Abstract
Keywords
Introduction
Pelvic lymph node dissection (PLND) is an integral part of the surgical treatment of localized intermediate-risk and high-risk prostate cancer (PCa) 1 ; roughly 70% of prostatectomies are accompanied by PLND. It provides important information for staging, risk assessment, and prognosis, helps to further determine the extent of disease and guide subsequent treatment.
Symptomatic lymphocele formation represents the most common complication of PLND. It is among the most frequent complications reported after radical prostatectomy, independently from approach (open, laparoscopic, and robot assisted). 2 The incidence reported in the present literature ranges from 2% up to 61%.3,4
In majority of the cases the development of lymphoceles is clinically asymptomatic.5,6
Lymphoceles, in general, can lead to significant morbidity resulting from infections and recurrences. As they increase in size or become superinfected, the retention of lymphatic fluid in the retropubic space along iliac vessels may cause a wide spectrum of sequelae such as pelvic pain, neurological symptoms, systemic infection, lower urinary tract symptoms, edema of lower extremities complicated by deep vein thrombosis until life-threatening conditions like urosepsis and pulmonary embolism.
Symptomatic lymphoceles after robot-assisted radical prostatectomy (RARP) with PLND occur in 2%to 15% of patients depending on the extension of nodes dissection.7–10
The risk of lymphocele formation and its consequences are an important aspect when counseling patients prior to surgery regarding risks and benefit of PLND. In order to identify those patients at higher risk to be affected by lymphovascular complications, many clinicopathological variables should be considered predictors of symptomatic lymphocele, representing valuable tools for patient counseling and management; many experiences helped in preventing this complication, but, although data from numerous studies evaluating risk factors are available, results are still conflicting.4–7
The best treatment option for pelvic lymphoceles resulting from PLND has not been defined. Conservative management with sclerosis and percutaneous drainage is often used as an initial management strategy. Subsequent, after failure of percutaneous approach, surgical excision is mandatory but, at the same time, challenging because of significant scarring from failed conservative attempts.
In the literature, a large amount of reports focused on laparoscopic management (fenestration or marsupialization) for symptomatic lymphocele, after radical prostatectomy performed with any approach (open, laparoscopic, or robotic) 5 but there is paucity of experiences reporting the resolution of this complication by robot approach.
The aim of this article is to analyse the current evidence on the treatment of symptomatic/infected lymphocele by robot-assisted approach.
Materials and Methods
Literature Search and Data Collection
Studies were identified by searching electronic databases for all studies listed in the databases, scanning reference lists of studies that were included, and consultation of experts in the field. A nonsystematic review was performed. A literature search was conducted up to January 1, 2022 using the Medline, EMBASE, and Cochrane databases.
The following terms were used to search the databases without language barrier: “lymphocele”, “symptomatic”, “infected”, “robot-assisted AND radical prostatectomy”, “robot-assisted”, “treatment”.
A data extraction sheet was developed and refined to the predefined outcome parameters.
One reviewer systematically extracted the data from included studies accordingly.
Extracted data were crosschecked by a second reviewer.
Eligibility assessment was performed independently in an unblinded manner by one reviewer. Studies were screened for title and abstract and if necessary the entire article was reviewed.
Eligibility Criteria
Types of Studies
We included original studies such as case series, case reports, retrospective, and prospective cohort studies, including studies for which a full text was not available in our institution. We did not include studies focusing on lymphatic fistulas, chylous ascites, or chylothorax.
A lymphocele is a cavity of any size containing lymphatic fluid; it is identified by radiological imaging. Laboratory analysis of the fluid was decisive in the absence of consensus on the etiology of the lymphatic fluid collection.
Lymphoceles were considered symptomatic in case of any discomfort, pain or compliants experienced by the patient and due to the compression of the lymphatic fluid collection on the adjacent structures; lymphoceles were considered infected if patients experienced fever and elevated white blood cell (WBC) count not related to other diseases.
Surgical Technique
The extensive adhesions often present from prior prostatectomy and treatment attempts require the need of comprehensive preoperative imaging and intraoperative ultrasound in order to identify landmarks useful in dissection. In order to prevent recurrence, it is mandatory to excise the medial lymphocele wall and surrounding fibrotic tissue; in order to minimize the risk of damage to the iliac vessels and obturator nerve, the lateral wall excision and bipolar coagulation must be performed very carefully. Intravesical instillation of dye represents another useful step, to confirm the integrity of the bladder at the end of the procedure. Dye injection could also be perfomed by percutaneous drains (when present) at the beginning of the procedure in order to localize and to better delineate the lymphocele walls, and therefore to facilitate the complete surgical excision.
Results
Evidence Synthesis
The search identified three series (Table 1) for a total of 8 cases focusing on the treatment with robot-assisted approach of symptomatic and/or infected lymphocele, while the most were reports on lymphocele prevention, and laparoscopic treatment (Figure 1).
Evidence synthesis focusing on the treatment by robot-assisted approach of symptomatic and/or infected lymphocele.

Abbreviations: CT, computed tomography; EBL, estimated blood loss; NR, not reported.
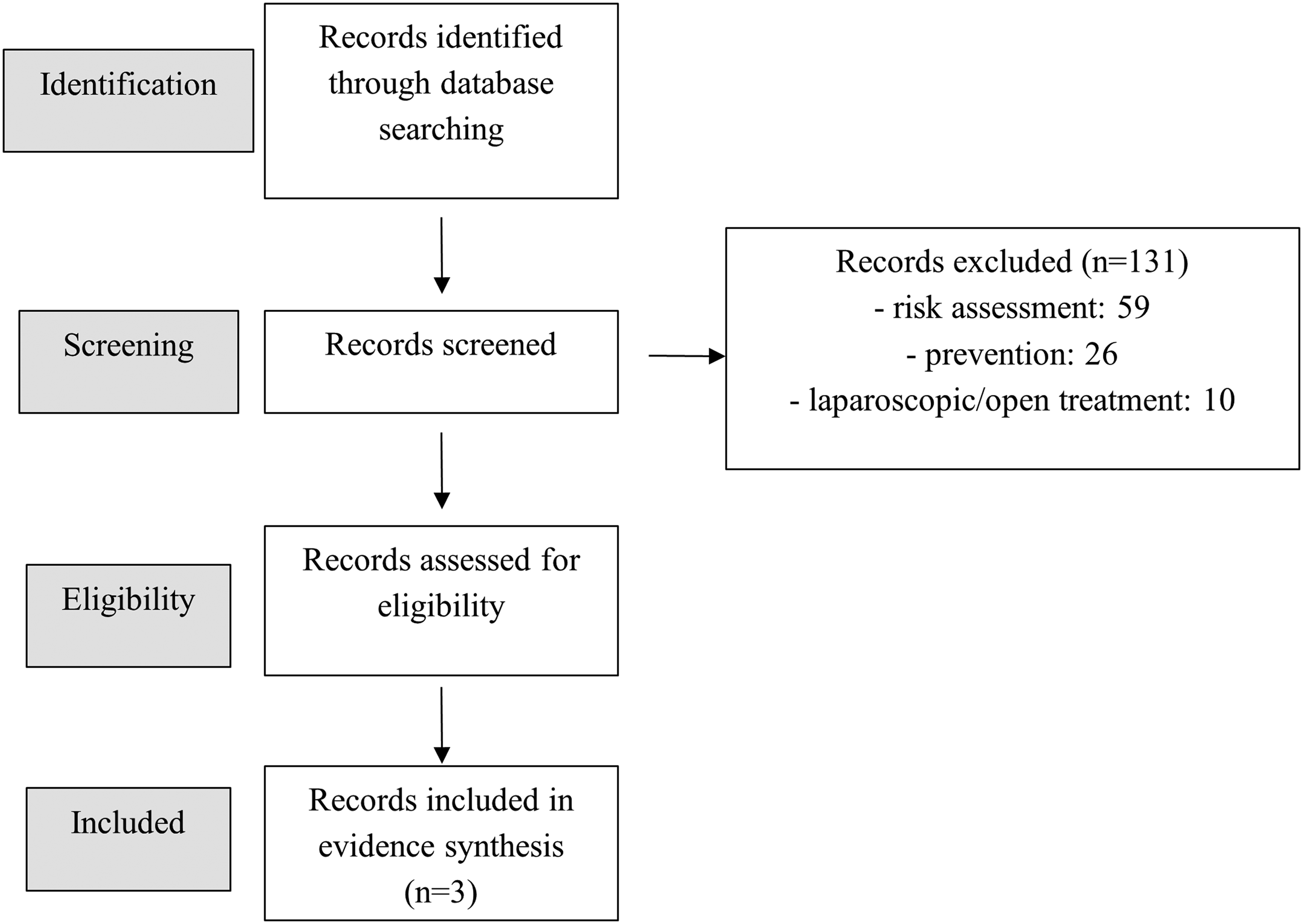
Flow Diagram of Selection Process.
The main and most frequent reason for performing the robotic treatment was infected lymphocele, followed by symptomatic lymphocele.
The median time from RARP + PLND to robot-assisted treatment of lymphocele was 118 days (range 30-240). All patients had experienced unsuccessful prolonged antibiotic courses and percutaneous drainage (with/without sclerosant agents) treatment of lymphocele. Robot-assisted approach proved effective in all cases.
Discussion
The exact role of PLND for the PCa management has not been completely defined, despite the clinical evidence and the introduction of several prediction models. In case of intermediate and high-risk PCa with estimated risk for lymph node invasion of at least 5%, current guidelines recommend an extended (iliac external, internal, and common) PLND, 1 which results in an improvement in the staging accuracy up to 94%. 11 PLND represents the most accurate approach for nodal staging, with presumable benefit for metastatic patients in terms of early adjuvant treatment.
The extended PLND surely improves the quality of the staging but with potential negative impact on intraoperative and perioperative outcomes (such as operating time, blood loss, and postoperative complications), which seem related to the presence of more extensive disease.
The management of lymphoceles is one of the most common re-interventions after radical prostatectomy, with different clinical manifestation, even non-specific. 12
Several studies focused to identify patient-related and surgical risk factors of symptomatic lymphocele, with a particular attention on its prevention.
Despite not always confirmed,5,6,9,13 age was reported as one of the most prominent patient-related predictors, with an increase of the risk of symptomatic lymphoceles by 5% with every additional year of age. 14 Even other patients-related factors were considered, such as an higher lymphatic flow in obese patients 15 or the use of low molecular weight heparin,5,16–18 but the conflicting results did not strictly correlate.
The relevant surgical risk factors may result in the choice of procedure (retropubic radical prostatectomy [RRP] vs RARP) and approach (extraperitoneal vs transperitoneal), different sealing techniques as extension of PLND. In a retrospective single-centre study, Zorn et al 4 showed no difference in lymphovascular complications (2% vs 2.5%, P =.9) comparing RARP and RRP. Transperitoneal approach creates a passage of communication between the peritoneal space and the afferent lymphatic flow, and this might prevent the formation of persistent lymphatic retention but Horovitz et al 19 reported no significant difference in terms of prevalence of symptomatic lymphoceles (1.49% vs 2.83%, P = .09) in a retrospective propensity score-matched analysis between transperitoneal and extraperitoneal RARP. Several studies confirmed these data. 20
Magistro et al in a retrospective single-centre study on 472 patients (241 patients treated with open RRP compared to 231 patients treated with RARP) associated the high-grade disease with a higher risk to develop symptomatic lymphocele irrespective of the technical approach. In addition to the number of dissected lymph nodes (n = 11) (odds ratio [OR] 1.1; 95% confidence interval [CI] 1.055-1.147; P = .001), the authors reported the Gleason score > 8 (OR 4.7; 95% CI 2.365-9.363; P = .001) and the total prostate specific antigen >10 ng/mL (OR 1.05; 95% CI 1.02-1.074; P = .001) as independent predictors for the development of symptomatic lymphocele. 21
From a pure technical point of view, the most crucial step is represented by the accurate preparation during the lymph node dissection but conflicting outcomes derived from the influences of clipping, coagulation, flap-mediated reconstructive techniques or the application of additional hemostatic sealants. In a prospective randomized study, Grande et al compared the frequency of postoperative complications after sealing lymphatic vessels from the leg to the abdomen using metallic clips or electrical coagulation during robot-assisted surgery for prostate cancer. They reported no statistically significant differences between groups regarding overall lymphocele incidence (47% vs 48%; difference −0.91%, 95% [CI] −2.6 to 0.7; P = .9) other than the rate of clinically significant lymphocele. 13
Despite some studies evaluating various sealing agents reported promising outcomes,22–24 In a recent review, Motterle et al reported no significant impact by different sealing techniques and agents or by surgical approach. In the same review, the authors showed a potential benefit of peritoneal reconfiguration to maximize the peritoneal surface of reabsorption. 25
Lebeis et al reported no lymphoceles after the creation of a peritoneal interposition flap fixed to the lateral bladder wall covering the perivesical adipose tissue. 26
Some studies suggested a correlation between lymphocele formation and the harvest of lymphnodes during PLND, with a predictive cut-off of 20 lymph nodes and an increase in the risk increased by 5% with every single lymph node removed. 14 Liss et al reported a higher rate of lymphocele after RARP with PLND compared to single prostatectomy, with a subsequent higher rate of re-intervention for the treatment of symptomatic lymphocele (3.4% vs 0%). 27 Other studies confirmed this trend, by reporting the lymphocele formation in 28% after PLND and in 14% without, 5 and the need of active treatment required only in those after PLND (5.9% vs 0%). However, other series were not able to confirm the correlation between an extended PLND and the occurrence of lymphoceles.27,28 Surely, PLND itself represents a clear risk factor for the development of symptomatic lymphoceles, but there are several others additional confounding factors still without an exact role, deserving to be further acknowledged.
Next to the large experience reported in literature regarding the laparoscopic management of lymphocele, 29 very few studies focused on the robotic approach.
The current analysis showed how patient deserving treatment of infected/symptomatic lymphocele should be considered for a robotic approach. Surely there are many concern in the robotic approach, due to the cost, the availability of the technology and, not at least, the surgeon’s experience. All the widely accepted advantages of the technique (including fewer incisions and better cosmesis) are surely useful even in case of a critical clinical situation like in a postsurgical complication. The impact of a re-intervention might affect both the patient’s and surgeon’s confidence but the awareness of surgery with a very high rate of success might mitigate the complexity of the clinical situation: the surgical approach allows to entirely remove and clear the thick purulent content of some infected lymphoceles, differently from small percutaneous drains with inherent limitations to drain this type of fluid. 30
Tremp et al in their case report that they showed how the infected/symptomatic lymphocele affects clinical management: the authors reported two cases, highlighting how the first approach should be conservative and only in case of failure should the invasive approach be considered. 31
Furthermore, Moschovas et al reported their experience by using the single-port system: in the robotic era, this is the further demonstration of the growing opportunities that the robotic approach might lead, with the challenges-associated single-port system similar to the multiport robot.30,32 Moreover, Moschovas et al emphasized the safety and feasibility of single-port lymphocelectomy by reporting no recurrence. 30
Robot-assisted approach could help surgeon, with its magnification, to identify and isolate the lymphocele especially when an adhesiolysis due to recent surgical procedure is needed.
The concept to create a communication between lymphocele cavity and peritoneal space is at the base of some recent techniques developed in order to prevent lymphocele formation after RARP.33–35
A communication between iliac vessels and peritoneal cavity created at the end of the procedure seems to be effective to reduce lymphocele formation.
Another kind of debate might arise about the general low tendency to report complications; the complication should always represent an important element to consider in order to improve the surgical technique and the clinical management and the basis for the further technical improvements and the increase in surgical indications. Considering minimally invasive approach allows by robot-assisted platform other than high success rate, surgical treatment for symptomatic lymphocele could be considered as a gold standard.
In the current analysis, certain limitations have to be acknowledged (very few studies and cases, lack of a lot of clinical data) but they do not appear to limit the opportunities to highlight the advantages of the robotic treatment of symptomatic/infected lymphocele.
Conclusion
The occurrence of lymphocele might be predicted and not always requires a treatment, but in case of infected or symptomatic lymphocele an active treatment is mandatory. The robotic approach allows a safe and feasible drainage and a definitive treatment of the infected and/or symptomatic lymphocele sac.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received “Ricerca Corrente” funding from the Italian Ministry of Health to cover publication costs.
