Abstract
Keywords
Introduction
Hepatocellular carcinoma (HCC), a prevalent type of cancer in China, has a notably high occurrence rate and is currently ranked as the fourth major cause of cancer-related mortality. Due to the distinct imaging manifestations associated with HCC, a conclusive diagnosis can often be made solely based on imaging findings, eliminating the need for invasive histopathological analysis in high-risk patients. In order to address this need, the American College of Radiology (ACR) released the Liver Imaging Reporting and Data System (LI-RADS) for the first time in 2011, aimed at standardizing the imaging assessment of HCC in screening, surveillance, diagnosis, and evaluating treatment response for high-risk populations with HCC. 1 Subsequently, after continuous improvement, the latest is LI-RADS version 2018. LI-RADS provides a detailed description of the major features(MF) of HCC and various ancillary features(AF), including those that are specific to HCC, and those that tend to be malignant but not HCC. The primary goal of the LR-5(definitely HCC) imaging guideline is to ensure high specificity in the diagnosis of HCC. A recent meta-analysis has validated that the LR-5 category in LI-RADS v2018 boasts an impressive specificity of up to 91% for diagnosing HCC. 2
Gadolinium ethoxybenzyl diethylenetriaminepentaacetic acid (Gd-EOB-DTPA) is increasingly utilized as a hepatic-specific contrast agent for its ability to provide hepatobiliary phase (HBP) image. Recent studies have shown that the reduced sensitivities of LR-5 criteria pose a greater challenge for EOB-MRI compared to extracellular contrast agent-enhanced MRI (ECA-MRI), 3 as LI-RADS was originally designed for ECA-MRI, 4 and it was reported with lower arterial vascular and parenchymal enhancement. 5 In clinical practice, arterial phase hyperenhancement (APHE) has always been the MF of HCC in all versions of LI-RADS (including v2018). 6 Nevertheless, a few cases of early-stage HCC have not experienced arteriolation7,8 and does not exhibit APHE manifestations. This could lead to the misclassification of the LR-5 category into other categories, potentially decreasing the sensitivity of LI-RADS for HCC. Threshold growth indicates the rate at which tumors grow and is often a reference indicator for differentiating between benign and malignant tumors. However, the use of threshold growth in the diagnosis of HCC is controversial, and there is overlap between HCC and non-HCC, such as intrahepatic cholangiocarcinoma(ICC). 9 Research has demonstrated that reclassifying threshold growth as an AF and substituting it with a more specific AF related to HCC, the diagnostic performance of LR-5 is comparable. 10
We hypothesized that threshold growth can be replaced by a AF favoring HCC in particular with greater frequency, APHE extended to hypointensity on precontrast images/isointensity in the arterial phase (AP), and expanding washout to hypointensity in the transitional phase (TP) (2-min) could enhance the sensitivity of diagnosing HCC without compromising the specificity. Therefore, the purpose of our research was to assess the effectiveness of adjusted LR-5 category in the diagnosis of HCC by examining the sensitivity, specificity and positive predictive value (PPV).
Materials and Methods
Patients
This retrospective study was approved by the Institutional Review Board of our hospital (2023-RE-262), and the need for patient informed consent was exempted. We have de-identified all patient details and the reporting of this study conforms to STROBE guidelines. 11 A retrospective analysis was conducted on 2321 consecutive patients deemed to be at elevated risk for HCC, who received Gd-EOB-DTPA-enhanced MRI between October 2019 to June 2023. Patients were included in our study according to the following inclusion criteria: (1) ≥ 18 years of age; (2) chronic hepatitis B viral infection, or cirrhosis, or a past history of surgical treatment for HCC; (3) having received an Gd-EOB-DTPA-enhanced MRI scan with lesions ≥ 1 cm; (4) the pathological diagnosis was obtained through surgery or biopsy within 3 months after MR examination. The exclusion criteria were as follows: (1) no pathological results were obtained; (2) cirrhosis resulting from genetic factors or abnormalities in blood vessels; (3) the lesion received local-region treatment such as radiofrequency ablation, radiotherapy, or systematic anti-tumor treatment; (4) presence of multiple lesions hindering accurate determination of the number and boundaries; and (5) inadequate image quality rendering evaluation impossible (Figure 1).

Flowchart of the inclusion and exclusion criteria of our study sample.
MRI Techniques
None of the patients were allowed to eat anything within 6 h before the examination. MRI was performed using 3.0-T scanners (Magnetom VERIO, Siemens Healthineers; and Discovery MR 750w, GE Healthcare). The initial MR imaging protocols included: fat-suppressed turbo spin-echo T2-weighted sequence, a single-shot echo-planar technique with two b values (50, 800 s/mm2) allowing free-breathing and in-phase and out-of-phase T1-weighted sequences; dynamic contrast–enhanced images obtained with either a three-dimensional volumetric interpolated breath-hold examination T1-weighted imaging with fat suppression (3D-VIBE-TIWI-FS) or a LAVA-Flex sequence; and the utilization of FluoroTrigger technology. The parameters related to MRI are shown in Tables 1 and 2 below.
MR Scanning Parameters (GE Healthcare).
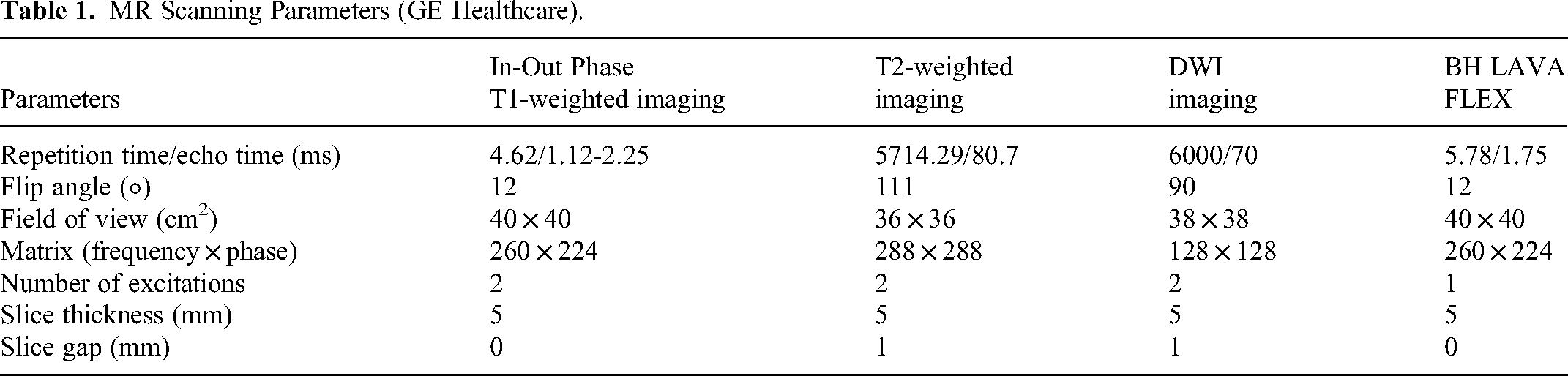
MR Scanning Parameters (Siemens Healthineers).

Dynamic contrast-enhanced imaging involved the administration of GD-EOB-DTPA (Primovist, Bayer Schering, Pharma AG, Berlin) through the cubital vein using a high-pressure syringe. The contrast agent was injected at a dose of 0.1 ml/kg at a flow rate of 1 mL/sec followed by a 10 ml saline flush. Images of dual-AP(including the early and late phases), portal venous phase (PVP), TP, and HBP were collected at 10 s, 60 s, 120 s and 20 min after the injection of contrast agent.
Image Analysis and Modification of the LI-RADS Category Assignment
Two abdominal MRI radiologists with 5 and 10 years of experience were tasked with assessing the findings without knowledge of the pathological results. If there was a discrepancy, a consensus review session of the two radiologists was performed to resolve any disagreements. The observation content included (a) tumor size: defined as the maximum axial diameter on PVP/TP images; (b) number of observations; (c) MF of HCC according to LI-RADS v2018: nonrim APHE, nonperipheral washout, and enhancing capsule; AF favoring HCC in particular: nonenhancing capsule, nodule-in-nodule, mosaic architecture, blood products within the mass, and higher fat content within the mass compared to the surrounding liver tissue; (d) for cases with previous examination enabling assessment for threshold growth: the radiologists measured and compared the longest diameter on the current and prior dynamic enhanced MRI in PVP/TP imaging, as well as PVP or delayed phase(DP) of CT (diameter increase ≥ 50% within ≤ 6 months); (e) modified major features: nonrim APHE extended to hypointensity on precontrast imaging and isointensity in the AP, nonperipheral washout extended to hypointensity in TP(2-min); (f) assessment of AP image artifacts and scoring according to 5-point scale: 1 point, no respiratory artifacts; 2 points, mild respiratory artifacts with negligible effect on diagnostic accuracy; 3 points, moderate respiratory artifacts with slight impact on diagnosis but not severe; 4 points, severe respiratory artifacts, poor image quality but diagnosable; 5 points, widespread respiratory artifacts rendering images undiagnosable.
Diagnostic Performance
The modified LI-RADS versions were as follows: LR-5A, a HCC-specific AF with greater frequency replaces threshold growth; LR-5B, a HCC-specific AF replaces threshold growth and nonrim APHE extending to hypointensity on precontrast imaging and isointensity in the AP; LR-5C, a HCC-specific AF replaces threshold growth, nonrim APHE extended to hypointensity on precontrast imaging and iso or hyperintensity in the PVP, and nonperipheral washout extended to hypointensity in TP(2 min).
Histopathologic Diagnosis
The standard of reference for histopathologic diagnoses was established through surgical resection or biopsy. Two pathologists, each with over 5 years of experience in liver pathology, primarily conducted our histopathological diagnosis, and the grading of pathology was determined following the WHO classification of tumors of the digestive system. 12
Statistical Analysis
The evaluation of parameter normal distribution was conducted using the Shapiro–Wilk test. Mean ± standard deviation was reported for count data, and either the independent sample t test or nonparametric Mann‒Whitney U test was utilized. Comparison of patients’ baseline characteristics involved the use of the χ2 test or Fisher's exact test for categorical variables. Student's t test or the Wilcoxon signed-rank test was applied to continuous variables. The sensitivity and specificity of the original LI-RADS v2018 based on GD-EOB-DTPA MRI with the modified LR-5 Strategies (LR-5A, LR-5B, LR-5C) for diagnosing HCC using McNemar's test. Moreover, a 95% confidence interval (95% CI) was calculated. Interreader agreement for MF and AF favoring HCC, AP image quality and image scoring was determined using Cohen's kappa(k) test. Agreement levels were interpreted as follows: slight(0.01-0.20), fair(0.21-0.40), moderate (0.41-0.60), substantial(0.61-0.80), and excellent(0.81-0.99). Statistical analyses were conducted with SPSS version 20 (IBM), with statistical significance set at p < .05.
Results
Patient Characteristics
This group included 412 patients at a heightened risk for HCC, including 78 cases of cirrhosis due to hepatitis B, 4 patients of cirrhosis from alcoholic liver disease, 1 patient of cirrhosis resulting from hepatitis C, 1 patient due to schistosomes, and 383, 24 and 5 patients classified as Child‒Pugh class A, B and C, respectively. Among the patients without cirrhosis, 323 patients had chronic hepatitis B, 1 patient had hepatitis E combined with hepatitis B, and 4 patients had hepatitis C combined with hepatitis B. None of the patients were diagnosed with liver cirrhosis stemming from nonalcoholic fatty liver disease. Among the cohort of 412 patients, there were a total of 445 observations with 381 patients having 1 observation and 32 patients having 2 observations. Of these cases, 391 observations were HCC, 39 were non-HCC malignant tumors, and 15 were benign lesions. (Table 3)
Clinical Characteristics of the 412 Study Subjects at Risk of Hepatocellular Carcinoma (HCC).

HCC,hepatocellular carcinoma; IMCC,intrahepatic mass-forming cholangiocarcinoma; cHCC-CCA,combined HCC-choangiocarcinoma; FNH, focal nodular hyperplasia; AML, angiomyolipoma
LI-5 Major Features and Ancillary Features Favoring HCC and AP Image Quality Scores: Interreader Agreement
The 2 radiologists interpreted the MF and AF favoring HCC for all lesions. The κ values were greater than 0.6 (0.631-0.911) except for the nonenhancing capsule (κ = 0.588). The average κ value for all lesions was 0.684 ± 0.169(Table 4). Both radiologists identified artifacts in a total of 151 AP images from 412 patients, but there was a discrepancy in the quality scores between early and late arterial images, with κ values of 0.752 and 0.856, respectively. This indicates a high level of agreement between the 2 radiologists. (Table 5)
The Frequency of Major Features and Ancillary Features Favoring HCC According to LI-RADS v2018 by Readers with Interobserver Agreement Analysis.

Interobserver Agreement Analysis of Arterial Phase Image Artefacts and Image Quality Scores.

The Diagnostic Performance of Major and Ancillary Features Favoring HCC in HCC and Non-HCC Malignancy
Utilizing EOB-enhanced MRI, a total of 354 observations revealed non-APHE, with 37 observations demonstrating hypointensity in the precontrast images and isointensity in AP. 309 observations displayed washout during PVP, while 82 exhibited iso- or hyperintensity. Furthermore, 8 observations demonstrated iso- or hyperintensity in TP and 6 observations in HBP. Compared to non-HCC malignancy, the prevalence of these three MFs (APHE, washout, and enhancing capsule) was higher in HCC, except for threshold growth, with statistically significant variations (p < .001). Concerning AF favoring HCC in particular, blood products in the mass were significantly more frequent in HCC compared to non-HCC malignancy (p < .001). The remaining four AF favoring HCC did not exhibit statistically significant difference between the two groups (p > .05). (Table 6)
The Difference of MF and AF in HCC and no-HCC Malignancy.

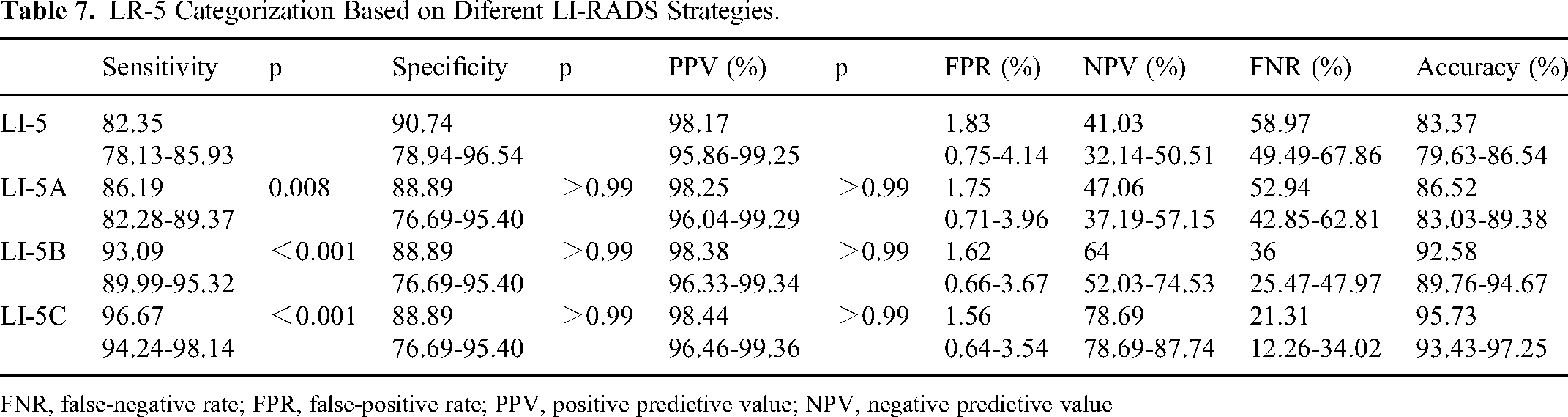
Compared to LI-RADS v2018, 1 LR-3 nodule, 3 LR-4 nodules, and 16 LR-M nodules were reassigned to LR-5 using LR-5A (Figure 2); 2 LR-3 nodules and 22 LR-4 nodules were reassigned to LR-5 using LR-5B, based on LR-5A (Figure 3); and 5 LR-3 nodules and 15 LR-4 nodules were reassigned to LR-5 using LR-5C, based on LR-5B (Figure 4). There were no other LI-RADS category changes. Despite the modified LI-RADS strategy showing a marginally decreased specificity compared to the original LI-RADS strategy, the difference was not notable. The sensitivity differences between the four groups were statistically significant, with the highest sensitivity in the LR-5C strategy. (Table 7)

Hepatocellular carcinoma in a 43-year-old man. A-C T2WI showed a moderate hyperintensity mass(A), with hypointensity and blood products in mass on precontrast T1WI(B) and diffusion weight image showed target appearance(C). D-F The dynamic Gd-BOPTA-enhanced scanning showed isoenhancement in early(D)、late arterial phase(E) and hypointensity in portal venous phase, transitional phase, and enhanced capsule can be seen (F,G). According to LI-RADS v2018 criteria, the lesion was classified as LR-M. However, upon utilizing blood products in mass to replace threshold growth as a major feature and nonrim arterial phase high enhancement extended to hypointensity on precontrast imaging and isointensity in arterial phase, the lesion was re-diagnosed as HCC(LR-5).

Hepatocellular carcinoma in a 58-year-old man. The mass appeared hypointense on the precontrast T1WI(A). The Gd-BOPTA-enhanced arterial phase showed isoenhancement(B,C) and hypoenhancement in portal venous phase(D). Following the LI-RADS v2018 criteria, the lesion was classified as LR-4. However, when nonrim arterial phase high enhancement extended to hypointensity on precontrast imaging and isointensity in arterial phase, the lesion was reclassified as LR-5.

Hepatocellular carcinoma in a 71-year-old man.T2WI(A) showed 19-mm lesion with circular, uniform, moderate hyperintensity and hypointensity on precontrast T1WI(B). Gd-BOPTA-enhanced MRI showed the lesion with hypoenhancement in early arterial phase(C) and hyperenhancement in late arterial phase(D)(although there was respiratory artifact), but without the presence of washout and enhancing capsule in the portal venous phase(E). The lesion was hypointensity in transitional phase(2 min)(F) and enhancing “capsule” can be seen, whlie the lesion was isointensity in transitional phase(5 min)(G) and moderate hypointensity in hepatobiliary phase(H). According to LI-RADS v2018 criteria, the lesion was categorized as LR-4. However, upon the extension of washout appearance from portal venous phase to transitional phase(2 min), the lesion was reclassified as LR-5.
LR-5 Categorization Based on Diferent LI-RADS Strategies.
FNR, false-negative rate; FPR, false-positive rate; PPV, positive predictive value; NPV, negative predictive value
Discussion
The results of this study showed that when the most common AF blood products in the mass replaced the threshold growth as MF, nonrim APHE was redefined as hypointensity on precontrast imaging and isointensity in AP, and when the appearance of nonperipheral washout extended to TP (2 min), the sensitivity of the revised LR-5 in diagnosing HCC rose, while specificity remained largely unchanged.
LI-RADS v2018 provides a detailed description of major imaging features of HCC, while also standardizing many AFs. The trio of nonrim APHE, nonperipheral washout, and enhanced capsule display a solid underlying physiological rationale in HCC development. These features have been corroborated in HCC detection and are integrated into other major imaging-based HCC diagnostic algorithms, such as the Asian Organization, the American College of Radiology (LI-RADS), and the European Association for Liver Research (EASL).13,14,15 In contrast, according to LI-RADS v2018, the definition of threshold growth is primarily derived from expert viewpoints and aims to align with the Organ Acquisition and Transplantation Network (OPTN) algorithm. 16 Threshold growth, as the main sign for diagnosing HCC, lacks a strong pathological basis, as the rate of HCC growth can differ depending on the histological differentiation, initial size, and pathological type of the lesion.17,18 Moreover, the optimal timeframe between the two scans to assess threshold growth remains undetermined. Substituting original threshold growth with tumor growth rate may enhance LI-RADS sensitivity in diagnosing HCC (≤3.0 cm). 19 Research indicates that when threshold growth is reclassified as AF and replaced by HCC favorable AF (fat-in-nodule), the diagnostic accuracy of LR-5 is comparable. 10 It is suggested that threshold growth is more indicative of the malignancy level of the tumor rather than being specific to HCC. Therefore, we alternative AF turned to blood products in the mass, as HCC is a tumor with high vascularity and prone to hemorrhage within the lesion. Up to 18% of HCC have blood products in the mass. 20 On the other hand, the majority of cholangiocarcinoma-containing tumors (MCCs) with liver cirrhosis exhibit noticeable desmoplastic and hypovascularized tumor stroma, leading to limited internal hemorrhage. 21 For non-MCC patients, much blood products may be important predictors. 22 In a study that did not impose restrictions on tumor size revealed that the incidence of intratumoral bleeding was as high as 47.6%. 23 Our findings showed that blood products within the mass were seen in 129 lesions (32.99%). Despite the occurrence of three cases of cHCC-CCA combined with hemorrhage, each exhibited a primary HCC element, which was similar to Deng's findings. 24 We speculate that the presence of blood products within the lesion remains linked to the significant HCC component. Our research revealed a heightened prevalence of blood products within the mass among the 5 AFs favoring HCC specifically, which is different from Park’s study. 10 This may be related to the size of the lesion. In our research, the average diameter of the lesion was greater (4.80 cm vs 3.28 cm).
The basis of APHE is that HCC is mainly supplied by arterial blood, while the liver parenchyma is predominantly nourished by the portal vein and with high specificity (97%-99%). However, the sensitivity is limited, given that 30% of HCC cases show an atypical enhancement pattern due to the presence of immature neovascularization,25,26 which is usually seen in small, well-differentiated HCC. 27 In contrast to conventional gadolinium chelation, the activity of Gd-EOB-DTPA is weak and short, leading to atypical enhancement patterns in some HCC in the AP, although it can improve sensitivity to small HCC lesions (<2 cm). 28 Researchers have proposed improved diagnostic accuracy 29 and sensitivity 30 when using subtraction images for assessing arterial enhancement level. However, subtraction imaging is susceptible to registration inaccuracies caused by liver motion during respiration, posing a challenge to its sensitivity. Previous study has shown that patients are more prone to transient severe respiratory motion artifacts (TSMs) following the administration of disodium gadolinate, with an incidence rate of approximately 4.8% to 26.7%. 31 In our study, 151(36.65%) patients were found to have artifacts in early and/or late AP, including 15 (36.39%) cases showing hypointensity in precontrast images and isointensity in AP. The utilization of subtraction algorithms may result in inaccurate image alignment, compromising both the quality of diagnostic images and the precision of subtraction images. Despite employing visual techniques to evaluate signal characteristics in AP, we achieved a high level of interobserver agreement (κ = 0.865). When we expanded nonrim APHE to hypointensity on precontrast imaging and isointensity in AP, the specificity for diagnosing HCC remained constant, while the sensitivity increased. Therefore, although the number of HCC cases with hypointensity on precontrast imaging and isointensity in AP was not lager in this group, additional investigations are warranted to ascertain if modifications to the LI-RADS v2018 criteria are necessary to achieve comparable efficacy based on GD-EOB-DTPA-enhanced MRI.
In order to attain the utmost specificity in LI-RADS, when it comes to gadoxetic acid–enhanced MRI, the evaluation of washout should be only in PVP, 32 the presence of hypointensity in TP or HBP lesions is only considered an AF favoring malignancy, thus preventing an elevation to LR-5 in classification, 33 potentially diminishing diagnostic sensitivity. However, other major Asian recommendations suggest utilizing hypointensity in the TP and/or HBP as substitute explanations for washout.12,34 Studies have shown that expanding the appearance of washout to TP(180 s) or HBP (rather than limited to PVP) can improve the sensitivity without reducing the specificity. 35 Readjusting LR-5, extending the washout period to the TP(150 s), and using arterial subtraction images to interpret APHE classification methods can substantially enhance the sensitivity of HCC diagnosis without reducing the specificity. 36 Per the CT/MRI LI-RADS v2018 guidelines, 37 TP is chosen to be 2-5 min after injection of the contrast agents. In our study group, 29 patients underwent TP for 2 and 5 min simultaneously. Three cases showed hypointensity at 2 min and isointensity at 5 min, and 5 cases showed hypointensity at 2 min and patchy iso- or hyperintensity within the lesion at 5 min. Currently, the exact time at which liver cells begin to absorb contrast agents remains unknown. It is uncertain whether the different signal intensities of these 8 observations at 2 and 5 min can be interpreted as the contrast agent being predominantly washed out at 2 min, similar to the DP of extracellular contrast agent enhancement, while the later time starts with intracellular enhancement in the liver cells. We need to increase the size of the samples to determine if there exists a notable disparity in the signal intensity between TP(2-min) and TP(5-min), and nonperipheral washout can be extended to TP(2-min), just as washout can be observed at DP when an extra cellular contrast agent is used.
Our study had several limitations. First, this study is retrospective, and the imaging assessment of patients included is restricted to GD-EOB-DTPA-enhanced MRI, potentially introducing selection bias. Nonetheless, we aimed to enroll as many patients as feasible. Second, our findings are subject to regional constraints, as chronic hepatitis B is the primary reason for HCC in our country. None of the chosen patients had nonalcoholic fatty liver disease. Third, all imaging features were assessed using GD-EOB-DTPA-enhanced MRI, while LI-RADS was originally designed for MRI with extracellular contrast agents. This might limit the generalizability of our modified diagnostic strategies. Fourthly, only 37 observations showed hypointensity on precontrast imaging and isointensity in AP in this study, which may limit the statistical power of our study. However, the proportion of HCC with this appearance is not high, and we made efforts to reduce this constraint by enrolling a significant cohort of study participants. Lastly, this is a single center study, the only statistically significant difference observed between HCC and non-HCC groups was in the levels of blood products in the mass. The remaining four AFs had no difference between the two groups. This may be attributed to the considerable size of the lesion within this particular group and the prevalence of these four AFs in cHCC-CCA cases characterized by a predominance of HCC component. This type of tumor was also difficulty to identify. In the future, we plan to carry out a multicenter research to gather a larger sample size for statistical analysis and validate our results.
Conclusion
Based on GD-EOB-DTPA-enhanced MRI, when threshold growth was replaced by blood products within the mass, nonrim APHE extended to hypointensity on precontrast imaging and isointensity in AP, nonperipheral washout extended to hypointensity in TP (2 min) may increase the sensitivity of the modified LR-5 to HCC without compromising the specificity and PPV.
Footnotes
Acknowledgement
Medical Research Ethics Committee of the First Affiliated Hospital of the University of Science and Technology of China (Anhui Provincial Hospital), 2023-RE-262
English language, grammar, punctuation, spelling, and overall style were edited by one or more of the highly qualified native English speaking editors at SNAS.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Innovation Program of Anhui Province (grant number No. GXXT-2021-065).
