Abstract
Aberrant expression of microRNAs (miRNAs) has been reported to play a role in tumorigenesis. Dysfunction of miR-1296 was found in a variety of cancers, however, the function of miR-1296 in the progression of glioma remains largely understood. Here, our results showed that miR-1296 was significantly down-regulated in glioma tissues and cell lines. Decreased expression of miR-1296 was associated with the tumor size, WHO grade and karnofsky performance scale (KPS) of glioma patients. Low expression of miR-1296 was significantly correlated with the shorter 5-year overall survival of glioma patients. Overexpression of miR-1296 inhibited the proliferation, colony formation, migration and induced apoptosis of glioma cells. MiR-1296 was found to bind the 3’-untranslated region (UTR) of ABL proto-oncogene 2 (ABL2) and subsequently repressed both the mRNA and protein expression of ABL2. ABL2 was overexpressed in glioma tissues and inversely correlated with that of miR-1296. Ectopic expressed ABL2 could reverse the inhibitory effects of miR-1296 on glioma cell proliferation. Our results illustrated the novel tumor-suppressive function of miR-1296 in glioma via repressing ABL2, suggesting a potential application of miR-1296 in the treatment of glioma.
Introduction
Glioma is one of the most common malignant brain tumors with high morbidity and lethality in adults. 1 -3 Currently, surgical resection combined with radiotherapy and chemotherapy have been widely applied in the treatment of glioma. 4,5 However, due to the low differentiation, fast proliferation and invasion of glioma cells, the prognosis of glioma patients still remains poor. Therefore, investigating the underlying mechanisms that involved in glioma development and exploring novel biomarkers for the early diagnosis and treatment of glioma are critical.
MicroRNAs (miRNAs) are a class of highly conserved, non-coding, single-stranded RNA molecules with the length of approximately 22 nucleotides. 6,7 miRNAs act as negative post-transcriptional regulators of gene expression via binding the 3’-untranslated region (UTR) of target mRNAs, leading to the degradation or translational inhibition of mRNAs. 8,9 miRNAs play important roles in various cellular processes, such as cell proliferation, differentiation, invasion and migration. 7 Interestingly, increasing evidence demonstrates that miRNAs are critical regulators in cancer progression and are suggested as possible prognostic biomarkers for cancers. 10 -15 Aberrant expression of miRNA has been found in glioma, which positively or negatively modulated the development of glioma. 16 -19 However, understanding the mechanisms explaining the function of miRNAs in the progression of glioma, particularly how they affect cancer cell survival call for more evidence.
The coding gene of miR-1296 is located at chromosome 10 of human genome. 20 miR-1296 was found as a novel cancer-related miRNA in various cancers and played suppressive roles in the development of cancers, including prostate cancer, hepatocellular carcinoma, breast cancer and gastric cancer. 20 -23 For example, miR-1296 inhibits the migration and invasion of hepatocellular carcinoma cells by suppressing the PI3K/AKT pathway. 23 Additionally, miR-1296 negatively modulates the progression of triple-negative breast cancer via targeting cyclin D1. 21 Recent studies also showed that miR-1296 inhibits the migration and invasion of gastric cancer cells by modulating the ERBB2/Rac1 signaling. 22 These findings indicated the important tumor-suppressive roles of miR-1296 in cancers, however, to our best knowledge, the function of miR-1296 in glioma and the potential targets of miR-1296 in glioma have not been fully illustrated.
ABL proto-oncogene 2 (ABL2) is an ABL family non-receptor tyrosine kinase. ABL2 was ubiquitously expressed and regulated many cellular processes, such as cell proliferation, adhesion and migration. 24,25 Recent studies suggested that ABL2 is up-regulated in several cancers including pancreatic cancer, colorectal cancer and NSCLC. 26,27 Overexpression of ABL2 promoted the malignant progression of cancer cells, indicating ABL2 was a promising target to disrupt the cancer development. Recent study showed that silencing of ABL2 inhibited the invasion of glioma cells, which indicated ABL2 as a potential therapeutic candidate to block the progression of glioma. 28 Therefore, it would be necessary to identify the negative regulators of ABL2 to down-regulate its function in glioma.
The aim of this study was to explore the function of miR-1296 in the malignancy of glioma and identify the underlying molecular mechanism.
Materials and Methods
Carcinoma Samples
The sample size of this study was calculated with the G. Power based on 0.9 power to detect a significant difference. Paired glioma tissue samples and non-cancerous brain tissues were obtained from a total of 50 glioma patients at the First Affiliated Hospital of Guangxi University of Chinese Medicine between January 2011 and September 2013. The pathological classification of all these patients were identified as astrocytoma. Tissues were snap-frozen in liquid nitrogen and maintained at -80ºC before further analysis. None of the patients received chemotherapy or radiotherapy prior to the tissue collection. This study was approved by the Ethics Committee of the First Affiliated Hospital of Guangxi University of Chinese Medicine (Approval number 2014-01-3523A). Written informed consents were obtained from all involved patients.
RNA Extraction and RT-qPCR
Total RNA was extracted from the cell lines or tumor samples using the TRIzol reagent (Thermo Fisher Scientific Inc.) according to the manufacturer’s instruction. RNA concentration was monitored with the NanoDrop (Thermo Fisher Scientific Inc.). Reverse transcription was performed using the Hairpin-itTM miRNA quantitative detection kit (GenePharma, Shanghai, China). qPCR was conducted on the ABI7500 platform with the SYBR Premix Ex TaqTM II PCR Kit (Takara, Dalian, China) following the manufacturer’s protocol. The primers of miR-1296 (F: 5′-GTTAGGGCCCTGGCTCC-3′ and R: 5′-CAGTGCGTGTCGTGGAGT-3′), ABL2 (F: 5′-GTGATGAGACTACTGCAGCATCC-3′ and R: 5′-CGGTTACCGTGTCGTCCATGAT-3′), GAPDH (F: 5′-CCATGTTCGTCATGGGTGTGAACCA-3′ and R: 5′-GCCAGTAGAGGCAGGGATGATGTTC-3′) and U6 RNA (F: 5′-GCTTCGGCAGCACATATACTAAAAT-3′ and R: 5′-CGCTTCAGAATTTGCGTGTCAT-3′) were designed and synthesized by GenePharma (Shanghai, China). U6 RNA and GAPDH was detected as the internal reference of miR-1296 and ABL2, respectively. The relative expression of miR-1296 was analyzed with the 2-ΔΔCq method.
Cell Counting Kit-8 (CCK-8) Assay
Cells expressing miR-1296 mimic or miR-NC were seeded into the 96-well plate with 1,000 cells per well. Three replicates were set. 10 µl of CCK-8 solution (Sigma, St. Louis, MO, USA) was added at the indicated time points (0-, 1-, 2-, 3-, 4- and 5 days) and incubated for 4 h at 37ºC. The OD values of each well at the wavelength of 450 nm were measured by the microplate reader.
Colony Formation Assay
Cells expressing miR-1296 mimic or miR-NC were seeded into the 6-well plate with 500 cells per well. Cells were cultured with DMEM containing 10% FBS for 2 weeks. Then cells were washed with PBS and fixed with 4% paraformaldehyde for 15 min at room temperature (RT). Cells were stained with 0.1% crystal violet (Beyotime, Shanghai, China) for 10 min at RT. Colonies were counted using a light microscope.
Wound-Healing Assay
U87 and SWO-38 cells transfected with miR-1296 mimic or miR-NC were seeded into the 6-well plate. After cultured overnight, the wound was generated by scratching cells with a 100-µl tip. The cell debris were removed by washing cells with medium. Cells were then cultured for 24 h and the wound healing was observed with the light microscope.
Cell Invasion Assay
The invasion of glioma cells was detected using the transwell chambers (8-µm pore size, Corning, New York, USA). U87 and SWO-38 cells transfected with miR-1296 mimic or miR-NC were seeded into the upper chamber and cultured with 200 µl of DMEM medium. The lower chamber was added with medium supplemented with 10% FBS. After cultured for 48 h at 37°C, cells migrated to the lower surface of the membrane were fixed with 4% paraformaldehyde at RT for 15 min. Cells were then stained with 0.1% crystal violet (Tiangen, Beijing, China) for 15 min at RT. The invaded cells were counted with an inverted light microscope.
Prediction of miR-1296 Targets
The potential targets of miR-1296 were predicted using the miRDB (http://www.mirdb.org/). The parameters were set as follows: species = Human, and miRNA name = miR-1296.
Western Blot Analysis
Total proteins were extracted from glioma cells using the radioimmunoprecipitation assay (RIPA) buffer (Beyotime, Shanghai, China) containing protease inhibitor (Millipore, Billerica, MA, USA). Equal amount of proteins was separated by 15% sodium dodecyl sulphate-poly-acrylamide gel electrophoresis (SDS-PAGE) and transferred onto the polyvinylidene difluoride (PVDF) membrane. The membranes were blocked with 5% skimmed milk for 1 h at RT followed by incubating with primary antibodies against ABL2 (1:2000 dilution; ab134134; Abcam, USA) or GADPH (1:5000 dilution; ab181602; Abcam, USA) at 4ºC overnight. After washing twice with TBS-Tween 20 (0.1%), membranes were incubated with horseradish peroxidase-conjugated-conjugated secondary antibody for 1 h at RT. The bolts were analyzed using the enhanced chemiluminescence (ECL) substrate (Pierce, Thermo Fisher Scientific, Inc.) according to the manufacturer’s instructions.
Dual-Luciferase Reporter Assays
The wild-type (WT) or mutant (MUT) 3’-UTR fragments of ABL2 that contained the predicted binding sites of miR-1296 were inserted into the pGL3 Luciferase vector (Promega, Madison, WI, USA). Cells were co-transfected with miR-1296 mimic and WT-3’-UTR of ABL2 or Mut-3’-UTR of ABL2 using the Lipofectamine 2000 (Thermo Fisher Scientific, Inc.). The luciferase activity was measured using the dual-luciferase reporter assay kit (Promega, Madison, WI, USA) after transfection for 48 h. The activity of Renilla was also determined as the normalization of firefly activity.
Cell Apoptosis Analysis
Glioma cells were seeded into the 35 mm dishes and transfected with miR-1296 mimic or miR-NC. After transfection for 48 h, cells were trypsinized and stained with the Annexin V-FITC/PI Detection kit (Sigma, Merck KGaA, USA) according to the manufacturer’s instructions. The apoptosis of cells was determined by flow cytometry (Becton, USA). The FITC+/PI- and FITC+/PI+ fractions were considered as early and late apoptosis, respectively. Data was analyzed using the FlowJo software (Version X.10.0.7-1).
Statistical Analysis
Data were presented as mean ± standard deviation and analyzed using the SPSS 19.0 (SPSS Inc., Chicago, IL, USA). Difference between 2 groups was determined using 2-tailed paired Student’s t test and 1-way analysis of variance (ANOVA) for comparisons of multiple groups. P < 0.05 was considered as statistically significant.
Results
miR-1296 Expression Was Down-Regulated in Glioma Tissues and Cells
Given the limited understanding for the role of miR-1296 in glioma, the expression of miR-1296 in paired glioma tissues and matched non-tumor tissues was detected by RT-qPCR. The data indicated that the expression of miR-1296 in glioma tissues was notably decreased compared with that in adjacent non-cancer tissues (Figure 1A). To further characterize the dysregulation of miR-1296 in glioma, those 50 patients were divided into miR-1296-high expression group and miR-1296-low expression group according to the median expression value of miR-1296. Association of miR-1296 level with the clinicopathological data of glioma patients showed that lower expression of miR-1296 was significantly correlated with the tumor size, WHO grade and Karnofsky performance scale (KPS) of the patients (Table 1). These results suggested the potential involvement of miR-1296 in the progression of glioma. To further support the dysfunction of miR-1296 in glioma, the expression of miR-1296 in several glioma cell lines was also detected. As indicated in Figure 1B, the expression of miR-1296 was significantly down-regulated in glioma cells compared with that of normal cell NHA. To provide more evidence about the potential clinical significance of miR-1296, the correlation between miR-1296 expression with the 5-year overall survival rate of those glioma patients was analyzed. The data showed that the 5-year overall survival rate of patients with relative lower expression of miR-1296 was worse than those harboring higher level of miR-1296 (Figure 1C). To explore the function of miR-1296 in the malignancy of glioma, both U87 and SWO-38 cells, which harbored relative lower level of miR-1296 among all the cell lines we tested in Figure 1B, were transfected with miR-1296 mimic or miR-NC. The overexpression of miR-1296 in U87 and SWO-38 cells was confirmed by RT-qPCR (Figure 1D) and subsequently used in the following experiments.

miR-1296 was down-regulated in glioma. (A) The expression of miR-1296 was significantly down-regulated in glioma tissues compared with adjacent normal tissues. (B) The expression of miR-1296 was lower in glioma cell lines compare with that in the normal cells. (C) The correlation between miR-1296 expression with the 5-year overall survival rate of glioma patients was analyzed with the log rank test. (D) U87 and SWO-38 cells were transfected with miR-NC or miR-1296 mimic and the relative expression of miR-1296 was detected by RT-qPCR. **P < 0.01; *** P < 0.001.
Association of miR-1296 With the Clinicopathological Characteristics of Patients With Glioma.
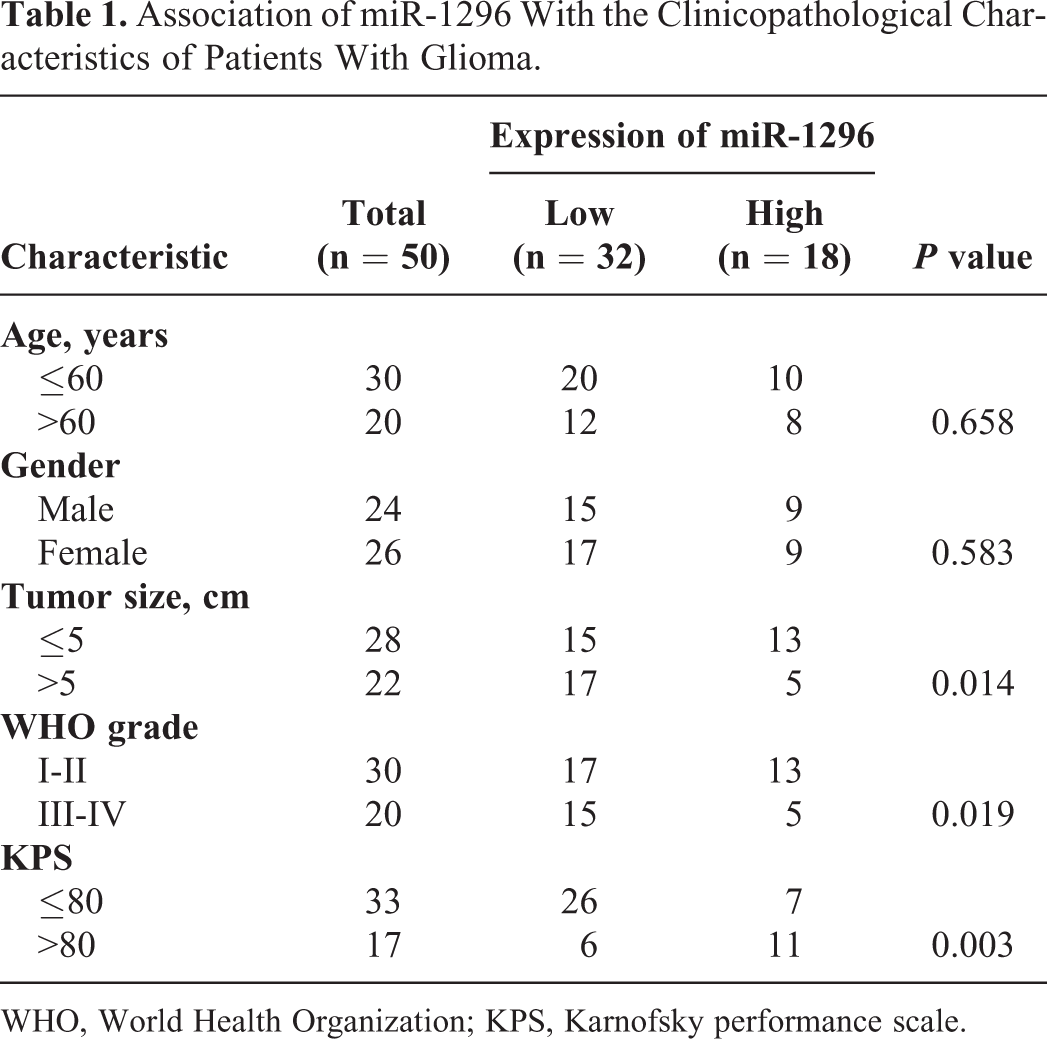
WHO, World Health Organization; KPS, Karnofsky performance scale.
miR-1296 Inhibited the Proliferation and Induced Apoptosis of Glioma Cells
To investigate the biological effects of miR-1296 on the growth of glioma cells, CCK-8 assay was performed with U87 and SWO-38 cells expressing miR-1296 mimic or miR-NC. The results showed that overexpression of miR-1296 significantly inhibited the proliferation of glioma cells compared with cells carrying miR-NC (Figure 2A and 2B). We also evaluated the effects of miR-1296 overexpression on the colony formation of glioma cells. The result showed that less colonies were observed with cells expressing miR-1296 mimic in comparison with that of cells expressing miR-NC (Figure 2C). Additionally, flow cytometry was performed to determine the roles of miR-1296 in the apoptosis of glioma cells. The results indicated that overexpression of miR-1296 significantly increased the apoptosis of both U87 and SWO-38 cells (Figure 2D). To further validate the suppressive role of miR-1296 in the malignant phenotypes of glioma cells, the wound healing assay was performed with overexpression of miR-1296. As indicated in Figure 2E, transfection of miR-1296 significantly suppressed the wound healing capacity of both U87 and SWO-38 cells compared with cells expressing control vector. Furthermore, overexpression of miR-1296 also significantly reduced the invasion of glioma cells (Figure 2F). Overall, miR-1296 exerted tumor-suppressive function in regulating the malignant behaviors of glioma cells.

Overexpression of miR-1296 inhibited the growth of glioma cells. (A, B) Cell proliferation was suppressed with the overexpression of miR-1296 relative to the control cells. (C) Transfection of miR-1296 significantly inhibited the colony formation of glioma cells. (D) The apoptosis of both U87 and SWO-38 cells were increased with the overexpression of miR-196 compared with the control cells. (E) Overexpression of miR-1296 inhibited the wound-healing capacity of glioma cells. (F) The migration and invasion ability of glioma cells was reduced with the transfection of miR-1296. *P < 0.05; **P < 0.01; *** P < 0.001.
ABL2 Was a Target of miR-1296 in Glioma Cells
It is well documented that miRNAs exert their functions by down-regulating the expression of target genes. To explore the functional mechanism underlying the suppressive role of miR-1296 in glioma, the potential targets of miR-1296 were predicted using the miRDB database. Complementary binding site of miR-1296 was found within the 3’-UTR of ABL2 (Figure 3A), suggesting ABL2 was a possible candidate target of miR-1296. To verify the association between miR-1296 and ABL2, luciferase reporter assay was performed by co-transfecting ABL2-3’-UTR-WT or ABL2-3’-UTR-MUT and miR-1296 mimic into glioma cells. As indicated in Figure 3B, overexpression of miR-1296 significantly decreased the luciferase activity of cells carrying ABL2-3’-UTR-WT, however, on obvious effect was observed on that of the ABL2-3’-UTR-MUT (Figure 3B and 3C). These results suggested the specific binding between the ABL2 3’-UTR with miR-1296 in glioma cells. To investigate the regulation of miR-1296 in the expression of ABL2, the mRNA level of ABL2 in both U87 and SWO-38 cells expressing miR-1296 mimic or miR-NC was detected by RT-qPCR. The result showed that ABL2 mRNA expression was significantly reduced with miR-1296 overexpression (Figure 3D). Furthermore, consistent with the decreased mRNA abundance, the protein level of ABL2 was also suppressed upon the overexpression of miR-1296 (Figure 3E). These findings identified ABL2 as a target of miR-1296 in glioma cells. To further validate this conclusion, the expression of miR-1296 was down-regulated by transfecting miR-1296 inhibitor into both U87 and SWO-38 cells (Figure 3F). As indicated in Figure 3G and 3H, down-regulation of miR-1296 significantly promoted the proliferation of both U87 and SWO-38 cells. Additionally, both the mRNA and protein levels of ABL2 were increased with the down-regulation of miR-1296 (Figure 3I and 3 J). These findings supported the tumor-suppressive role of miR-1296 and the negative regulation of ABL2 by miR-1296 in glioma.

miR-1296 inhibited ABL2 expression by binding its 3’-UTR. (A) Schematic representation of miR-1296 and its binding sequence within the 3’-UTR of ABL2. (B, C) The luciferase activity of cells expressing WT but not Mut 3’-UTR of ABL2 was decreased following the transfection of miR-1296. (D, E) Overexpression of miR-1296 decreased the mRNA and protein expression of ABL2 in glioma cells. (F) Cells were transfected with miR-1296 inhibitor or scramble miRNA, and the down-regulation of miR-1296 was confirmed by RT-qPCR. (G, H) Knockdown of miR-1296 significantly promoted the proliferation of both U87 and SWO-38 cells. (I, J) Depletion of miR-1296 increased both the mRNA (I) and protein abundance (J) of ABL2 in glioma cells. *** P < 0.001.
ABL2 Was Overexpressed in Glioma Tissues and Cell Lines
To further illustrate the function of ABL2 in glioma, the mRNA expression of ABL2 in paired glioma tissues and non-cancerous tissues was examined by RT-qPCR analysis. Compared with adjacent non-cancerous tissues, the level of ABL2 was significantly increased in glioma tissues (Figure 4A). Additionally, the expression of ABL2 was also frequently up-regulated in glioma cells in comparison with that of the normal cell NHA (Figure 4B). The correlation between ABL2 expression and the 5-year overall survival rate of glioma patients was also determined. As shown in Figure 4C, higher ABL2 abundance was significantly correlated with the worse prognosis of glioma patients. Because ABL2 was identified as a target of miR-1296, the correlation between the expression of ABL2 and miR-1296 in glioma tissues was analyzed by Spearman test. As shown in Figure 4D, the expression of miR-1296 in glioma tissues was inversely correlated with that of ABL2, which supported the conclusion that ABL2 was negatively regulated by miR-1296 in glioma cells.

The level of ABL2 in glioma tissues was inversely correlated with miR-1296. (A) RT-qPCR analysis of the relative expression of ABL2 in glioma tissues and paired adjacent non-cancer tissues. (B) The level of ABL2 was increased in glioma cells compared with that of the normal cells. (C) The correlation between the expression of ABL2 and the 5-year overall survival rate of glioma patients was determined by the log rank test. (D) The inverse relationship between the expression of ABL2 and miR-1296 in glioma tissues determined by the Pearson’s correlation test. *** P < 0.001.
Overexpression of ABL2 Markedly Reversed the Inhibitory Effects of miR-1296 on Glioma Cells
To explore the involvement of ABL2 in miR-1296-mediated growth inhibition of glioma cells, overexpression vector of ABL2 and miR-1296 mimic was co-transfected into both U87 and SWO-38 cells. The expression of ABL2 was confirmed by RT-qPCR and western blot, respectively. Transfection of ABL2 significantly increased the expression of ABL2 in glioam cells compared with control group (Figure 5A and 5B). As shown in Figure 5C and 5D, restoration of ABL2 reserved miR-1296-mediated inhibitory effects on the proliferation of both U87 and SWO-38 cells. To confirm this, colony formation assay was also performed with glioma cells expressing miR-1296 mimic or overexpressed ABL2. Overexpression of miR-1296 decreased the colony formation of both U87 and SWO-38 cells, while transfection of ABL2 significantly attenuated the suppressive role of miR-1296 on the colony formation capacity of glioma cells (Figure 5E). Collectively, these findings demonstrated that miR-1296 inhibited the malignant behaviors of glioma cells at least partially via regulating ABL2.

ABL2 reversed the inhibitory regulatory effects of miR-1296 on glioma. (A, B) Glioma cells were transfected with miR-NC, miR-1296 mimic or miR-1296 mimic +ABL2. The level of ABL2 was detected via RT-qPCR and western blot. (C, D) Restoration of ABL2 significantly reversed the inhibitory effects of miR-1296 on the proliferation of glioma cells. (E) Overexpression of ABL2 rescued the colony-forming ability induced by miR-1296 in both U87 and SWO-38 cells. *** P < 0.001.
Discussion
Glioma has been considered as the most common brain tumor with poor prognosis. 1 Current therapeutic strategies show low efficacy for certain glioma patients, which calls for novel and effective therapeutic options. Increasing evidence has reported that miRNAs contribute to the progression of various tumors including glioma via acting as tumor suppressors or oncogenes. 10,11,13,14,29,30 For example, recent study showed that miR-346 suppresses the growth of glioma cells by targeting NF1B and serves as a possible biomarker for the diagnosis of glioma patients. 31 miR-181 was significantly down-regulated in glioma tissues, and overexpressed miR-181 inhibited the proliferation, drug sensitivity and invasion of glioma cells. 32 In this study, our results showed that miR-1296 was decreased in glioma tissues and associated with the advanced progression of patients with glioma. The potential involved functional mechanisms of miR-1296 in glioma were also explored.
The dysfunction of miR-1296 has been reported in several human cancers. Significantly down-regulated miR-1296 was found in non-small cell lung cancer (NSCLC) tissues and cells, which exhibited a notable association with the lymph-node metastasis of NSCLC patients. 33 Overexpressed miR-1296 inhibited the proliferation and invasion of NSCLC cells by regulating the Wnt signaling. 33 The tumor-suppressive role of miR-1296 was also demonstrated in triple-negative breast cancer. 21 In this study, overexpression of miR-1296 significantly inhibited the proliferation, colony formation, migration, invasion and triggered the apoptosis of glioma cells. Along with the evidence form the current study, miR-1296 is predominantly acting as a tumor suppressor in human cancers.
The overexpression and oncogenic function of ABL2 have been reported in many types of cancers, which plays an important role in cancer development. 27,34 In this study, our results showed that miR-1296 bound the 3’-UTR of ABL2 and decreased the expression of ABL2 in glioma cells. Consistent with the down-regulation of miR-1296, the expression of ABL2 was significantly higher in glioma tissues and negatively correlated with that of miR-1296. Moreover, ABL2 restoration obviously reversed the suppressive role of miR-1296 in regulating the proliferation of glioma cells. Our results uncovered the novel function of miR-1296 in glioma via targeting ABL2.
In summary, the present study demonstrated that miR-1296 expression was reduced in glioma tissues and cells. Lower expression of miR-1296 was associated with the advanced progression of patients with glioma, suggesting miR-1296 as a possible prognostic predictor for glioma. miR-1296 inhibited the growth of glioma cells by directly targeting ABL2. Therefore, miR-1296 might be a new promising therapeutic target for glioma. The major limitation of the current study is lacking of in vivo evidence. Thus, to better understand the role of miR-1296 in glioma, in vivo xenograft mouse model needs to be established to validate the tumor suppressive function of miR-1296 in the malignancy of glioma as well as the negative regulation of ABL2 by miR-1296. Additionally, the involvement of miR-1296 in the distant metastasis and drug resistance of glioma also calls for more investigation. In addition to ABL2, many targeting candidates of miR-1296 are also predicted in the present study, their roles in miR-1296-mediated growth inhibition of glioma remain to be answered.
Conclusions
Our study uncovered the down-regulation of miR-1296 in glioma, which was significantly correlated with the advanced progression of glioma patients. miR-1296 acted as a tumor suppressor in the progression of glioma at least partially via targeting ABL2. These findings indicated miR-1296 as a potential target for the treatment of glioma.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study was approved by the Ethics Committee of the First Affiliated Hospital of Guangxi University of Chinese Medicine (Approval number 2014-01-3523A).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science Foundation of China (No. 8166150307).
