Abstract
Background and Aims
Thyroid cancer, a prevalent endocrine malignancy, often presents as thyroid nodules, whose benign or malignant nature is challenging to determine. This study aims to identify circulating miRNA panels that may distinguish between benign nodules, papillary thyroid cancer, and normal thyroid conditions, building on extensive research into miRNAs as potential thyroid cancer biomarkers.
Materials and Methods
As a cross-sectional case-control study the study revealed the quantification of the 17-miRNA panel was evaluated using qRT-PCR method on 60 blood samples, comprising 25 patients diagnosed with PTC, 24 patients with benign lesions, and 11 healthy controls.
Results
We found that seven miRNAs, including miR144-3p and miR190a-5p, exhibited statistically significant variations in their expression levels, and miR144-3p exhibited the most superior diagnostic capability in distinguishing between malignant and healthy groups, as well as between benign and healthy groups. The concurrent utilization of miR144-3p, miR148a-3p, and miR190a-5p resulted in an enhancement of sensitivity from 76% to 88%.
Conclusion
Our study suggests that miR144-3p and miR190a-5p exhibit potential as biomarkers for distinguishing between benign and malignant thyroid nodules and healthy individuals, and further investigation is necessary to evaluate their clinical significance.
Introduction
Thyroid cancer is a neoplasm originating from the thyroid gland's cellular components. According to Sung et al, thyroid cancer is widely recognized as the prevailing form of endocrine malignancy, accounting for many cases and fatalities globally in 2020, with an estimated 567 000 new incidences and 41 000 deaths. The global prevalence of thyroid cancer has exhibited a notable upwards trend in recent decades. 1 Thyroid cancer exhibits a higher prevalence among females than males and is characterized by a female-to-male ratio of approximately 3:1. The prevalence of thyroid cancer indicates geographical disparities, with developed nations such as the United States, Canada, and Japan displaying elevated rates. 2 According to Vaccarella et al, there has been a global rise in the prevalence of thyroid cancer, with an average annual percentage increase of approximately 5% between 1990 and 2016. The rise in occurrence is believed to be attributed to enhanced diagnostic methodologies, heightened monitoring efforts, and the influence of environmental variables. 3
Thyroid cancer ranks as the seventh most prevalent cancer among women in Egypt, constituting approximately 3% of all female cancer cases. 4 The prevalence of thyroid cancer in Egypt has exhibited a notable upwards trend in recent decades. The incidence rate of thyroid cancer in Egypt in 2018 was 2.1 per 100 000 population after being age-standardized. In Egypt, the prevalence of thyroid cancer exhibits a notable gender disparity, with females being more affected than males, as evidenced by a female-to-male ratio of approximately 7:1. The median age of diagnosis for thyroid cancer in Egypt is reported to be 50 years. 5
Thyroid cancer is associated with several risk factors, including exposure to ionizing radiation, a familial predisposition to thyroid cancer, and specific genetic mutations. 6 The association between exposure to ionizing radiation and the development of thyroid cancer has been firmly established in the scientific literature. 7 The presence of a familial history of thyroid cancer is an established risk factor for the development of this particular disease. According to, 8 specific genetic mutations, such as those occurring in the BRAF and RAS genes, have been linked to an elevated susceptibility to thyroid cancer.
Papillary thyroid carcinoma (PTC) is the predominant form of thyroid cancer, constituting approximately 80%-85% of all cases of malignant thyroid neoplasms. 9 The diagnosis of PTC is commonly established through the integration of clinical manifestations, ultrasound imaging, fine-needle aspiration biopsy (FNAB), and histopathological analysis. 10 Nevertheless, fine-needle aspiration biopsy (FNAB) exhibits certain limitations in terms of its precision and dependability, thereby necessitating the identification and incorporation of supplementary biomarkers to enhance the diagnostic capabilities for PTC.
MicroRNAs (miRNAs) are a class of small RNA molecules that play a crucial role in the regulation of gene expression at the posttranscriptional level. The roles of these factors in cancer development and progression have been demonstrated, and their dysregulation has been documented in different cancer types, such as PTC. 11 Numerous studies have been conducted to examine the diagnostic utility of miRNAs as biomarkers for PTC. As an illustration, a study conducted by Gu et al who identified a panel consisting of six microRNAs (miR-222, miR-221, miR-146b, miR-181b, miR-155, and miR-224) that exhibited a notable level of diagnostic accuracy for PTC. 12 A separate investigation conducted by Wang et al has revealed a notable decrease in the expression of miR-375 within PTC tissues. This finding suggests that miR-375 has the potential to function as a biomarker for PTC. Previous studies have reported a correlation between miR-134 and thyroid cancer. 13 According to Gong et al, the investigation revealed the downregulation of miR-134 in both PTC tissues and cell lines. Furthermore, the study demonstrated that the overexpression of miR-134 effectively suppressed the proliferation and invasion of PTC cells. 14 In a separate investigation conducted by Liu et al, it was observed that miR-134 effectively suppressed the proliferation and invasion of PTC cells through the specific targeting of KRAS. 15
The research conducted by Sun et al revealed a notable decrease in the expression of miR-144 in both PTC tissues and cell lines. This downregulation was found to be significantly correlated with more advanced tumor stage and an unfavorable prognosis. Furthermore, the upregulation of miR-144 was found to suppress the proliferation of PTC cells and trigger cell cycle arrest. 16
A separate investigation revealed that the expression of miR-148a was notably diminished in both PTC tissues and cell lines. Furthermore, this reduced expression was found to be correlated with more advanced tumor stage and unfavorable prognosis. Furthermore, the upregulation of miR-148a was found to suppress the proliferation and invasion of PTC cells while also inducing cell cycle arrest. 17 According to Cantara and his colleagues, their study proposes that miRNA190 could potentially function as a dependable and sensitive indicator for distinguishing between various types of thyroid nodules within a Caucasian demographic. 18
This study aimed to assess the expression levels of 17 miRNAs associated with PTC to identify a circulating miRNA profile biomarker with the potential to distinguish between PTC, benign thyroid nodules, and healthy individuals.
Patients and Methods
Patients and Sample Collection
This study was approved by the Institutional Review Board and the Ethical Committee of the Faculty of Medicine at Mansoura University in Mansoura, Egypt, under the code number R.21.08.1415. All participants in the study provided written, informed consent before the collection of samples. Information revealing the patient's identity has been avoided. All patients have been identified by numbers or aliases and not by their real names.
This study aimed to assess the efficacy of miRNA blood markers in diagnosing thyroid gland cancer while ensuring compliance with the ethical standards outlined in the Declaration of Helsinki. The conduct and reporting of this study adhere to the STARD guidelines for transparency and completeness in diagnostic accuracy research. 19 Prior to the collection of samples, all participants in the study were required to provide written informed consent. A total of 25 individuals diagnosed with PTC and 24 individuals with benign thyroid nodules (BTNs) were included in the research study. Table 1 presents a comprehensive summary of clinicopathological characteristics pertaining to patients, encompassing factors such as tumor size and number of lesions. The patients in the study underwent diagnostic assessments using pathological examinations conducted by skilled pathologists at OCMU, Mansoura University, located in Egypt.
General Characteristics of the Study Participants.

IQR: Interquartile range, NLR: Neutrophil-Lymphocyte Ratio, PLR: Platelet-Lymphocyte ratio. P value by Kruskal-Wallis test for continuous variables and Chi-Square test for categorical variables.
Total RNA Isolation/Quantitative Real-time Polymerase Chain Reaction (qRT–PCR) Samples and RNA Extraction
Venous blood was drawn from all subjects, including healthy controls, on the day of surgery and stored in vacuum tubes. Samples underwent initial centrifugation at 1900xg for 10 min at 4 °C within an hour of collection. To minimize cell-derived nucleic acids, the supernatant was further centrifuged at 12 000xg for 10 min at 4 °C. Samples were then stored at −80 °C until RNA extraction, excluding any affected by hemolysis. Total RNA, including microRNA, was isolated from 200 µl of serum using Qiagen's protocol, and eluted with 50 µl of RNase-free water. The RNA was stored at −80 °C. Quantity and quality of the RNA were assessed using a NanoDrop 1000 spectrophotometer (NanoDrop Technologies, Thermo Fisher Scientific, Wilmington, DE, USA).
miRNA Expression Analysis
Using a miScript II RT kit (Qiagen), total RNA (250 ng/µl) was converted to cDNA as per the manufacturer's protocol. The relative expression of 17 miRNAs was quantified using a miScript SYBR Green PCR kit (Qiagen), with 1 µL of diluted RT product serving as the template in a 10 µL PCR reaction containing 1X SYBR Green master mix, 200 nM miRNA-specific forward primer, and 200 nM universal primers. qRT‒PCR conditions were set at 95 °C for 10 min, followed by 40 cycles of 95 °C for 15 s and 60 °C for 30 s. All RT‒qPCRs were conducted on a ViiA 7 real-time PCR system (Applied Biosystems, Foster City, CA, USA). miR-16 was used as an endogenous control to normalize the relative expression levels of miRs. Real-time PCR experiments were performed in triplicate. Primers were designed using the Primer BLAST tool on the NCBI website, and sequences were sourced from Eurofins Genomics (Hamburg, Germany).
Fold Change Calculation
The threshold cycle (CT) was determined for individual miRNAs. The (2−ΔΔCT) method was used for the FC calculation. First, ΔCT was calculated by subtracting the numerical CT value of the miRNA of interest from the housekeeping gene (ΔCT = CT sample of interest – CT housekeeping gene). Second, the results were normalized against control samples using the formula (ΔΔCT = ΔCT sample of interest – ΔCT control sample). Third, the fold change was calculated as follows (FC = 2− ΔΔCT). Finally, base 2 logarithmic transformation was calculated using the formula (Log2 FC).
Statistical Analysis
The statistical analysis of the data was conducted using IBM SPSS v28.0. The type of the study is cross-sectional case-control study where the presentation of continuous data involved the utilization of the median and interquartile range (IQR), whereas categorical data were represented using numerical counts and corresponding percentages (%). The assessment of data normality was conducted through visual examination of histograms and Q‒Q plots, as well as through the application of the Kolmogorov‒Smirnov test. The statistical analysis involved conducting comparisons between continuous variables using the Kruskal‒Wallis test, followed by Dunn-Bonferroni procedure if a significant result was found in the Kruskal-Wallis test to identify significance difference between pairwise comparisons. The chi-square test was employed to evaluate disparities in categorical variables. Receiver operating characteristic (ROC) curve analysis was employed to evaluate the diagnostic efficacy of the measured miRNAs. The measurements of the area under the curve (AUC), standard error (SE), sensitivity, and specificity were acquired. The determination of the cut-off point was computed based
Results
Clinical Characteristics
The study encompassed a cohort of 49 participants. The study population consisted of 25 individuals diagnosed with malignant thyroid neoplasms and 24 individuals with benign lesions. There were no statistically significant differences observed in the clinical parameters among the various groups of subjects, as indicated in Table 1. The predominant lesion observed in the benign group was colloid goiter, accounting for 58% (n = 14) of cases, whereas in the malignant group, papillary carcinoma was the prevailing condition, representing 92% (n = 23) of cases (Table 2).
Clinical Characteristics of Participants with Thyroid Lesions.

Relative miRNA Expression
A significant difference in expression level was found in 7 out of 17 tested miRNAs, namely, miR134-5p, miR144-3p, miR148a-3p, miR190a-5p, miR221-3p, miR222-3p and miR34b-3p (P value < .05) (Table 3). The highest observed difference in expression level between groups was found for miR144-3p followed by miR190a-5p (P value < .005) (Table 3, Figure 1A-G). A heatmap showing the expression of miRNAs in the whole sample is presented in Figure 2.

Box plots showing expression level of different miRNAs in the malignant, benign and healthy control groups, respectively. A: miR190a-5p, B: miR34b-3p, C: miR134-5p, D: miR144-3p, E: miR148a-3p, F: miR222-3p, G: miR221-3p. P values obtained by post-hoc Mann-Whitney U-test following Kruskal-Wallis test. Bonferroni correction was applied to account for multiple comparisons.

Heatmap showing the relative expression of miRNAs in different study groups represented in log fold change.
Relative Expression of the Different Tested miRNAs in the Study Participants.
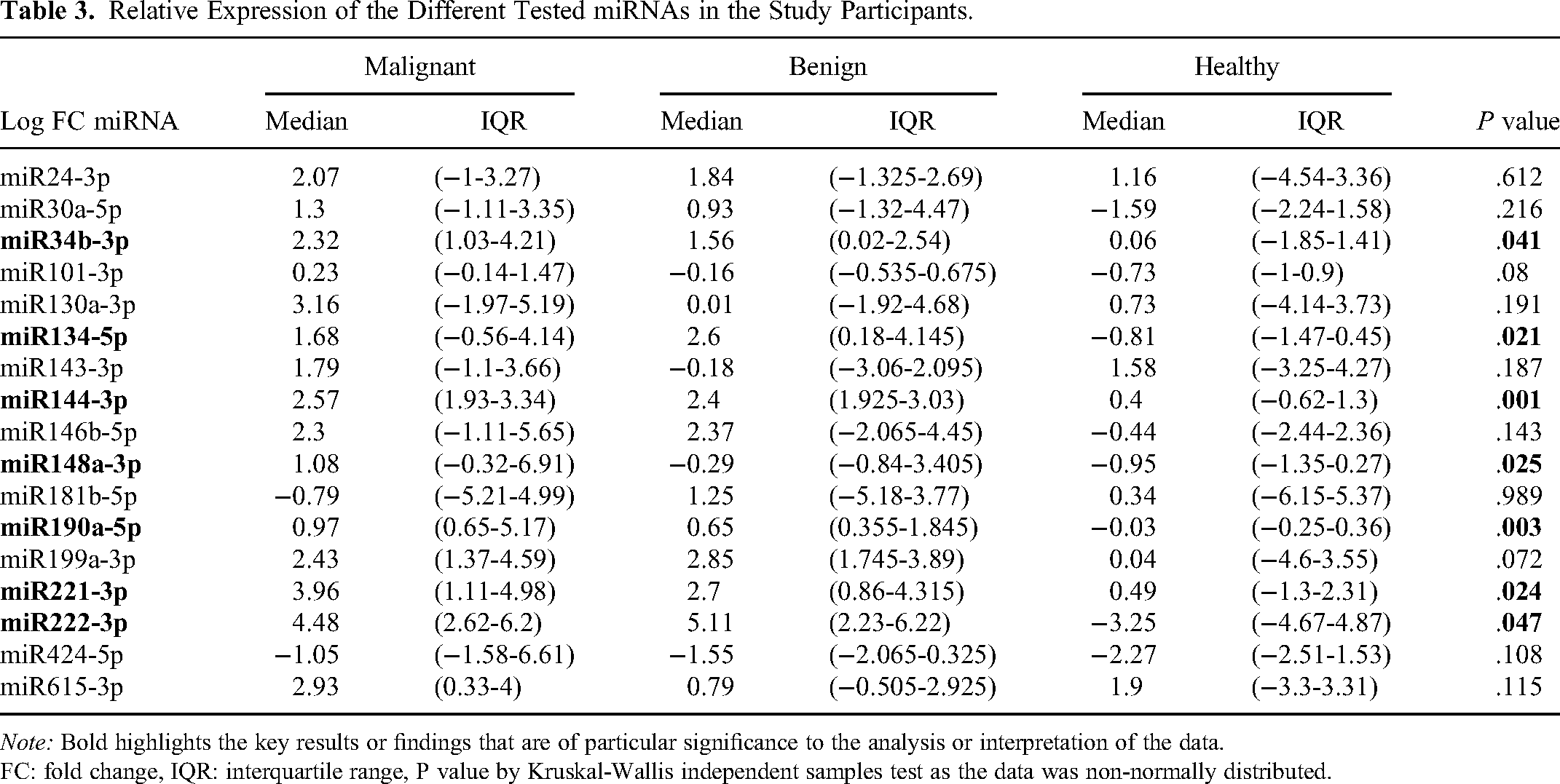
Note: Bold highlights the key results or findings that are of particular significance to the analysis or interpretation of the data.
FC: fold change, IQR: interquartile range, P value by Kruskal-Wallis independent samples test as the data was non-normally distributed.
The Diagnostic Power of Tested miRNAs
The diagnostic power of the differentially expressed miRNAs was assessed using ROC curve analysis. miR144-3p showed the highest diagnostic ability in differentiating malignant versus healthy and benign versus healthy groups (AUC 0.9 and 0.96, P < .0001), followed by miR190a-5p (AUC 0.83 and 0.8, P < .001). miR-148a-3p and miR-34b-3p showed nonsignificant ROC curves in differentiating benign thyroid lesions versus healthy controls, indicating poor diagnostic ability. None of the measured miRNAs were able to discriminate between patients with malignant versus benign lesions of the thyroid gland (AUC range 0.44-0.63, P > .05 in all cases) (Table 4, Figure 3A-C).

ROC curves showing the diagnostic ability of select miRNAs between different groups of study subjects. A: Malignant versus Healthy, B: Benign versus Healthy, C: Malignant versus Benign. Curves closer to the upper left point represent better diagnostic ability. Figure 2C shows inability of measured miRNAs to discriminate between malignant and benign thyroid lesions.
Diagnostic Power of the Measured miRNAs in Between Different Groups of the Study Subjects.
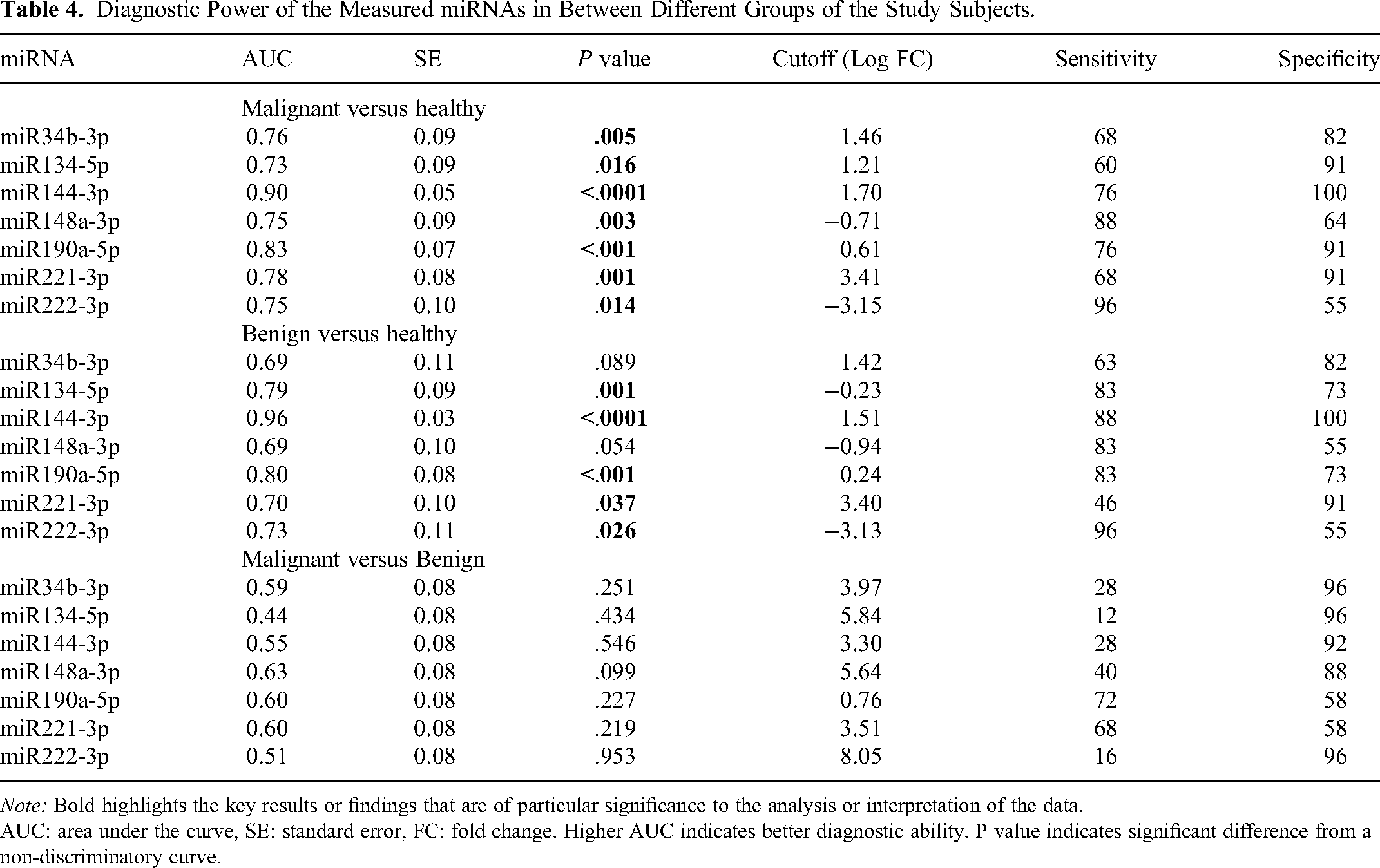
Note: Bold highlights the key results or findings that are of particular significance to the analysis or interpretation of the data.
AUC: area under the curve, SE: standard error, FC: fold change. Higher AUC indicates better diagnostic ability. P value indicates significant difference from a non-discriminatory curve.
Diagnostic Ability of the Combination of Different miRNAs
The combination of miRNAs was chosen according to stepwise logistic regression for comparison. Regarding malignant versus healthy, the combination of miR144-3p, miR148a-3p, and miR190a-5p improved the AUC from 0.9 to 0.956, and the sensitivity increased from 76 to 88; however, the increase in AUC was nonsignificant (P = .37). For benign versus healthy patients, the combination of miR-134-5p + miR-144-3p was found to be a favorable option compared to miR-144-3p alone; however, the difference was once more nonsignificant. Regarding malignant versus benign, the combination of miR148a-3p, miR134-5p, and miR34b-3p was found to have a better Youden's index than miR148a-3p alone; however, the difference in AUC was still nonsignificant (Table 5).
Diagnostic Ability of Combination of miRNAs Versus the Single Best miRNAs in Each Between-Groups Comparison.

Note: Bold highlights the key results or findings that are of particular significance to the analysis or interpretation of the data.
AUC: area under the curve, SE: standard error, Cutoff in Log fold change for single miRNA and in predicted probability for multiple miRNAs. The selection of miRNAs combination was based on stepwise logistic regression.
Discussion
Thyroid nodules represent a prevalent issue in clinical practice, and the differentiation between benign and malignant nodules poses a significant diagnostic obstacle. Colloid goiter emerged as the prevailing lesion within the benign category, whereas papillary carcinoma exhibited dominance within the malignant group. The aforementioned findings align with prior research that has documented papillary carcinoma as the predominant form of thyroid cancer, constituting approximately 80% of all instances of thyroid cancer. 23 In the present study, it was observed that there were no statistically significant variations in clinical parameters across the three groups. The absence of substantial disparities in clinical parameters observed among the three cohorts in our study underscores the necessity for additional exploration into the utilization of molecular markers in the identification and prediction of thyroid nodules.
The present study aimed to examine the relative expression levels of 17 microRNAs (miRNAs) in cancerous tissues and their corresponding adjacent normal tissues among cancer patients. Significant differences were observed in the expression levels of seven out of the seventeen miRNAs that were tested, specifically miR134-5p, miR144-3p, miR148a-3p, miR190a-5p, miR221-3p, miR222-3p, and miR34b-3p. The most notable disparity in expression level between the groups was observed in miR144-3p, with miR190a-5p exhibiting the second-highest difference. According to a study conducted by Sun et al, miR-144 exhibited a high frequency of downregulation in cases of PTC. 16 Furthermore, the expression of miR-144 was found to be correlated with larger tumor sizes. Additionally, it was demonstrated that miR-144 functions as a tumor suppressor by impeding the proliferation of IHH4 cells through its targeting of WWTR1, a transcription regulator containing the WW domain. The present findings are incongruent with our own results, which indicated an upregulation of miRNA-144 in cancer patients relative to healthy individuals. This discrepancy may be attributed to variations in patient characteristics, differences in experimental methodologies employed, or even discrepancies in the sample size of patients.
In addition, our analysis did not identify any statistically significant correlations between tumor size and the expression levels of the miRNAs investigated, as indicated in Supplementary Table S1.
However, the findings for miR-190a differ from previous research which supports the involvement of miR-190a in facilitating the epithelial-mesenchymal transition (EMT) in gastric cancer cells and demonstrated that miR-190a downregulates the expression of PHLDB2, resulting in the activation of AKT3. 24 This activation subsequently promotes EMT, as well as cell migration and invasion. Moreover, the observed upregulation of miR134-5p aligns with prior studies indicating its association with the initiation and advancement of cancer which documented the overexpression of miR-134 in both oral potentially malignant disorders (OPMDs) and oral squamous carcinoma (OSCC) compared to control subjects. 25
According to Liu et al, a mutation occurring in the 3′-UTR of GOLPH3, which is a highly influential oncogene, has been identified as a potential factor contributing to the upregulation of miR-134 and the subsequent advancement of the disease. The luciferase reporter, which contains the miR-134 binding site in the 3′-untranslated region (3′-UTR) of GOLPH3, exhibited repression when miR-134 was overexpressed. However, this effect was nullified when a mutation was introduced. 26
Conversely, an upregulation of miR148a-3p was observed, consistent with the results of Pereira et al documented an elevated expression of miR148a in gastric cancer and adjacent tissues relative to normal tissues. Moreover, Bcl2 is a frequent target of miR148a. 27
In both the malignant and benign groups, the expression levels of miR221-3p and miR222-3p were found to be upregulated when compared to the healthy group. This finding aligns with a previous study that demonstrated the upregulation of miR-221 and miR-222 in PTC when compared to normal thyroid gland tissue from the same patient, as well as in PTC when compared to nodular goitre. 28
Furthermore, the observed upregulation of miR34b-3p in our study aligns with previous findings that miR-34b is regulated by p53 and plays a role in the regulation of cell proliferation and adhesion-independent growth. Corney et al reported that miR-34b is a target of the tumor suppressor protein p53 and functions in cooperation with p53 to control cell proliferation and adhesion-independent growth. 29
The levels of miRNA expression and the observed differences in expression across different stages of cancer did not exhibit statistically significant variation. This suggests that miRNA expression may not be a reliable indicator for distinguishing between the various stages of cancer within the studied group (Supplementary Table S2).
The diagnostic efficacy of miRNA differential expression in thyroid nodules was assessed through the utilization of ROC curve analysis. miR144-3p demonstrated the most robust diagnostic capability in distinguishing between malignant and benign thyroid nodules as well as healthy controls, with miR190a-5p exhibiting the second-highest diagnostic ability. On the other hand, the diagnostic capacity of miR-148a-3p and miR-34b-3p in distinguishing benign thyroid lesions from healthy controls was found to be limited. The findings of this study indicate that miR144-3p and miR190a-5p have the potential to be utilized as biomarkers for differentiating between malignant and benign thyroid lesions in comparison to healthy individuals. Previous studies have identified these miRNAs as potential diagnostic markers. Sun et al demonstrated the diagnostic value of miRNA-144 in distinguishing between PTC and noncancerous tissue, with an area under the curve (AUC) of 0.743 and a significance level (P) less than 0.001. 16 Lv et al reported that the upregulated expression of a competing endogenous RNA (ceRNA) consisting of the NR2F1-AS1/miR-190a/PHLDB2 axis promoted the epithelial-mesenchymal transition (EMT) process, which may be an early biological event facilitating the invasive ability of gastric cancer (GC) cells. 24
However, the measured miRNAs failed to distinguish between patients with malignant and benign thyroid lesions, as indicated by AUC values between 0.4 and 0.6 and p values exceeding 0.05. We evaluated the diagnostic potential of differentially expressed miRNAs in differentiating malignant and benign thyroid lesions and healthy controls, using stepwise logistic regression to identify optimal miRNA combinations. The use of various miRNAs shows promise in improving thyroid nodule diagnosis. Concurrent use of miR144-3p, miR148a-3p, and miR190a-5p increased sensitivity, but the AUC improvement was not statistically significant. Similarly, the combined use of miR134-5p and miR144-3p did not significantly improve the AUC. The combination of miR148a-3p, miR134-5p, and miR34b-3p showed a better Youden's index, but again, the AUC improvement was not significant. These findings suggest that combined miRNA use may serve as diagnostic markers for thyroid nodules. However, the lack of significant AUC improvement could be due to patient demographic variations, differences in experimental methods, or sample size discrepancies. Given these results, miRNA combinations show potential for thyroid nodule identification. However, further research is needed to refine miRNA selection and determine their clinical effectiveness in improving thyroid nodule diagnosis.
This study is limited by a small sample size, largely due to funding constraints, which reduces the statistical power and generalizability of the findings. While miRNA expression showed diagnostic potential, the lack of significant improvement in AUC when combining miRNAs suggests that further validation with larger cohorts is needed to confirm their clinical utility.
Conclusion
In summary, our research indicates that miR144-3p and miR190a-5p exhibit promise as biomarkers for distinguishing between benign and malignant thyroid nodules, as well as individuals without any thyroid abnormalities. The expression levels of these miRNAs exhibited notable distinctions in blood samples obtained from individuals diagnosed with papillary thyroid cancer, individuals with benign nodules, and individuals without any thyroid abnormalities. The inclusion of miR144-3p, miR148a-3p, and miR190a-5p in the test resulted in an enhanced level of sensitivity. The findings of this study may underscore the potential of utilizing circulating miRNAs as a diagnostic modality for thyroid cancer. However, additional investigation is required to validate their clinical efficacy.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241291637 - Supplemental material for The Implications for Clinical Practice of Circulating miR144-3p and miR190a-5p as Promising Diagnostic Biomarkers for Thyroid Nodule Differentiation
Supplemental material, sj-docx-1-tct-10.1177_15330338241291637 for The Implications for Clinical Practice of Circulating miR144-3p and miR190a-5p as Promising Diagnostic Biomarkers for Thyroid Nodule Differentiation by Basima Hassan, Mohamed M. Omran, Magdy M. Youssef, Manar Refaat, Amr Abouzid, Abdel Hady A. Abdel Wahab and Yasser M. Bakr in Technology in Cancer Research & Treatment
Footnotes
Acknowledgements
We would like to express our deepest gratitude to Khaled T. Dardeer, whose expertise and insights as a statistician were invaluable to this research. His rigorous analysis and thoughtful interpretation of the data have significantly enhanced the quality and credibility of our work. His dedication and professionalism have not only made this paper possible but also instilled in us a greater understanding of statistical methodologies. We are truly fortunate to have had the opportunity to work with him.
Authors’ Contributions
Basma Hassan and Yasser M Bakr has performed all the lab work and wrote the manuscript. Magdy M.Youssef, Manar Refaat, Amr Abouzid and Tarek M.Emran are the clinical oncologist and surgeon who was responsible for providing all samples and clinical data. Mohamed M. Omran is the principal investigator and the main supervisor of this research work in accordance with Abdel Hady A. Abdel Wahab where both of them had read, revised and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study was approved by the Institutional Review Board and the Ethical Committee of the Faculty of Medicine at Mansoura University in Mansoura, Egypt, under the code number R.21.08.1415.
Ethics Approval Declaration
The protocol was approved by the Institutional Review Board and the Ethical Committee of the Faculty of Medicine at Mansoura University in Mansoura, Egypt (ID: R.21.08.1415). All participants in the study provided written, informed consent before the collection of samples.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Human Ethics and Consent to Participate Declarations
The research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. Information revealing the patient's identity has been avoided. All patients have been identified by numbers or aliases and not by their real names.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
