Abstract
Objective
To compare the ability of gadolinium ethoxybenzyl dimeglumine (Gd-EOB-DTPA) and gadobenate dimeglumine (Gd-BOPTA) to display the 3 major features recommended by the Liver Imaging Reporting and Data System (LI-RADS 2018v) for diagnosing hepatocellular carcinoma (HCC).
Materials and Methods
In this retrospective study, we included 98 HCC lesions that were scanned with either Gd-EOB-DTPA-MR or Gd-BOPTA-M.For each lesion, we collected multiple variables, including size and enhancement pattern in the arterial phase (AP), portal venous phase (PVP), transitional phase (TP), delayed phase (DP), and hepatobiliary phase (HBP). The lesion-to-liver contrast (LLC) was measured and calculated for each phase and then compared between the 2 contrast agents. A
Results
Between Gd-BOPTA and Gd-EOB-DTPA, significant differences were observed regarding the display efficiency for capsule enhancement and the LLC in the AP/PVP/DP (
Conclusion
Gd-BOPTA and Gd-EOB-DTPA are liver-specific contrast agents widely used in clinical practice. They have their own characteristics in displaying the 3 main signs of HCC. For accurate noninvasive diagnosis, the choice of agent should be made according to the specific situation.
Introduction
Primary hepatic carcinoma (PHC) is the fourth most common malignant tumor in China and the second most common cause of cancer death, posing a serious threat to life and health.1,2 Hepatocellular carcinoma (HCC) is the most common PHC 3 and the second leading cause of death due to cancer worldwide, with an annually increasing incidence and mortality. 4
HCC has no obvious signs or symptoms at the early stage, for which magnetic resonance imaging (MRI) plays a pivotal role in detection.5-7 Multiphase dynamic enhanced scanning with hepatocyte-specific magnetic resonance contrast agents is widely used, as hypointensity in the hepatobiliary phase (HBP) is considered an important sign for the diagnosis of HCC.8,9
According to the guidelines of the American Association for the Study of Liver Diseases (AASLD) and the European Association for the Study of the Liver (EASL), HCC in high-risk patients can be diagnosed based solely on imaging. Gadobenate dimeglumine (Gd-BOPTA) and gadolinium ethoxybenzyl dimeglumine (Gd-EOB-DTPA) are the 2 most commonly used hepatocyte-specific contrast agents in clinical practice. Previous studies have confirmed that MR employing liver-specific contrast agents such as Gd-BOPTA and Gd-EOB-DTPA is superior to nonspecific dynamic enhanced MR in the noninvasive diagnosis of liver cancer.10-14 Although Gd-BOPTA and Gd-EOB-DTPA are both taken up by hepatocytes, they differ in terms of drug composition and metabolism.15-19 The major features recommended by the Liver Imaging Reporting and Data System (LI-RADS 2018v), which was issued by the American College of Radiology (ACR), 20 for detecting HCC are nonrim arterial hyperenhancement (nonrim APHE), capsule enhancement, and nonperipheral washout. Each is closely related to the signal contrast between the lesion and surrounding liver tissues caused by the uptake of contrast agents in each phase of dynamic scanning. In previous studies, the ability of the 2 specific contrast agents to display these features was evaluated separately, with both confirmed to have effective diagnostic ability.
The purpose of this study was to evaluate the ability of the above 2 contrast agents to display capsule enhancement, nonrim APHE and nonperipheral washout by comparing the signal contrast between lesions and liver tissues in each phase of dynamic enhancement to provide a better contrast agent selection scheme for clinical practice.
Materials and Methods
Patients
We collected 505 patients with PHC confirmed by pathology from January 2015 to October 2022 in our hospital; 187 patients who only underwent biopsy were excluded. Thirty-nine patients who underwent only extracellular contrast MR examination preoperatively and 190 patients who underwent only enhanced computed tomographic examination preoperatively were also excluded. Finally, 89 patients who underwent hepatocyte-specific contrast-based magnetic resonance examinations within 2 weeks preoperatively were enrolled, and 81 patients were confirmed to have HCC by pathology, including 42 in whom Gd-EOB-DTPA was used and 39 in whom Gd-BOPTA was used. In cases of multifocal liver cancer, only different pathological grades were selected for evaluation by 2 doctors with more than 10 years of abdominal MR film-reading experience. A total of 98 lesions were evaluated (Figure 1).

Flowchart of lesion collection.
MRI
Liver MRI was performed using a 3.0 T (Skyra MR, Siemens Healthineers/Architect MR, GE Healthcare; n = 41) or 1.5 T (Avanto MR, Siemens Healthineers; n = 40) scanner. All scanners were equipped with an 8-channel phased-array body coil. The routine liver MRI protocol used for all patients included an axial respiratory-triggered fat-saturated T2-weighted fast spin‒echo sequence, respiratory-triggered diffusion-weighted imaging with a single-shot echo-planar sequence with
Basic Parameters of Liver Specific Contrast Agent Scanning Sequence.

The contrast agent injection programs of the contrast agent for Gd-EOB-DTPA (Primovist ®, Bayer Healthcare) and Gd-BOPTA (MultiHance®, Bracco) are provided were as follows: The dose of A 0.1 ml/kg dose of Gd-EOB-DTPA was 0.1 mL/kg manually injected, followed by 20 mL of normal saline flow rate of 2 mL/s. A 0.2 mL/kg The dose of Gd-BOPTA was 0.2 mL/kg with injected at a flow rate of 2 mL/s, followed by injection of 20 mL normal saline at a flow rate of 2 mL/s.
Image Analysis
When using Gd-BOPTA, the HBP needs to be relocated for scanning. All, and all regions of interest (ROIs) were selected and measured manually by 2 abdominal radiologists (with 16 and 6 years of experience in abdominal MR imaging). Both were aware of the study protocol and independently measured all the MR images in 2 review sessions separated by 4 weeks. In the first session, Gd-BOPTA-MR images were measured by Reviewer 1, and Gd-EOB-DTPA-MR images were measured by Reviewer 2; this order was reversed in the second session. The 2 senior radiologists were not blinded to the MR contrast agent used because they can detect the presence of HBP.
The lesion-to-liver contrast (LLC) assesses lesion conspicuousness during different phases and is defined as LLC = SIlesion−SIliver (absolute value taken). The ROI for measuring SIlesion was selected in the area with the most obvious signal change inside of the lesion in each sequence, and the ROI for measuring SIliver was selected in the adjacent normal liver parenchyma away from the tumor. The SI was obtained from AP/portal venous phase (PVP)/TP/DP/HBP. The typical ROI size per slice was approximately 2 cm2, and large intrahepatic vessels and artefacts were avoided.
The 2 reviewers also evaluated whether the lesion was visible in the 5 enhancement phases; both considered an absolute LLC value below 20 as the lesion being invisible in that phase. In other words, when the absolute value of the LLC was greater than 20, they reported a 1, and when it was less than 20, they reported a 0. Therefore, each lesion had a maximum score of 5 and a minimum score of 0. A total score of less than 3 was evaluated as poor display ability and was marked 0, while a total score of 3 or above was evaluated as good display ability and was marked as 1.
Capsule enhancement, a major feature of HCC, was evaluated on PVP, DP or TP images. The minimum value of the 3 measurements was recorded as the diffusion-weighted imaging/apparent diffusion coefficient value.
When the results of the 2 radiologists were inconsistent, a third abdominal radiologist (with 20 years of experience in abdominal MR imaging) further evaluated the images and decided which result to use.
Histopathologic Diagnosis
The histopathological lesion diagnosis after surgical resection was used as the standard of reference. Histopathologic analysis was performed by two pathologists with more than 5 years of experience in liver pathology.
Statistical Analysis
The data description is provided in Appendix 1, and the data is not distributed according to a normal distribution., a rank sum test was conducted. Statistical analyses were performed using SPSS, version 26 (IBM). A P value < 0.05 was considered to indicate statistical significance. Evaluation of the efficiency of Gd-BOPTA-MR and Gd-EOB-DTPA-MR for HCC was assessed by receiver operating characteristic (ROC) curve analysis.
Results
Patients and Lesions
The basic clinical data and grouping proportions of the 81 patients are as follows. A total of 42 patients (42/81, 51.9%) underwent Gd-EOB-DTPA-MR examination, with an average age of 55.45 years (standard deviation 12.28), 40 of whom had a chronic hepatitis history and 24 of whom had elevated alpha-fetoprotein (AFP) levels. The other 39 patients (39/81, 48.1%) underwent Gd-BOPTA-MR examination and were older (average age 56.95 years, standard deviation 10.51); 34 had a history of chronic hepatitis, and 24 had elevated AFP levels. The difference in pathological and clinical liver function grading of the 106 lesions between the 2 groups was assessed by the Mann‒Whitney
Assessment of Consistency Between the 2 Groups of Contrast Agents.

Abbreviations: Gd-EOB-DTPA, gadolinium ethoxybenzyl dimeglumine; HCC: hepatocellular carcinoma.
Imaging Features of HCC
Capsule Enhancement
There was a significant difference between the 2 contrast agents (
Evaluation of Capsule Enhancement Differences Between the 2 Groups of Contrast Agents.

Abbreviations: Gd-EOB-DTPA, gadolinium ethoxybenzyl dimeglumine; Gd-BOPTA, gadobenate dimeglumine.
Lesion-to-liver Contrast
None of the data from the 5 groups were consistent with a normal distribution, so the Mann‒Whitney U test was used for comparison. The LLC in the TP and HBP was not significantly different between Gd-BOPTA and Gd-EOB-DTPA, but there was a significant difference between the 2 groups in the LLC in the other dynamic enhancement phases, as indicated in Table 4 and Figures 2 and 3.
Skyra MR, siemens 3.0 T. A 67-year-old male with Child‒Pugh Classification A and surgically confirmed HCC (medium grade). Gd-BOPTA-MR shows obvious nonrim APHE in the AP, capsule enhancement in the TP and DP, nonperipheral washout during the AP to DP, and hypointensity in the HBP. The signal intensity between the lesion and the surrounding normal liver tissue had good contrast. Abbreviations: Gd-BOPTA, gadobenate dimeglumine; HCC: hepatocellular carcinoma; AP: arterial phase; TP: transitional phase; DP, delayed phase; HBP, hepatobiliary phase; nonrim APHE, nonrim arterial hyperenhancement.

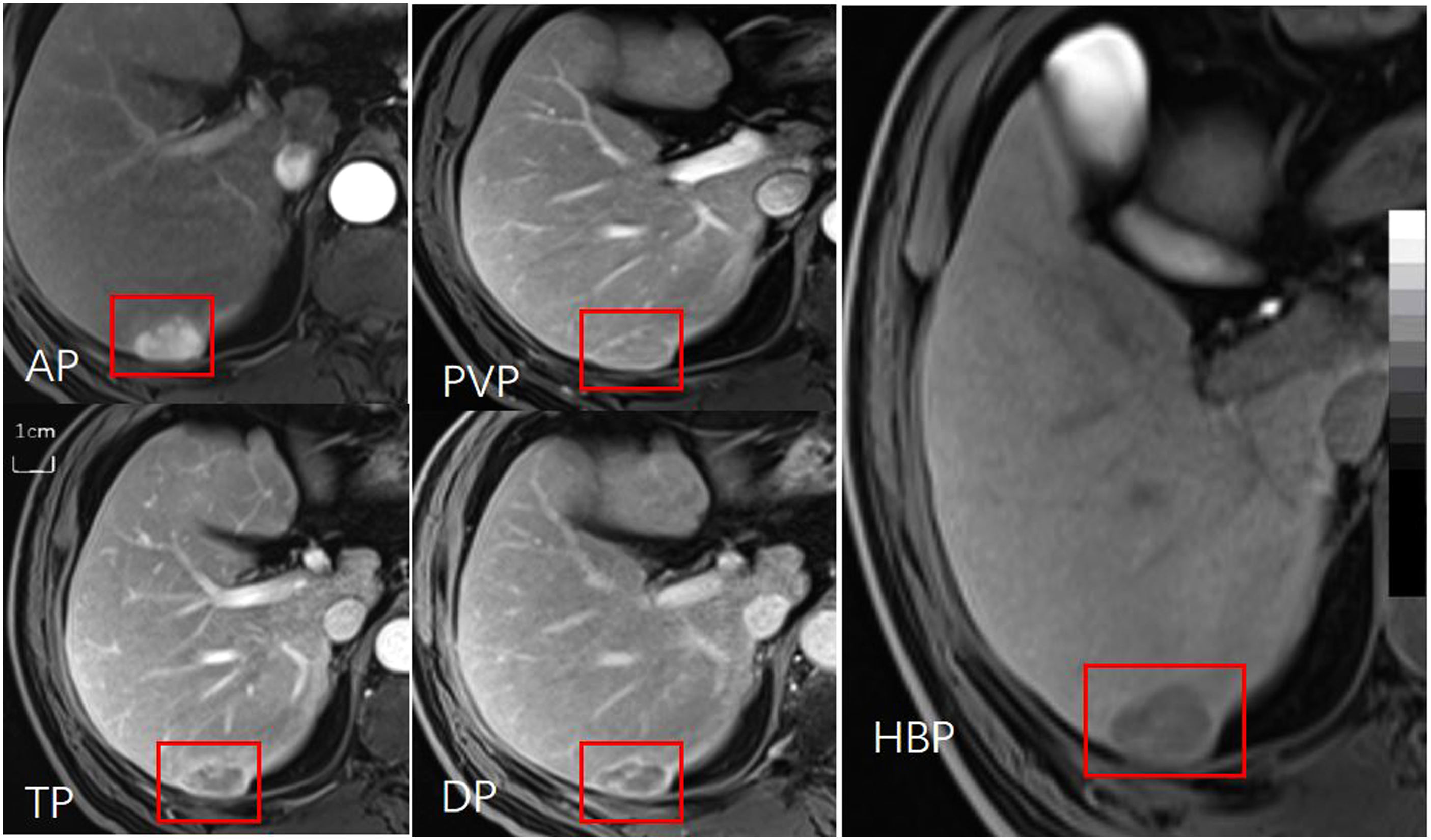
Skyra MR, siemens 3.0 T. A 72-year-old male with Child‒Pugh Classification A and surgically confirmed HCC (medium grade). Gd-EOB-DTPA-MR shows slightly nonrim APHE in the AP, no capsule enhancement in the PVP, TP or DP, no nonperipheral washout during AP to DP (LLC less than 20), and hypointensity during the HBP. Abbreviations: Gd-EOB-DTPA, gadolinium ethoxybenzyl dimeglumine; HCC: hepatocellular carcinoma; AP: arterial phase; PVP: portal venous phase; TP: transitional phase; DP, delayed phase; HBP, hepatobiliary phase; LLC, lesion-to-liver contrast; nonrim APHE, nonrim arterial hyperenhancement.
Difference Enhancement* in LLCR Between the 2 Contrast Agents in Each Phase of Dynamic Agents.

Abbreviations: Gd-EOB-DTPA, gadolinium ethoxybenzyl dimeglumine; Gd-BOPTA, gadobenate dimeglumine; AP: arterial phase; PVP: portal venous phase; TP, transitional phase; DP, delayed phase; HBP, hepatobiliary phase.
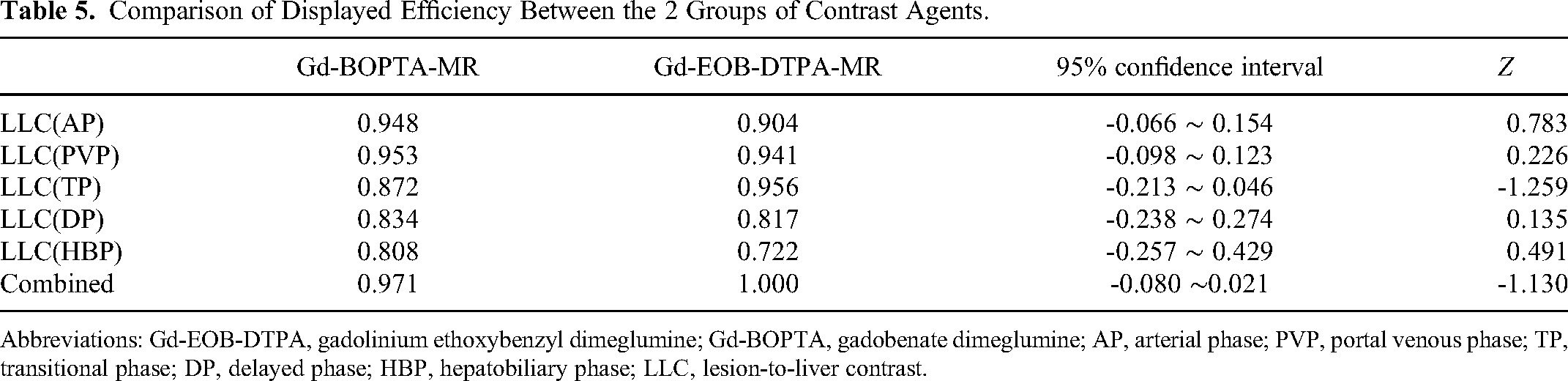
Differences in display efficiency between Gd-EOB-DTPA MR and Gd-BOPTA MR in terms of the LLC were assessed by ROC curve analysis. Gd-EOB-DTPA and Gd-BOPTA both have high display efficiency in each phase(AP/PVP/TP/DP/HBP) Gd-EOB-DTPA:0.904/0.941/0.956/0.817/0.722, Gd-BOPTA:0.948/0.953/0.872/0.834/0.808. As depicted in Table 5 and Figure 4(a/b/c/d/e/f).

ROC curves of the display efficiency of Gd-BOPTA and Gd-EOB-DTPA in different phases. (A) Arterial phase, (B) portal venous phase, (C) transitional phase, (D) delayed phase, (E) hepatobiliary phase, and (F) the combination of the 5 phases. Abbreviations: Gd-EOB-DTPA, gadolinium ethoxybenzyl dimeglumine; Gd-BOPTA, gadobenate dimeglumine; ROC, receiver operating characteristic.
Comparison of Displayed Efficiency Between the 2 Groups of Contrast Agents.
Abbreviations: Gd-EOB-DTPA, gadolinium ethoxybenzyl dimeglumine; Gd-BOPTA, gadobenate dimeglumine; AP, arterial phase; PVP, portal venous phase; TP, transitional phase; DP, delayed phase; HBP, hepatobiliary phase; LLC, lesion-to-liver contrast.
Intra/interclass correlation coefficients (ICCs) were used to calculate the consistency between reviewers, and the interobserver agreement of all measurements between the 2 reviewers was excellent, with ICC values greater than 0.80.
Discussion
Capsule enhancement is a major feature of HCC, 20 with an incidence between approximately 43% and 64%; such capsules can be divided into normal capsules and pseudocapsules based on the pathology and are composed of sinusoids, fibrosis, or both. 21 In general, capsule enhancement is defined as a smooth and uniform edge around the lesion, which is usually displayed on PVP or TP/DP images. In our study, the incidence of enhanced capsules was approximately 47% (46/98); the display rate with Gd-BOPTA-MR (36/47, 76%) was significantly higher than that with Gd-EOB-DTPA-MR (10/51, 20%), which is consistent with previous research. 22 The 2 contrast agents have pharmacokinetic and pharmacodynamic differences that impact their ability to display capsule enhancement; hepatocytes only selectively take up 3% to5% Gd-BOPTA but 50% Gd-EOB-DTPA. As a result, the intensity and speed of signal enhancement in the liver parenchyma are higher after injection of Gd-EOB-DTPA than after injection of Gd-BOPTA, 23 which may lead to the display of an enhanced capsule being masked to a certain degree. In addition, the dose of Gd-BOPTA in this group was twice that of Gd-EOB-DTPA, which may also explain the good visibility of the capsule.
The other 2 major features of HCC proposed in LI-RADS 2018v are nonrim APHE and nonperipheral “washout,” the display of which both depend on the signal difference between the lesion and liver parenchyma in each phase of dynamic liver enhancement.
In this study, there was a significant difference in the LLC between the 2 contrast agents during the dynamic enhancement AP; although both had good display efficiency, Gd-BOPTA was superior to Gd-EOB-DTPA, which is consistent with previous research. 24 First, Gd-EOB-DTPA and Gd-BOPTA both exhibit high T1 relaxation, nearly twice as high as that of conventional extracellular contrast agents, but the recommended dose of Gd-EOB-DTPA is only 1/2 that of Gd-BOPTA,25,26 which may lead to a reduction in its arterial peak. As previous Gd-EOB-DTPA dynamic enhancement studies have reported that the peak enhancement of HCC occurs approximately 14 s after the contrast agent arrives at the aorta, 27 the acquisition time in the AP is key for peak lesion enhancement. Furthermore, Gd-EOB-DTPA will lead to blurring and motion artefacts on dynamic enhanced MR imaging, especially in the AP,28,29 with a great impact on observation of the arterial-phase focus enhancement peak. Overall, the imaging quality of the Gd-EOB-DTPA in AP can be improved to a certain extent with the application of 3.0 T MR multiarterial-phase fast-imaging sequences and an emphasis on breath-hold training before scanning. 30
We also found that the LLC of the 2 contrast agents significantly differed during the PVP/DP, compared with Gd-EOB-DTPA, Gd-BOPTA results in higher which is consistent with previous research in healthy volunteers, 31 but during TP, which Gd-EOB-DTPA had superior display efficiency. We consider that these are all related to the pharmacokinetics and acquisition time of the 2 contrast agents. According to the LI-RADS 2018v, the PVP is defined as the time during which the portal vein is filled with contrast medium while the liver parenchyma is slightly enhanced, beginning approximately 60 s after contrast medium injection. The TP is defined as the time during which the signal of the hepatic blood vessel is similar to that of the hepatic parenchyma, and the scanning time begins approximately 2 to5 min after contrast medium injection. For determination of nonperipheral “washout,” some Asian guidelines allow the use of both the PVP and TP, although most major international guidelines only allow the use of the PVP for this purpose. 32 The LLC in the PVP/TP is composed of 2 components: the washing out of contrast agent within the lesion and progressive enhancement of the surrounding liver parenchyma. 33 Approximately 3 min after injection of Gd-EOB-DTPA, hepatocytes begin to take up the agent, and the liver parenchyma reaches maximum enhancement within 10 to20 min. However, the uptake of Gd-BOPTA by hepatocytes starts relatively late, and the liver parenchyma reaches its maximum enhancement in approximately 2 h, so the display efficiency of Gd-EOB-DTPA is significantly improved at the PVP over that at the AP, reaching its optimal level in the TP and surpassing that of Gd-BOPTA. The DP sequence is collected within 5 to10 min after contrast injection, as defined by our hospital. In our study, we found that LLC during the DP, Gd-BOPTA slightly outperformed Gd-EOB-DTPA in displaying the lesions,which is consistent with previous research. 34 Due to the time delay associated with this phase, the uptake of specific contrast medium by hepatocytes increases, and thus, the injection dose of Gd-BOPTA is twice that of Gd-EOB-DTPA. Previous studies have found that higher concentrations of contrast agent in the blood might help to increase portal vein blood flow and consequently total hepatic flow, 35 which might explain the slightly higher display efficiency obtained with Gd-BOPTA than with Gd-EOB-DTPA.
During the HBP, the LLC of the 2 contrast agents was not significantly different, which is consistent with the results of Park and colleagues’ previous retrospective study on 47 patients with HCC lesions. 36 The display efficiency of Gd-EOB-DTPA decreased, possibly due to hepatic function, as the liver damage caused by chronic liver disease increased, and it is difficult to obtain sufficient liver parenchymal signal elevation even after waiting for more than 20 min after contrast administration. 37 Although there was no significant difference in the incidence of hepatitis B or cirrhosis or in the Child‒Pugh grades between the 2 groups, the injection dose of Gd-BOPTA was twice that of Gd-EOB-DTPA, and the acquisition time was 2 h later.
Compared with BOPTA, EOB uses a lower dose, and most of it is excreted through bile; therefore, this is a relatively safe choice for patients at risk of nephrogenic systemic fibrosis, 38 with a short scanning time and high patient tolerance. 39 On the other hand, Gd-BOPTA is not recommended for patients with Child–Pugh grade C liver function, for whom the enhancement signal of the liver parenchyma is reduced, nor for patients with renal insufficiency. Due to the blurring and motion artefacts of Gd-EOB-DTPA is not recommended for patients who are unable to complete breathing training or if the examination equipment cannot perform multiple AP scans.
This study has several limitations. First, it was a retrospective, single-centre study rather than a large-scale, prospective, multicentre study. Second, we did not perform a crossover study (with both contrast agents used in the same patient population), but there was no significant difference in age and liver function grading, among others, between the groups. Third, because it was measured at different series, at different times, and with different machines, the LLC could be influenced by different factors. Although the overall difference was not significant and the scanning time and sequence met the standards for liver dynamic enhancement, a more objective and accurate assessment of LLC should be conducted after standardization. Fourth, the sample size was small, and larger sample sizes are needed to better assess the parameters.
Conclusion
Both Gd-BOPTA and Gd-EOB-DTPA are widely used in clinical practice as hepatobiliary-specific contrast agents; they both have good display efficiency for HCC, but each of them has its own characteristics. The agent should be selected according to the actual situation. With additional research, we believe more detailed personalized suggestions will emerge in the future. Consequently, when selecting preoperative examinations for HCC, we can offer more tailored recommendations to clinicians, and further improve the diagnostic accuracy of noninvasive HCC.
Highlights
Gd-BOPTA and Gd-EOB-DTPA can be independently used in the noninvasive diagnosis of HCC as representatives of hepatocyte-specific contrast agents.
Gd-BOPTA and Gd-EOB-DTPA display the major features of noninvasive diagnosis of HCC based on LI-RADS 2018v differently. Combined with the material differences between the 2 contrast agents, the choice of Gd-BOPTA and Gd-EOB-DTPA should be optimized according to the specifics of each case in clinical practice.
Footnotes
Abbreviations
Acknowledgements
Thanks to Dr Yu-Feng Liu (Southern Medical University, Guangzhou, China) for his help in this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was reviewed and approved by the ethics committee of our institution (Ethics approval number: HKUSZH2023119).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangdong Basic and Applied Basic Research Foundation (grant number 2021A1515220131), Guangdong Medical Science and Technology Research Fund Project (No. A2024475), and Clinical Research Project of Shenzhen Second People's Hospital, China (No.20223357022).
Appendix 1.
The figure all above depicts the distribution of LCC values across the 5 scanning phases, with AP/PVP/TP/DP/HBP arranged from top to bottom.
The LCC value in AP, the median of the Gd-BOPTA group is 154, the quartile is 52, while the median of the Gd-EOB-DTPA group is 51, and the quartile is 20, with an overall median of 89 and a quartile of 28.75.
The LCC value in PVP, the median of the Gd-BOPTA group is 107, the quartile is 40, while the median of the Gd-EOB-DTPA group is 51, and the quartile is 21, with an overall median of 68 and a quartile of 29.
The LCC value in TP, the median of the Gd-BOPTA group is 80, the quartile is 14, while the median of the Gd-EOB-DTPA group is 92, and the quartile is 34, with an overall median of 84 and a quartile of 32.75.
The LCC value in DP, the median of the Gd-BOPTA group is 101, the quartile is 36, while the median of the Gd-EOB-DTPA group is 167, and the quartile is 119, with an overall median of 149.5 and a quartile of 89.
The LCC value in HBP, the median of the Gd-BOPTA group is 211, the quartile is 168, while the median of the Gd-EOB-DTPA group is 221, and the quartile is 185, with an overall median of 214.5 and a quartile of 175.5.
The overall distribution does not conform to the normal distribution, hence the rank sum test is chosen.
