Abstract
Tumor growth and metastasis rely on angiogenesis. In recent years, long non-coding RNAs have been shown to play an important role in regulating tumor angiogenesis. Here, we review the multidimensional modes and relevant molecular mechanisms of long non-coding RNAs in regulating tumor angiogenesis. In addition, we summarize new strategies for tumor anti-angiogenesis therapies by targeting long non-coding RNAs. The aim of this study is to provide new diagnostic targets and treatment strategies for anti-angiogenic tumor therapy.
This review summarizes the multidimensional modes and relevant molecular mechanisms of lncRNAs in regulating tumor angiogenesis, and new strategies for tumor anti-angiogenesis therapies by targeting lncRNAs.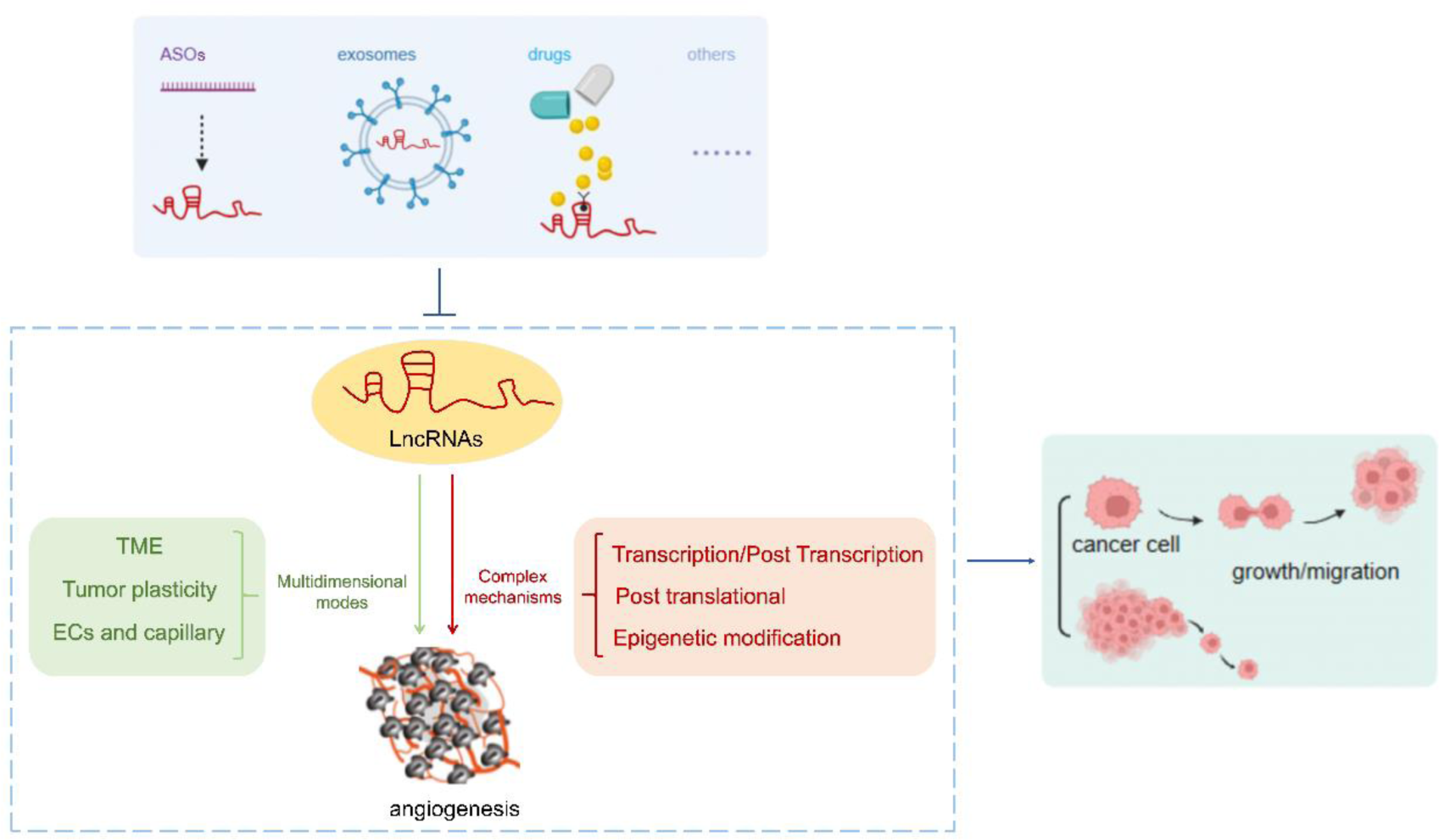
Introduction
In 1971, Folkman proposed that tumor growth and metastasis depend on angiogenesis. 1 Angiogenesis is the formation of new blood vessels on the basis of preexisting capillaries or postcapillary venules, which is one of the hallmarks of cancers associated with malignancy and the poor prognosis. 2 Physiological angiogenesis is a short-term, controlled process that occurs during embryogenesis, tissue repair, and regeneration. In contrast, pathological angiogenesis is associated with disease and involves uncontrolled endothelial cells (ECs) proliferation, migration, and degradation of the extracellular matrix (ECM). 3 The vascular morphology and function in tumor tissues are quite different from those in normal tissues. 4 Neovascularization in tumor tissue lacks complete coverage of pericytes, ECs and basement membrane (BM), therefore these blood vessels have tortuous, dilated, irregular morphology and high leakage characteristics, which are more conducive to tumor metastasis. 5
Patterns of angiogenesis in tumor could be summarized into three categories: budding and proliferation of ECs, tumor cell vasculogenic mimicry (VM), and endothelial transdifferentiation of cancer stem cells (CSCs). 6 Angiogenesis is a complex process controlled by many different proangiogenic and antiangiogenic factors, such as vascular endothelial growth factor (VEGF), angiopoietin (ANGPT), platelet-derived growth factor (PDGF), fibroblast growth factor (FGF) and other pro-angiogenic factors. 7 Meanwhile, endostatin, angiostatin, platelet factor 4 (PF4), interferon (IFN) and other inhibitory angiogenic factors prevent excessive angiogenesis. 8 In hypoxia tumor microenvironment (TME), the balance between pro-angiogenic and anti-angiogenic factors is broken. ECs are activated and proliferated, while ECM is degraded by various proteolytic enzymes. As a result, neovascularization occurs. 9
Angiogenesis-associated factors and signaling pathways have become promising targets for anti-cancer therapy; however, their long-term efficacy is not satisfactory because of drug resistance and alternative angiogenesis modes. 6 To treat tumors more effectively, we need to further explore the mechanisms of tumor angiogenesis and identify more effective therapeutic targets and strategies.
Long non-coding RNAs (LncRNAs), initially considered non-functional evolutionary garbage, have gradually been discovered to play key roles in many pathological processes, especially cancer. 10 LncRNAs are expressed in almost all types of cancers and are potential cancer regulators, which are mainly located in the nucleus but could also located in the cytoplasm. According to their subcellular localization, lncRNAs may have different functions.11,12 They could participate in transcription and post-transcription processes, epigenetic chromatin remodeling, protein function, small RNA maturation, and other important biological processes, including gene regulation.13–16 They also affect the biological behavior of tumor cells, such as stem cell maintenance, cell proliferation, apoptosis, cell cycle, invasion, and migration, and play an important role in regulating the occurrence and development of tumors.17–20 It is worth mentioning that lncRNAs are involved in regulating tumor angiogenesis, while the mechanism is extremely complex.
Here, we reviewed the different mechanisms of lncRNAs-mediated angiogenesis and the potential value and feasibility of targeting lncRNAs for anti-angiogenic tumor therapies, and provided a summary and future prospects.
LncRNAs Play a Multidimensional Regulatory Role in Tumor Angiogenesis
An increasing number of studies have found that lncRNAs play a direct or indirect regulatory roles in angiogenesis and participate in tumor angiogenesis in multi-dimensional manners, including the regulation of the TME, impacts on tumor cells themselves, and direct effects on vascular endothelial cells (VECs).
LncRNAs Affect the TME
Malignant tumor tissues have complex structures consisting of malignant and non-transformed cells by recruiting normal cells to the tumor tissue. 21 TME is consists of tumor cells, ECs, immune cells, fibroblasts, macrophages, ECM, and secreted substances such as cytokines and growth factors. 22 TME could be regulated by lncRNAs and involved in tumor angiogenesis through different manners such as ECM remodeling, macrophage polarization, angiogenic factors secretion, and cell-to-cell communication (Figure 1A).

LncRNAs regulate tumor angiogenesis in different manners. (A) LncRNAs regulate angiogenesis by affecting the TME, including the secretion of MMPs for participating in the remodeling of the ECM, the polarization of TAMs, the secretion of angiogenic factors, and the secretion of tumor-derived exosomes; (B) LncRNAs regulate angiogenesis by affecting the plasticity of tumor cells, promoting the differentiation of CSCs into VECs, or promoting the formation of VM in tumor cells; (C) LncRNAs regulate angiogenesis by directly influencing VECs and maintaining the biological behaviors of VECs, such as proliferation, migration, and tubular formation.
LncRNAs Affect the Remodeling of the Tumor ECM
ECM remodeling and tumor progression are two highly interrelated processes. ECM provides a mechanical scaffold for cells and mediates the diffusion of signaling molecules to maintain cell function, and the multifaceted signals generated by tumor cells and immune cells recruited in TME induce changes in the surrounding matrix, which in turn affects the homeostasis of the ECM, 23 resulting in a “cancerous” microenvironment that supports tumor growth and metastasis. 24 ECM is also a key component, allowing cell-to-cell crosstalk and storing many angiogenic factors such as VEGF, PDGF, and FGF. 25 After the degradation of the ECM, angiogenesis factors are released, which induce the migration of ECs to participate in tumor angiogenesis. 26 In addition, ECM components also play a vital role in the development of blood vessels. One study found that angiogenesis is dependent on the secretion and subsequent extracellular deposition of type IV collagen. 27 Laminin also appears to be involved in the regulation of angiogenesis which could inhibit ECs proliferation and maintaining vascular stability. 28 Different types of cells in tumor could produce enzymes, include matrix metalloproteinases (MMPs) and a disintegrin and metalloproteinases (ADAMs) to degrade ECM. 29 Among ECM-degrading enzymes, MMPs play the most important role. They can degrade various protein components in the ECM, destroy the histological barrier of tumor cell invasion, and play a key role in tumor invasion, metastasis and angiogenesis. 26
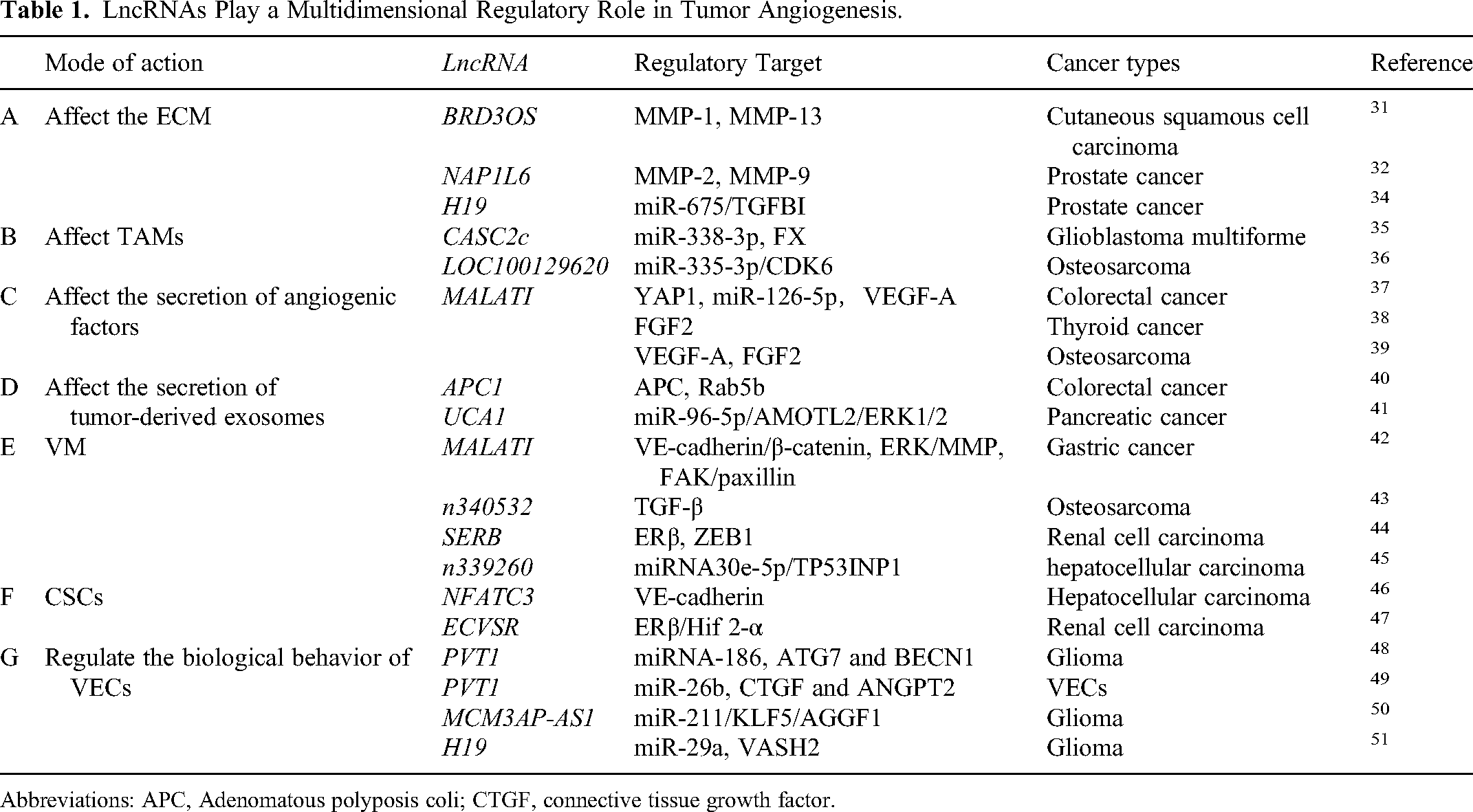
LncRNAs have been shown to promote tumor progression by disrupting the basal layer of the ECM and facilitating the release of metastatic cells into the blood or lymphatic vessels. 30 In cutaneous squamous cell carcinoma (cSCC), BRD3OS (also known as LINC00094) could upregulate the expression of the MMP-1 and MMP-13 and promotes the invasion of cSCC cells into type I collagen and angiogenesis, which is downstream of super enhancer (SE). However, the specific mechanism by which BRD3OS regulates MMP-1 and MMP-13 has not been specified. 31 In prostate cancer (PCa), the lncRNA NAP1L6 promotes the transcription of MMP-2 and MMP-9 to activate the MMP pathway, thereby promoting the malignant progression of tumors, including angiogenesis. 32 Transforming growth factor-beta-induced (TGFBI) is an ECM protein involved in adhesion, migration and chemotaxis. 33 LncRNA H19 plays a key role in the progression and angiogenesis of PCa through the H19/miR-675/TGFBI axis, in which H19 is the primary miRNA precursor of miR-675 and positively correlated with miR-675 34 (Table 1A).
LncRNAs Play a Multidimensional Regulatory Role in Tumor Angiogenesis.
Abbreviations: APC, Adenomatous polyposis coli; CTGF, connective tissue growth factor.
LncRNAs Affect the Polarization of Tumor-Associated Macrophages (TAMs)
TAMs are the main components of inflammatory cells in the TME and are associated with tumor growth, local recurrence, and immunosuppression. 52 Macrophages can be divided into M1 and M2 types according to their activation stage and functional status. Subtype M2d of type M2, which is activated in the presence of tumor-related factors, promotes tumor growth and angiogenesis, and is the main component of TAMs in the TME. 53
Coagulation factor X (FX) can induce macrophages polarization towards M2 subtype, and promotes tumor growth and angiogenesis. In glioblastoma multiforme (GBM) cells, lncRNA CASC2c could inhibit the secretion of FX through the miR-388-3p/FX axis, which interacts with miR-338-3p and inhibits each other. Alternatively, CASC2c could also bind to FX directly and inhibit its secretion. As a result, CASC2c inhibits macrophage M2 polarization and GBM angiogenesis. 35 In osteosarcoma (OS), lncRNA LOC100129620 promote angiogenesis and macrophage polarization through LOC100129620/miR-335-3p/CDK6 signaling axis, in which LOC100129620 regulates the function of miR-335-3p through sponge adsorption and miR-335-3p mediates the regulatory effects of LOC100129620 on CDK6 36 (Table 1B).
LncRNAs Affect the Secretion of Angiogenic Factors in Tumor Cells
Tumor microenvironment possesses a variety of protein peptides and factors, some of which play critical roles in promoting tumor angiogenesis such as VEGF, FGF, and PDGF. 54 VEGF was first identified as a key regulator in tumor angiogenesis 55 and remains the most specific inducer of angiogenesis. 22
As an important member of the lncRNA family, MALATI is expressed in a variety of tumor cells and is involved in regulating the biological behavior and angiogenesis of tumor cells. As an up-regulated oncogene in colorectal cancer (CRC), yes-associated protein 1 (YAP1) interacts with transcription factor 4 (TCF4)/β-catenin and formed a compound to promote the expression of MALAT1 in CRC. According to base complementary pairing, MALAT1 promotes the expression of angiogenesis-related molecule VEGFA and regulates the angiogenesis of CRC by sponging miR-126-5p. 37 In addition, MALAT1 could also promotes proliferation, migration and angiogenesis of thyroid cancer (TC) cells through increase FGF2 secretion. However, the underling mechanism of MALAT1 promoting FGF2 secretion needs to be further studied. 38 Similarly, some researchers considered MALAT1 as a new therapeutic target to prevent OS progression, as its positive regulating roles in VEGF-A and FGF2 expression and inducing angiogenesis 39 (Table 1C).
LncRNAs Affect the Secretion of Tumor-Derived Exosomes
Exosomes are nanovesicles with a diameter of 30-150 nm derived from various cells (including tumor cells) 56 that participate in cell-to-cell communication by transmitting intracellular components, such as DNA, RNA, and proteins. 57 Tumor cells can release millions of exosomes to reprogram the TME and aggravate tumor progression. Tumor-derived exosomes play an important role in tumor angiogenesis. 58
LncRNAs can affect the secretion of tumor-derived exosomes. Adenomatous polyposis coli (APC) gene plays a key role in the pathogenesis of CRC; APC-activated lncRNA APC1 inhibits the production of exosomes by directly binding and reducing the stability of Rab5b mRNA. While when APC is mutated, CRC-derived exosome production would increase and resulting in angiogenesis, tumor growth and metastasis. 40 In addition, tumor-derived exosomes promoted the regulation of angiogenesis by lncRNAs. LncRNA UCA1 is highly expressed in exosomes derived from hypoxic pancreatic cancer (PC) cells, which could metastasize to the recipient cells through exosomes. UCA1 was reported to promote angiogenesis and tumor growth through miR-96-5p/AMOTL2/ERK1/2 axis. Specifically, UCA1 acted as a sponge of miR-96-5p, relieving the repressive effects of miR-96-5p on the expression of its target gene angiomotin-like protein 2 (AMOTL2), which further mediates the activation of the extracellular signal-regulated kinase (ERK) signaling pathway 41 (Table 1D).
LncRNAs Affect Tumor Cell Plasticity
LncRNAs could regulate angiogenesis by remodeling tumor cells, including the formation of VM and participating in regulating the differentiation of CSCs (Figure 1B).
VM refers to highly plastic tumor cells, which undergo de-differentiation to acquire endothelial properties and form a self-organized vascular-like structure. This alternative neovascularization process in tumor development was first discovered in melanoma in 1999 59 it also has the ability to transport oxygen and nutrients 60 and aggravates the development of tumors.
Recently, several lncRNAs have been shown to be involved in tumor VM formation. Li et al found that the expression of lncRNA MALAT1 in clinical specimens of gastric cancer (GC) was closely related to the density of VM and the vascular endothelium. MALAT1 was found to up-regulate the expression of vascular endothelial-cadherin (VE-cadherin), β-catenin, MMPs 2 and 9, membrane type 1 (MT1)-MMP, p-ERK, p-focal adhesion kinase (FAK), and p-paxillin to promote VM and angiogenesis in GC. 42 LncRNA n340532 was reported to have potent VM-promoting and pro-metastasis effects in vivo and in vitro through upregulating the transforming growth factor-β (TGF-β) signaling pathway and promoting the release of angiogenesis-related factors in OS. However, the specific mechanism by which n340532 regulates TGF-β is not described in detail. 43 In addition, lncRNA SERB can act as an upstream regulator of estrogen receptor β (ERβ) to control the VM formation and angiogenesis of renal cell carcinoma (RCC). Mechanistically, SERB could bind to the promoter area of ERβ, which further transcriptionally regulates zinc finger E-box binding homeobox 1 (ZEB1) to induce VM formation. 44 LncRNA n339260 may enhance snail expression by decreasing the expression of miRNA30e-5p, thereby reducing tumor protein p53-inducible nuclear protein1 (TP53INP1) expression. Therefore, a potential n339260/miRNA30e-5p/TP53INP1 regulatory axis was associated with VM formation and hepatocellular carcinoma (HCC) progression 45 (Table 1E).
CSCs are a subpopulation of tumor cells with the capacity for self-renewal and multilineage differentiation, which play a pivotal role in tumor angiogenesis. LncRNAs are known to be involved in regulating CSC differentiation. 61
In HCC, Zhao et al found that the co-expression of c-Myc and sex determining region Y box protein 9 (SOX-2) could induce the expression of lncRNA NFATC3. And NFATC3 was positively correlated with the expression of the CSC marker CD133, confirmed that the expression of NFATC3 correlated with the CSC phenotype. The author further illustrated the role of NFATC3 in VM formation and angiogenesis through promoting the development of CSC subsets and upregulating the expression of VE-cadherin, which is a transmembrane glycoprotein expressed in adherens junctions between VECs and is a marker of VM formation. And the increased expression of vascular-like structures in VM also further indicates that the angiogenesis capacity of the tumor is enhanced. 46 The expression of lncRNA ECVSR is up-regulated in RCC, which could increase CSC phenotype and promote the formation of VM through the ECVSR/ERβ/HIF 2-α signaling axis. In terms of mechanism, ECVSR could directly bind to ERβ mRNA to enhance the stability of ERβ mRNA and upregulate the transcription of hypoxia-inducible factor (HIF) 2-α. In addition, ERβ can also further feedback and regulate the expression of ECVSR, forming a feedback loop between the two 47 (Table 1F).
A Direct Effect on VECs
In recent years, there has been increasing evidence that lncRNAs could regulate the biological behavior of VECs, such as the formation of new blood vessels, to sustain specialized VECs phenotypes and functions. In addition, tumor cells can change the expression of lncRNAs in VECs by stimulating VECs, but what are the determinants of tumor stimulation and how these factors change the expression of lncRNAs in VECs still need to be further studied. It has been reported that conditioned media from tumor cells can stimulate VECs and change the expression level of lncRNAs in VECs,48,51 and we speculate that chemokines, growth factors and others secreted by tumor cells may induce such changes (Figure 1C).
The lncRNA PVT1 has previously been reported to affect the angiogenesis of glioma microvascular endothelial cells (MVECs) by regulating the expression levels of miRNA-186. When glioma-conditioned medium stimulates human cerebral MVECs, PVT1 is highly expressed in glioma VECs. In terms of mechanism, PVT1 binds to miR-186 and inhibits its expression, thus blocks the silencing effect of miR-186 on its targets autophagy related protein 7 (ATG7) and beclin 1 (BECN1). Subsequently, the increased expression of ATG7 and BECN1 protein would increase the proliferation, migration, and angiogenesis of the glioma VECs. 48 In another study, PVTI has been found to directly interact with miR-26b and negatively regulate the expression of miR-26b as a regulator of angiogenesis. 62 Zheng et al found that PVT1 activates connective tissue growth factor (CTGF)/ANGPT2 by binding and degrading miR-26b, triggering the proliferation, migration, and capillary formation of VECs. 49 Moreover, lncRNA MCM3AP-AS1 is over-expressed in glioma associated VECs, which could upregulate the expression of Krüppel-like factors 5 (KLF5) by negatively regulating the expression of miR-211. KLF5 could bind to the promoter region of angiogenic factor with G-patch and FHA domain 1 (AGGF1) and initiate the expression of AGGF1 to promote glioma angiogenesis by triggering the PI3 K/AKT and ERK1/2 signaling pathways 50 ; LncRNA H19 is upregulated in relevant VECs cultured in glioma-conditioned medium. H19 negatively regulates the expression of miR-29a, which in turn upregulates the expression of vasohibin2 (VASH2), thereby promoting glioma-induced angiogenesis in vitro 51 (Table 1G).
Molecular Mechanisms of Tumor Angiogenesis Regulation by lncRNAs
Regulation of Tumor Angiogenesis at the Transcriptional and post-Transcriptional Levels
The lncRNAs can regulate the expression of pro-and anti-angiogenic factors at the transcriptional and post-transcriptional levels and affect tumor angiogenesis. The competitive endogenous RNA (ceRNA) network, formed by the interaction of lncRNA/miRNA/mRNA, is a new regulatory mode of gene expression between RNAs and is a typical post-transcriptional mode. LncRNA may function as ceRNA, which sponges miRNA via complementary miRNA binding sites, and this sponging function antagonizes miRNA availability to mRNAs and attenuates the suppressive roles of miRNA on its targets, 63 which, in turn, plays an important role in tumor angiogenesis (Figure 2A).

Molecular mechanisms of lncRNAs in regulating tumor angiogenesis. (A) LncRNAs regulate the expression level of angiogenic factors at the transcriptional and post-transcriptional levels, including binding to transcription factors or acting as ceRNAs, which competitively occupied the shared binding sequence of miRNA, sequestered miRNA and changed the expression of its downstream target genes; (B) LncRNAs are involved in the post-translational ubiquitination and phosphorylation modification of proteins, and regulates the expression level and activity of proteins; (C) LncRNAs could also regulate tumor angiogenesis by epigenetic modifications, including histone methylation and acetylation.
The lncRNA MAGI2-AS3 is localized in the nucleus, which is low expressed in clear cell RCC (ccRCC) and associated with poor patient survival. MAGI2-AS3 binds to the transcriptional repressor HEY1, reducing the enrichment of HEY1 in the aminoacylase 1 (ACY1) promoter region and thus increasing the transcription of the ACY1 gene. By regulating the HEY1/ACY1 signaling pathway, the expression of VEGF was down-regulated, and anti-angiogenic and tumor progression inhibition activities were exerted in ccRCC. MAGI2-AS3 could also reduce vimentin, N-cadherin, MMP-2, and MMP-9 secreted by tumors during invasion and metastasis. However, the expression of MAGI2-AS3 was downregulated in ccRCC, leading to the inhibited expression of ACY1, thus promoting angiogenesis and tumor growth. 64
In GC, LINC01410 is overexpressed, which could promote the expression of miR-532-5p target neutrophil cytosolic factor 2 (NCF2) mRNA by binding and inhibiting the activity of miR-532-5p. NCF2 could subsequently activate the nuclear factor kappa-B (NF-κB) pathway by increasing the level of p65 protein in the nucleus, and reduce the expression levels of VEGF-A and microvessel density (MVD) in tumor tissues. NCF2 can also regulate the expression of LINC01410 through the NF-κB pathway, forming a positive feedback loop of LINC1410/miR-5325p/NCF2/NF-κB, which plays an important role in tumor angiogenesis and progression. 65 The upregulated expression of the lncRNA NKX2-1-AS1 in GC serves as a potential prognostic index and therapeutic target, as it promotes tumor angiogenesis and development. In terms of mechanism, NKX2-1-AS1 competitively binds to miR-145-5p and upregulates the expression of serpin family E member 1 (SERPINE1), which is the upstream of VEGFR-2 signaling pathway. 66
In breast cancer (BC) cells, the lncRNA NR2F1-AS1 is highly expressed, increasing the expression of insulin-like growth factors 1 (IGF-1) by sponging miRNA-338-3p, upregulating the phosphorylation of IGF-1R and ERK1/2 in VECs, activating the IGF-1R and ERK pathways, and promoting tumor angiogenesis through the NR2F1-AS1/IGF-1/IGF-1R/ERK axis. IGF-1 has been shown to have a biological effect in angiogenesis and can be used as one of the pro-angiogenic factors by inducing VEGF or nitric oxide (NO). 67
The lncRNA MYLK-AS1, which is frequently upregulated in HCC tissues and cell lines, is positively correlated with tumor progression and poor prognosis, and directly targets miR-424-5p, while miR-424-5p specifically targets E2F transcription factor 7 (E2F7), activating the VEGFR-2 signaling pathway via E2F7 and promoting tumor angiogenesis and tumor cell proliferation, thus promoting tumor progression. 68
In ovarian cancer (OV), lncRNA DANCR directly binds to miR-145, and promotes the expression of VEGF in OV. Therefore, promoting tumor angiogenesis and tumor growth 69 ; the lncRNA NEAT1 is overexpressed, sponging miR-365, and upregulating the expression of FGF9, which is the inducer of cell proliferation and related angiogenesis, promoting tumor cell proliferation and tumor angiogenesis. 70 In glioma, the expression of the lncRNA H19 is increased, sponging miR-138 and upregulating the expression of HIF-1α, which, as one of the upstream molecules of VEGF, regulates the expression of VEGF, thereby promoting tumor angiogenesis. 71 The upregulated lncRNA PVT1 competes for the adsorption of miR-1207-3p, while miR-1207-3p, through targeted binding, regulates the expression of hepatocyte nuclear factor 1 beta (HNF1B) and promotes tumor angiogenesis by up-regulating the expression of epithelial-mesenchymal transition (EMT)-related genes (Vimentin and N-cadherin) and mitogen-activated protein kinase (MAPK) pathway-related genes (foxm1, VEGF-A and plakophilin 4), resulting in poor tumor prognosis. 72
In addition to promoting tumor angiogenesis, overexpressed lncRNAs could also inhibit tumor angiogenesis. For example, in laryngeal squamous cell carcinoma (LSCC), the lncRNA IGKJ2-MALLP2 can regulate the proliferation, colony formation, migration, and invasion of laryngeal cancer cells as well as angiogenesis around these cells, with its expression in laryngeal cancer tissues being lower than that in adjacent normal tissues. When IGKJ2-MALLP2 is overexpressed, it competes as a ceRNA to bind miR-1911-3p, upregulates the expression of p21, downregulates VEGF-A expression, reduces angiogenic activity, and inhibits LSCC progression 73 (Table 2A).
Molecular Mechanisms of Tumor Angiogenesis Regulation by lncRNAs.

LncRNAs Mediated Protein post-Translational Modifications on Tumor Angiogenesis
The lncRNAs are closely related to proteins and participate in the post-translational modification that affect the expression, activity and degradation of proteins. 85 They can also regulate the activity of proteins by phosphorylating modification. 86 LncRNAs can interact with various proteins to mediate angiogenesis within tumors (Figure 2B).
In cervical cancer (CC), lnc-CCDST acts as a scaffold to enhance the interaction of the E3 ubiquitin ligase mouse doubleminute 2 homolog (MDM2) with the oncogenic protein DExH-box helicase 9 (DHX9). The lnc-CCDST-dependent interplay facilitates DHX9 degradation by MDM2 and inhibits angiogenesis. However, in HPV-infected CC, the virus-encoded E6 and E7 proteins diminish lnc-CCDST expression and elevate DHX9 abundance to induce malignant behaviors. 74
The lncRNA PVT1 is significantly upregulated and is associated with late progression and poor prognosis in GC. PVT1 induces tumor internal angiogenesis through activating the STAT3 signaling pathway and increasing the expression of VEGF-A. In terms of mechanism, it interacts with nuclear phospho-signal transducer and activator of transcription 3 (p-STAT3) protein and protects it from polyubiquitination and proteasome-dependent degradation, thus improves its stability and promotes the nuclear accumulation of p-STAT3. High levels of PVT1 combined with VEGF-A can predict an adverse prognosis of GC, while STAT3 can occupy the promoter of PVT1 to promote its transcription, forming a positive feedback regulatory loop to continuously maintain its carcinogenic behavior. 75 In contrast to the lncRNA PVT1, lncRNA NBAT1 has tumor inhibitory activity. Sex determining region Y box protein 9 (SOX-9) plays a key role in driving tumor development, including proliferation, migration, invasion, and angiogenesis.87–89 NBAT1 could interact with SOX-9 to reduce its stability by promoting SOX-9 polyubiquitination and proteasome-dependent degradation, resulting in suppression of tumor angiogenesis, and subsequently malignant progression of GC. 76
LINC00908 is a downregulated cytoplasmic lncRNA in triple-negative breast cancer (TNBC) regulated by ERα, which is associated with tumor progression. It encodes a small regulatory peptide of STAT3 (ASRPS), which could inhibit tumor angiogenesis by directly bind to STAT3 through the coiled helical domain to inhibit STAT3 phosphorylation and VEGF expression. 77
In GC, LINC01021 could regulate kisspeptin 1(KISS1) through cyclin dependent kinase 2 (CDK2), which promotes phosphorylation and nuclear export of caudal type homeobox 2 (CDX2). Silencing LINC01021 downregulates VEGF and inhibits GC migration, invasion, and angiogenesis through reducing the binding between CDK2 with CDX2 and blocking CDK2-dependent CDX2 phosphorylation and promoting specific binding between CDX2 and KISS1 promoters. Taken altogether, high LINC01021 expression in GC promotes malignant cell migration and angiogenesis by downregulation of KISS1 through CDK2-mediated CDX2 phosphorylation 78 (Table 2B).
LncRNAs-mediated Epigenetic Modifications on Tumor Angiogenesis
Epigenetics refers to the chromatin machinery that is important in regulating gene expression without involving changes in the DNA sequence itself.90–92 Epigenetic mechanisms include DNA methylation, histone modification, and RNA-based mechanisms, including lncRNAs. 93 The lncRNAs are often enriched in the nucleus as epigenetic modifiers of gene expression and work by regulating the epigenetic state of protein-coding genes, 94 which can bind to DNA-binding proteins, such as chromatin modification complexes and transcription factors, and induce epigenetic changes (Figure 2C).
The lncRNA SNHG14 is highly expressed in HCC tissues and cells and is mainly localized in the nucleus, promoting tumor cell proliferation, migration, and angiogenesis by regulating poly(A) binding protein cytoplasmic 1 (PABPC1) in Hep3B and HepG2 cells. It positively regulates PABPC1 through lysine 27 on histone H3 (H3K27) acetylation in the PABPC1 promoter and negatively regulates it through SNHG14/PABPC1 in the development of HCC, regulating the phosphatase and tensin homolog (PTEN) signaling pathway, PTEN is involved in tumor cell proliferation or angiogenesis through negative regulation of phosphatidylinositol 3-kinase (PI3 K)/Akt signaling or VEGF expression, respectively. 79
The hypoxic signaling pathway plays a key role in tumor progression and angiogenesis. 95 The transcription factor HIF is a heterodimer composed of α and β subunits. Under normal oxygen conditions, the HIF-α protein is degraded through the proteasome pathway 96 ; in the hypoxic TME, the HIF-α protein escapes degradation and translocates to the nucleus, forms heterodimers with HIF-β, combines with the hypoxia-response element (HRE), activates the transcription of downstream target genes, 97 such as VEGF, stimulates the development of neovascularization inside the tumor, and enables tumor cells to adapt to the hypoxic environment. The lncRNA HOTAIR, a hypoxia-responsive gene, is overexpressed in colon cancer (CA) and several other types of tumors. Under hypoxic conditions, HIF-1α binds to the HRE in the HOTAIR promoter, and together with the HIF-1α, the histone methylase mixed-lineage leukemia 1 (MLL1) and the histone acetylase p300 are enriched at the HOTAIR promoter. This is accompanied by an increase in the levels of h3k4-trimethylation and histone acetylation, namely, the histone methylase MLL1 and the histone acetylase p300 coordinate and co-regulate, together with HIF-1α, the expression of hypoxia-induced HOTAIR, playing an important role in tumor development. 80
Many lncRNAs have been shown to bind to chromatin-modifying complexes, accelerating epigenetic activation and silencing target gene expression. For example, lncRNA PVT1, which is mainly localized in the nucleus, is overexpressed in cholangiocarcinoma (CCA) cells by binding to the H3K27me3-specific histone methyltransferase enhancer of zeste homolog 2 (EZH2) in the epigenetic modification polycomb repressive complex 2 (PRC2), and coregulates the expression of the target gene ANGPTL4, while EZH2, which directly binds to the promoter region of ANGPTL4, regulates histone methylation of the ANGPTL4 promoter by mediating H3K27me3 demethylation, epigenetically silencing the expression of ANGPTL4 and promoting tumor angiogenesis, proliferation, and apoptosis. 81
Many studies have demonstrated that a variety of lncRNAs in CRC can regulate the expression of VEGF by promoting methylation of gene promoter regions and inhibiting their transcription, thereby promoting angiogenesis and tumor progression. For example, LINC01116, by recruiting EZH2 (a transcription factor of TPM1), promotes the methylation of the tropomyosin α-1 chain (TPM1) promoter region, inactivating the promoter and inhibiting its transcription, thereby promoting the proliferation and angiogenesis of CRC cells 82 ; and ICMT-DT can recruit DNA methyltransferase 1 (DNMT1) to the promoter region of calponin 1 (CNN1), and subsequently methylate the promoter region of CNN1 and inhibit its transcription, promoting tumorigenesis and angiogenesis of CRCs. 83 The novel antitumor protease a disintegrin and metalloproteinase with thrombospondin 12 (ADAMTS12) plays an important role in controlling the epigenetic silencing mechanism in tumor progression. The lncRNA AK001058 regulates hypermethylation in the ADAMTS12 promoter region, downregulates ADAMTS12 protein and mRNA expression, promotes the expression of VEGF-A and angiotensin II (Ang II), reduces tumor cell apoptosis, and promotes angiogenesis and tumor growth 84 (Table 2C).
As mentioned earlier, lncRNAs could participate in tumor angiogenesis and progression at the genetic level through epigenetic modification, which has been validated by in vivo animal experiments.79,82–84 DNA methylation and histone acetylation are two important research hotspots. However, the therapeutic application of targeting lncRNAs to exert anti-angiogenic at the epigenetic level is really limited. Study reported that flavonoids can regulate the epigenetic modification of GBM at the gene level through lncRNAs (such as HOTAIR), improving the metabolic abnormalities of GBM, and inhibiting the angiogenesis of GBM cells. 98 However, other relevant studies have not been reported and clinical research is still lacking, which is a hot issue that we need to continue to research.
Potential Value and Feasibility of Targeting lncRNAs Anti-Angiogenesis for Tumor Therapy
Most lncRNAs have tumor-promoting effects and are rapidly becoming promising anti-angiogenic therapeutic targets for tumor treatments. In addition, some anti-tumor lncRNAs can be transferred to specific cells for gene therapy. For example, plasmid BC-819 (DTA-19) takes advantage of the high specificity of lncRNA H19 expression in tumors and clones the diphtheria toxin A subunit gene under the H19 promoter. Upon transfecting this plasmid into tumor cells, diphtheria toxin is expressed which would lead to tumor shrinkage without affecting normal tissue. 99
Antisense Oligonucleotides (ASOs)
In the past few years, ASOs-based therapies have led to breakthroughs in the treatment of various tumors. ASOs are chemically synthesized short single-stranded oligonucleotides with a sequence of 15-25 nucleotides that can bind to complementary RNA. After recruiting RNase H, ASO triggers RNA degradation and alters the expression of downstream proteins. 100 ASOs-based drugs have been approved for the treatment of a variety of neurological diseases, mainly in the treatment of Parkinson's disease (PD), Alzheimer's disease (AD) and tumors. 101 In addition, subcutaneous delivery of MALAT1 phosphorothioate-modified ASO successfully inhibited primary tumor differentiation and reduced lung metastasis in a mouse model of BC, which offers preliminary evidence that MALAT1 ASO is a therapeutic drug able to inhibit BC progression, indicating the clinical prospect of ASOs-targeted lncRNAs therapy. 102 LNA gapmeR, is a class of ASOs that can efficiently inhibit lncRNA function. By degrading MALAT1, it has shown superior antitumor activity in a humanized mouse multiple myeloma (MM) model, which provided preclinical evidence for the use of this new effective ASOs-targeting lncRNAs for the treatment of MM. 103
In recent years, research on lncRNAs-targeting by ASOs against angiogenesis in tumor treatment have been achieved. The LINC00173.v1 regulates VEGFA expression by sponging miR-511-5p, promoting angiogenesis and the progression of lung squamous cell carcinoma (SQC). Chen et al injected SQC cells into the caudal veins of BALB/c-nu mice in animal xenograft tumor experiments and then designed ASOs targeting LINC00173.v1 as antagonists to inhibit endogenous LINC00173.v1 expression. Monitoring the intraperitoneal injection of low-dose, high-dose, or negative controls in vaccinated mice, they found that the growth of lung tumors was inhibited and the cumulative survival was prolonged. Since anti-angiogenic therapy is usually combined with chemotherapeutic drugs, such as cisplatin, to treat lung cancer (LC), the combined efficacy of LINC00173.v1 ASO and cisplatin was further evaluated. The LINC00173.v1 ASO significantly improved the therapeutic sensitivity of SQC cells to cisplatin in vivo and even achieved a combined therapeutic effect of bevacizumab and cisplatin, indicating that LINC00173.v1 ASO can be used as a chemotherapy sensitizer in the treatment of SQC. Thus, its combination with cisplatin may be a reasonable treatment for SQC. 104
The lncRNA CRNDE indirectly regulates angiogenesis in HCCs by regulating M2 polarization in macrophages. Han et al constructed a tumor-bearing mouse model using H22 cells to observe the growth of the mice and the formation of subcutaneous solid tumors. After the appearance of subcutaneous solid tumors, they injected CRNDE ASO, which inhibited the expression of CRNDE in tumor-bearing tissues, and found that the expression of CD163, CD31 and M2 macrophage key signaling proteins and key angiogenesis-related proteins in tumor-bearing tissues were downregulated, which inhibited macrophage M2 polarization and tumor angiogenesis. The volume of subcutaneous solid tumors in the nude mice was significantly reduced, which demonstrated the therapeutic effect. 105
LINC00482 upregulates the expression of MMP15 by recruiting forkhead box protein A1 (FOXA1) and promoting the inflammation and angiogenesis associated with bladder cancer (BCa). Wang et al selected the cell lines with the highest level of LINC00482 for their experiments during the cell-log phase. Cells were transfected with the plasmid LINC00482-ASO. The LINC00482-ASO treatment effectively inhibited the expression of LINC00482 in the cells and reduced the number of tubes in the tumor cells in the tube formation assay. And tumor cell invasion, inflammation, and angiogenesis were all inhibited. Moreover, the xenograft experiments with nude mice confirmed the conclusion that silencing the expression of LINC00482 suppresses inflammation and angiogenesis in vivo. 106
As previously mentioned, ASOs targeting lncRNAs are promising strategies for the treatment of tumors in clinic. ASOs could specifically target any known RNA sequence, which could be artificially manufactured with versatility and adaptability. 107 Because of the chemical properties, ASOs possess disadvantages of low cell permeability and poor. Thus, remarkable progress has been made in the improvement of their pharmacological properties. 100 In recent years, more than 100 ASOs-based drugs have been enrolled in various phases of clinical trials. However, ASOs may have potent off-target effects and difficulties for targeted delivery. The nucleic acid delivery system may prove to be an important missing link between ASOs and its use in cancer treatment. Most vector systems need to be compliance for safety, immunogenicity, and stability in clinical settings. 108 Chemical modifications could improve delivery potency. 109 In addition, formulations of covalently conjugated and nanoparticles targeted to cells have been developed to address ASOs delivery challenges. 107 Therefore, although there are many preclinical applications of targeted lncRNAs, how to enhance potency and safety, and reduce the risk of dose-dependent toxicity, still need further technical exploration.
Exosomes
There is increasing evidence that exosomes are of great value in the targeted therapy of tumors. 110 Alvarez-Erviti et al proposed the concept of exosomes as nucleic acid drug delivery vehicles for the very first time. 111 Exosomes, as promising delivery vehicles, can be engineered to restore or knock down the expression of pathogenic lncRNAs in lncRNAs-dominant diseases. Additionally, they are being gradually explored in tumor treatments targeting lncRNAs against angiogenesis.
RCC-derived exosomes induce macrophage polarization by delivering lncARSR through the STAT3 pathway and promote tumor angiogenesis and progression. Zhang et al changed the level of lncARSR in macrophages by overexpressing lncARSR or reducing its expression using small interfering RNA (siRNA), showing that their secretion, phagocytosis, and pro-angiogenesis capacity were inversely inhibited in macrophages treated with lncARSR siRNA. In addition, Caki-1 cells were treated with lncARSR siRNA, and the total exosomes were isolated from the supernatant and injected into specific pathogen-free female BALB/C nude mice through the tail vein after pretreatement. Subsequently, Caki-1 cells were injected into the renal capsules of the mice, establishing an orthotopic transplantation model. The mice were divided into a no-pretreatment group, an exosome pretreatment group, a lncARSR siRNA exosome pretreatment group, and a normal control group. The results showed that the tumor size of the nude mice in the lncARSR siRNA exosome pretreatment group was significantly reduced, and the tumor development process was delayed. 112
The distinctive properties of exosomes, such as innate stability, low immunogenicity, biocompatibility, and proper biomembrane penetration capacity, allow them to function as superior natural nanocarriers for efficient delivery. 113 Exosomes can be integrated with liposomes to improve the specificity and controllability of delivery systems. 114 For example, exosome-liposome hybrids have been used successfully for in vivo and in vitro delivery in CRISPR-Cas9 systems, 115 and surface modification is also a practical tactic for exosome delivery optimization. Most essentially, targeted exosomes can be obtained by displaying targeting molecules on the outer surface of exosomes, such as peptides or antibody fragments recognizing target antigens. 116 Huang et al combined lncRNA-enriched exosomes with tumor-targeted cRGD peptides, which have a good targeting capability against tumor. 117 Moreover, as mentioned earlier, Alvarez-Erviti et al have shown the efficient use of exosomes in siRNA delivery. 111 Exosomes have been used in several ways to transport siRNA in vitro and in vivo.118–120 However, the in vivo stability, toxic effects on healthy cells, selectivity for tumor cells, non-specific immunostimulatory effects, and off-target effects of siRNAs are also the biggest obstacles to the implementation of siRNA therapeutics. And new strategies need to be found to optimize their distribution at target locations and maximize their potential. 121
Targeting Drugs
The advent of anti-angiogenic drugs has led to extensive studies on anti-angiogenic strategies for the treatment of tumors. 122 In addition, the application of certain therapeutic drugs targeting lncRNAs is receiving increasing attention and may play an important role in antitumor angiogenesis.
Xia et al showed that thalidomide inhibits angiogenesis and immune escape in non-small cell lung cancer (NSCLC), exhibiting a dual anticancer role. In terms of mechanism, thalidomide targets the lncRNA FGD5-AS1 and relies on the FGD5-AS1/miR-454-3p/ZEB1 axis to exert anti-tumor activity in NSCLC, which is an effective treatment for NSCLC. Xia et al constructed a FGD5-AS1 overexpression A549 xenograft tumor model in nude mouse, and divided these mice into A549 alone, A549 + PBS, and A549 + thalidomide treatment groups. They found that thalidomide reversed the effect of FGD5-AS1, and the tumor size of mice was significantly reduced, as was the tumor growth curve. Angiogenesis marker expression was downregulated, and tumor-infiltrating CD8 + T cell-mediated antitumor activity was enhanced. 123
Earlier studies reported that sunitinib was designed to inhibit angiogenesis by inhibiting EC growth, which in turn slows the progression of RCC. In general, sunitinib is well tolerated, with the most common side effects occurring in more than 20% of patients, including hypertension, hypothyroidism, hand-foot syndrome, and neutropenia, etc, which have been explored as potential biomarkers of sunitinib's efficacy. 124 Fortunately, hypertension, as a well-known systemic adverse reaction, can be countered with conventional antihypertensive drugs. 125 Although the side effects are manageable, there are serious toxicities associated with sunitinib that require careful management and dose reduction. 126 Meanwhile, the patients may become insensitive or develop resistance toward sunitinib treatment. He et al found that sunitinib could have adverse effects of promoting RCC progression by increasing VM formation of RCC cells, and VM formation might be a compensation for endothelium deprivation. Mechanism dissection revealed that sunitinib can increase the expression of lncRNA ECVSR, thereby enhancing the stability of ERβ mRNA. Subsequently, the increased ERβ expression can then up-regulate the transcription of Hif 2-α. Notably, sunitinib-increased ECVSR/ERβ/Hif 2-α signaling resulted in an increased CSC phenotype, thereby promoting VM formation. In preclinical studies of RCC mouse xenografts, 4-[2-phenyl-5,7-bis(trifluoromethyl)pyrazolo[1,5-a]pyrimidin-3-yl]phenol (PHTPP), as a complete antagonist of estrogen ERβ receptor, when combined with sunitinib, it can effectively block the ECVSR/ERβ/Hif 2-α signaling axis, improve the efficacy of sunitinib, reduce the formation of VM. 47
Cancers with a high capability for angiogenesis are frequently regarded as being difficult to treat and anti-angiogenesis drugs are considered the primary therapy for these types of cancers. Anti-angiogenesis drugs mainly targets VEGF or receptor tyrosine kinases, 127 and there is currently less research on anti-angiogenesis drugs that target lncRNAs. And the clinical efficacy of these drugs is limited, mainly due to congenital or acquired resistance. However, the resistance of anti-angiogenic drugs can be affected by lncRNAs dysregulation, so targeting lncRNAs may be a potential way to overcome anti-angiogenic resistance. 128 The combination of lncRNAs-targeted and anti-angiogenic drugs has higher clinical prospects.
Summary and Outlook
In this review, we summarized the multiple mechanisms by which lncRNAs induce tumor angiogenesis and revealed the complexity of lncRNAs in promoting tumor angiogenesis. The same lncRNAs may trigger tumor angiogenesis through different mechanisms, such as PVT1. In glioma, PVT1 acts as a ceRNA 72 ; In GC, PVT1 interacts with proteins and affects their stability 75 ; While in CCA, PVT1 epigenetically silences target gene expression. 81 Similarly, different lncRNAs can regulate angiogenesis in the same tumor. For example in GC, LINC01410 acts as a ceRNA 65 ; NBAT1 mediates protein degradation and affects protein stability 76 ; LINC01021 participates in proteins phosphorylation. 78 This diversity reveals that lncRNAs are rapidly emerging as promising drug targets or drug candidates.129–131
Since angiogenesis is an important process in cancer pathogenesis and treatment, new therapeutic targets are needed to inhibit angiogenesis. LncRNAs are multi-functional regulators of tumor angiogenesis, are better therapeutic targets than existing protein-coding genes due to their specific expression in different cell types and diseases,132–134 have clinical application prospects. Ye et al demonstrated that aniticin can inhibit the ceRNA function of lncRNA MEG3 and thus inhibit angiogenesis in OV 135 ; Flavonoids can inhibit angiogenesis in GBM by influencing the epigenetic modification function of lncRNAs (such as HOTAIR) 98 ; Some studies have found that targeting lncRNAs can improve the resistance of existing anti-angiogenic drugs 127 ; In addition, lncRNAs have the potential to serve as diagnostic biomarkers. For example, MALAT1 mediates tumor angiogenesis136–138 and plays important roles in regulation of tumor biological behaviors, 139 anti-tumor immune responses 140 and drug resistance, 141 which has been effectively used to diagnose PCa. 142
For silencing lncRNAs expression, three small molecule tools (ASOs, exosomes, drugs), have also been shown to have potential for clinical applications. Among the three molecular tools that target lncRNAs against angiogenesis, exosomes are expected to be more widely used in clinical treatment. As mentioned earlier, exosomes are involved in the regulation of tumor angiogenesis by lncRNAs, and the relevant mechanism of translocation of lncRNAs to VECs through exosomes to mediate angiogenesis is also guaranteed. 58 Moreover, exosomes as natural carriers, have proper molecular transport properties and biocompatibility, 143 which can protect lncRNAs targeted trugs from degradation and reducing non-targeted cytotoxicity. 110 In addition, exosomes contain transmembrane and membrane-anchored proteins that enhance endocytosis and thus facilitate the transfer of their contents. 144 Today, accumulating evidences confirm that exosomes have instrumental value in disease diagnosis and targeted therapy. We believe that exosomes-based cancer treatment strategies will make great progress for the benefit of cancer patients.
However, at this stage, the clinical research on targeting lncRNAs against tumor angiogenesis is very limited, and the function of most lncRNAs is poorly understood. Their effective use as therapeutic targets still requires a lot of research, such as studying the toxicity, pharmacokinetics, and biological properties of lncRNAs. At present, the therapeutic potential of lncRNAs in terms of diagnosis, prognosis, and therapeutic ability, as well as the therapeutic application of anti-angiogenic drugs in tumors, are being widely studied.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241273239 - Supplemental material for Regulation and Therapeutic Application of Long non-Coding RNA in Tumor Angiogenesis
Supplemental material, sj-docx-1-tct-10.1177_15330338241273239 for Regulation and Therapeutic Application of Long non-Coding RNA in Tumor Angiogenesis by Shuo Zhang, Yunxiu Xia, Weiwei Chen, Hongliang Dong, Bingjie Cui, Cuilan Liu, Zhiqiang Liu, Fei Wang and Jing Du in Technology in Cancer Research & Treatment
Footnotes
Author's Degree Information
Shuo Zhang, postgraduate; Yunxiu Xia, postgraduate; Weiwei Chen, postgraduate; Hongliang Dong, postgraduate; Bingjie Cui, postgraduate; Cuilan Liu, postgraduate; Zhiqiang Liu, postgraduate; Fei Wang, postdoctor; Jing Du, doctor.
Author Contributions
SZ: Writing – original draft. FW: Writing – review & editing. JD: Writing – review & editing. YXX: Writing – review. WWC: Writing – review. HLD: Writing – review. BJC: Writing – review. CLL: Writing – review. ZQL: Writing – review.
Acknowledgments
Sincerely thanks to the irreplaceable contribution by the authors who helped edit and revise the manuscript.
Data Availability Statement
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
This study was supported by the National Natural Science Foundation of China (31900441, 82373097; to JD), China Postdoctoral Science Foundation (2023M742089; to FW), Natural Science Foundation of Shandong Province (ZR2019MC026, ZR2023QH080, ZR2022QH192; to JD, FW and WWC), Qilu Outstanding Young Talents in Health Project, Double-Hundred Project and Taishan Scholarship.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
