Abstract
Introduction
Colorectal cancer (CRC) is the third most common malignant cancer class and the second leading cause of tumor-related death worldwide. 1 The liver is the most common site of metastasis for colorectal cancer, and approximately 50% of colorectal cancer patients will develop liver metastasis. 2 Unfortunately, most patients with colorectal cancer liver metastases (CRLMs) are not eligible for surgery or local thermal ablation. However, treatment with irinotecan drug-eluting beads may be considered in patients with liver-only or liver-dominant metastatic disease with tumor loads of no more than 60%. 3 Irinotecan is a derivative of camptothecin, the prodrug of SN-38. It acts through irreversible binding to topoisomerase-I, an enzyme involved in DNA replication. This binding interferes with DNA replication and induces DNA single-strand breaks. 4 Drug-eluting bead TACE (DEB-TACE) preloads chemotherapeutic agents into embolic microspheres and then releases them in a controlled and sustained manner in tumor vessels. 5 Pharmacokinetic studies in both animals and humans have confirmed that DEB-TACE can deliver high concentrations of chemotherapeutic agents consistently in tumors while reducing systemic toxicity.6–8 Several clinical studies have demonstrated the efficacy and safety of irinotecan-loaded DEB-TACE in patients with unresectable CRLMs.9,10 Various microspheres have been introduced into the clinic, such as DC Beads (BTG, UK), CalliSpheres (Callisyn Biomedical-Suzhou, China), Tandem (Boston Scientific, MA), and HepaSpheres (Merit Medical, Utah); however, these DEBs are invisible under x-ray, MR, and ultrasound, which poses a challenge to the concept of precision therapy, as the invisibility of microspheres during and after administration is not conducive to the precise delivery of drugs to the target area and the timely assessment of embolic efficacy. Thus, there is a clinical need for the use of visualized microspheres to assist in interventional procedures, which could contribute to improved efficacy and procedural safety. Several studies have attempted to fabricate this type of platform. Visualization under x-ray can be achieved by attaching metallic components to microspheres, such as iodine substances, barium sulfate nanoparticles, and tantalum nanoparticles,11–13 Microspheres combined superparamagnetic iron oxide nanoparticles or holmium ions, which are used to achieve visualization via MR,14,15 or by the simultaneous combination of multiple imaging materials for visualization via multiple modalities, In Kim et al's study, microspheres could be visualized via MR and CT by combining iron oxide nanoclusters and gold nanorods. 16 In our previous study, we developed a drug-loaded microsphere that can be visualized under CT/MR/Raman spectroscopy, which showed good drug-loading capacity for doxorubicin. 17 To validate the clinical value of NAMs, the pharmacokinetics of three microsphere-delivered irinotecan agents, DC Beads, CalliSpheres and NAMs, were compared. This study will compare the similarities and differences of the three microspheres in terms of drug-loading capacity, drug release profiles, diameter variation and morphological characteristics of irinotecan under equivalent conditions.
Materials and Methods
Materials
All materials were used as received without further purification. The following microspheres were evaluated in this study: CalliSpheres (100-300 μm) were purchased from Jiangsu Hengrui Medicine Co., Ltd (Suzhou, China); DC Beads (100-300 μm) were purchased from Biocompatibles UK Limited (United Kingdom); chloroauric chloride (HAuCl4·4H2O) and polyacrylic acid (PAA) were purchased from Sino-pharm Chemical Reagent Co., Ltd (Shanghai, China); and 4-nitrothiophenol (4-NBT, 90%) was purchased from Fluorochem Ltd (Derbyshire, United Kingdom). Iron oxide (Fe3O4) nanoparticles, polyethyleneimine (PEI, 50%), Tris-HCl buffer, sodium polystyrene sulfonate (PSS), sodium hydroxide, sodium citrate dihydrate, and dopamine hydrochloride (PDA, 98%) were obtained from Aladdin Chemical Reagent Co., Ltd (Shanghai, China). Irinotecan hydrochloride, sorbitol, and lactic acid were purchased from Macklin Chemical Reagent Co., Ltd (Shanghai, China). Embospheres (100-300 μm) were purchased from BioSphere Medical (United States). Ultrapure water (UPW, 18.2 MΩ) was used for all experiments.
Synthesis of NAMs
Based on our previous study, we synthesized multimodal imageable nanoassembled microspheres. Briefly, we first synthesized multimodal visible nanospheres containing Fe3O4 nanoparticles, gold nanoparticles, the Raman signal molecule 4-NBT and a dopamine coating. Then, we modified the surface of Embospheres with PAA. Embospheres, which are collagen-coated microspheres with a trisacryl core, are frequently used in the clinic as blank embolization microspheres and cannot load drugs on their own. 18 Subsequently, we assembled nanospheres with Embospheres to synthesize NAMs, and sulfonic acid groups were modified on the surface of the microspheres by reacting the NAMs with PSS to achieve loading with irinotecan.
Determination of the Standard Curve for Irinotecan
An irinotecan solution (10 mg/ml) was added, the pH was adjusted to 3.2–3.6 with 0.2 mol/L sodium hydroxide solution, the solution was diluted to 20, 40, 60, 80, and 100 µg/mL, and the pH was adjusted to 3.2–3.6 with 0.02 mol/L sodium hydroxide solution. For each concentration, 3 mL of solution was taken, and the absorbance was measured at 369 nm by UV spectrophotometry, with 3 groups in parallel. The concentrations of lactic acid and sorbitol were 0.0009 g/mL and 0.0225 g/mL, respectively.
Irinotecan Loading
The clinical application is based on one bottle of commercially available microspheres; therefore, this study compared the irinotecan loading of one bottle of DC Beads, one bottle of CalliSpheres, and one bottle of NAMs, with microsphere volumes of 2 ml, 1 ml, and 1 ml, respectively. Irinotecan exists as a closed-ring lactone and an open-ring carboxylate, of which only the lactone is active, and this equilibrium is controlled by pH. The carboxylate is the predominant form in neutral and alkaline environments, while acidic conditions shift the equilibrium toward the active lactone form. 19 Based on its method of use in the clinic, 4 mL of 25 mg/mL irinotecan hydrochloride solution with a lactic acid concentration of 0.0009 g/mL and a sorbitol concentration of 0.0225 g/mL was added, and the pH was adjusted to 3.2–3.6 by the addition of 2 mol/L sodium hydroxide. One bottle of microspheres was mixed with 4 mL of irinotecan solution at a concentration of 25 mg/mL and then incubated at 37 °C with gentle rotation. At 0, 30, 1, 2 and 24 h, 30 µL of each supernatant was mixed with 1.77 mL of solution (solution composition: 0.885 mL of 0.0018 g/mL lactic acid + 0.885 mL of 0.045 g/mL sorbitol). The absorbance of the supernatant at 369 nm at different times was determined using a UV spectrophotometer, and the amount of irinotecan was calculated by applying a standard curve (y = 0.031x + 0.0409, R2 = 0.9991). The loading capacity was calculated as follows: loading capacity = initial irinotecan amount - sum of the amount of irinotecan remaining in the supernatant at different time points.
Irinotecan Release
A bottle of drug-eluting beads containing 4 ml of irinotecan at a concentration of 25 mg/ml was placed in 250 ml of PBS at pH = 5.6 and pH = 7.4 and then placed in an oven at 37 °C for spin release of the drug. A 200 ml sample was collected at 30 min and after 1, 2, 4, 6, 8, 24, and 48 h and was then supplemented with 200 ml of solvent. The concentration of irinotecan was determined by methods described previously. The release at each time point was calculated as follows: Release amount = Cn × V − Cn – 1× residual volume, Cumulative release = sum of releases at each time point.
Morphology and Physical Properties
The morphological properties of each drug-eluting bead (DEB) were obtained using an optical microscope and a JSM-IT800 scanning electron microscope (SEM, JEOL, Japan). The diameters of the microspheres before and after loading and after drug release were evaluated via the dynamic light scattering method using a Mastersizer 2000 (Maerwen Co.), and the shrinkage percentage was calculated as follows: percentage of shrinkage (%) = (diameter of the original DEBs - the diameter of the DEBs after loading or elution)/diameter of the original DEBs.
Statistical Analysis
The data are presented as the mean ± standard deviation. Comparisons between two groups were performed by t tests, and paired t tests were used for pre–post comparisons of the same group. SPSS Statistics 23.0 and GraphPad Prism 8.0 were used for statistical analysis and plotting. P < 0.05 was considered significant.
Results and Discussion
Loading
The loading of irinotecan by the three types of DEBs is shown in Figure 1. Overall, all three types of microspheres could be rapidly loaded with irinotecan and could reach plateau levels within 30 min. The drug-loading amount of DC Beads in 30 min was 98.39 ± 1.44 mg, which was greater than those of NAMs (89.96 ± 0.68 mg) and CalliSpheres (88.39 ± 0.87 mg). The drug-loading capacity of DC Beads was significantly greater than those of the other two kinds of microspheres (P < 0.05), and there was no significant difference in the drug-loading capacity of NAMs and CalliSpheres (P = 0.068). The drug-loading capacities of the three DEBs varied, and the loading amounts and loading efficiencies of all three DEBs were acceptable considering the clinical application dosage. Both DC Beads and CalliSpheres are composed of nondegradable hydrogel microspheres of polyvinyl alcohol (PVA), which have smooth surfaces, and the resulting microspheres are mainly characterized by the presence of a large amount of negatively charged AMPS (2-acrylamido-2-methylpropanesulfonate). The charge‒charge interactions between the positively charged irinotecan drug and the negatively charged sulfonic acid groups of the microspheres represent the primary mechanism of binding.4,20,21 NAMs also rely on surface sulfonic acid groups to bind irinotecan. Although the three microspheres were loaded with irinotecan using similar mechanisms, the amounts of drug loaded were different, probably because we were comparing microspheres based on the clinical application setting, but there were also some differences in the volume and quality of each brand of microsphere.Differences in the amount of AMPS caused differences in the amount of drug loaded.

Irinotecan loading of NAMS, DC beads, and CalliSpheres in 25 mg/ml solution. Error bars indicate standard deviations (n = 3).
Release
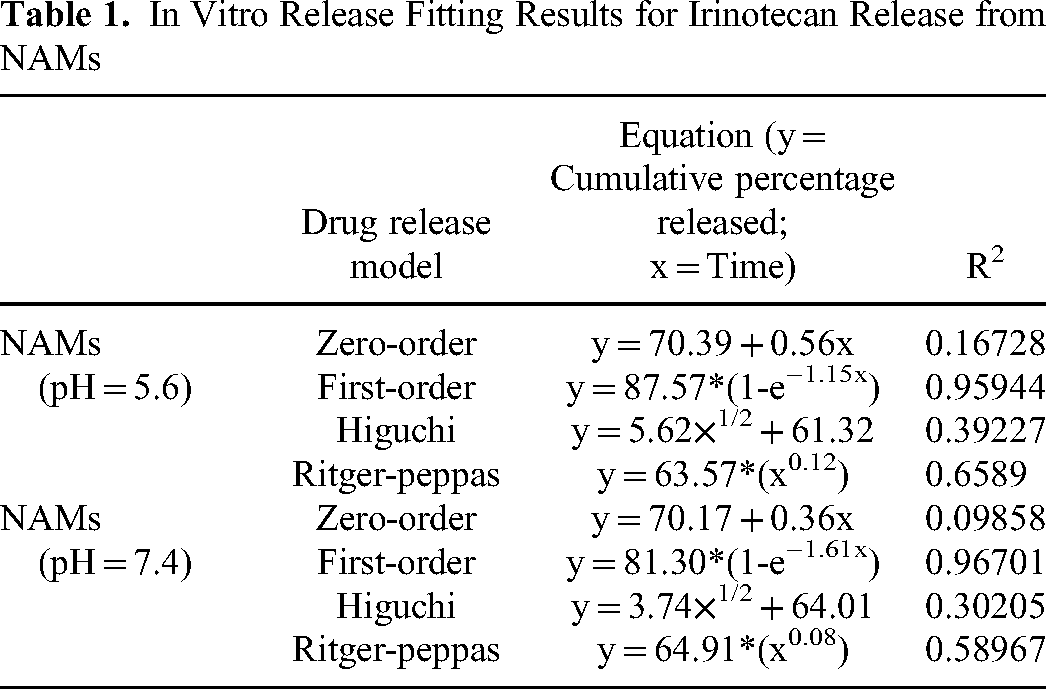
The cumulative amounts of irinotecan released at both pH values are shown in Figure 2 and Figure 3. Consistent with previous studies, initial burst effects were observed for irinotecan release from DC Beads and CalliSpheres microspheres.22–24 At both pH values, the DC Beads were released within two hours, and the CalliSpheres were released within one hour. The interaction between the irinotecan tertiary amine and DEBs was weak, and higher drug loads led to significant shrinkage of DEBs and a shortening of the drug diffusion pathway, resulting in faster drug release. 25 NAMs had the advantage of sustained and slow release of irinotecan compared to DC Beads and CalliSpheres, which may be related to two factors: the more hydrophobic, lower water-content polymer matrix had an impact on slowing the diffusion of the drug, and the relative roughness of the surface slowed the release of the drug. Therefore, NAMs were able to release irinotecan continuously for 48 h. In PBS at pH 5.6, the cumulative release amounts of NAMs, DC Beads and CalliSpheres were 79.09 ± 4.50 mg, 77.46 ± 3.96 mg and 62.07 ± 0.34 mg, respectively, within 48 h. In PBS at pH = 7.4, the cumulative release amounts of NAMs, DC Beads, and CalliSpheres were 72.60 ± 4.66 mg, 74.44 ± 2.42 mg and 61.86 ± 2.25 mg, respectively, within 48 h. There was no significant difference in the cumulative amounts of irinotecan released from the three types of DEBs in PBS at either pH (NAMs: p = 0.158; DC Beads: p = 0.323; and CalliSpheres: p = 0.882). There was no significant difference in the release of DC Beads or NAMs at either pH (pH = 5.6, p = 0.662; pH = 7.4, p = 0.578), and the release amounts of DC Beads or NAMs were significantly greater than that of CalliSpheres (pH = 5.6, DC Beads vs CalliSpheres, p = 0.021; NAMs vs CalliSpheres, p = 0.003; pH = 7.4, DC Beads vs CalliSpheres, p = 0.003; NAMs vs CalliSpheres, p = 0.023). The release mechanism of NAMs loaded with irinotecan tended to follow first-order kinetics (Table 1), suggesting that drug release was concentration dependent, primarily due to diffusion, and that the rate of drug release was proportional to the amount of drug remaining, with the rate of release initially increasing and then decreasing over time.

Irinotecan release of NAMS, DC beads and CalliSpheres in PBS at ph 5.6. The error bars indicate the standard deviation (n = 3).

Irinotecan release of NAMs, DC beads and CalliSpheres in PBS at ph = 7.4. Error bars indicate standard deviations (n = 3).
In Vitro Release Fitting Results for Irinotecan Release from NAMs
Morphology and Physical Properties
The optical microscopy results are shown in Figure 4. NAMs were black before and after irinotecan loading due to the multimodal nanovisible spheres assembled on their surface, which means that NAMs can be visualized in suspension without the addition of dye. The DC Beads and CalliSpheres were both transparent blue spheres, and after drug loading, they appeared green. SEM images before and after irinotecan loading are shown in Figure 5. NAMs had round and uneven surfaces before loading, and after loading, NAMs had a slightly altered appearance but still resembled a round shape, and their surfaces become flatter relative to those before loading. The DC Beads and CalliSpheres were round and smooth before drug loading, and DC Beads and CalliSpheres were round and had smoother surfaces after drug loading. The surfaces of all three DEBs became smoother after drug loading, possibly because small drug molecules stuck to the surfaces of the microspheres. SEM images at both pH values are shown in Figure 6. There was no significant difference in the morphology of the DC Beads when they were released at the two pH values, as both maintained round and smooth surfaces. There was no significant difference in the appearance of the NAMs after release at the two pH values, though their shape changed slightly to an oval shape. CalliSpheres maintained a round appearance when released at both pH values but had a smoother surface at pH = 7.4 than at pH = 5.6. Subsequently, as shown in Figure 7, the dynamic light scattering method was used to determine the diameters of the unloaded, irinotecan-loaded and post-eluted NAMs, DC Beads and CalliSpheres. Before loading, the diameters of the NAMs, DC Beads and CalliSpheres were 253.23 ± 0.71 μm, 333.76 ± 4.84 μm and 331.36 ± 4.85 μm, respectively. After loading, the diameters of the NAMs, DC Beads and CalliSpheres were 270.28 ± 4.23 μm, 212.29 ± 3.61 μm and 198.64 ± 2.42 μm, respectively, and the shrinkage percentages of the NAMs, DC Beads and CalliSpheres were −6.73%, 36.39% and 40.05%, respectively. After release, the diameters measured at pH 5.6 were 267.54 ± 7.49 μm, 342.11 ± 4.39 μm and 324.67 ± 3.67 μm, respectively, and the shrinkage percentages of NAMs, DC Beads and CalliSpheres were −5.65%, −2.50% and 2.02%, respectively. The diameters of the NAMs, DC Beads and CalliSpheres were determined to be 263.19 ± 6.18 μm, 336.01 ± 3.87 μm, and 323.01 ± 3.24 μm at pH 7.4, respectively, and the shrinkage percentages of the NAMs, DC Beads and CalliSpheres were −3.93%, −0.67% and 2.52%, respectively. The DC Beads and CalliSpheres showed significant shrinkage after drug loading due to the charge neutralization and water displacement induced by hydrophobic drug–drug and drug–bead interactions. 25 The size of the NAMs did not change significantly after drug loading and release. The visualization components of the NAMs are the rigid and hydrophobic Fe3O4@PEI@Au nanoparticles, which are distributed on the surface of the microspheres, which may be why the NAMs did not shrink after drug loading. The only commercially available visualization microsphere, DC Bead LUMI, has a core chemistry and interacts with drugs in the same way as DC Beads, but the average size of the microspheres does not change significantly after loading with drugs, which is consistent with NAMs.11,26

Morphologies of unloaded and irinotecan-loaded NAMs, DC beads and CalliSpheres under an optical microscope. The scale bars are 100 μm.

SEM images of NAMs, DC beads and CalliSpheres microspheres before and after irinotecan loading.

SEM images of NAMs, DC beads and CalliSpheres microspheres after irinotecan release in PBS (pH = 5.6, pH = 7.4). The left scale bars are 50 μm, and the right scale bars are 5μm.

Changes in the diameter of NAMs, DC beads and CalliSpheres microspheres before and after irinotecan loading and release.
Discussion
An important aim of using DEBs is to achieve sustained drug release and reduce systemic toxicity. Irinotecan acts primarily through its conversion to the active metabolite SN-38. Both DC Beads and CalliSpheres demonstrated an initial burst release of irinotecan in this study, and it is expected that NAM-based delivery will afford a longer phase of low-level SN-38 exposure; this may be advantageous because prolonged exposure to low levels of SN-38 provides greater antitumor activity. 27 Due to limitations, only in vitro studies were performed, and in vivo pharmacokinetic studies are needed to characterize the relative differences in vivo. It is expected that a slow and sustained release would reduce drug clearance and increase drug bioavailability. Conventional DEBs are not visible by either CT or digital subtraction angiography. A contrast agent can be mixed with DEBs during the procedure to observe where the DEBs are located; however, the contrast agent quickly disperses with the bloodstream; thus, the final location of the DEBs cannot be confirmed. Radiopaque beads have been successfully visualized under CT in the human liver, revealing the distribution of the beads in real time, which means that the surgeon can keep abreast of the embolization effect and adjust the surgical plan during the procedure. 28 With the advancement of interventional procedures, the use of intraoperative imaging devices such as cone beam CT and combined angiography and CT systems has become increasingly popular. These devices can be utilized in conjunction with radiopaque beads to obtain additional quantitative information to assist in surgery; for example, it has been found that it may be possible to estimate drug doses based on the measured attenuation of radiopaque beads. 29
Imaging follow-up is necessary for postsurgical patients; however, radiopaque beads may be difficult to distinguish from iodinated contrast agents because they both appear to have high density on CT images, which interferes with the imaging physician's judgment of tumor status (ie, complete response, partial response, stable disease and progressive disease). 30 However, we have developed NAMs that can be visualized under CT and MR; thus, when there is a disagreement in the CT judgment of the tumor condition, the NAMs can be evaluated on MR. Due to the Fe3O4-based materials on the surface of the microspheres, NAMs are good T2-weighted MR contrast agents, showing low signal intensity on T2-weighted MR, and have little effect on T1-weighted MR. Gadolinium-based contrast medium is a T1-weighted MRI contrast agent that shows high signal intensity on T1-weighted MR images. Therefore, NAMs may not significantly interfere with the development of gadolinium-based contrast medium.
Drug-eluting beads must be nontoxic and easy to deliver, and in our previous study, we demonstrated that different concentrations of NAMs had negligible cytotoxicity. 17 We also demonstrated good suspension stability of NAMs in a suspension of 1 ml of NAMs and 10 ml of 300 mg I/mL iohexol contrast agent (Yangzijiang Pharmaceutical Group Co., Ltd), and during embolization of a porcine hepatic artery, the microspheres smoothly passed through a 2.7F microcatheter (MC-PC2715/, Terumo Corporation, Japan) to reach the target location without aggregation or blockage of the catheter, which will be mentioned in future studies.
There are several limitations to this study. Though drug elution systems can characterize drug release profiles in vitro, the in vivo environment is much more complex and does not provide insight into drug release in vivo, and the final location of the DEBs delivered to the liver depends on the physical interaction of the beads with the tissue. The spatial distribution of the drug depends on the drug elution profile and drug-tissue interactions. 31 Most of the clinically used microspheres are nondegradable, and the microspheres are deposited in tumors and tumor blood vessels through blood flow. Due to their nondegradability, the blood vessels are occluded, and the drug is slowly released. Ischemia and drug toxicity continue to exert antitumor effects. Because NAMs are stable, with no significant shape change or fragmentation observed after loading or releasing the drug, the microspheres will likely never move again and will permanently occlude the vessel; this increased capacity for visualization facilitates subsequent monitoring of the microspheres’ position. In DEB-TACE, by embolizing the major arterial vasculature, there are instances in which tumors are not completely killed. Hypoxia induces the expression of angiogenesis-promoting genes such as vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF), which stimulate neovascularization and restore blood supply to the tumor. 32 Clinical management of this condition is tricky because NAMs are not degradable and cannot reenter the previous artery for treatment, the newly formed vessel is small and difficult to access through the microcatheter, and the physician may use nonselective treatment, which in this case increases the risk of hepatic impairment.
Conclusions
In conclusion, the present study identified the similarities and differences between multimodal imaging NAMs and two commercially available microspheres loaded with irinotecan. NAMs had a similar drug-loading capacity to DC Beads and CalliSpheres and provided a relatively sustained and slow release of irinotecan compared to the other two microsphere bursts of irinotecan. In addition, NAMs can be visualized under MR and CT. We believe that NAMs have good application prospects in TACE treatment of colorectal cancer liver metastases.
Footnotes
Acknowledgments
We are grateful to the National Natural Science Foundation of China (Nos. 82072032 and 61905057) and the Zhejiang Provincial Natural Science Foundation of China (LY21A040002) for their financial support.
Author Contributions
Y.Z. and G.S. designed and supervised the experiments. Y.Z. and G.S. provided funding. J.W., S.Z., Y.H., W.S., X.Z., Z.X., Q.M., Y.Y., and Z.S. conducted the experiments and analyzed the experimental data. J.W. and S.Z. wrote the original draft. Y.Z. proofread the manuscript. All authors have read and agreed to the published version of the manuscript and agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval Statement
The research conducted in this paper did not involve any human or animal research, nor did it use any sensitive data requiring special ethical considerations. In accordance with the Measures for Ethical Review of Life Science and Medical Research Involving Human Beings formulated by the Chinese government and the relevant parts of the Declaration of Helsinki of the World Medical Association, this study did not need to be approved by an ethical review committee.
Funding
This research was funded by the National Natural Science Foundation of China (Nos. 82072032 and 61905057) and the Zhejiang Provincial Natural Science Foundation of China (LY21A040002).
