Abstract
Purpose
The aim of this study was to describe the safety and efficacy profiles of TACE using DC Beads LUMI.
Materials and methods
We retrospectively analyzed 90 patients with HCC who underwent TACE with DC Bead LUMI™ between November 2018 and November 2020 at Fondazione IRCCS Cà Granda Policlinico Hospital in Milan, Italy. Patient- and tumour-related factors were registered, including the number of treated lesions, dose of DC Beads LUMI™, dose of Epirubicin, DC Beads LUMI™ target tumour coverage (LC) according to the percentage of target nodule involvement (LC1-0%–25%, LC2-25%–50%, LC3-50%–75%, LC4 75%–100%). Treatment efficacy was obtained through reviewing the follow-up imaging for evidence of response in target lesion(s), according to modified response criteria in solid tumours (mRECIST) criteria with the following outcomes: complete response (CR), partial response (PR), stable disease (SD) and progressive disease (PD). Safety assessment was based on the quantitative and qualitative recording of the adverse events, classified according to CIRSE classification.
Results
Seventy-two patients were enrolled, and 95 procedures were carried out. We observed a target tumour response rate at 1 month with CR in 68%, PR in 10.3% 11.8%, SD in 13%, PD in 7.2%, and an overall tumour(s) (whole liver) response at 1 month with CR in 58.9%, PR in 12.6%, SD in 10.5% and PD in 18%. We found a significant association (p < 0.01) between tumour response CR or CR + PR and the number of the target lesion(s). CIRSE classification grade I and grade II complications were recorded, respectively, in 11 (11.6%) and 6 (6.3%) procedures. No grade III-IV-V complications occurred.
Conclusion
TACE using DC Beads LUMI is a safe and effective treatment option for patients with HCC.
Introduction
Hepatocellular carcinoma (HCC) accounts for 85%–90% of primary liver cancers and approximately 6% of all cancers, 1 being the fourth major cause of cancer-related deaths globally. 2
Therapeutic approaches for the treatment of HCC can be classified into three categories: potentially curative, palliative and symptomatic.
Potentially curative treatments include liver resection, transplantation and image-guided locoregional treatments.
Locoregional treatments comprising thermal ablation, trans-arterial chemoembolization (TACE), and radioembolization with Yttrium-90 embedded microspheres. 3
The shortage of organ donors and high costs limits the application of liver transplantation, raising demands for image-guided locoregional embolotherapy. 4
According to the Barcelona Clinical Liver Cancer (BCLC) system, TACE is the first-line treatment for intermediate stage disease (BCLC-B), which includes asymptomatic patients with limited unresectable multinodular lesions, without vascular invasion or extrahepatic spread and who have well-preserved liver function (Child-Pugh stage A-B and Eastern Cooperative Oncology Group performance status of 0). 5
The BCLC system also provides for TACE to be used in patients with very early stage (BCLC-0) and early stage (BCLC-A) in whom the recommended treatments are not feasible or have failed. 5
The 2022 BCLC version stratifies the BCLC-B stage into 3 groups of patients according to tumour burden and liver function. The first subgroup within BCLC-B includes patients with well-defined HCC nodules, who are candidates for liver transplantation (LT); the second subgroup comprises patients, candidate for TACE, without the option of LT but who have preserved portal flow and defined tumour burden, suggesting the feasibility of selective access to feeding tumour arteries; the third subgroup includes patients with diffuse, infiltrative, extensive HCC liver Involvement, for which is recommended the systemic therapy. 5
Conventional TACE (cTACE) consists of the intra-arterial injection of chemotherapeutic drug emulsified in an oil-based radiopaque agent (Lipiodol) into the tumour target, while drug-eluting bead (DEB)-TACE involves the use of microspheres that actively sequester and then slowly release the cytotoxic drug inside the target lesion.6,7
The advantage of DEB-TACE compared to cTACE is its lower concentration of chemotherapeutic reaching the systemic circulation, with fewer attendant side effects. Nevertheless, unlike cTACE, in which the oil-based radiopaque agent (Lipiodol) may be retained by tumour cells for several months with a possibility of visualization on post-operative computed tomography (CT), 8 following DEB-TACE the embolized territory cannot be usually directly visualized. 7
DC Bead LUMI™ (Biocompatibles UK Ltd., UK) is a bead with iodine incorporated into its chemical structure resulting in permanent radiopacity, 9 thus combining the advantage of low systemic drug absorption and visualization on post-operative imaging.
DC Beads LUMI™ is approved by the manufacturer, Boston Scientific, for loading with Doxorubicin, Epirubicin and Irinotecan.
Nowadays, there is no univocal agreement on the optimal chemotherapy to use in TACE, though Irinotecan is most used in colorectal cancer, while Doxorubicin and Epirubicin are the most used agents in HCC. 10
The aim of this study was to describe the safety and efficacy profiles of TACE using DC Beads LUMI™ in patients with HCC.
Material and Methods
Patients
For this retrospective observational study, patients with HCC treated with TACE with DC Beads LUMI™ between November 2018 and November 2020 were included.
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of IRCCS Ca’ Granda Policlinico di Milano (Project identification code: OSMAMI-23/05/2023-0021935-U).
The reporting of this study conforms to STROBE guidelines. 11
Written informed consent was obtained from all patients.
Indication to treatment was given in each case by a multi-disciplinary tumour board including surgeons, hepatologists and interventional radiologists. In addition, each patient had a visit with an interventional radiologist before procedure in which informed consent was taken.
All patients were deemed suitable for TACE.
Inclusion criteria for TACE with DC Beads LUMI™, according to Barcelona Clinical Liver Cancer (BCLC) guidelines, were Stage B BCLC (BCLC-B) and, when the recommended treatments were not feasible or have been failed, in patients with Stage 0 or Stage A of BCLC (BCLC-0, BCLC-A). 5
Exclusion criteria include patients who underwent additional treatments for HCC following TACE in the first month following TACE, patients without follow-up imaging available and patients out of indications, patients BCLC-C and BCLC D, patients with bi-lobar involvement, patients with a tumour size > 9 cm and patients with contraindications to TACE.
Absolute contraindications to TACE include portal vein neoplastic thrombosis or hepato-fugal blood flow, impaired hepatic function (Child-Pugh B8 or greater), poor performance status (ECOG P2 or greater) and contraindication for arteriography.
Outcome Measures
We evaluated as primary outcome measures the following: number of treated lesions, dose of DC Beads LUMI™, dose of Epirubicin, DC Beads LUMI™ target tumour coverage (LC) according to the percentage of target nodule involvement (LC1 0%–25%, LC2 25%–50%, LC3 50%–75%, LC4 75%–100%), TACE-related complications according to Cardiovascular and Interventional Radiological Society of Europe (CIRSE) classification 12 target lesion's response rate and overall tumour(s) response rates according to modified response evaluation criteria in solid tumours (mRECIST) criteria, with outcomes classified as complete response (CR), partial response (PR), stable disease (SD) or progressive disease (PD).13,14
Liver function laboratory data were assessed pre- and post-procedure.
CT or MRI imaging follow-up was done as follow-up at 1 month and then at 3, 6, 9, 12 months post- DC Bead LUMI™ TACE and we considered overall (or liver) tumour(s) progression and disease-free survival.
Procedure and Technical Considerations
DC Bead LUMI™ has the same core chemical structure as DC Bead™, a polyvinyl alcohol-2-acrylamido-2-methylpropane sulphonic acid hydrogel bead, with the difference in iodine- containing-moiety of 23,5-triiodobenzaldehyde, added to the PVA backbone. 13 This iodine-containing-moiety modifies the bead's physical proprieties in three ways: makes the beads radiopaque, increases the beads’ density by 30% and reduces the beads’ compressibility by two orders of magnitudes compared to the DC Bead™. 10 The increased density leads to a rapid sedimentation; for this reason, is recommended to mix the DC Bead LUMI™ is mixed with a high-viscosity contrast agent to maintain a workable suspension and to flush the microcatheters with contrast agent rather than saline to ensure a bead clearance. 9 We used DC Bead LUMI™ with a size range of 75–150 um.
Beads were loaded with Epirubicin by hospital pharmacy staff according to protocols developed by the manufacturer: each vial of DC Beads LUMI™ (2 mL of beads) was loaded with 70 mg of Epirubicin.
Written informed consent was obtained from all patients. All procedures were performed in accordance with the 1964 Helsinki Declarations and its later amendments. 15
All procedures were performed with the same protocol. The procedures were performed in an angiographic suite using digital subtraction angiography (DSA) and cone-beam-CT (CBCT).
Through a 5-Fr transfemoral introducer sheath and a 5-Fr Cobra or Simmons 1 catheter (Cordis, Miami Lakes, Florida), a preliminary selective angiography of the celiac trunk and a catheterization of the common hepatic artery was achieved.
In all patients, a 2.7 F microcatheter (Progreat™, Terumo, Tokyo, Japan) was used to achieve a super-selective micro-catheterization of tumour vessels.
DC beads LUMI™ was loaded with Iopromide 350, as contrast media, with a ratio of 2 mL beads: 20 mL contrast agent.
The mixture was shaken between two syringes to obtain a uniform suspension and then slowly injected from a 2 mL syringe under direct fluoroscopic guide.
The mixture was injected under fluoroscopic guidance and the endpoint was a “tree-in-winter” appearance with occlusion of small tumour-feeding radicals. (Figures 1, 2, 3)
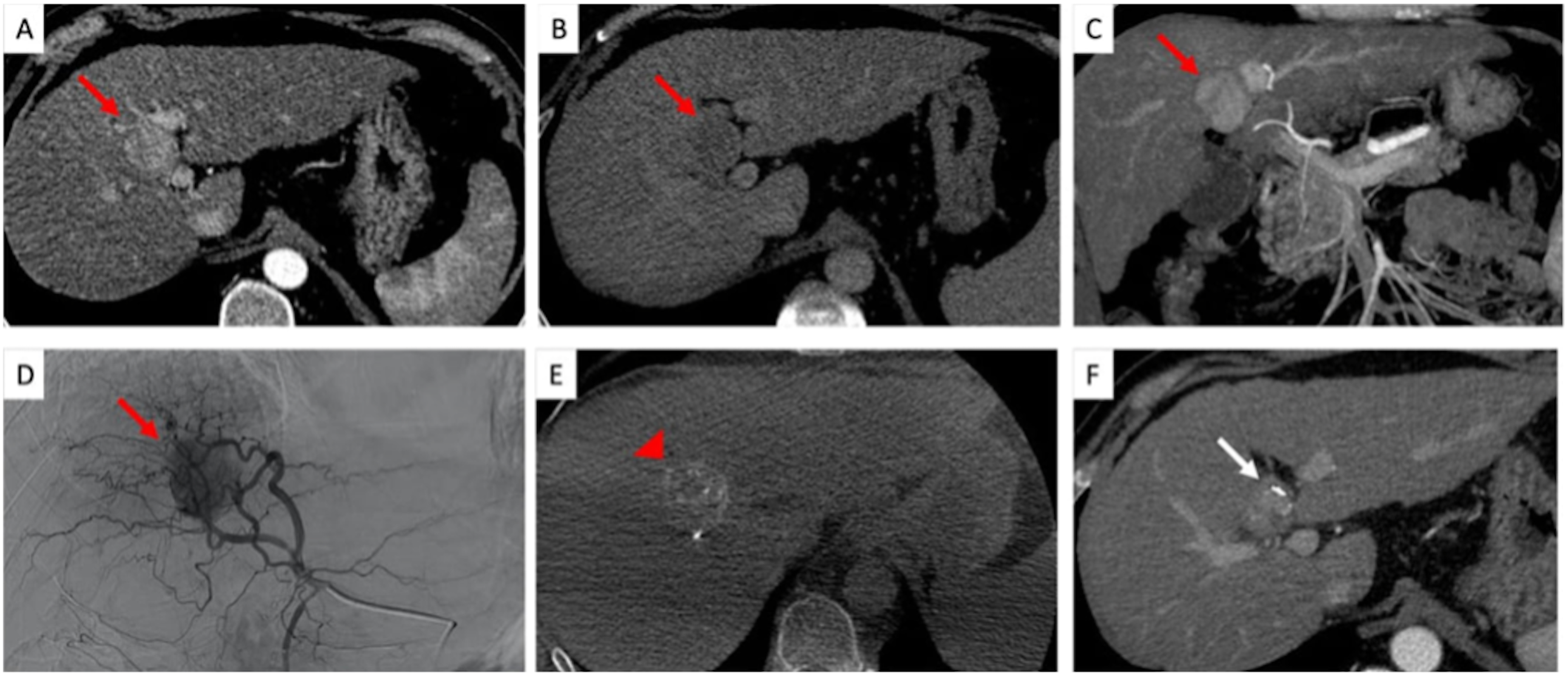
TACE treatment of an HCC nodule using LUMI beads with residual disease at follow-up. Pre-procedural CECT shows a typical HCC lesion (red arrow) with wash-in in arterial phase (A) and wash-out in late phase (B). (C) MIP reconstructions in the coronal plane are performed to study the lesion (red arrow) vascularization. (D) Intraprocedural angiographic study from the left hepatic artery demonstrates the lesion with its feeding vessels (red arrow). (E) Non contrast-enhanced CBCT performed after treatment shows hyperdensity within the lesion corresponding to focal LUMI beads deposition within the target lesion. (F) Follow-up CECT at 1 month shows the presence of contrast enhancement within the treated lesion corresponding to residual disease. Abbreviations – CBCT: cone-beam computed tomography; CECT: contrast-enhanced computed tomography; HCC: hepatocellular carcinoma; MIP: maximum intensity projection.

TACE treatment of an HCC nodule using LUMI beads with no residual disease at follow-up. The pre-procedural CECT exam shows a subcapsular lesion measuring 2.6 cm with wash-in (A) and wash-out (B), consistent with HCC. (C) Intraprocedural angiographic study performed from the right hepatic artery shows the tumor and its feeding vessels (D) The non-contrast-enhanced CBCT performed at the end of treatment demonstrates hyperdense material within the lesion compatible with LUMI beads accumulation (F) Follow-up CECT at 1 month shows no signs of residual HCC. Abbreviations - CBCT: cone-beam computed tomography; CECT: contrast-enhanced computed tomography; HCC: hepatocellular carcinoma.
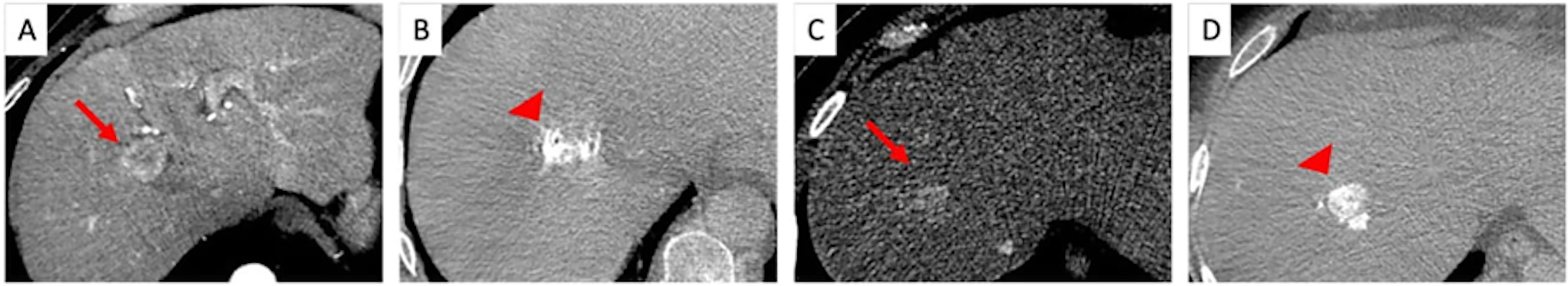
Target lesion coverage by LUMI at the end of TACE procedure. In one patient the pre-procedural CECT shows the HCC target lesion hyperenhancing in arterial phase (arrow, A), while the non-contrast-enhanced CBCT performed after TACE reveals that the hyperdense area corresponding to the desposition of LUMI beads is covering 50% of the target lesion (arrow head, B). In another patient, the pre-procedural CECT shows an HCC lesion with wash-in in arterial phase (arrow, C), with the non contrast-enhanced CBCT performed at the end of TACE procedure demonstrates that the hyperdensity caused by the LUMI deposition is covering nearly the entire target lesion volume (arrow head, D). Abbreviations - CBCT: cone-beam computed tomography; CECT: contrast-enhanced computed tomography; HCC: hepatocellular carcinoma; TACE: transarterial chemoembolization.
Sometimes for the accomplishment of tumour arterial devascularization, it requires the use of additional embolic agents during the procedure, such as unloaded microspheres of different size or Spongostan (absorbable hemostatic gelatin sponge).
Laboratory Analysis Data
As pre- and post-treatment laboratory data, we have collected the following: albumine, bilirubin, alphafeto protein, prothrombin time (PT), activated partial thromboplastin time (aPTT) and INR (International Normalized Ratio). The ALBI (Albumin-Bilirubin) Grade score was also assessed.
Follow-up Imaging
Standard imaging follow-up was quadruple-phase CT (pre-contrast, arterial, portal venous and delayed venous phases) at 1 month post-TACE, and then at 3-month intervals thereafter.
Magnetic resonance imaging (MRI) was reserved for patients allergic to CT contrast media agents.
Statistical Analysis
The descriptive data was defined as mean, median, standard deviation and range for continuous variables and as counts and percentages for categorical variables.
Logistic regression analysis was used to identify the correlation between tumour response at 1 month according to mRECIST guidelines and LC coverage and with the number of treated lesions.
Time to progression and survival were all analyzed using Kaplan–Meier.
Results
Patients
Between November 2018 and November 2020, 90 consecutive patients with HCC were submitted to TACE with DC Beads LUMI™. Eighteen patients were censored: two were out of indication (one Child-Pugh C11 and one bi-lobar multifocal disease), five underwent additional treatments for HCC in the first month following TACE (four underwent radiofrequency thermal ablation (RFTA) and one LT) and 11 didn’t have follow-up imaging available (we assumed that the subsequent follow-up was done in different hospitals). Therefore, a total of 72 patients took part in the research project, of which 19 were female (26.4%) and 53 were male (73.6%). The median age at first treatment was 68.0 (mean 67.71 ± 11.46, range 36–88).
The disease characteristics, including etiology of liver disease, extra-hepatic disease and Child-Pugh classification, are summarized in Table 1. The major risk etiological factors in our patients were hepatitis B virus (HBV), hepatitis C virus (HCV), nonalcoholic steatohepatitis (NASH), alcoholic hepatitis and primary biliary cholangitis (PBC) (Table 1). Moreover, in our populations, the patients also had extra-hepatic disease as hypertension, renal failure, diabetes heart disease and extra-hepatic tumours (Table 1). We found nine patients had extra-hepatic tumours: three had stage I Kidney cancer, three had stage IA lung cancer and three had stage I breast cancer.
The Disease Characteristics Including Etiology of Liver Disease, Concurrent Liver Disease, Cirrhosis/Chronic Hepatitis/Steatosis/Steatohepatitis, Extra-Hepatic Disease and Child-Pugh Classification.

Abbreviations: HBV, hepatitis B virus; HCV, hepatitis C virus; NASH, nonalcoholic steatohepatitis; PBC, Primary biliary cholangitis.
Treatment Data
Ninety-five TACE procedures with DC Beads LUMI were carried out in 72 patients, a mean of 1.32 ± 0.55 procedures per patient (median 1.00, range 1–3). Most patients received 1 (52%–72.2%) or 2 (17%–23.6%) TACE with DC Beads LUMI; only 3 (4.2%) patients received three TACE with DC Beads LUMI.
Among all patients, 26 (36.1%) of them did not receive any previous treatment before TACE with DC Beads LUMI, 46 patients received previous treatment (63.9%) and are classified according to the type of treatment(s) administered and the status of the lesion in Table 2.
Type of Previous Treatment(s) and Previous Treatments According to Lesion status.

Abbreviations: TACE, trans-arterial chemoebolization; MWTA, microwave thermal ablation; RFTA, radiofrequency thermal ablation; PEI, percutaneous ethanol injection; TAE, trans-arterial embolization; TARE, transarterial radioembolization.
The use of additional embolic agents in this study was limited to only 15 treatments (15.8%).
Drug
The mean Epirubicin dose was 25.91 ± 16.33 mg (range 9.38–75 mg, median 18.75 mg).
According to Person correlation, the Epirubicin dose and the size of the target lesion(s) are directly correlated (R = 0.47, p < 0.0000034) (Figure 4).
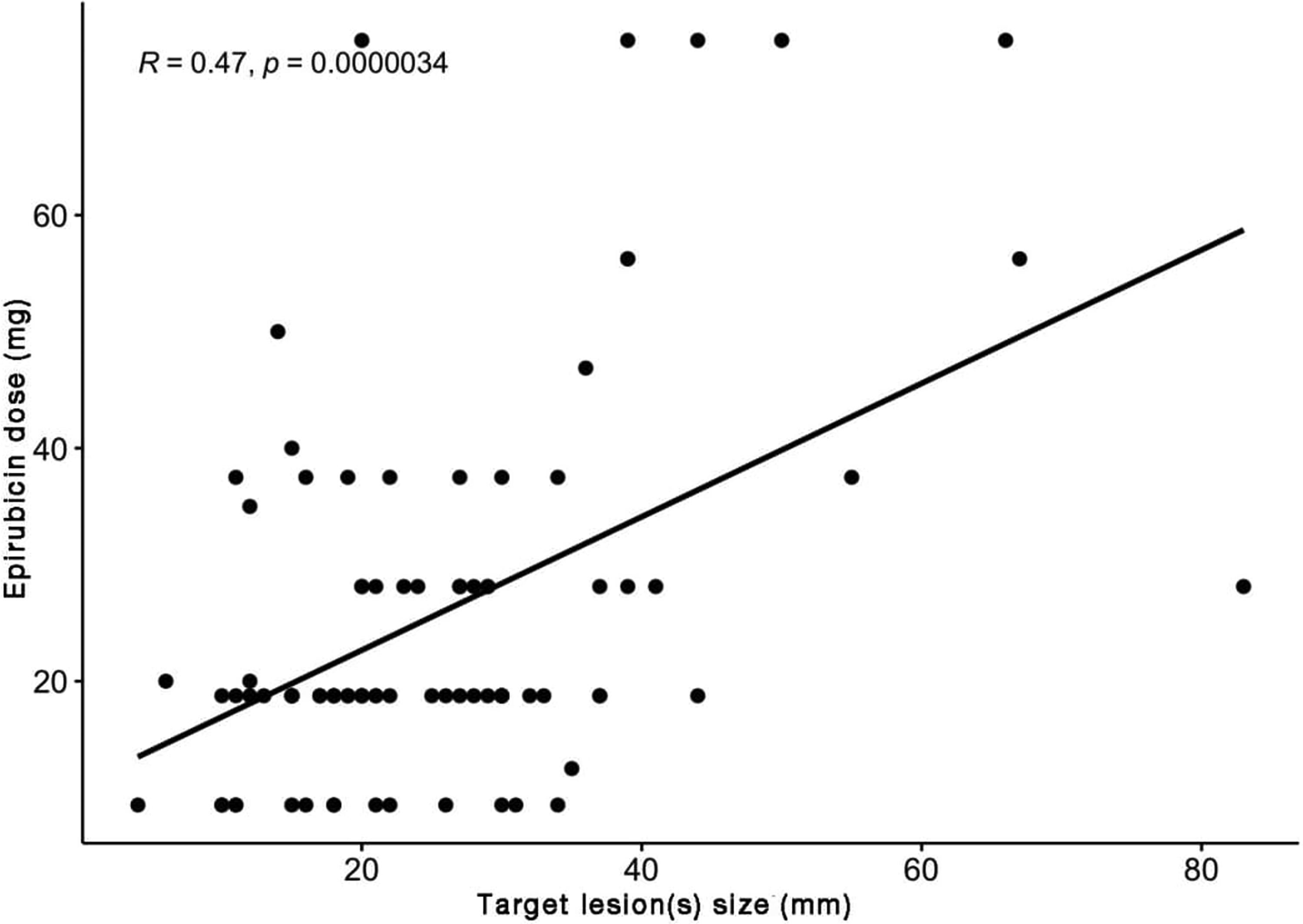
Scatterplot of Epirubicin dose as function of target lesion(s) size.
Lesion Characteristics
The mean number of tumours per patient was 1.54 ± 1.14 (range 1–3, median 1). Most patients only had one lesion (65–69.9%), 18 (19.4%) patients had two lesions, five (5.4%) had three lesions, two (2.2%) patients had four lesions and three patients (1%–1.1%) had respectively 5–6–8 lesions.
Most patients treated one lesion (71%–74.7%), 20 (21.1%) patients treated two lesions and four (4.2%) patients treated three lesions. In addition, we observed that in most cases (89.2% of patients) the number of treated lesions did coincide with the number of total lesions throughout the liver.
As for the lesion size, the mean diameter of target lesions was 25.42 ± 13.29 mm (range 4–83 mm, median 22 mm).
Of the 95 lesions treated, 76 (80%) had an arterial phase non-rim enhancement and 19 (20%) had an arterial phase hypo- or iso-enhancement.
Safety
The safety assessment of TACE with DC Beads LUMI™ was based on the quantitative and qualitative recording of the adverse events, classified according to CIRSE classification (Table 3).
Cardiovascular and Interventional Radiological Society of Europe (CIRSE) Classification of Adverse Effects

The median time to observation of adverse events was 3 months post-procedure, even though all adverse events occurred on the same day or the day after the procedure.
All 95 procedures were completed as planned with no intra-operative complications. Thirty-day mortality was 0. The large majority of patients (n = 78; 82.1%) experienced no adverse events; according to CIRSE classification of adverse effects, grade I and grade II complications were recorded, respectively, after 11 (11.6%) and 6 (6.3%) procedures.
The most common side effect was pain (n = 7; 41.3%), followed by fever nausea and/or vomiting (n = 3; 17.6%); all these adverse events belong to the so-called post-embolization syndrome. Other complications were hypotension (n = 3; 17.6%) and dyspnea (n = 1; 5.9%).
Laboratory Analysis Data
Table 4 summarizes the pre- and post-treatment laboratory data evaluated for each patient and ALBI Grade score. (Table 4)
Laboratory Data and Albumin-Bilirubin (ALBI) Score

Imaging Follow-up
CT, as standard imaging follow-up at 1- and 3 months post DC Bead LUMI™ TACE, was used in 70 patients. On the other hand, MRI, was used in two cases, due to allergy to CT contrast media agent, in which, was impossible to calculate the distribution of DC Bead LUMI. On CT images, the embolized territory was clearly visible on the unenhanced scan due to DC Bead LUMI™ radiopacity. The distribution of DC Bead LUMI™ was classified according to the percentage of target nodule involvement: 0%–25% of coverage for 14 nodules (15.1%), 25%–50% of coverage for nine nodules (9.7%), 50%–75% of coverage for 19 nodules (20.4%), 75%–100% of coverage for 51 nodules (54.8%). Using a cut-off of 50% of coverage we evaluated the correlation between the target tumour coverage by DC Bead LUMI™ and the target tumour response rate at 1 month: we found a not statistically significant correlation, using a Pearson Chi-square test (p = 0.605958).
Lesion Response Rate
The efficacy assessment of TACE with DC Beads LUMI was based on the reviewing of the follow-up imaging for evidence of response in the target lesion(s), according to mRECIST criteria.
Median follow-up time was 377 days (range 45–810 days, mean 391.77 ± 226.28 days); for patients who underwent more than 1 TACE, the follow-up period was deemed to start on the date of the TACE treatment. No patient died during the study observation period. One-month follow-up was available for 72 patients (95 treatments).
Target tumour(s) response rates and overall tumour(s) (whole liver) response rates at 1 month are summarized in Table 5.
Target Tumor(s) Response Rate and Overall Tumor(s) Response Rate at 1 Month and Association Between Tumor Response and the Number of Target and Total Lesion(s)

Abbreviations: CR, complete response; PR, partial response; SD, stable disease; PD, progression disease.
Using logistic regression, we found a significant association (p < 0.01) between tumour response CR or CR + PR and the fact that number of target lesion(s) = number of total lesion(s). The probability of having CR is about 20% when the number of target lesion(s) ≠ number of total lesion(s) and 80% when the number of target lesion(s) = number of total lesion(s). (Table 5)
Progression and Survival
At 3, 6, 9 and 12-months follow-up we considered the following parameters: overall (or liver) tumour(s) progression and disease-free survival.
Median overall tumour(s) progression time was 424 days (14.1 months). At 12 months, the probability of observing hepatic progression was around 56%. (Fig.5) The median disease-free survival time was 385 days (12.8 months). (Fig.6)

Overall tumor(s) progression.
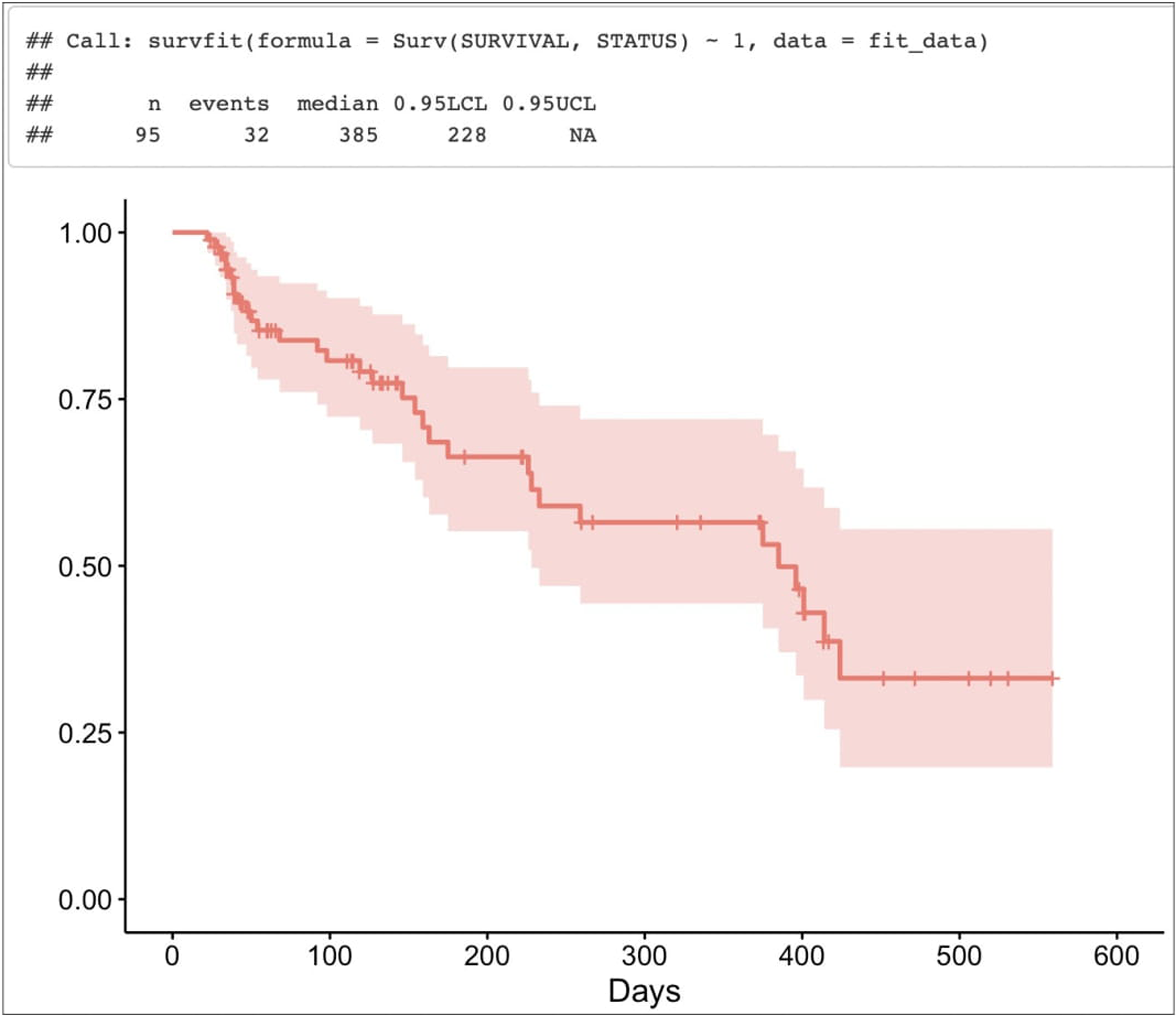
Disease-free survival.
Discussion
According to Barcelona Clinical Liver Cancer (BCLC), TACE is recommended primary therapy for the intermediate stage HCC (BCLC-B). 4 DEB-TACE, conversely to c-TACE, is generally preferred due to its lower concentration of chemotherapeutic agent that reaches the systemic circulation, with fewer attendant side effects, but hasn’t radiopaque proprieties. DC Bead LUMI™ (Biocompatibles UK Ltd., UK) with its chemical structure ensuring that it is permanently radiopaque, combines the advantage of DEB-TACE, with its low systemic drug absorption, and c-TACE, with a better visualization on post-operative imaging. The opacity of DC Bead LUMI™ is different from the radiopacity of lipiodol, because it remains in the vessel and does not interfere with the contrast medium CT scan, with the possibility of visualization of residual tumour or progression of disease. 16
Moreover, the radiopacity of beads, as demonstrated by Reicher et al, in selected cases, ensures the visualization of the embolized lesion in post-operative CT, giving the possibility to use LUMI as a marker of the lesion in case of reintervention, such as for CT-guided ablation. 10
A significant advantage of DC Bead LUMI™ is their direct visualization during the procedure, which allows identifying both intraprocedural and on 1-month CT of the area of treatment.
We found a DC Beads LUMI™ target tumour coverage (LC) of 75%–100% in 54.8% of nodules, but without a significant association on logistic regression analysis between tumour response and LC.
Tolerability analysis showed pain was the most common side effect (7%–41.3%), followed by fever, nausea and/or vomiting (3%–17.6%), hypotension (3%–17.6%) and dyspnea (1%–5.9%). In our study post-embolization syndrome occurs in 17.6% of patients; the comparison with other studies is challenging given the differences in data collection. The most of patients (78%–82.1%) experienced no adverse events. According to CIRSE classification Grade I and II complications were recorded, respectively, in 11 (11.6%) and 6 (6.3%) procedures. Our findings are similar to the published literature: Reicher et al reported 14.5% CIRSE Grade I complications and 7.3% CIRSE Grade II and III complications; Lakhoo et al reported 6.1% CIRSE Grade I and II complications and 6.8% CIRSE Grade III and above complications.3,10
The efficacy assessment of TACE with DC Beads LUMI was based on the reviewing of the follow-up imaging for evidence of response in the target lesion(s), according to mRECIST criteria.
In our study, the analysis of tumour response was limited by the following factors: the limited period of time follow-up and the lack of a control group. On that note, we observed a target tumour response rate at 1 month with CR in 68%, PR in 10.3% 11.8%, SD in 13%, PD in 7.2% and an overall tumour(s) (whole liver) response at 1 month with CR in 58.9%, PR in 12.6%, SD in 10.5% and PD in 18%. As for the comparison with the other two clinical studies which have examined TACE with DC Beads LUMI, Reicher et al reported a lesion response rate at 1 month with CR in 45.7%, PR in 45.7%, SD in 5.7% and PD in 2.9%; Lakhoo et al reported an overall tumour response at 1 month with CR in 19.5%, PR in 28.0%, SD in 29.3% and PD in 22.0% and a target tumour response at 1 month with CR in 30.5%, PR in 25.6%, SD in 41.5% and PD in 2.4%.3,10 CR in our study is better than CR reported in other studies with LUMI. However, our early response rate falls within the expected range previously given by other studies following DEB-TACE of between 44% and 99.5%.16–18
We found a significant association (p < 0.01) between tumour response CR or CR + PR and the fact that the number of target lesion(s) = number of total lesion(s). The probability of having CR is about 20% when the number of target lesion(s) is different from the number of total lesion(s) and 80% when the number of target lesion(s) is the same as the number of total lesion(s).
No patient died during the study observation period. One-month follow-up was available for 72 patients (95 treatments).
The limitations of this study are its retrospective nature, limited sample size and the absence of a control group, which means the impossibility of demonstrating the relative performance of the LUMI with other beads. Moreover, another limitation regarding complications is the retrospective nature of that study, which compared with other studies, underestimate the post-embolization symptoms compared with the study that has questionnaires for all patients. 16
Conclusions
In conclusion, our study demonstrates the efficacy and safety of DC Bead LUMI™ in TACE for the treatment of HCC.
