAE22 . Meanwhile, Perreta et al conversely selected 11 patients with invasive carcinoma smaller than 1 cm for VAE followed by PLA.23 The equipment used was an Nd:YAG laser. The power was programmed at 6 W, and the target power ranged between 1200 and 1800 J. After ablation, the patients underwent imaging with US, MRI, or contrast-enhanced MMG (cMMG). Of the 11 selected cases, 10 (90.9%) had complete pathologic response. Collectively, the post-ablation imaging methods, in this study with a small number of cases, displayed a sensitivity and negative predictive value (NPV) of 100%.23
Treatment Success Rates
When evaluated collectively, the studies present quite variable findings regarding the ablation rate. The quantification of the response, when provided, was presented in two distinct ways: as the average rate of tumor ablation area or as the complete response rate. Haralsdóttir describes the average tumor ablation area of 33% (range from 0-100%). 7 Schwartzberg, on the other hand, describes that among cases that did not achieve complete response, the average tumor ablation rate was 88%.4 Among the authors using the complete response rate to assess treatment efficacy, we have the descriptions of Dowlatshahi, achieving a 70% success rate (38 out of 54 patients), 6 Van Esser describing 50% (7 out of 14 patients),8 Schwartzberg with an 84% complete ablation rate (51 out of 61 patients)4 which allow us to calculate an overall complete ablation rate of 74.4% (96 out of 129 patients in whom this data was analyzed). If we consider the data described by Perreta, 23 with complete ablation in 10 out of 11 cases undergoing APL after VAE, we see an increase in the complete ablation rate to 75.7%
Pathological Findings After Ablation
According to the descriptions of the studies, pathological alterations can be split into three main zones (Figure 2):
The central charring zone is composed of charred necrotic debris. Immediately adjacent to this zone, signs similar to electrocautery artifacts are observed;17,24 The intermediate or “pseudo-viable” zone, wherein cells seem morphologically normal or “mummified”,24 displays no signs of necrosis, increased apoptosis, or inflammation. The nuclei of these cells are hyperchromatic. The cytoplasm is eosinophilic and no longer exhibits its usual characteristics.
17
In this area, further evaluation with IHC is essential to confirm cell viability4,17,24 CK8/18 and ER are favored, simpler methods.8,24 In the hemorrhagic/thrombotic zone, a hemorrhagic halo with severe cell damage is observed, with hypocolored and hyperchromatic nuclei. These cells are interspersed with proliferative fibroblasts, thrombosed blood vessels, and extravasated red blood cells.17,24
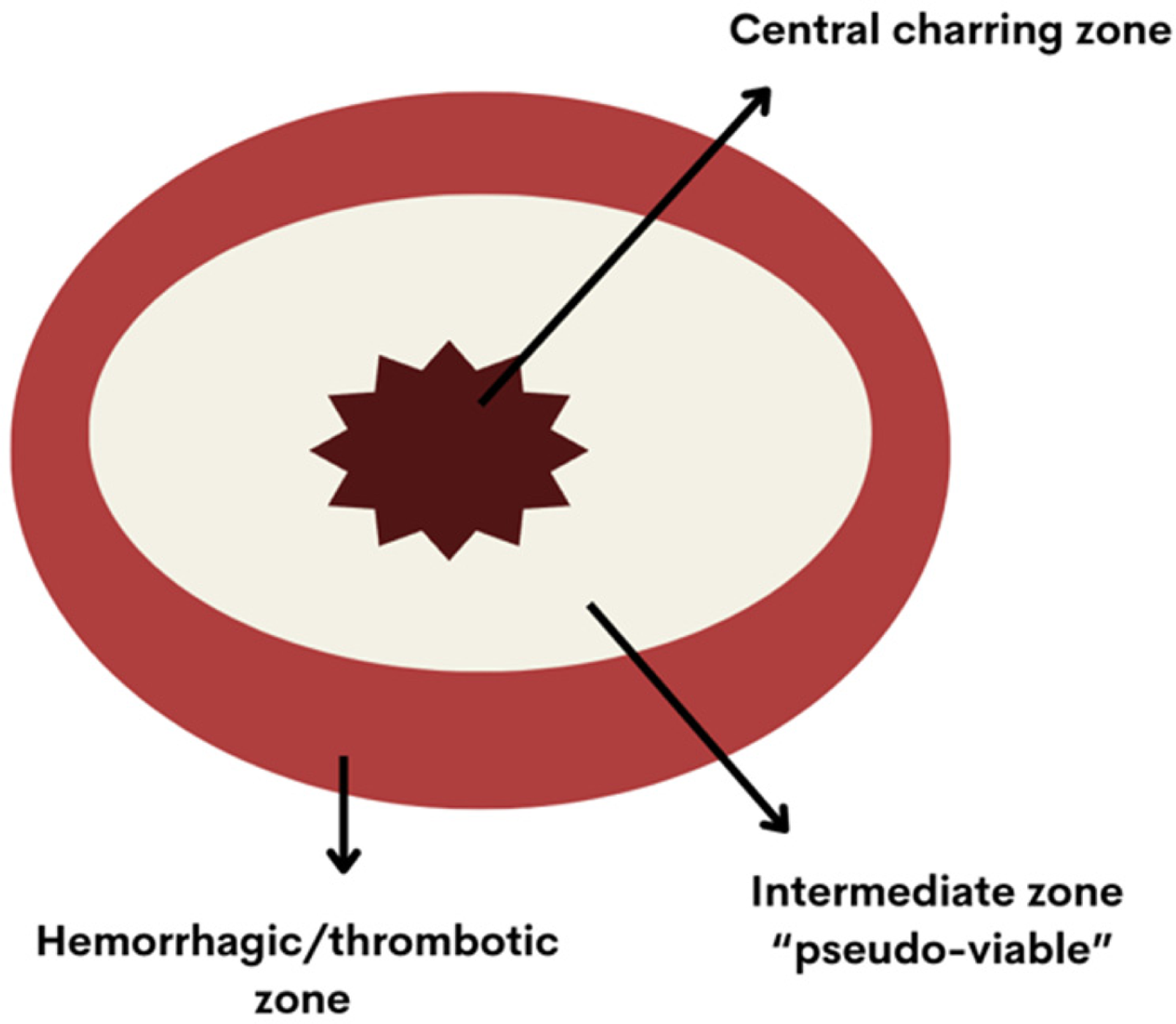
Bloom et al suggest that the pathological effects of laser have two key mechanisms: the direct transfer of thermal energy, similar to electrocautery, and the induction of thrombosis in small and medium-sized peripheral tumor vessels. In the so-called “pseudo-viable” zone, the effects are possibly due to the rapidity of the damage added to the absence of blood flow, which produces a cellular “mummification” with consequent apoptosis of such cells of the intermediate layer.24
Radiological Evaluation
Suitable imaging methods in the ablative procedure have three central steps and characteristics:4,7,8,17
In studying candidate lesions for ablation, the shape and dimensions of the nodule, potential multicentricity, and association with microcalcifications and enhancements that could indicate the presence of an associated Proper visualization of the lesion is performed for the ablative procedure. A reliable correlation between the area of necrosis (and eventual residual lesion) is determined to safely assure the replacement of the surgical procedure by the percutaneous ablative method.
Among the studies evaluated, only Schwartzberg et al.4 used MRI for patient selection, demonstrating improved rates of complete ablation and a higher average percentage of the treated tumor area. Although most studies had tumors smaller than 2 cm as inclusion criteria, the mean diameters observed in the surgical specimens ranged from 0.5 to 6.0 cm. This highlights the limitations of MMG and US in accurately estimating tumor size.
Most of the studies mentioned here used US to guide PLA. Despite the high availability, low cost and dynamism of the technique, the alterations in the echogenicity of the lesion resulting from the release of gases from tumor necrosis blur the image and hinders visualizing any residual tumor.8,18,24 Consequently, no method suitable for real-time visualization of the ablative procedure has been established. In terms of the exam to assess the effectiveness of the ablative method, MRI best correlated with the area of tumor necrosis and any residual tumor, with an NPV between 92% and 100% and correlation coefficients (r2) of 0.8 and 0.86 for determination of complete ablation and occurrence of residual tumor, respectively.8,15,17,23
Side Effects
Seven studies have documented the occurrence of side effects, which are summarized in Table 2.4,6,7,8,15,18,20
Side Effects Described in PLA* Studies

*PLA: Percutaneous Laser Ablation
Skin burns were the most frequently observed complication, occurring in 6% of patients. Other complications included hematomas/bleeding, pain, nodulation, erythema, seromas, and steatonecrosis. However, the frequency of occurrence cannot be inferred from the descriptions.4 6,7,8,15,18,20
Two severe adverse events were described: one case each of gas rupture of the tumor18 and pneumothorax.8 The gas rupture occurred in a tumor with a diameter exceeding 4 cm under 3 W power ablation when the energy reached 5400 J. The patient experienced sudden pain and subcutaneous emphysema. The complication was resolved with analgesia after the immediate suspension of the procedure.18 Pneumothorax was observed during the ablation of a lesion adjacent to the pectoralis muscle. The procedure was halted, and conservative treatment was administered.8
Discussion
Given the advantages of early breast cancer diagnosis, the necessity for aggressive treatments has been questioned, consequently prompting a continual quest for less invasive approaches, in particular percutaneous treatments such as PLA, cryoablation, RFA, microwave and HIFU ablation. None of the ablative methods mentioned are widely used, as their applicability has not yet been authorized in clinical practice. PLA, cryoablation, RFA, and microwave ablation are percutaneous and more accessible methods. HIFU uses a collimator to ablate the tumor, often guided by MRI, and requires more expensive equipment. With the exception of cryoablation, all methods destroy the tumor through heat, which leads to the formation of gases from necrosis and makes real-time monitoring of the ablation more challenging. Cryoablation, by generating an “ice ball,” appears to offer better real-time visualization of the tumor and requires less anesthesia. But the appropriate selection of patients who are candidates PLA, as well as the improvement of imaging methods can mitigate such difficulties on real- time visualization and allow results with a very high rate of complete ablation.20
PLA was employed in the treatment of around 310 patients in previous studies, demonstrates promising results as a method for locally treating early-stage breast cancer. However, the lack of standardized protocols hinders its reproducibility and challenges its immediate application in clinical practice.
The average tumor ablation rates found in this review ranged from 33% to 87%, and the studies that describe a complete tumor ablation reported an average rate of 64.7%. As with other conservative treatments, the accurate selection of patients for ablative procedures is paramount for their success. Utilizing MRI to more accurately estimate tumor dimensions, along with excluding histological subtypes such as ductal carcinoma in situ (DCIS), and invasive lobular carcinoma (ILC), are correlated with increased success rates of the procedure.4,5,8
Among the 4 studies that documented the rate of complete ablation followed by surgery, 3 used diode laser and observed, on average, 65% complete ablation,4,6,7,8 compared to 50% in the study that used Nd:YAG laser.8 Therefore, a diode laser coupled to a thermistor and aiming for a temperature above 60 °C at all points of the sensor (and not a predetermined energy level for the end of the procedure) seems to has the highest possibility of ablative success.4,6,7 Concerning the best power to be calibrated, the studies with higher ablation rates reported power between 5 and 10 W.4,6,8,23. Developing an improved method to guide the procedure, with real-time monitoring of residual lesions, would also be indispensable in improving results.
In the studies analyzed, no evaluations were specifically directed to the potential increase in the rate of tumor necrosis with time elapsed since the procedure. Bloom et al described that the size of the pseudo-viable zone apparently decreased as the days passed, while growth in the external necrotic zone was noted.24 The immune alterations after exposure to tumor antigens may contribute to the increase in apoptosis long after the ablative procedure. 7 Schwartzberg et al reported the best rates of ablation with excision 28 days after ablation, but most studies conducted excision after a shorter interval.4 The excellent results observed by Nori et al, wherein the ablative procedure was conducted as a single local treatment and no recurrence was observed after a mean follow-up of 28 months, reinforce this hypothesis.5 Dowlatshahi et al also reported the success of the procedure as a single treatment, without subsequent surgical excision, for two patients. One patient was monitored for six months, with subsequent loss to follow-up, while the other was followed for three years without local or distant recurrence.22
Regarding the post-ablation imaging evaluation, the inclusion of MRI would be paramount in future studies to evaluate the efficacy of ablation, as well as PLA as a single therapeutic procedure.4,17 cMMG following PLA was evaluated in a singular study wherein the ablative procedure was conducted after VAE. However, data regarding its applicability remain inconclusive.23
The table below outlines characteristics compiled through this review that seem to correlate with increased rates of successful ablation. These factors can guide the design implementation of future studies aimed at improving ablative techniques for breast cancer treatment (Table 3).
Proposal of Criteria for Optimization of Procedure in Future Studies

Future Directions
The indication of most ablative methods for breast cancer treatment remains restricted to highly individualized cases and research protocols.11 In PLA specifically, further studies aimed at standardizing the technique are crucial in advancing this procedure as a standalone local treatment option in selected cases.
Concerning ablation as the exclusive treatment of early-stage breast cancer, an important issue permeates its applicability: the need for prognostic evaluation of axillary lymph nodes conducted through surgical treatment, with sentinel lymph node biopsy (SLNB) as the technique utilized in most cases. As of the writing of this document, the omission of SLNB has been considered safe only for patients over 70 years old, with clinical stage I tumors, positive hormone receptors, and absent HER-2 overexpression.25,26 Moreover, the SOUND study published in September 2023 indicates safety in extending the potential omission of a lymph node approach to patients over 50 years old with tumors exhibiting previously identified characteristics.27 Other ongoing studies are also analyzing the broader indication of SLNB omission.25,27–30 Considering that one of the primary objectives of studies involving ablative techniques is not to expose the patient to the surgical procedure, the broader indication of the omission of the axillary approach makes the search for minimally invasive percutaneous treatments more plausible.
As more robust data from studies becomes available, other applications that have been previously underexplored or scarcely assessed should also be contemplated. For instance, one such application could involve utilizing tumor bed ablation after neoadjuvant systemic treatment or after VAE. This approach may not only promote the ablation of any residual foci but also promote a possible increase in the local immune response.21,24
Conclusion
This study is the only evaluation of the technical aspects encompassing studies published up to January 2024 regarding percutaneous laser ablation (PLA) for the ablative treatment of breast cancer. Although the application of this procedure remains restricted to cases with specific indications or within the context of research protocols, addressing the highlighted points in future studies could potentially validate this promising technique for the localized treatment of early breast cancer management.
Finally, the authors propose criteria for optimizing procedures in future studies.
Footnotes
No Disclosures
No financial support for this research
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
