Abstract
Keywords
Introduction
In 2014, an experiment employing ultra-high dose rate (UHDR), with an average dose rate above 40 Gy/s, electron beams to irradiate lung tumors in mice showcased the normal tissue-sparing effect of UHDR while maintaining tumor-killing efficacy compared to conventional dose rate (CONV).
1
This method of delivering radiation doses at UHDR is called FLASH radiotherapy. The radiobiological observation of normal tissue sparing and effective tumor killing by UHDR irradiation is known as the FLASH effect.1,2 FLASH radiotherapy not only significantly reduces the treatment time for patients, but also enhances radiotherapy benefits.3,4 The normal tissue sparing of the FLASH effect is reflected in two aspects. Firstly, it induces less structural damage to tissues and organs, preserving their structural and functional integrity more effectively.
5
Secondly, FLASH radiotherapy reduces the infiltration of inflammatory cells and the secretion of inflammatory cytokines, thereby mitigating the inflammatory response induced by radiation.1,6 Over the past decade, FLASH radiotherapy has achieved many encouraging results, with observations of the FLASH effect in both
Ion irradiations, which involve using proton and carbon ion beams, represent a promising treatment approach for cancer patients. For some tumors that are not sensitive to x-ray therapy, such as chondrosarcoma and chordoma, ion radiation can achieve better tumor control. Numerous clinical centers have incorporated ion irradiation systems for tumor treatment. Compared with electron beams and x-rays, protons have a larger energy range and wider range, enabling continuous or pulsed UHDR irradiation with significant potential in UHDR radiation. Among current radiotherapy systems, proton radiotherapy equipment emerges as one of the prime candidates for FLASH radiotherapy. 7 Recently, Walter Tinganelli et al successfully realized FLASH radiation using the RF-knockout method at the HIT medical synchrotron in Heidelberg, 8 which not only verified that UHDR carbon ion can achieve FLASH effect, but also opened new avenues for studying FLASH radiation. Currently, carbon ion is mainly generated by synchrotrons. The RF-knockout method is considered the optimal choice for synchrotron FLASH irradiation. 9
Several studies on FLASH ion irradiations suggest that UHDR ion irradiations can potentially decrease radiation-induced normal tissue toxicity.5,10 However, these investigations also highlight that UHDR ion irradiations may not completely alleviate all radiation-induced normal tissue toxicity; instead, it tends to mitigate specific aspects of toxicity.11,12 For example, the UHDR proton beam has been shown to reduce pericardial edema in zebrafish embryos, but it does not alleviate spine curvature compared to the CONV. 13 Similar findings have been reported in studies involving UHDR photon and electron irradiation.14,15 The variability in outcomes could be attributed to factors such as radiation dose or specific outcome indicators. Therefore, this systematic review and meta-analysis aims to evaluate the experimental results of UHDR ion irradiations on animals and comprehensively outline the similarities and differences among various experimental outcomes.
Materials and Methods
Protocol Registration and Ethics Statement
The protocol was pre-registered in PROSPERO under the registration number: CRD42023425106. This systematic review and meta-analysis study were following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA-2020) guidelines. 16 This is a meta-analysis using data from published articles and does not require ethical approval.
Search Strategy
Two investigators (YL.C. and ZQ.L.) independently searched articles published and indexed in PubMed, Web of Science, and EMBASE databases, with retrieval dates from the time of each databases inception to 20 March 2023. We used a broad combination of search terms relating to UHDR, proton radiation or carbon ion radiation. The full search strategy is described in Additional file: Table S1. Relevant reviews and references were screened to identify additional papers of relevance that may have been missed during the database search process. Only full-text studies in English were included.
Eligibility Criteria
The inclusion criteria for this study included investigations involving the irradiation of animals with CONV and UHDR proton or carbon ion beams, excluding
Population/Animals, Intervention, Comparison, Outcome, and Study Design (PICOS) Strategy for Systematic Review.

Study Selection, Data Extraction and Risk of Bias Assessment
The articles and indexes retrieved from the mentioned database were imported into Endnote X9 software in RIS format files. Duplicate articles and indexes were identified and removed using the Find Duplicates feature in Endnote X9. Subsequently, potential eligible articles underwent an initial screening based on titles and abstracts, guided by the predefined inclusion and exclusion criteria. A thorough full-text review followed, with both processes independently executed by two reviewers (Q.W. and MY.T.). Discrepancies in assessments were resolved through consultation with a third reviewer (TQ.D.).
Information extraction from the articles included in this systematic review was carried out independently by two researchers (HT.L. and RF.L.). The extracted details encompassed the first author's name, publication year, country, animal species, sample size, particle type, cell line, irradiation dose, dose rate, and outcomes. For continuous variables, mean and standard deviations (SD) were obtained. In cases where data were presented graphically, the numerical values were extracted using GetData Graph Digitizer software (version 2.26). The alluvial diagram was generated using RAWGraphs.
The SYRCLE risk of bias tool for animal studies was employed to assess the risk of bias. 17 Two trained researchers (JH.T. and SL.S.) independently evaluated each included study and any disagreements were resolved by consulting a third researcher (KH.Y.).
Statistical Analysis
To provide a concise understanding of the research status in this area, we utilized RAWGraphs (https://app.rawgraphs.io/) to construct an alluvial diagram and visualize the interconnectedness among three key categories: animal models, irradiation sites, and outcome indicators. Statistical analyses were conducted using R statistical software (version 4.3.0). Continuous data were summarized with mean, standard deviation (SD), and sample size. Survival data were processed using the method outlined by Tierney et al to calculate hazard ratio (HR).
18
Differences were expressed as standard mean differences (SMD) with 95% confidence intervals (CI). Meta-analyses were executed employing a random-effects model with the restricted maximum likelihood method. The
Results
Search Results
The study selection process was summarized using a PRISMA flowchart (Figure 1). Initially, 2867 records were identified in the search. After removing 623 duplicate records and screening the titles and abstracts of the remaining articles, 34 articles remained eligible for full-text screening. Figure 1 illustrates the reasons for excluding 17 studies. Ultimately, 17 studies met the inclusion criteria, and data were extracted. Among these, only one study utilized a carbon ion beam, while the remaining studies employed a proton beam.5,10–13,19–30

PRISMA flowchart.
Study Characteristics
Table 2 provides a summary of the characteristics of all included studies. Notably, all radiation doses administered were delivered in a single fraction. The murine model emerged as the predominant animal model, with eleven studies conducted on mice, encompassing six strains.5,10,11,20–23,27–30 Additionally, two studies were conducted on rats involving two strains.12,24 The primary outcomes assessed in these studies included survival, soft tissue toxicity, inflammatory response, body weight, and tumor volume. Furthermore, four studies reported experimental findings on zebrafish embryos,13,19,25,26 focusing on outcomes such as survival, pericardial edema, body length, and spinal curvature. Some studies also reported additional experimental results, covering aspects like lymphedema, cell proliferation, DNA damage, cognitive and behavioral testing, as well as tumor metastasis (Figure 2).

Alluvial diagram of included studies on the research landscape. The alluvial diagram visually connects three categories - animals, irradiation sites, and outcomes - to illustrate normal tissue sparing in various tissues at UHDR irradiation. The width of the colored curve is positively related to the frequency of outcomes connecting two categories, and the height of the black rectangle is directly proportional to the frequency of outcomes inside each subgroup and category.
Characteristics of Included Studies.

Risk of Bias
The results of the risk of bias assessment for each included study are depicted in Figure 3. The most prevalent domain with an unclear risk was noted in 55% of cases, primarily due to a lack of a precise procedure overview. Among the studies, one implemented blinding of caregivers and/or investigators during the experiment and five incorporated blinding of outcome assessors. Only one study explicitly described the use of a random number table for grouping rats, while eleven studies did not provide details on the generation and application of the allocation sequence, resulting in a high risk of bias in the sequence generation domain. Two of the seventeen studies reported similar baseline characteristics, while the remaining studies did not specify the differences in baseline characteristics between the UHDR and the CONV groups. Random housing of animals was observed in 6 out of 17 studies, and only one study illustrated animals being selected randomly for outcome assessment. Seven studies provided reasons for incomplete outcome data, while four studies did not. The risk of reporting bias across all included studies was deemed unknown, as none explicitly stated a review of selective outcome reports.
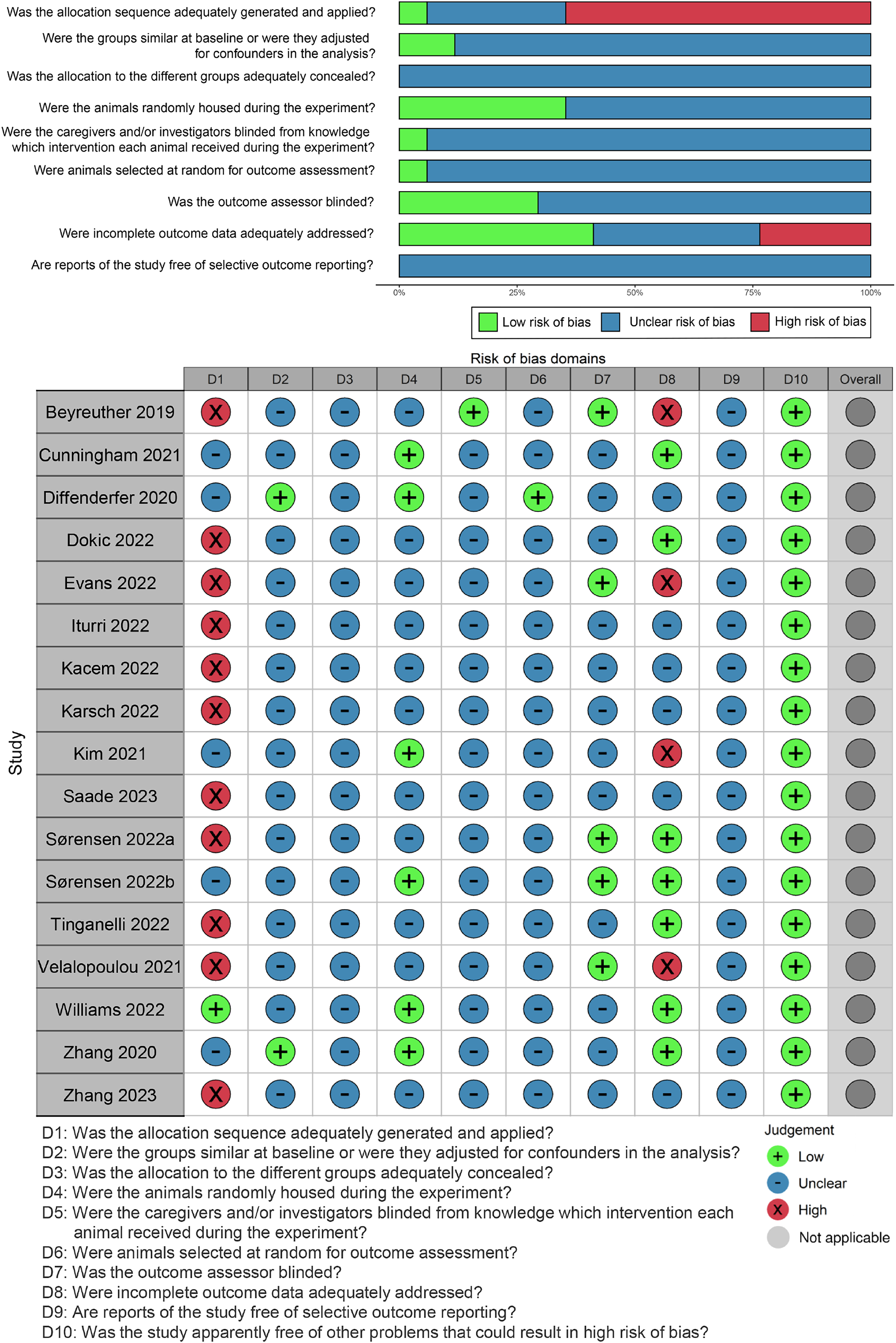
SYRCLE's risk of bias tool for animal studies.
Survival
A total of 8 studies utilizing proton beams reported survival outcomes post-irradiation, involving mice (n = 4) and zebrafish embryos (n = 4).13,19,23,25–27,29,30 Among these, one study specifically detailed the survival of zebrafish embryos after 30 Gy and 40 Gy irradiation.
13
Forest plots displaying the meta-analysis of survival across different species are presented in Figure 4a and 4b. The meta-analysis for mouse models included four studies,23,27,29,30 revealing superior survival in the UHDR group compared to the CONV group (HR 0.48, 95% CI 0.29 to 0.78,

Forest plots for survival. (a) Mouse. (b) Zebrafish embryo. * numerical values were extracted by GetData Graph Digitizer software.

Subgroup analysis for the survival of zebrafish embryos. Based on the irradiation dose, those studies with irradiation doses of 30 Gy or less were included in the 30 Gy group, while the remaining were included in the 40 Gy group. * numerical values were extracted by GetData Graph Digitizer software.

Box-and-whisker plot of irradiation dose in normal tissue sparing of ultra-high dose rate. The upper and lower limits of the box, represent the 75th and 25th percentiles, respectively. The line within the box represents the 50th percentile (median), and the whiskers represent the ranges.
Tumor Volume/Control
Six studies encompassing five types of tumors (oral carcinoma, pancreatic tumor, osteosarcoma, sarcoma, and mammary carcinoma) reported alterations in tumor volume and control following irradiation (Figure 7 and Additional file: Table S3).5,20,21,27–29 Notably, all transplanted tumors were established subcutaneously. Across most studies, no significant difference was observed in tumor control between the UHDR and CONV groups. However, in one study employing a carbon ion beam, superior tumor control was evident in the UHDR group compared to the CONV group.

Heatmap for tumor volume/control, body weight and soft tissue toxicity in murine models. Green indicates that UHDR irradiation have the normal tissue-sparing effect compared to CONV irradiation, while tangerine indicates that UHDR irradiation has a stronger inhibitory effect on tumors. Blue indicates no difference between the two irradiation dose rates.
Body Weight Soft Tissue Toxicity
The body weight of mice after irradiation at different dose rates was documented in two included studies,12,30 with no significant differences observed between the two groups (Figure 7 and Additional file: Table S4).
Soft tissue toxicity was reported in a total of six studies utilizing mouse models (Figure 7 and Additional file: Table S5).5,10,11,20,28,29 Skin toxicity, both early (n = 4) and late (n = 1), was the most commonly employed indicator for assessing normal tissue toxicity in mouse models.10,11,20,28,29 Four studies reported changes in leg contracture, tissue fibrosis, muscle atrophy, morphology, and structure post-irradiation to reflect soft tissue toxicity.5,20,28,29 Only one proton beam study indicated that early skin toxicity between the UHDR and the CONV groups was comparable at 45 Gy. 29 However, across the remaining studies, whether utilizing proton or carbon ion beams, soft tissue toxicity was consistently milder in the UHDR group than in the CONV group.
Embryo Length, Spinal Curvature and Pericardial Edema
Three studies reported the embryo length of zebrafish embryos post-irradiation (Figure 8 and Additional file: Table S6).13,25,26 One study indicated that the impact of ion irradiations at different dose rates on embryo length was comparable. 25 However, two studies suggested that the embryo length was longer in the UHDR group.13,26

Heatmap for embryo length, spinal curvature and pericardial edema in zebrafish embryos. Green indicates that UHDR irradiation have the normal tissue-sparing effect compared to CONV irradiation, while blue indicates no difference between the two irradiation dose rates.
Three studies were conducted for spinal curvature and pericardial edema assessment in zebrafish embryos (Figure 8 and Additional file: Table S7).13,19,26 One study concluded that spinal curvature was lower in the UHDR group, 26 while two reported equivalent spinal curvature endpoints between groups.13,19 Pericardial edema in zebrafish embryos was significantly influenced by irradiation dose. In all three studies, zebrafish embryos exposed to UHDR exhibited milder pericardial edema at radiation doses of only 23 Gy and 30 Gy compared to CONV irradiation. At radiation doses of 10 Gy, 15 Gy, 31 Gy, 40 Gy and 42 Gy, there was no significant difference in pericardial edema between the two groups.
Cytokine and Immune Cell
Four studies reported outcomes related to the inflammatory response, encompassing cytokines and immune cells, following varied irradiation dose rates in murine models (Table 3).20,22,24,29 One study found no differences in infiltrating immune cells between the two dose rates, whether in the tumor, blood, or normal brain tissue. 24 Conversely, two studies demonstrated that UHDR irradiation reduced the activation of microglia and myeloid cells in normal tissues.22,29 Except for some cytokines (CXCL1, IL-6, GM-CSF, G-CSF, HMGB1, TGF-β) that exhibited differences in both groups, most cytokines showed no significant difference after irradiation.20,22,29
Studies Reporting Inflammatory Response in Murine Models.
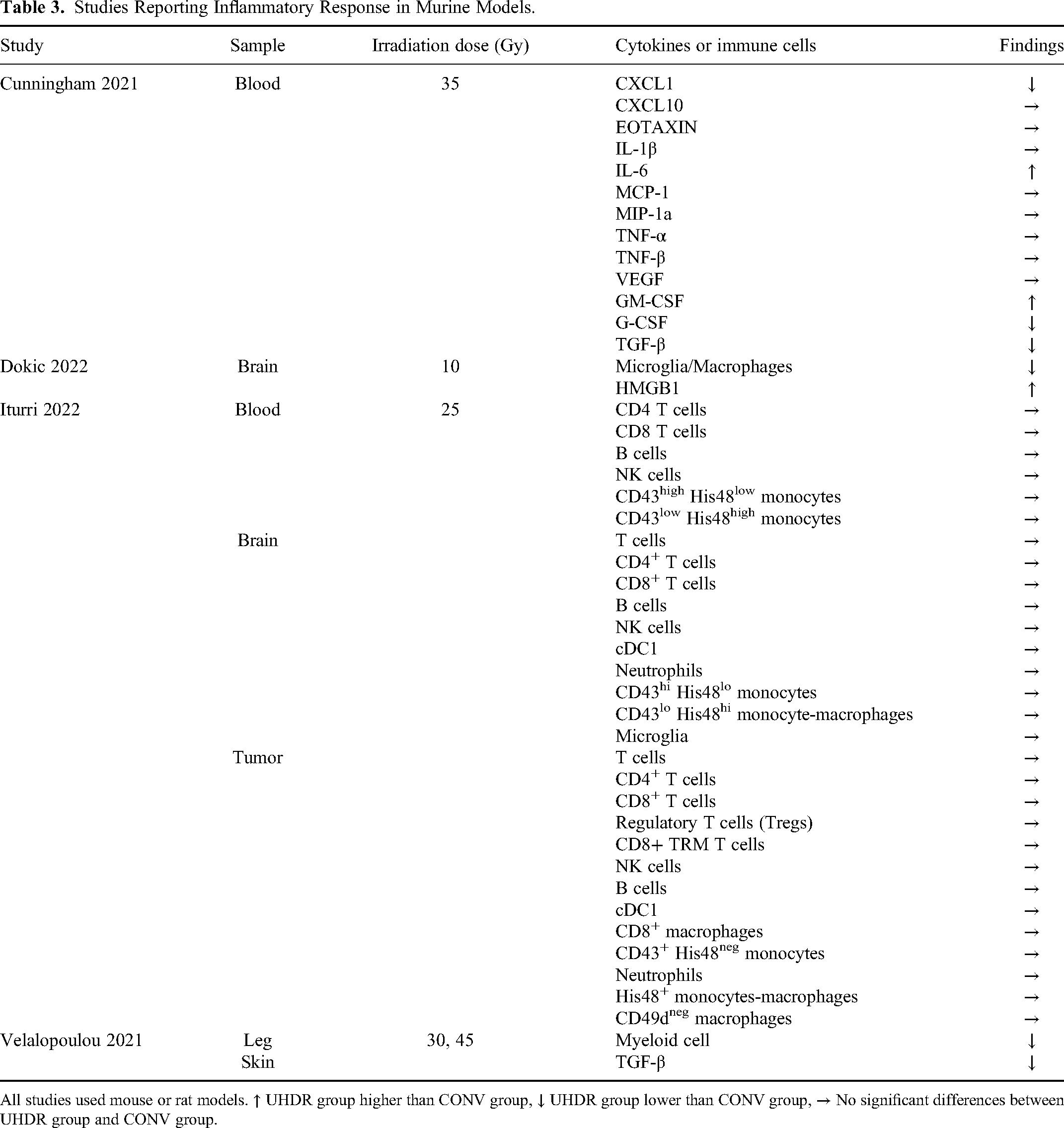
All studies used mouse or rat models. ↑ UHDR group higher than CONV group, ↓ UHDR group lower than CONV group, → No significant differences between UHDR group and CONV group.
Discussion
Summary of Key Findings
In this study, we conducted a comprehensive review and analysis of current evidence from animal studies investigating the impact of UHDR ion irradiations on normal tissues and tumors. Our analysis comprised 17 original studies involving two species, namely murine (mouse and rat) and zebrafish embryos, meeting the specified inclusion criteria. Despite inherent heterogeneity across these studies, a pooled analysis indicates that the utilization of UHDR leads to improved survival and comparable or even enhanced tumor control in animals, with most of the evidence originating from studies on mice. In murine models, consistent reports on changes in body weight between the two groups indicated that the irradiation dose rate did not influence body weight. Despite variations in damage assessment methods and outcome indicators for evaluating soft tissue toxicity, most studies results support that UHDR ion irradiations can achieve normal tissue sparing. The results on embryo length were inconclusive, with two out of three studies suggesting that the embryo length of zebrafish embryos was longer after UHDR irradiation than CONV irradiation. Similarly, findings regarding spinal curvature and pericardial edema in zebrafish embryos were inconsistent between the two groups. In mouse models, a limited number of cytokines and immune cells differed at different dose rates, adding complexity to our understanding of the immunological response to UHDR irradiation.
The FLASH effect of UHDR photon and electron has been confirmed in various experimental animal models, spanning mice, rats, zebrafish embryos, pigs, cats, and dogs, and involving multiple organs such as lungs, skin, intestines, and brain.1,15,31–34 However, the 17 studies on UHDR ion irradiation included in this study involved only two animal models, namely murine and zebrafish embryos. Therefore, the current evidence may not comprehensively represent the effects of UHDR ion irradiations across diverse animal models and strains, necessitating additional research before widespread clinical applications. Furthermore, there are inconsistencies in assessment and measurement methods across some studies, raising concerns about the comparability and reliability of results. Standardization of assessment and measurement methods is imperative to facilitate more robust comparisons and draw reliable conclusions from various UHDR ion irradiations studies.
The findings of the present study highlight the unsatisfactory reproducibility of normal tissue sparing by UHDR ion irradiations, with the sparing effect observed in some studies but not consistently replicated in others. This phenomenon was notably pronounced in zebrafish embryos. The observed variability in results is a complex interplay of multiple factors. While all included studies indicated that UHDR ion irradiations can achieve normal tissue-sparing effects under specific experimental designs and conditions, this sparing effect is partial and not universally applicable based on existing evidence. The manifestation of normal tissue sparing varies under distinct experimental designs and conditions, and in some cases, it may not occur at all. Survival is a comprehensive index reflecting the effects of irradiation, and meta-analysis demonstrated that UHDR ion irradiations significantly improved survival in mice compared to the conventional group. However, this result was not consistently reproduced in zebrafish embryos. Subgroup analysis further indicated that the effect of UHDR ion irradiations on the survival of zebrafish embryos was associated with the irradiation dose, with a tendency for improved mean survival rates at a 30 Gy irradiation dose, whereas a 40 Gy dose did not exhibit the same trend.
Possible Mechanisms
Several hypotheses have been proposed to elucidate the FLASH effect, encompassing radiolytic oxygen consumption, radical-radical interactions, differential DNA damage, and differential immune response. Notedly, there is still no consensus on these hypotheses.
Radiolytic oxygen consumption: This mechanism posits that intracellular oxygen is rapidly consumed and cannot be replenished after UHDR irradiation, creating a hypoxic environment that enhances cell irradiation resistance. Ionizing radiation primarily generates radicals by hydrolyzing water in the cytoplasm, leading to their reaction with oxygen and the production of secondary radicals and reactive oxygen species (ROS).
35
This process consumes oxygen, decreasing oxygen concentration within the cell.36,37 When the oxygen concentration of tissues or organs is artificially increased, the FLASH effect will not occur.
38
Therefore, oxygen concentration is a critical factor influencing the FLASH effect. While the oxygen consumption mechanism was initially considered the primary explanation for the FLASH effect, it is now being scrutinized. For example, it fails to account for equivalent tumor control, challenging its status as the primary mechanism driving the FLASH effect. Radical-radical interactions: The hypothesis of radical-radical interactions builds upon the oxygen consumption hypothesis as the key to the FLASH effect. Following UHDR irradiation, local high concentrations of radicals interact with each other and trigger chain reactions. But some of these radicals interact to produce stable molecules that terminate the chain reaction of radicals.
39
This process significantly reduces the amount of ROS available to react with biological targets such as lipids and DNA. Consequently, the detrimental effects of ionizing radiation on cells are mitigated. Notably, the distinct metabolic characteristics of normal cells and cancer cells also play a pivotal role in this hypothesis. Normal cells possess robust antioxidant reserves and can efficiently regulate the levels of antioxidant enzymes and active metal ions (eg, Fe2+) to eliminate ROS; therefore, they can handle the increased ROS generation without increased damage. In contrast, cancer cells lack such antioxidant reserves to cope with additional ROS, which increases their oxidative stress levels. Moreover, the quantity of active metal ions in cancer cells is typically higher than in normal cells, and the oxidation chain reaction triggered by the Fenton reaction can sustain the toxicity of ionizing radiation in cancer cells. Therefore, the damage of UHDR radiation to cancer cells is at least maintained.40,41 Differential DNA damage: There is evidence to suggest that the mechanism involved in DNA damage formation may be related to the occurrence of FLASH effect.
1
Other authors have demonstrated that the DNA damage sites and numbers indicated by γ-H2AX and TRP53 binding protein 1 (53BP1), widely used double-stranded (ds) DNA break markers, are consistent after tumor cells are exposed to CONV and UHDR radiation.14,42 Therefore, cancer cell death induced by radiation at different dose rates is similar. For normal cells, the sites and quantities of γ-H2AX and 53BP1 are not consistent after UHDR irradiation.6,14 One study indicated that reducing the production of 53BP1 through free radical recombination is considered the reason for the sparing effect of UHDR radiation on normal cells,
38
but the inconsistency between γ-H2AX and 53BP1 was not explained. Some researchers believe that the γ-H2AX are indicators of the initiation of the dsDNA break repair process, not of dsDNA break explicitly. If the nature of DNA damage between CONV and UHDR differs, then γ-H2AX cannot be used as a direct comparison indicator.
43
Differential immune response: The extremely short irradiation time associated with UHDR irradiation has been suggested to significantly reduce the number of circulating immune cells exposed to irradiation, potentially alleviating immune system damage.
44
Irradiation with different dose rates can also differentially activate immune cells and alter the tumor microenvironment.45,46 Compared to CONV irradiation, UHDR irradiation may differentially regulate the expression of certain cytokines, such as a reduction in transforming growth factor-β (TGF-β) expression.1,20,29 However, the specific regulatory mechanism underlying these differential effects remains unclear. Indeed, the hypothesis of a differential immune response is primarily derived from observational results following UHDR irradiation, and there is a lack of direct evidence establishing a causal relationship between the immune response and the FLASH effect. Consequently, the role of the differential immune response after UHDR irradiation as a catalyst for the FLASH effect remains uncertain.
Limitations and Future Research
The limitations of this review should be taken into consideration when interpreting the results. Firstly, restricting the language of included studies to English may introduce a potential bias, as relevant research in other languages may have been overlooked. Secondly, most of the included studies did not explicitly report blinding or differences in baseline characteristics between the experimental and control groups, raising concerns about potential performance and selection biases. Thirdly, the reliability of our findings is compromised by the inconsistency in the tools used for outcome evaluation across studies. Addressing this issue through harmonizing outcome evaluation tools is critical for advancing research in this field.
FLASH radiotherapy is regarded as one of the most promising new radiotherapy modalities due to the normal tissue sparing under UHDR radiation. At present, there are at least three clinical trials of UHDR radiation (FAST-01: NCT04592887, IMPulse: NCT04986696, and FAST-02: NCT05524064) that are under consideration. However, several challenges may prevent the widespread clinical adoption of this technology, partially due to the inadequate understanding of the cell damage mechanisms under UHDR radiation. Enhancing research on the physicochemical processes occurring under UHDR radiation will help to understand the biological mechanisms of the FLASH effect. To achieve this, it is necessary to first clarify the radiation source, corresponding technical parameters, and experimental conditions and standardize them, which will ensure the stable occurrence of the FLASH effect and accurate comparison between different experiments. Moreover, confirming the occurrence of the FLASH effect in patients and real-time dose monitoring are crucial in clinical practice for the clinical translation of FLASH radiation, guiding treatment plans, and improving efficacy and safety.
Conclusion
UHDR ion irradiation shows promise in reducing radiation-induced toxicity, although the reproducibility of normal tissue sparing remains a concern. In addition, standardizing the technical parameters and experimental conditions of FLASH radiation is necessary to ensure the stable occurrence of the FLASH effect and accurate comparison between different experiments.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241289990 - Supplemental material for Examining the Occurrence of the FLASH Effect in Animal Models: A Systematic Review and Meta-Analysis of Ultra-High Dose Rate Proton or Carbon Ion Irradiation
Supplemental material, sj-docx-1-tct-10.1177_15330338241289990 for Examining the Occurrence of the FLASH Effect in Animal Models: A Systematic Review and Meta-Analysis of Ultra-High Dose Rate Proton or Carbon Ion Irradiation by Xun Wu, MD, Hongtao Luo, MD, Qian Wang, MD, Tianqi Du, MD, Yanliang Chen, MD, Mingyu Tan, MD, Ruifeng Liu, MD, Zhiqiang Liu, MR, Shilong Sun, MR, Kehu Yang, PhD, Jinhui Tian, PhD, and Qiuning Zhang, MD in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
All authors sincerely thank Professor Wang Xiaohu for his guidance in this paper.
Author Contributions
Xun Wu: Conceptualization, Methodology, Software, Writing - Original Draft, Hongtao Luo: Validation, Funding acquisition, Qian Wang: Investigation, Tianqi Du: Formal analysis, Yanliang Chen: Data Curation, Mingyu Tan: Investigation, Ruifeng Liu: Data Curation, Funding acquisition, Zhiqiang Liu: Formal analysis, Shilong Sun: Data Curation, Resources, Kehu Yang: Visualization, Jinhui Tian: Visualization, Formal analysis, Qiuning Zhang: Project administration, Funding acquisition, Writing - Review & Editing, Supervision.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics Statement
This is a meta-analysis using data from published articles and does not require ethical approval.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Central guidance local science and technology development program (Grant No. 24ZYQA029), the National Key Research and Development Program of China (No. 2022YFC2401500), Science and Technology Project of Lanzhou City (Grant No. 2023-1-9), Gansu Province Project of Science and Technologies (Grant No. 20JR10RA680, 22CX8JA149) and the Lanzhou heavy Ion Accelerator High-end user Project (HIR20GY007). The role of the funder: The funder had no role in the design of the study, the collection, analysis and interpretation of the data, writing of this manuscript or the decision to submit it for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
