Abstract
Through meticulous examination of lymph nodes, the stage and severity of cancer can be determined. This information is invaluable for doctors to select the most appropriate treatment plan and predict patient prognosis; however, any oversight in the examination of lymph nodes may lead to cancer metastasis and poor prognosis. In this review, we summarize a significant number of articles supported by statistical data and clinical experience, proposing a standardized evaluation protocol for lymph nodes. This protocol begins with preoperative imaging to assess the presence of lymph node metastasis. Radiomics has replaced the single-modality approach, and deep learning models have been constructed to assist in image analysis with superior performance to that of the human eye. The focus of this review lies in intraoperative lymphadenectomy. Multiple international authorities have recommended specific numbers for lymphadenectomy in various cancers, providing surgeons with clear guidelines. These numbers are calculated by applying various statistical methods and real-world data. In the third chapter, we mention the growing concern about immune impairment caused by lymph node dissection, as the lack of CD8 memory T cells may have a negative impact on postoperative immunotherapy. Both excessive and less lymph node dissection have led to conflicting findings on postoperative immunotherapy. In conclusion, we propose a protocol that can be referenced by surgeons. With the systematic management of lymph nodes, we can control tumor progression with the greatest possible likelihood, optimize the preoperative examination process, reduce intraoperative risks, and improve postoperative quality of life.
Background
Standards for Examined Lymph Node Counts
The examined lymph node (ELN), a prognostic factor for solid tumors, such as colorectal cancer, lung cancer, and gastric cancer, represents whether or not lymph node dissection at the surgical site was adequately performed. This factor is important for cancers characterized by more lymph node metastasis, more recurrence and poorer five-year overall survival and is considered a key indicator for evaluating surgical quality. 1 The guidelines from the National Comprehensive Cancer Network (NCCN) recommend that a minimum number of examined lymph nodes should be established for each tumor. It allows standardization and proceduralization, ensures surgical quality control, and reduces operative time and postoperative complications. For example, as early as 1990, the World Congress of Gastroenterology in Sydney recommended at least 12 ELNs for colorectal cancer. This standard was not only endorsed by the College of American Pathologists (CAP) and the American Joint Committee on Cancer (AJCC) but also incorporated into NCCN evidence-based guidelines.2-4 Recently, there has been another opinion about escalating this standard to 15 lymph nodes. 5 For gastric cancer, the Japanese Classification of Gastric Cancer from the Japanese Gastric Cancer Association (JGCA) and the eighth edition of the AJCC Manual of Gastric Cancer both recommend that the number of ELNs be at least 16.6,7 Some researchers have also questioned the appropriateness of this standard. Further investigations revealed that 20 nodes are needed, while others reported that 25 nodes are required.8,9 For non-small-cell lung cancer (NSCLC), the International Association for the Study of Lung Cancer (IASLC) suggested a minimum of 6 ELNs for precise nodal staging, which included 3 at the N1 station and 3 at the N2 station. 10 The European Society for Thoracic Surgery (ESTS) suggested that at least 6 hilar or mediastinal lymph nodes be dissected. 11 A study published in the Journal of Clinical Oncology stated that the optimal number of nodes used to stratify node-negative and node-positive NSCLC should be 16. 12 However, a subsequent study argued for lowering the standard to 10. 13 For head and neck squamous cell carcinoma, the optimal number ranges from 16 to 18 to 19 to 20. A conclusion has not yet been reached.14-18 For renal cell carcinoma, the threshold can be as low as 3. 19 The minimum number of ELNs for pancreatic cancer has not been set, as there is still a great deal of variation between guidelines. First, it was not addressed in the NCCN guidelines. Second, according to the AJCC and the Union for International Cancer Control (UICC), a minimum of 10 lymph nodes should be dissected for staging. Third, according to the European Society for Medical Oncology (ESMO), 15 or more is favored. 20 A study published in the Annals of Surgery proposed a minimum number of 11 and an optimal number of 19 as new validated standards for intraoperative node dissection and postoperative patient stratification in pancreatic cancer patients. 21 For esophageal squamous cell carcinoma, the minimum number and the optimal number were 14 and 18, respectively. 22 (Table 1).
Standards for ELN Count in Various Cancers.

Proper management of ELNs can greatly improve the prognosis of cancer patients. In contrast, it can have consequences, including occult node metastasis, lymph node micrometastasis, stage migration and immune impairment.
Occult Node Metastasis and Lymph Node Micrometastasis
Occult node metastasis refers to positive lymph node (PLN) missed during preoperative palpation and imaging but accidentally found in postoperative tissue sections, which commonly occurs in patients at clinical stage N0/N1.23-25 Similarly, lymph node micrometastases are clusters of microscopic tumor cells sneaking in lymph nodes; these clusters are too small to be detected on preoperative examination and are even overlooked during routine macroscopic pathology. These clusters of cells are defined by the AJCC as being between 0.2 mm and 2.0 mm in diameter. Further examination requires more sensitive techniques, such as immunohistochemical staining, flow cytometry, and reverse transcriptase polymerase chain reaction. 7 Unfortunately, even with the application of the above technologies, a high detection rate cannot be achieved. Research indicates that among patients tested for lymph node-negative status through carcinoembryonic antigen-specific nested RT‒PCR, 54% end up with micrometastatic disease at surgery. 26 It is vital to detect lymph node micrometastasis as much as possible to make accurate clinical decisions and to provide prompt intervention in early-stage patients with this risk factor.
Stage Migration
Occult nodal metastasis and lymph node micrometastasis are characteristic of stage migration, with an insufficient number of ELNs being the cause of stage migration. Stage migration is a type of Will Rogers phenomenon, which in this context refers to changes in the N stage. More specifically, a group of node-positive patients may be misclassified as node-negative in clinical practice due to inadequate preparation or operation, representing the occurrence of missed diagnoses.27,28 With further treatment, micrometastasis is identified, and patients previously categorized as “good stage” are assigned to “bad stage”. Although the prognosis of reclassified patients was worse than that of the remaining patients of “good stage”, it was better than that of other patients of “bad stage”. Survival outcomes appeared to improve in each group, while the individual outcomes remained the same.
The medical oncologist determines whether postoperative radiotherapy or chemotherapy is necessary for a patient according to his or her N stage. Those prone to stage migration will neither receive an appropriate treatment plan from the oncologist nor provide credible treatment feedback with the oncologist. Statistically, the reliability of survival data for the pN0 population is compromised. In short, stage migration tampers with the whole picture.
Immune Impairment
Immune impairment refers to the weakening of anti-tumor immunity due to the expanded dissection of tumor-draining lymph nodes (TdLNs). This finding is contrary to the generally accepted theory that “the more lymphadenectomy, the better.” Since these nodes undergo primary tumor antigen exposure and subsequent antigen-specific immune activation, the roles of harboring effector lymphocytes cannot be exaggerated. 29 In addition to resulting in increased susceptibility to postoperative infections, immune impairment further hampers the implementation of adjuvant immunotherapy due to the lack of appropriate treatments.
These three consequences cover three phases: preoperative examination, intraoperative lymphadenectomy, and postoperative therapy for cancer patients. The questions to be answered include how to detect as many PLNs as possible by noninvasive approaches preoperatively, how to remove as many PLNs as possible and how to preserve as many immune-dependent lymph nodes as possible intraoperatively. These three questions have received close attention from oncologists in recent years. This review summarizes multi-institutional, multinational, retrospective and prospective cohort studies over the past two decades, covering the entire research process from patient selection to data analysis to outcome assessment. It is distilled into a comprehensive evaluation protocol for cancer patients, aimed at providing guidance for clinical research and practice (Figure 1).

The evaluation protocol for examined lymph nodes in cancer patients. The whole protocol involved preoperative examination, intraoperative lymphadenectomy and postoperative immunotherapy. Preoperative examination aided by deep learning models categorizes patients into three groups: clinically node-negative, low-risk and high-risk in clinically node-positive. Those at risk will receive the necessary neo-adjuvant therapy. Following the preliminary screening, patients will undergo further surgical procedures. Lymphadenectomy guided by statistical models categorizes patients into three groups: pathologically node-negative, low-risk and high-risk in pathologically node-positive. Those at risk will receive the necessary postoperative immunotherapy.
Preoperative Examination
Ultrasound, positron emission tomography (PET), computed tomography (CT) and magnetic resonance imaging (MRI) are commonly used as noninvasive preoperative approaches for screening lymph nodes in various cancers. The combination of PET/CT has been recognized as the most reliable diagnostic solution for the diagnosis of occult lymph nodes in lung cancer and gastric cancer, with higher sensitivity and specificity in staging than CT alone and higher sensitivity than PET alone.30-32 This is because CT provides anatomical information to determine the location of the node, and PET provides metabolic information to determine whether the node is positive. The two complement each other to form a more complete picture. In head and neck squamous cell carcinoma, a meta-analysis showed no difference in the sensitivity or specificity of the above imaging approaches, except that CT displayed greater specificity than ultrasound. 33 However, in addition to intratumor heterogeneity, there are inherent limitations in human vision. Subjective differences only make it more difficult to achieve reproducibility and consistency in examination. 34 Relying solely on manual inspection will result in a large number of missed cases of occult node metastasis. 35 In NSCLC, 12.9%-39.3% of lymph node metastases are not identified by PET/CT.36-39 In gastric cancer, diffusely growing and mucus-containing tumor types are likely to be falsely identified as negative for low uptake of fluorodeoxyglucose. 40 In head and neck squamous carcinoma, either PET/CT or MRI lacks accuracy in the diagnosis of occult lymph nodes. 41 With the development of molecular imaging toward quantitative imaging, radiomics under the background of deep learning has integrated multiple disciplines, such as medical imaging, clinical medicine, computer science, and statistics. Convolutional neural networks are able to extract high-throughput features from images in an automated and repeatable manner, converting them into high-dimensional data; however, these features are unobtainable under human vision. With respect to the detection of occult lymph nodes, the performance of radiomics has been proven to be superior.42-44
For model establishment and model validation, an internal cohort and an external cohort are necessary for the patient population, with the internal cohort divided into a training set at 80% and a validation set at 20%. The additional prospective cohort allows observation of whether the model is feasible for clinical use and enables further validation. First, the input data included the region of interest of the original image and various parameters, such as tumor size, center location, merging rate and metabolic value. Subsequently, a deep learning model is used for spatial feature extraction, including texture features, first-order statistical features, and higher-order statistical features. Popular neural network structures for image recognition include ResNet, ImageNet, U-Net, and DenseNet. The multimodal algorithms used integrated image information, clinical information, and follow-up records for quantitative analysis. It is important to note that overfitting and poor generalization should be avoided when using multimodal data.
Current PET/CT radiomics research on occult lymph nodes is focused on lung cancer. In the study by Wallis et al, a model based on the U-net not only identified PLNs at the expert level but also localized the thoracic region and suspicious regions in the complete image without previously outlining the ROI by a radiologist. 45 A review on PET and CT in the detection of occult lymph nodes in lung cancer patients involved two studies that included the combination of PET/CT data, one based on deep learning and the other based on semi-automated machine learning that required manual extraction of image features. Both demonstrated that compared to using either one kind of imaging data or clinical-histopathological data alone, their models have the best potential to predict lymph node micrometastasis.46-48
A recent study selected PET/CT images from a cN0 population who had undergone curative surgery for primary NSCLC as data resources, and the deep learning architecture used was finalized after comparison with other architectures, such as ResNet18. 49 The model is named Deep Learning Nodal Metastasis Signature. The largest Area Under the Precision-Recall Curve was achieved not only in comparison with PET images alone and CT images alone but also with the prediction model-based clinical variables and the scoring systems of senior and junior radiologists, suggesting that the ability of the model to differentiate between positive and negative cases was excellent. 50 The calibration curves and the decision curve showed that the model was quite close to the real situation and was clinically useful. In addition to effectively stratifying early-stage patients, it provides reliable clinical opinions on whether sublobectomy or lobectomy, elective nodal dissection or systematic nodal dissection (SND), or postoperative adjuvant therapy or a lack of postoperative adjuvant therapy should be adopted for low-risk and high-risk patients.
Apart from PET/CT, deep learning models have demonstrated superiority in predicting lymph node micrometastasis in MRI and ultrasound examinations for cervical cancer and breast cancer.51-54
In the detection of cervical lymph nodes, a ResNet50-based deep learning model was robustly able to distinguish between lymph node metastasis and lymphoma using PET/CT images in a patient population with a chief complaint of enlarged lymph nodes. 55 Although this is not a study targeting lymph node micrometastasis, the developed model can be one of the preferred options for a variety of cancers that initially present with enlarged superficial lymph nodes, such as head and neck squamous carcinoma, thyroid cancer, and breast cancer. Subsequently, other deep learning models for the detection of occult lymph nodes can be used to refine the overall lymph node examination, thus maximizing the use of whole images for preoperative examination. Invasive examinations such as ultrasound-guided fine needle aspiration cytology have advantages in the examination of superficial lymph nodes, but drawbacks are also evident. First, they are not applicable for lymph nodes smaller than 7-8 mm in diameter, ie, they have limitations in the diagnosis of lymph node micrometastasis. Second, informed consent from the patient for invasive procedures is needed. Third, the procedure is highly dependent on the physician's skill. Fourth, it is likely to irritate the tumor site.41,56-59
Intraoperative Lymphadenectomy
Multivariate Regression Analysis of the ELN Count
At the beginning of this century, multiple retrospective studies confirmed a close relationship among ELN count, survival and lymph node metastasis by multivariate regression analysis. Patient data were obtained from a single institution, the National Cancer Database (NCDB), and the Surveillance, Epidemiology, and End Results (SEER) database. The total size of the SEER database is smaller than that of the NCDB database, but it is open to the public, easily accessible, and widely used in domestic studies. All patients were admitted early for primary surgery. The demographic characteristics included sex, age, race and ethnicity; clinical characteristics, such as comorbidities, primary tumor site, and tumor size; and pathological characteristics, such as Tumor Node Metastasis (TNM) classification, number of ELNs, number of PLNs, and involvement of margins. Inclusion criteria usually consisted of age greater than eighteen years, tumor confined to the primary site, and resection of at least one or more lymph nodes; exclusion criteria usually consisted of failure to undergo surgery, perioperative death, presence of distant metastasis, history of neoadjuvant therapy or history of cancer, and failure to follow-up; and evaluation criteria usually consisted of disease-free survival, disease-specific survival and overall survival. To prevent stage migration, some researchers excluded patients with few ELNs when performing multiple sensitivity analyses to control for statistical bias. 9
Researchers have reached a consensus that the greater the ELN count is in early-stage patients with pathologically negative nodes, the lower the recurrence rate and the higher the survival rate, as well as the greater the likelihood of detecting occult lymph nodes. This suggests that incomplete lymphadenectomy, inaccurate pathological evaluation, and incorrect histopathological staging have already lurked in the original patient population, leading to missed diagnoses of patients with PLNs and failure to receive postoperative adjuvant therapy; on the other hand, the improvement in survival due to the increase in ELN count reflects an increase in correct staging and a decrease in occult lymph nodes.18,60-64 In patients with pathologically positive nodes, researchers agree that the greater the PLN count or the greater the ratio of PLN count to ELN count is, the lower the survival rate. After controlling for PLN count, the survival rate still increased with ELN count.65-67 Thus, the ELN count is considered an important prognostic variable. The greater the number of nodes dissected is, the greater the likelihood of accurate staging, and the better the prognosis.
In addition to lymph node count being positively associated with survival, studies on colorectal cancer have shown that lymph node size (maximum long-axis diameter) is also positively associated with survival, with one study identifying 10 mm as the threshold based on the difference in five-year overall survival, which may be due to the stronger anti-tumor immune response of larger lymph nodes.68,69 Further research is required to confirm the correlation between the size of the lymph nodes and the occurrence of lymph node metastasis. Emerging single-cell analysis may provide novel insight into the relationship between the two from aspects such as transcriptomics and proteomics and establish optimal thresholds that can distinguish the size of lymph nodes according to the most typical difference.
Thresholds of the ELN Count
Despite the clarification of the relationship between ELN count and prognosis, guidance for practice is needed. In addition to the exhaustive method of selecting thresholds by multivariate regression analysis mentioned above, researchers have used larger population datasets based on more statistically convincing methods to explore thresholds of ELN count for each type of solid tumor. They aim to distinguish between clinically node-negative and clinically node-positive patients and to eliminate stage migration effects with the greatest likelihood of success. Larger population datasets are often obtained from multiple databases and multiple low- to high-volume medical centers. Typical examples of the various statistical methods used to calculate thresholds are listed below (Table 2).
Characteristics of the Models for Evaluating the ELN Count Thresholds.

Hypergeometric Distribution-Based Model
This model was pioneered in 1992 in a study by Kiricuta et al 70 Based on hypergeometric distribution and Bayes’ theorem, the number of residual PLNs after axillary lymph node dissection (ALND) can be estimated with 90% certainty. A total of 1446 T1 (n = 831), T2 (n = 565), and T3 (n = 50) breast cancer patients who underwent ALND were included. After categorizing the calculation according to T stage, the model yields, on the one hand, the minimum number of lymph nodes that need to be sent to avoid the presence of residual PLNs for each T stage with 90% certainty, and on the other hand, the number of PLNs that may be left after a certain number of lymph nodes have been sent and a certain number of PLNs have been identified for each T stage with 90% certainty. The mathematical model provides both preoperative instructions and postoperative evaluation of lymphadenectomy. It is able to identify pN0 patients with 90% certainty and to supplement adjuvant therapy for patients with incompletely removed PLNs.
In 2000, Iyer et al used this model for the evaluation of patients with detected PLNs. 71 Patients with primary breast cancer are usually categorized into three levels according to PLN count: pathologically negative, 1-3 PLNs, and greater than or equal to 4 PLNs, with the third level being an indication for postoperative radiotherapy.67,80 According to their study, the minimum ELN counts required to achieve 90% accuracy were 19, 20, and 20 for T1 patients who were predicted to have 1-3 PLNs, respectively; the minimum ELN counts were all 20 for T2 patients who were predicted to have 1-3 PLNs. For T1 and T2 patients in whom 1-3 PLNs were already found, the study also recommended the minimum ELN count to ensure that no more (greater than or equal to 4) PLNs remained. This study aimed to accurately stratify breast cancer patients and to ensure the necessity of supraclavicular regional radiotherapy after mastectomy. Moreover, as mentioned in section 2.1, lymph node size is associated with survival rates. In 2001, Suzuma et al noted that Kiricuta et al neglected the impact of lymph node size. 72 The mathematical model established in their research was inconsistent with the previous model. In addition to the ELN count and PLN count, they also incorporated the parameter of the maximum long-axis diameter of the lymph nodes. When data were collected from patients after ALND, the size of each node was recorded under a microscope. Since the study included T1 and T2 breast cancer patients with lymph node metastasis, the results of the study illustrate the minimum ELN count and the corresponding maximum long-axis diameter of the lymph node required to detect a PLN with 90% certainty rather than to avoid the presence of residual PLNs. The results complement those of the study by Iyer et al Together, these three studies complete the guidance for ALND in early-stage breast cancer patients.
In 2003, the model of Kiricuta et al was also applied to estimate the number of residual positive mesenteric lymph nodes in patients with colorectal cancer. 73 Compared with the previous study, this study included patients at intermediate and terminal stages, basically T3 and T4; it only accomplished the first half of Kiricuta et al's work—a minimum of 40 ELNs to achieve an 85% probability of pathological negative results in T3 patients and a minimum of 30 ELNs in T4 patients. The status of patients with residual PLNs was not determined, probably because postoperative adjuvant therapy is routinely implemented for colorectal cancer patients at intermediate and terminal stages.
Beta-Binomial Distribution-Based Model
This model is based on the maximum likelihood estimation, which is used to find the phylogenetic tree capable of generating observed data with a higher probability, ie, it is used to determine the ELN count, by which surgeons perform lymphadenectomy to have a greater probability of avoiding missed diagnoses of PLNs. 81 As early as 1973, Griffiths first used the beta-binomial distribution as the basis for modeling the incidence of non-communicable diseases. 82 In 2009, maximum likelihood estimation was applied to the prediction model of lymph node metastasis in colorectal cancer, and the Nodal Staging Score (NSS) was formulated on the premise that nodal staging follows a beta-binomial distribution, marking the beginning of this type of research. 74 In addition to a series of validation studies, studies of signet ring cell carcinoma, gallbladder carcinoma, esophageal squamous cell carcinoma, and papillary thyroid carcinoma were carried out, all of which confirmed the prognostic value of the model in pN0 patients.75-78
The key in the NSS is to estimate the probability of false negatives (P(FN)). This step is performed in the population of patients with positive nodes, so the data cleansing needs to start by filtering out the population of positive nodes from the total population and excluding the population of patients who may undergo lymph node biopsy rather than lymph node dissection; in other words, the ELN count is required to be more than one. According to the beta-binomial distribution model, P(FN) and the ELN count follow the formula:
After deriving the probability of false negatives corresponding to each number of ELNs, the number of false negatives in each group can be derived to calculate the corrected prevalence of true positives and to estimate the true prevalence of nodal disease in the entire study population. Finally, the number of true positives was subtracted from the total number of patients to calculate the node staging score of true negatives as a function of the ELN count. Furthermore, to achieve accurate staging, the total population was divided into different subgroups according to the T1, T2, T3, and T4 stages during data cleansing, and then applied to the NSS. According to the different T stages, the minimum number of ELNs was determined for the different populations. Following this number can achieve no residual PLNs with 90% certainty. 79
In the section discussing the quantitative relationship between ELNs and residual PLNs, the establishment of a beta-binomial distribution-based model and the derivation of the shape parameters are robust and reasonable, while Assumption 2, which the model relies on, is controversial in practice compared to the other two assumptions. This is because in most public databases, such as the SEER database or the NCDB, information on the ELN does not include information on lymph node sites. During END, surgeons usually have different sampling preferences for lymph nodes at different sites, eg, lymph nodes that are closer to the tumor site are more likely to be dissected than those further from the tumor site. A study by Lee et al also suggested that different approaches to the management of lymph nodes may result in different lymph node yields, so the heterogeneity of intraoperative dissection and microscopic examination should be taken into account. 17 However, Gil et al reported that there was no significant correlation between the ELN count and the PLN count (correlation coefficient R = 0.3), which eliminates the suspicion that the status of the lymph nodes is affected by selection bias. 83 Although the lymph node site has no effect on the PLN count, it does have an impact on patient prognosis. One study showed that the status of lymph nodes in the right colon was more closely related to the risk of death than that of lymph nodes in the left colon. 84 This highlights the need for separate thresholds for lymph node subsites.
Survival Data-Based Model
Both of the above mathematical models estimate a threshold based on the distribution of the ELN count, which is subsequently verified with survival data. Other researchers have analyzed the ELN count and survival data in the first step. In the second step, locally weighted regression or X-tile statistical analysis software was used to perform structural break point analysis or cutoff value analysis of the effect of the ELN count on overall survival without any assumptions, and in the third step, the calculated threshold was used to verify the effect on survival in a secondary way.12,13,19,21,22
Locally weighted regression (LOWESS) is a non-parametric regression method used in statistics and machine learning that captures complex nonlinear relationships in a dataset. It achieves a localized fit by assigning higher weights to points near the prediction point and lower weights to points far from the prediction point. The presence of structural break points in a dataset is subsequently determined by comparing the regression coefficients of the fitted curves on both sides of each potential structural breakpoint by the Chow test for significance. In 2017, the method was first applied by Liang et al for NSCLC. 12 The patient populations included were early-stage, multi-institutional, foreign and domestic NSCLC patients. There were 38 806 patients from the SEER database and 5705 patients from Chinese institutions. The structural break point from the training set passed independent internal and external validation. It is important to note that this study can only demonstrate that dissecting lymph nodes by the ELN count in the vicinity of the structural break point improves patient mortality but does not support that the ELN count exceeds the structural break point too much. In the curve to the left of the structural breakpoint, an increase in the ELN count avoids erroneous staging and reduces residual PLNs; in the curve to the right of the structural breakpoint, an increase in the ELN count is associated with a poorer prognosis.
X-tile is a graphical statistical analysis software designed to help identify which cutoff values separate patients into clusters with different survival outcomes by analyzing biomarker expression levels and survival data from a large number of patient samples. 85 Zhu et al considered the standard set by Liang et al to be questionable and included more early-stage NSCLC patients with the same inclusion and exclusion criteria—52 099 from the SEER database and 3002 from Chinese institutions. 13 They employed X-tile software to exhaustively search for various numbers of ELNs and eventually identified the number of ELNs that showed the most significant difference (the largest chi-square value, relative risk, and smallest P value) as the cutoff value. This cutoff value was confirmed to be an independent prognostic factor after excluding confounders and homogenizing the baseline by Propensity Score Matching. Overall survival in early-stage NSCLC patients also decreased in accordance with the results of Liang et al's research. They considered this cutoff value to be up-to-date and attributed the difference to the database version and the TNM classification version; however, they did not mention a possible impact of the methodological difference between the two studies.
Lymph Node Ratio or Lymph Node Density
The lymph node ratio (LNR) or lymph node density (LND) (PLN count/ELN count) is becoming a viable prognostic option for node-positive patients.83,86-94 The superiority of this fraction lies in its ability to simultaneously evaluate three factors: 1. tumor factors (PLN count); 2. treatment factors (ELN count); and 3. staging factors (completeness of the sampling procedure, including those related to the surgeon and pathologist).
83
Like for ELNs, the corresponding threshold for each cancer is also available through multivariate regression analysis and X-tile software. The LNR has also been proven to be superior to the traditional TNM classification for predicting disease-free survival, disease-specific survival and overall survival. For example, a patient with 1 PLN out of 20 ELNs (LND = 0.05) has a better prognosis than a patient with 1 PLN out of 5 ELNs (LND = 0.2). Although both patients had the same N stage, the latter were more likely to have occult lymph nodes. In contrast to the ELN, it can only be applied for prognostic evaluation of node-positive patients at intermediate and terminal stages, and it is considered an unfavorable prognostic factor; the larger the ratio is, the poorer the survival. Based on the LNR, the weighted lymph node ratio (ωLNR) and the log odds of positive lymph nodes (LODDS) were derived as similar assessment tools95,96: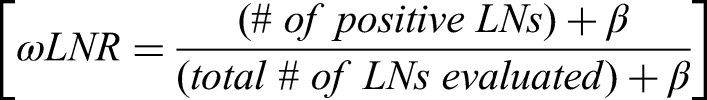

It is important to note that the LNR cannot be used as a prognostic factor independent of the PLN count. Researchers have argued that from the perspective of surgery and anatomy, the same LNR does not necessarily indicate that the extent of surgery and lymph node metastasis are similar. There might even be significant differences. 66 To put it more conservatively, the LNR is not reliable without knowing the exact PLN count.
Machine Learning-Based Model
The greatest advantage of machine learning is its ability to help doctors make better decisions through the use of large amounts of digitized medical information. In addition to being used for preoperative auxiliary analysis of occult lymph nodes, machine learning is also being used for postoperative auxiliary analysis of the residual risk of occult lymph nodes. Research in this area is still limited, and the relevant literature is focused on oral cancer.98-100 Patients included were also those at early stages without clinical evidence of lymph node metastasis who had undergone primary tumor resection, but the outcome indicator was pathologically confirmed lymph node metastasis within 2 years. Clinical variables included age, sex, race and ethnicity, body mass index, smoking history, and primary tumor site; pathological variables included the maximum size of the tumor, depth of invasion, muscle invasion, submucosal invasion, histological grade, involvement of margins, perineural invasion, and lympho-vascular invasion. In addition to clinical-pathological data, some researchers also included gene expression profiles, ie, the mRNA expression levels of genes most relevant to lymph node metastasis. 101 By inputting the above feature variables into training, the researchers constructed supervised machine learning models based on different classification algorithms and selected the models that best matched the real situation and predicted lymph node metastasis. Such models are also promising in both reducing the number of pathologically node-negative patients undergoing unnecessary cervical lymph node dissection and identifying those at high risk of occult lymph nodes. As Farrokhian et al reported, the model correctly spared 1 patient from END for every 10.6 patients screened and correctly identified 1 additional patient with occult lymph nodes for every 21.0 patients screened compared with the traditional depth of invasion model. 98 In addition, Xu et al combined preoperative PET/CT variables with postoperative pathological variables to construct a nomogram for the prediction of the risk of occult lymph nodes, and the results demonstrated that the combination showed better predictive performance than the application of either alone. 100 However, they directly adopted the logistic regression algorithm without explaining why they did not choose other algorithms or comparing the pros and cons with other algorithms.
Postoperative Immunotherapy
There have been earlier clinical trials and systematic reviews that yielded conflicting results. Some have suggested that SND only prolongs surgery and increases bleeding without increasing postoperative hospitalization, complications, or decreasing survival rates 102 ; some have suggested that harvesting a greater number of lymph nodes is either not associated with survival or is harmful to overall survival.103-105 A recent study performed a Restricted Cubic Splines analysis and demonstrated that the correlation between the ELN count and hazard ratio was not linear, as expected, but rather a U-shaped, dose‒response and nonlinear correlation, suggesting that excessive lymphadenectomy is actually detrimental to survival. 106 The reasons for the increased hazard ratio might be, on the one hand, as suggested by Jiang et al, that patients with so many lymph nodes dissected were in advanced stages. Despite eliminating lymph node micrometastasis, the risk posed by potential distant metastasis has surpassed that posed by lymph node micrometastasis. This is corroborated by other literature: extended lymph node dissection provides a survival benefit by eliminating stage migration in early-stage patients but does not improve survival in advanced-stage patients. 107 On the other hand, excessive lymphadenectomy can cause immune impairment (Figure 2). This opinion was first proposed by Liang et al in 2023, who followed their previous clinical study in 2017, inspired by basic medical research on the impact of removing TdLNs on immune infiltration and immunotherapy. 29 Basic studies have confirmed the adverse effects of TdLN removal on immune infiltration and immunotherapy, as well as the importance of TdLNs in the antitumor immune response.108-111

Balance of lymph node dissection. While less dissection may miss occult lymph nodes, excessive dissection is at risk of causing immune impairment.
Currently, immunotherapy primarily focuses on immune checkpoint blockade (ICB), with anti-PD-1/PD-L1 and anti-CTLA-4 monoclonal antibodies being the most common.112-114 The principle of ICB is to enhance endogenous anti-cancer T cell metabolism by blocking inhibitory signals in the metabolic reprogramming process of T cells, thus supporting the energy demand, biosynthesis, and cellular signaling processes needed for effector activities. Some reviews have indicated that CD8 memory T cells in the TdLN can be activated and function upon administration of ICBs.115-117 Through transformation to the Th1 phenotype, high levels of TdLN CD8 central memory T cells/effector memory T cells are associated with better ICB efficacy. Besides, CD8 T cells are also related to oncolytic virus therapy in immunotherapy. Kaufman et al described that oncolytic viruses achieve tumor elimination by inducing systemic innate immunity and tumor-specific adaptive immune responses. 118 Following oncolytic cell death, tumor cells release tumor-associated antigens, pathogen-associated molecular patterns (PAMPs) and additional cellular danger-associated molecular pattern signals and cytokines, which promote the maturation of antigen-presenting cells (APCs). APCs subsequently activate antigen-specific CD4+and CD8 T cell responses. Once activated, CD8 T cells can expand into cytotoxic effector cells with the ability to travel through lymphatic flow to established tumor growth sites, where they mediate anti- tumor immunity upon antigen recognition.
In the latest study by liang et al, the included NSCLC patients were from multiple domestic institutions who experienced tumor recurrence during follow-up and received PD-1 immunotherapy. 29 They reproduced the locally weighted regression fitting curves and still identified 16 as the structural break point. To explain the immunological mechanism of immune impairment, bulk RNA sequencing of the ELNs was performed. xCell and CIBERSORT algorithms were applied to assess immune cell type and abundance. Based on the observed prolonged recurrence-free survival, they mapped the immune memory cell landscape and hypothesized that this phenomenon may be attributed to the presence of long-lived memory cells within the ELNs. The memory cells that predominantly influenced the efficacy of immunotherapy were ultimately designated CD8 central memory T cells based on the results of survival analysis and immune infiltration. It is consistent with previous theories about the influence of CD8 memory T cells on immunotherapy.
Conclusion
The management of lymph nodes in cancer patients involves preoperative examination, intraoperative lymphadenectomy and postoperative immunotherapy. It is a multi-modal, multi-omics, multi-stage (3 M) evaluation protocol. Preoperatively, the combination of radiomics can detect occult lymph nodes more efficiently and accurately than manual inspection. With further development of convolutional neural network architectures for image recognition, we can foresee deep learning models that achieve even higher specificity and sensitivity. Intraoperatively, we recommend that surgeons take END and SND into consideration at all times. The development of lymph node dissection will focus on individualization and precision. SND certainly allows for accurate staging, but it does not always result in a favorable outcome. For END, various guidelines are updating the minimum number of ELNs for each type of tumor, known as the threshold of ELN count. Postoperative immunotherapy is necessary because preoperative examination or intraoperative lymphadenectomy cannot detect all PLNs at 100%. Inevitable missed cases require complementary adjuvant therapy. The paradox is that less dissection may result in more residual PLNs, and more dissection may cause immunotherapy failure, which seems to result in a poor survival outcome. We therefore suggest that until there is more evidence from randomized clinical trials that extended lymph node dissection does have a therapeutic benefit, lymphadenectomy should be performed conservatively by the threshold of ELN count.
In addition, there are no validity evaluations, algorithm comparisons, or goodness-of-fit studies of various statistical methods. This review presents a longitudinal review of various evaluation protocols but does not include a cross-sectional comparison. In the future, in addition to developing more evaluation protocols, comparative methodological studies between protocols should be carried out before they are put into clinical practice to identify the best protocols suitable for various races, regions and types of cancers.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Nos. 81872196 and 81672690).
