Abstract
Objectives
The current therapeutic indications of radiosurgery are constantly expanding. Magnetic resonance imaging (MRI) has an important role in the diagnostic and post-therapeutic period of primary and secondary brain tumor formations.
Methods
A total of 66 patients with verified cancer disease and brain metastases were separated into two groups. The first group includes 34 patients with primary non-small cell lung cancer and the second one 32 patients with other types of primary cancer. All of them received high-dose radiotherapy in 1–5 fractions. The number, size, and location of the treated lesions responded to robotic stereotactic radiosurgery criteria. The Response Assessment Criteria for Brain Metastases (RANO-BM) is an international multidisciplinary group of experts who developed acceptable criteria for assessing brain metastases. Before treatment and on the first, third, sixth month after radiosurgery, a MRI and blood tests were performed.
Results
Treated lesions were separated into four groups depending on the results – complete response, partial response, progressive disease, and stable disease. In both groups of patients, the percentage of complete or partial response had increased in the third and sixth months.
Conclusion
The results give us a reason not to recommend an MRI 1 month after treatment if the patient doesn't have any new neurological symptoms, because there may be a pseudo-progression. MRI results valued by RANO-BM criteria give us a good option to evaluate brain metastases on the third and sixth month after after stereotactic radiosurgery.
Introduction
Cancer is a socially significant pathology with increasing frequency and importance in the last two decades. Оverall, cancer mortality has been steadily increasing since 1991. 1 It is a fact that scientific and technological progress has led to in-depth knowledge at the molecular genetic level. The innovations have made significant progress with the refinement of diagnostics and the ability to personalized treatment. Unfortunately, in the last decade (2008-2017), the trend of declining mortality has been “slowing down” in women with breast and colorectal cancer, being at a standstill in prostate cancer. On the other hand, the trend to reduce lung cancer mortality increased from 3% in 2008–2013% to 5% in 2013–2017 for men and from 2% to 4% for women. Despite this statistical fact, lung cancer led to more deaths in 2017 than breast, prostate, colon, and brain cancers combined.2–4
Lung cancer (over 40%), breast cancer (about 20%), and melanoma (20%) are the most common primary diseases for the development of brain metastases (BM). Pulmonary carcinoma is the most common oncological etiology (in 70% of cases), with debut-manifested BM symptoms as an unknown primary disease. BM's occurrence at each oncological site complicates and limits the therapeutic behavior due to pharmacokinetic specificity: single drugs can cross the blood-brain barrier and target brain lesions.5,6 The average survival for untreated patients with BM is about 1–2 months, and this period increases to 6–8 months after radiosurgery and chemotherapy.7–9
Stereotactic radiosurgery (SRS) delivers a high dose of ionizing radiation to a specific target while minimizing the dose to healthy brain tissue. Although SRS's concept dates back to the 1950s, this treatment option has not been clinically applicable for many years. BM have characteristics that make them ideal for such kind of treatment.10,11 Some of these criteria are pseudospherical shape, location in the gray-white matter, and a maximum diameter of most lesions <4 cm. Also, the vast majority of BM have clear pathological and radiographic boundaries, allowing easier delineation of the target. All of the above advantages allow accurate lesion identification, treatment planning, and using multiple intersecting beams that produce a steep dose gradient. Since the 1990s, several studies have been published on SRS results as a primary and adjuvant treatment for BM. 12 At realized doses of 15 to 24 Gy, the local control varies from 70% to 95%. The lack of local progression depends on the dose, volume, and histology. Also, SRS is effective for the treatment of all types of primary tumors, incl. those with radioresistant histology such as melanoma and renal cell carcinoma. 13
The ideal candidate for SRS is the subject of debate but generally is a patient with controlled or no extracranial metastases and ECOG performance status 0–1. The method finds significant therapeutic application in the context of understanding oligometastatic disease (OMD). To investigate the relationship between the maximum tolerated dose of SRS and the tumor's maximum diameter was performed a clinical trial – RTOG-9005 (phase 1-2). 14 It includes patients with recurrent primary and metastatic brain tumors. The maximum tolerated doses for lesions > 2 cm to 3 cm, and > 3 cm to 4 cm are respectively 18 Gy and 15 Gy; no maximum tolerated dose was determined for tumors ≤2 cm due to reluctance to increase the dose above 24 Gy. Since the risk of radiation damage increases with increasing tumor volume and diameter, the doses for BM are inversely proportional to the tumor's maximum diameter and volume.15,16 As a result, the norms for smaller targets’ local control are usually higher than those for larger targets, especially when whole-brain radiotherapy (WBRT) is not part of the treatment strategy. Radiosurgical targets are usually limited to a maximum diameter of 4 cm.17,18
Rapid technological progress in recent years has opened up new opportunities in both radiation oncology and imaging. The criteria for the application of radiosurgery have been increasingly expanded. Managing BM remains a major problem and for the patients with inoperable lesions stereotactic radiosurgery is the only option. For this reason, it is extremely important to determine when to evaluate the effect of the treatment. Based on our clinical protocols we decided to do a MRI on the first, third, sixth, and 12th month after treatment and to use Response Assessment in Neuro-Oncology Brain Metastases (RANO-BM) criteria for evaluation.
Material and Methods
Patients with oncological diseases treated in the Clinic of Radiation Oncology – University Hospital “Sv. Georgi”- Plovdiv for the period 2017–2020 has been included. The patients have an OMD and have performed robotic SRS. In recent years in clinical practice, the term “OMD” has emerged with the concept that oligometastatic disease requires a specific therapeutic approach, including the possibility of applying more radical methods for local control depending on the organ localization of the various metastatic lesions.
The research meets the criteria for scientific ethics of Medical University – Plovdiv and complies with the requirements of: the Declaration of Helsinki on ethics in science; Principle of Good Clinical Practice; Bulgarian laws and regulations for conducting clinical and scientific research involving humans – Opinion №P-9013 / 15.11.2017.
Patients were divided into two groups based on primary disease and the frequency of BM: the first group included 34 (24 male and 10 female) patients with Non-Small Cell Lung Cancer (NSCLC). The second group has 32 patients (21 male, 11 female) with other primary cancer types (OTPC) and metastatic brain disease manifestations. Structurally this group includes patients with breast cancer (14), colorectal cancer (10), kidney cancer (4), small cell lung cancer (4). The average age of patients in the first group is 59,6 (36-84) while in the second one is 65,6 (40-91). The groups have no statistical difference in terms of number of patients, age and gender distribution. The percentage of smokers with NSCLS is 100%, while in the other group it is 75%.
Selection criteria includes patients over 18 years old with histologically verified oncology disease, ECOG PS 1–3, data for OMD, and MRI-proven metastatic brain disease (up to 4 lesions) with localizations and sizes that meet SRS/SRT requirements. Exclusion criteria: not completed a full course of treatment; impossibility (due to refusal or ECOG status) to conduct an MRI at the first, third or sixth month after treatment.
Equipment
All patients received treatment for BM's local control with CyberKnife – a small linear accelerator located on a robotic arm, which allows irradiation from hundreds of different angles. This type of linear accelerator has a specific feature compared to con-ventional external beam radiotherapy machines, although both methods use the same x-rays. Radiosurgery provides highly concentrated radiation to a predetermined target - providing a high dose and precisely positioned radiation to the target with a steep dose gradient relative to the surrounding tissues. The number of fractions depends on the location, size, and number of BM, varying between 1 and 5 in most cases. The duration of each fraction was between 35–55 min. The treatment is non-invasive and painless. SRS was performed with the CyberKnife M6 system with fixed collimators or IRIS variable aperture collimators.
For treatment plan and following-up of patients we were using a MRI and CT with contrast, which are essential for verifying BM. 19 The role of MRI is crucial, as it is informative of the number, size, and prevalence of lesions. BM detection can be increased by using a triple dose of contrast. The use of F-deoxyglucose positro-emission tomography for the diagnosis is not recommended due to the richness of brain tissue with glucose. Historical data from 1970 to the early 1990s show that 10% of patients diagnosed by CT or MRI were asymptomatic. BM is most often localized in the cerebral hemispheres (80%), followed by the cerebellum (15%) and the trunk (<5%).20,21
Immobilization includes an open-faced antibacterial mask EFFICAST® by ORFIT, CIVCO footrest, and, if necessary, a belt to hold the hands to the body. Image fusion, delineation of tumor volumes and critical organs, as well as dosimetric planning, are performed on Accuray's MultiPlan program. The Gross Tumor Volume (GTV) covers the contrast-enhanced tissue of the BM. The infiltration area around the lesions is usually tiny, so there is no insurance area to the Clinical Target Volume (CTV), respectively GTV = CTV. The security zone to the Planning Target Volume (PTV) is 1–2 mm, respectively GTV + 1/2 mm = PTV. The following critical organs must be identified: eyes, lenses, optic nerves, optic chiasm, pituitary gland, cochlea, brainstem, cerebellum, and whole brain. The maximum permissible doses in critical organs are compared against the criteria of QUANTEC, “National Standard for Cranial and Extracranial Radiosurgery”, International Protocol - The report of TG101 and the ALARA principle.
Statistical Analyses and Evaluation Criteria
Data were processed by one-dimensional frequency analysis. Fisher's exact test was performed for statistical comparison of two proportions.
Historically, the development of criteria for diagnosis, assessment of therapeutic response, and follow-up of patients with brain metastatic disease corresponds to the updating of the equipment's technical capabilities. The Macdonald criteria were published in 1990 and aim to assess the effect of treatment in first-line treatment of glioblastoma multiforme. They include four types of response: complete and partial response, stable disease, progression. The RESIST (Response Evaluation Criteria in Solid Tumors) criteria updated the therapeutic assessment and were published in 2000. They expanded the possibilities by including a detailed description of ‘measurable’ and ‘immeasurable’ lesions. Their update was made in 2009, as RESIST 1.1.22–24 In 2010 were published RANO (Response Assessment in Neuro-Oncology) criteria. Include an assessment of first-line response to all histological variants of glioblastoma (not just glioblastoma multiforme), including low-grade astrocytoma. Again, the therapeutic effect is characterized as four types of response (complete and partial response, stable disease, progression) based on MRI objectification. In 2011 a working group was formed, which proposes new criteria for evaluating BM in particular – RANO-BM.25,26 The following-up of patients was with MRI through RANO-BM criteria which divide metastases into target and non-target lesions. The target lesions are treated with CyberKnife, and non-target lesions are newly diagnosed ones.
Complete response - the disappearance of all target lesions for at least 4 weeks; without the new metastases appearance; without corticosteroids; improved or stable clinical condition of the patient; Partial response - > 30% reduction in > 4 weeks; without new metastases; the same or lower dose of corticosteroids; improved or stable clinical condition; Stable disease - < 30% reduction and <20% increase in lesion size; without new metas-tases; Progressive disease - > 20% increase in existing lesions relative to baseline, at least one of the lesions must have increased by at least 5 mm. Complete response – the disappearance of all non-target lesions; without new metas-tases; Progressive disease – an increase of existing lesions or the appearance of new ones; Non-complete response or non-progressive disease - one or more of the non-target le-sions persist
Results
On the first, third, sixth, and 12th month after treatment, control MRI of the head and blood tests were performed. The MRI results are applied to assess the therapeutic response of BM after the application of SRS in both patient groups (with NSCLC and OTPC). One evaluation of MRI results characterizes the dynamics of target metastases according to the accepted RANO-BM criteria: 0 = complete response; 1 = partial response; 2 = progression; 3 = stable disease. The second assessment includes non-target lesions in three categories: 0 = complete response; 1 = incomplete answer; 2 = progression.
MRI Results of Target Lesions
The dynamic monitoring of metastatic brain disease in patients with NSCLC diagnosis is summarized in Figure 1. Three main trends are outlined. The first trend is an increase in patients’ percentage with a complete response from 12% at the first month to 24% at third month and 25% at sixth month. The increase between the first and sixth months amounted to 13% but did not reach statistical significance (p = 0.132).

Results of dynamic monitoring of targeted brain metastases in patients with NSCLC.
The second trend included an increase in patients’ percentage with a partial response from 44% at first month to 67% at third and sixth months, with a marginally significant difference of 23% (p = 0.051). Overall, patients with a complete and partial response at the sixth month accounted for 92%.
The third trend is observed in a significant decrease in patients with the stable disease from 46% at first month to 4% at sixth month, with a difference of 42% (p < 0.001).
Only in the category with a progressive disease, no significant dynamics were observed, with a minimal increase in patients’ percentage from 0% to 4% between the first and sixth month.
In patients with OTPC diagnoses, similar trends were found in metastatic brain disease dynamics between the first and sixth months. Figure 2 shows an increase in the percentage of patients who achieved a complete response from 9% in first month to 26% in the sixth month. The increase amounts to 17%, but the difference does not reach significance (p = 0.107).
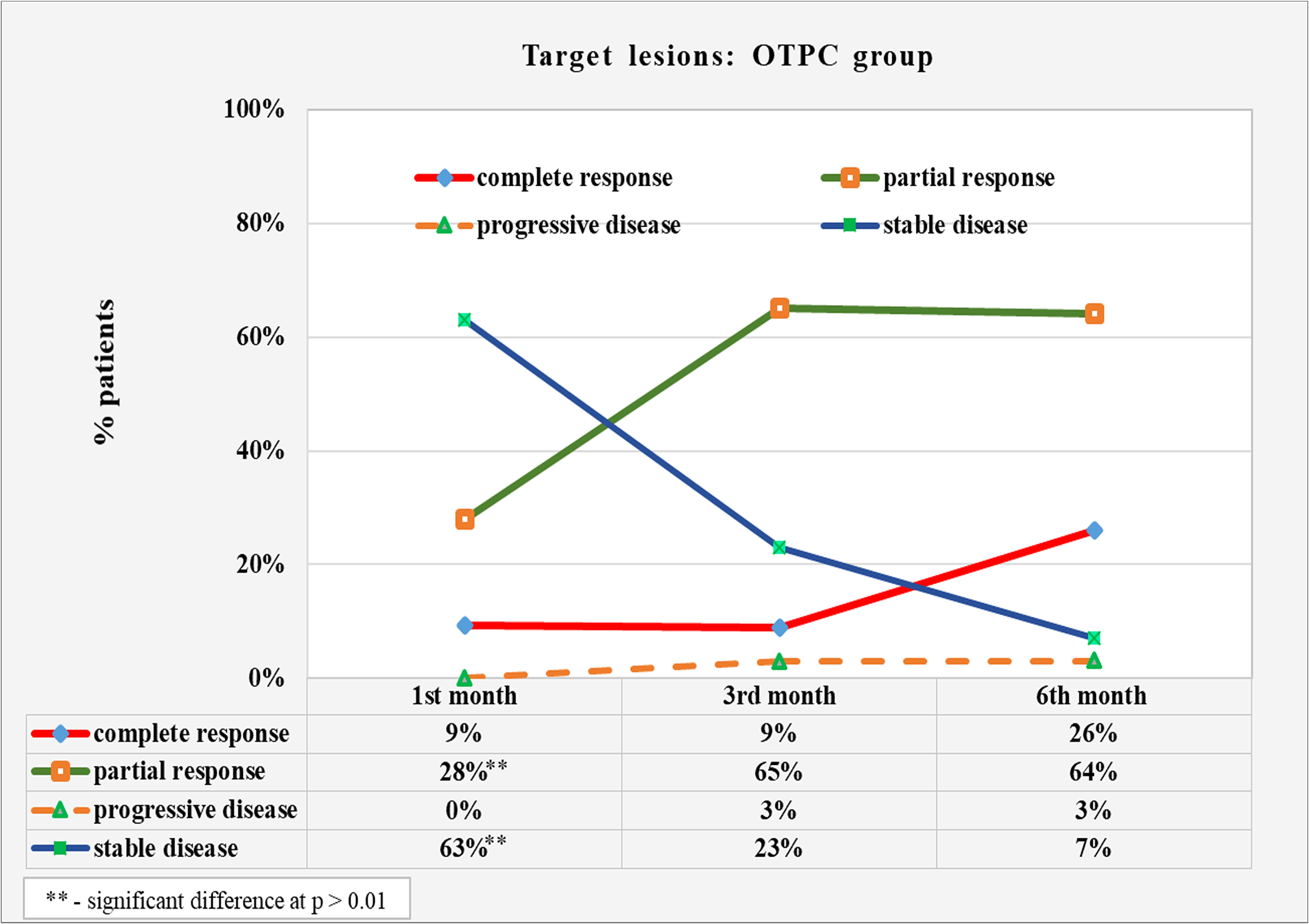
Results of dynamic monitoring of targeted brain metastases in patients with OTPC.
The next trend is an increase in patients’ percentage with a partial response from 28% at first month to 65% at third and 64% at sixth month, with a significant difference of 36% (p = 0.005) between first and sixth month. Overall, patients with a complete and partial response at the sixth month accounted for 90% of the group.
The third significant dynamics is observed in reducing the percentage of patients with the stable disease from 63% at first month to 27% at third and 7% at sixth month, with a significant difference of 56% (p < 0.001) between the st and sixth month.
The category of patients with the progressive disease did not show significant dynamics, with a minimal increase in the percentage of patients from 0% to 3% between the first and sixth month.
The dynamic monitoring of newly detected brain metastases in patients with NSCLC diagnosis is summarized in Figure 3. There was a slight decrease in the patient's percentage with no recently detected brain metastases from 77% at first month to 62% at third and sixth month in the category of complete response. The overall reduction rate in cases with a complete response was 15%, with no statistical significance of the change (p = 0.419). Over the same period, patients with incomplete response increased from 3% at first month to 12% at third and 25% at sixth month. The difference between the first and sixth months showed a significant 22% increase in incomplete response cases (p = 0.026). The percentage of patients with the progressive disease showed a slight fluctuation from 20% at first month to 25% at third month and decreased to 12% at sixth month, with no significant changes (p = 0.337).
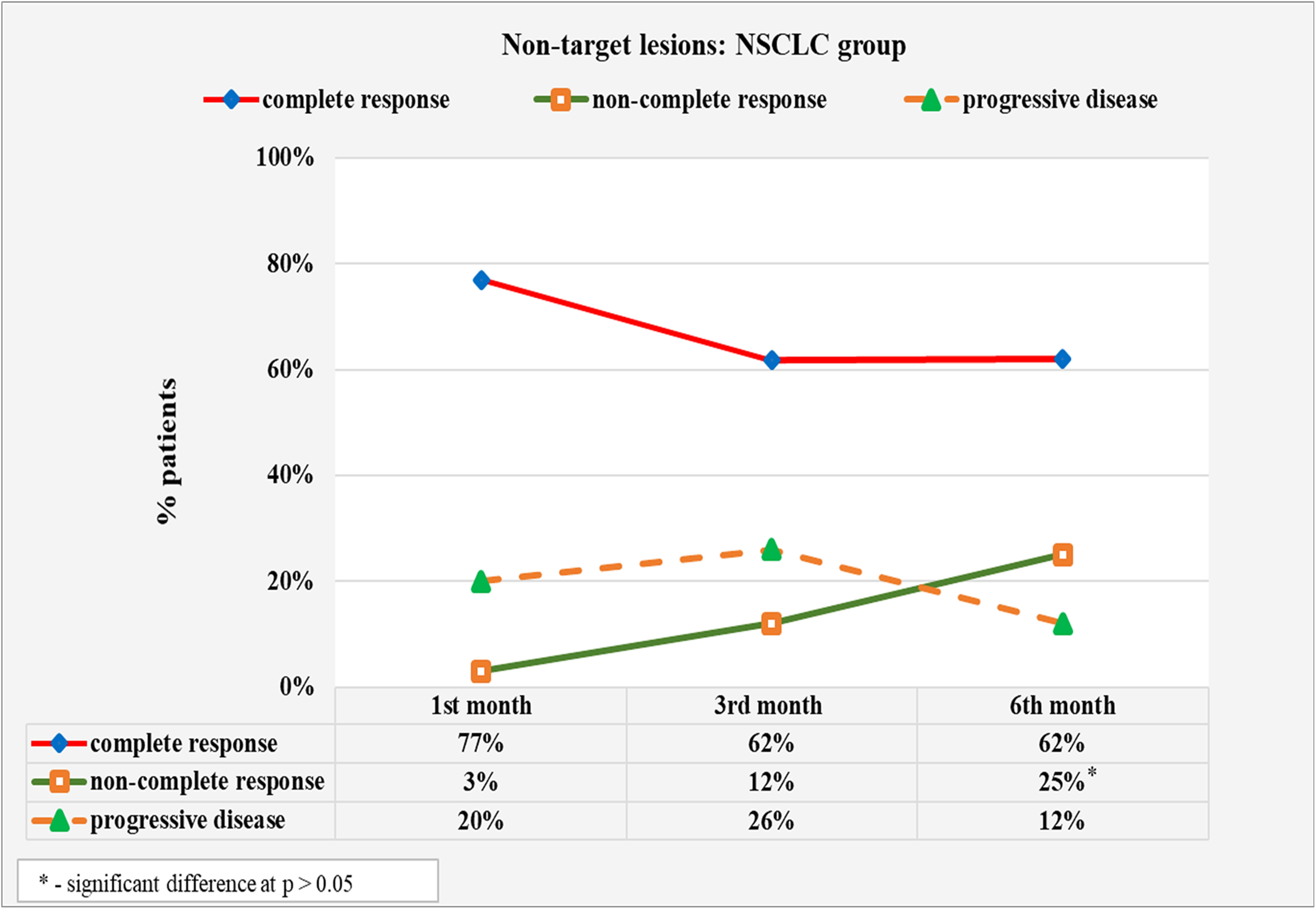
Results of dynamic monitoring of newly diagnosed (non-target) brain metastases in patients with NSCLC.
Two significant trends were identified in the group of patients with OTPC diagnoses (Figure 4). The percentage of patients with a complete response decreased from 84% at first month to 60% at third, and 56% at sixth month. The reduction in cases with a complete reaction between the first and sixth month amounted to 28%, with the change's statistical significance (p = 0.027). In the opposite direction, the percentage of patients with incomplete response changes, increasing from 0% in the first month to 12% in the third and 26% in the sixth month. The 26% increase in incomplete response cases between the first and sixth month was statistically significant (p = 0.005). The percentage of patients with the progressive disease increased from 16% in first month to 28% at third month, then decreased to 17% at sixth month, with no significant changes (p = 0.365).

Results of the dynamic monitoring of newly diagnosed (non-target) brain metastases in patients with OTPC.
In both patient groups, there is a steady trend of a significant increase in cases with a partial response between first month and sixth month. In the NSCLC group, the patients with partial response increased by 23%, and in OTPC, by 36%. In both patient groups, stable disease incidence decreased significantly between the first and sixth months. In the NSCLC group, the reduction of cases amounts to 42%, and in the OTPC group by 56%. In both patient groups, there was a lasting, but not significant, the trend of increasing cases with a complete response. The thin percentage increased by 13% in the NSCLC group and 17% in the OTPC group. In both groups, a significant increase in cases of incomplete response was found. In the NSCLC group, the patients with incomplete response increased by 22%, and in OTPC, by 26%. In the group of patients with OTPC diagnoses, there was a significant decrease in cases with a complete response by 28%. The NSCLC group also found a 13% reduction in these incidents, but the trend was not significant
The results show the best local control on the third and sixth months after SRS. In some cases, it is possible to see a pseudo-progression of the performed MRI in the first month. The overall survival of patients with treated brain metastases is 6–8 months. Due to the small number of surviving patients one year after treatment, there is no way to make a statistically reliable analysis of MRI results at 12 months. They are not presented and compared with the other results.
Brain is one of the most common organs for developing distant metastases from various types of cancers. Due to pharmacokinetic specificity radiosurgery is the best treatment option for inoperable lesions, and in some cases preferable by patients with operable metastases because of its non-invasive and painless approach. Management of the metastatic disease includes symptomatic treatment and definitive therapy, with the goal of stabilizing and improving neurologic function and survival. Advances in imaging and radiation oncology in recent years led to even better diagnostic and treatment options. Historically treatment of BM went through whole brain radiotherapy, intensity-modulated radiotherapy, volumetric modulated arc therapy, and now SRS/SRT, thanks to which we can perform a higher dose in tumor and minimize side effects. CyberKnife is a small linear accelerator design specifically for SRS/SRT, which can deliver a very high daily dose to a small volume with accuracy up to 1 mm. The ideal candidate for radiosurgery is a subject of debate but generally is a patient with controlled or no extracranial metastases and good ECOG performance status. The method finds significant therapeutic application in the context of understanding OMD.
Although MRI is a golden standard for diagnostic and following up of patients with BM, CT remains a vital tool for initial work-up and perioperative management. While certain findings on standard and advanced imaging are suggestive of a particular diagnosis, to date imaging is not able to reliably predict the histology of a brain metastasis. As research in the area advances, and the field of molecular imaging matures, this may become feasible in the future. 27 After introduction of RANO criteria there are strict indications for post-treatment monitoring of lesions, which are used in some clinical trials and gives us a chance to compare the results in different radiation oncology centers. In most of this articles RANO criteria are used to compare a response of immunotherapy versus SRS; WBRT versus SRS; results after reirradiation because of BM local failure; or for primary brain tumors.
Our primary objective is to demonstrate the practical application of the RANO-BM criteria in a clinical setting by categorizing lesions into target and non-target groups to achieve more precise results. This categorization enables us to compare both local tumor control after radiosurgery and the overall disease status, including the potential occurrence and development of new metastases. Standardizing the criteria for evaluating the effectiveness of radiosurgery on BM is crucial for comparing outcomes across different centers worldwide. Such standardization will facilitate comparisons of various doses and fractionation schedules, different irradiation techniques, and, if necessary, the revision of existing guidelines. Potentially we can use them to evaluate for early and late side effects too.
Our second objective is to determine the optimal timing for assessing the impact of radiosurgery. Based on our analysis, we have found that the most significant results are typically observed at the third and sixth months post-therapy. Consequently, our center has ceased routine MRI scans at the first month post-treatment due to the still minimal effect and potential for pseudo-progression. We now recommend additional imaging only if there is an intensification of symptoms or the emergence of new complaints. Given the low survival rate of patients with BM, typically 6–8 months, we have been unable to gather statistically significant data on treatment effects beyond one year. Moving forward, we are considering including an evaluation at the ninth month post-therapy to capture more comprehensive data on the long-term effects of treatment.
Conclusion
Our results show very good therapeutic control of brain metastases proven by MRI in patients with proven cancer and OMD. Data support the place of radiation oncology and, in particular radiosurgery, in the complex treatment of these patients. RANO-BM criteria are suitable for the staging of the lesions and follow-up of these patients. Our recommendation is to perform a control MRI on third and sixth month after treatment.
The utilization of radiosurgery in managing BM presents a promising avenue in oncology, offering patients a minimally invasive yet effective treatment modality. Our study sheds light on the efficacy of radiosurgery in this context, employing the RANO-BM criteria as a standardized method for evaluating treatment response. The findings underscore when we should evaluate the results and when we could expect the best local tumor control.
Moreover, the incorporation of standardized assessment methodologies on a certain period after treatment such as RANO-BM criteria enhances the comparability of different clinical studies. This standardization not only facilitates evidence-based practice but also enables researchers to conduct meaningful comparisons and meta-analyses, advancing our understanding of treatment outcomes in BM. The study's adherence to standardized assessment criteria contributes to the growing body of literature supporting the use of radiosurgery as a valuable therapeutic option in the management of BM.
Footnotes
Abbreviations
Author Contributions
Conceptualization: V.P. and Zh. G-P.; methodology: V.P. and G.R.; validation: V.G.; formal analysis: G.R.; investigation: V.P.; resources: P.U.; data curation: V.G. and P.U.; writing—original draft preparation: V.P.; writing—review and editing: V.P., G.R. and Zh. G-P.; visualization: V.G. and P.U.; supervision: G.R. and V.G.; project administration: Zh. G-P. All authors have read and agreed to the published version of the manuscript.
Data Availability
The data presented in this study are available on request from the corresponding author. The data are not publicly available due privacy restrictions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Institutional Review Board Statement
The study was performed according to the Declaration of Helsinki, and approved by the Research Ethics Committee of Medical University – Plovdiv (№P-3327//20.12.2017).
