Abstract
Single biofilm biomimetic nanodrug delivery systems based on single cell membranes, such as erythrocytes and cancer cells, have immune evasion ability, good biocompatibility, prolonged blood circulation, and high tumor targeting. Because of the different characteristics and functions of each single cell membrane, more researchers are using various hybrid cell membranes according to their specific needs. This review focuses on several different types of biomimetic nanodrug-delivery systems based on composite biofilms and looks forward to the challenges and possible development directions of biomimetic nanodrug-delivery systems based on composite biofilms to provide reference and ideas for future research.
Keywords
Introduction
Cancer is a public health problem worldwide and has become the most important disease threatening human health with a high mortality rate.1,2 Studies have found that early diagnosis and treatment are crucial to improving the survival rate of cancer patients. Cancer treatment methods include surgery, radiotherapy, immunotherapy, chemotherapy, and targeted therapy. 3 Treatment programs are usually tailored to the patient's specific situation, either by applying a certain treatment alone or by combining several methods in a comprehensive treatment. The complexity, diversity, and dynamics of cancer pose many difficulties for research and treatment. 4 Traditional treatments such as surgical resection, chemotherapy, or radiotherapy may not achieve the expected efficacy due to tumor recurrence and drug dependence. 5 Chemotherapeutic drugs greatly limit their clinical application because of drawbacks such as poor solubility, weak targeting ability to the tumor site, and severe adverse reactions. 6 Due to the difficulty of treating cancer and inflammatory diseases using traditional methods, more effective treatments are urgently needed.7–9
Nanotechnology shows great potential in the diagnostic and therapeutic fields of different diseases10,11 and is extensively used for drug delivery. It offers promising solutions to overcome the limitations of current therapeutic approaches. By encapsulating drugs in nanocarriers, the solubility, stability, and bioavailability of drugs can be improved, thereby increasing their efficacy and reducing their side effects. 12 Nanodrug delivery systems can also be used to achieve targeted drug release by adjusting their size, shape, and surface modifications, enabling more precise targeting of drugs to tumor cells or inflammatory sites. 13 However, for tumor therapy, an ideal nanodrug delivery system should also have properties such as prolonged drug blood circulation and tumor tissue targeting to achieve better antitumor efficacy. 14 Although tumor targeting can be achieved by modification of the nanocarrier surface, the nanodrug delivery system, as a foreign substance, is recognized and cleared by the immune system in the body. The outcome is a shorter in vivo circulation time and difficulty in achieving effective drug concentration at the target site. 15 Therefore, the solution of the biocompatibility problem of nanocarriers has become a research hotspot in recent years. In 2011, Zhang's team pioneered the development of a new biofilm biomimetic nanodrug delivery system, 16 setting a precedent in the field of biomimetic nanodrug delivery systems. This biomimetic nanodrug delivery system can encapsulate drugs or other substances in nanoparticles (NPs) and uses red blood cell (RBC) membranes as the surface encapsulation material. Erythrocyte membrane coating improves the stability of the delivery system and endows it with surface antigens consistent with erythrocytes. This system can facilitate their escape from macrophage recognition and clearance, possess long-circulating properties, and enrich tumor tissues through passive targeting effects. This research result provides new ideas and methods for developing the field of nanomedicine. Meanwhile, the study of biofilms originating from various natural biomolecules such as cells, exosomes, and bacteria is gradually expanding, 17 as shown in Figure 1. The criteria for biofilm selection primarily depend on the unique characteristics of different cells and the need for disease treatment. 18 On this basis, the technology developed for biofilm nanodrug-delivery systems has been extended to incorporate cell membranes from different types of cells to carry composite biofilms with the properties of both source cells, which can simultaneously amplify the original advantages of both cells. Composite biofilm biomimetic nanodrug delivery systems provide extended in vivo circulation, reduced immunogenicity, good biocompatibility, 19 high efficiency and safety, and greatly reduced immunotoxicity. 20 These features are due to the protection of biological components and the utilization of synthetic nanomaterials and natural materials, which provide a good platform for precise drug delivery, targeted therapy, and molecular imaging, 20 ,21as shown in Figure 2. In this study, the existing composite biofilm biomimetic nanodrug delivery systems are classified and summarized to provide references and lessons for subsequent studies.

Cell membranes from different types of natural cells were extracted. The figure was created using Biorender.com.

Composite biofilm biomimetic nanodrug delivery systems for cancer treatment. The figure was created using Biorender.com.
Preparation and Identification of a Biomimetic Nanodrug Delivery System Based on a Composite Biofilm
Preparation Methods for Composite Biofilm NPs
First, hypotonic lysis, 22 ultrasound, 23 or repeated freezing and thawing 24 are usually performed to obtain the cell membrane. Among them, repeated freezing and thawing is the most commonly used method, which has the advantage of not affecting cell membrane surface antigens. 25 Then, composite biofilms can be successfully prepared by the extrusion of different cell membranes, and extrusion and sonication can also be used to fabricate composite biofilms. Finally, the composite biofilm and NPs were extruded repeatedly by an extruder several times to obtain the composite biofilm NPs. Currently, some researchers use a simpler ultrasonic method to prepare composite biofilm NPs.26–28 In addition, ultrasonic analysis combined with extrusion and microfluidic electroporation technology was conducted to prepare composite biofilm NPs.29,30
Characterization and Identification of the Composite Biofilm NPs
The physicochemical structure and surface proteins of composite biofilm NPs play a crucial role in their function. 31 After the successful preparation of composite biofilm NPs, it is usually necessary to verify the physical and chemical properties and integrity of the surface proteins of the cell membranes to ensure the rationality and effectiveness of the membrane coating process design. Transmission electron microscopy and scanning electron microscopy can be used to observe the morphology of the composite biofilm NPs. In addition, dynamic light scattering and zeta potential were used to characterize the particle size and zeta potential of the cell membrane-coated NPs. The characterization data provide strong physical evidence for the successful encapsulation of composite biofilm NPs. 32 In addition, protein bands were detected by sodium dodecyl sulfate-polyacrylamide gel electrophoresis to verify the integrity of the protein on the membrane surface. At the same time, Western blotting was used to analyze the characteristic proteins on the cell membrane. Recently, fluorescent dyes have also been used to verify the successful preparation of composite biofilm NPs. Fan et al 33 used fluorescent dyes to label erythrocyte membranes and performed a visual analysis of membrane-coated NPs by laser scanning confocal microscopy. In addition, UV-vis absorption spectroscopy can also be used to characterize membrane biomimetic NPs.34,35 Ren et al 36 reported a type of magnetic NP coated with an erythrocyte membrane, and its new absorption peak at 400 nm was consistent with the characteristic absorption peak of erythrocyte membrane vesicles, indicating that the membrane coating process had no significant effect on the physicochemical characteristics of the membrane. Fusion of multiple cell membranes can be confirmed using the Forster resonance energy transfer strategy. Different cell membrane fusions were labeled with different fluorescent dyes, and the colocalization of fluorescent signals was observed under a confocal microscope. The unfused ones showed distinct monochromatic respective colors, further proving that multiple biofilms were retained on the NPs.37,38
Noncancer Cell–Noncancer Cell Composite Biofilm Biomimetic Nanodrug Delivery System
Erythrocyte–Platelet Composite Biofilm Biomimetic Nanodrug Delivery System
Cell membrane-based nanosystems with desirable properties have been extensively studied for many therapeutic applications. However, current research has focused on single-cell membranes, whereas multifunctional composite membrane materials from different cell membrane types are rare. RBC membranes are initially used to prepare composite membranes because of their biocompatibility and ability to escape by immune clearance. Although RBC membranes can prolong circulation time, the addition of targeted ligands can help improve localization to desired targets, such as tumors. Platelets, like RBCs, are an important component of mammalian blood. 39 Platelets play an important role in the fight against tumor metastasis because of the presence of associated antigens and functional proteins on platelets, which are associated with immune defense and targeting the damaged vascular system, as well as responding to invasive microorganisms. 40 Platelet membranes, such as RBC membranes, can prolong the blood circulation time of nanocarriers and evade immune clearance. 41 Because of the presence of specific target-recognition proteins on the membranes, platelet membrane-modified nanocarriers can be significantly uptake by tumor cells with active targeting ability. In turn, the enrichment of drugs in the tumor increases.42,43 Bionic nanocarriers based on platelet membranes have the advantages of good biocompatibility, low immunogenicity, and enhanced targeting.44,45 Zhang et al 46 reported that RBC membrane and platelet-membrane-coated NPs retain the surface membrane proteins from both cells, thereby combining the functions of 2 different cell types (Figure 3). Using a Förster resonance energy-transfer strategy, RBC membranes are confirmed to fuze with platelet membranes. Dual-membrane-encapsulated NPs also exhibit prolonged circulation and good biodistribution in a mouse model. 47 The composite membrane-coated NPs exhibit a combination of the physicochemical properties of the 2 single membrane-coated NPs compared with the RBC-coated and platelet-coated NPs. It can effectively avoid the metabolism and excretion of drugs, prolong the residence time of drugs in the body, improve the bioavailability of drugs, and enhance the efficacy of drugs. 48 This discovery brings new ideas and methods to the field of drug delivery, expands the research field of drug delivery, and provides new ideas for the design and development of novel drug delivery systems, which can be used to overcome the limitations of current NP-based therapy and imaging platforms. This approach also can be used to combine functions in different areas, including targeted drug delivery, immune regulation, and detoxification. 49

Fabrication of RBC-platelet hybrid membrane-coated nanoparticles (denoted [RBC-P]NPs). Schematic of membrane fusion and coating. The membrane material is derived from both RBCs and platelets and then fused. The resulting fused membrane was used to coat poly(lactic-co-glycolic acid) polymeric cores to produce [RBC-P]NPs. (Copyright © 2017 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim).
Platelet-White Blood Cell Composite Biofilm Biomimetic Nanodrug Delivery System
When the body undergoes an inflammatory response or when a tumor is generated, many immune cells, such as WBCs, are produced. WBCs are primarily divided into macrophages, granulocytes, and lymphocytes. Their main function is to play a defensive role for the organism, recognizing bacteria and viruses of extracorporeal origin, etc, and responding with an immune response to abnormal cells and tissues, which can recognize inflammation and accumulate in the area of lesions. 50 Therefore, the study of WBC membrane bionic drug-delivery systems for targeted drug delivery has gradually become a research hotspot. Liu et al 51 successfully developed a therapeutic biomimetic platform modified with specific antibodies (Figure 4). They fused platelet and WBC membranes and coated them with magnetic beads. They then modified their surfaces with specific antibodies for efficient and highly specific isolation of circulating tumor cells (CTCs). The prepared platelet–WBC composite membrane-coated immunomagnetic beads (HM-IMBs) have enhanced binding to cancer cells (CCs) because of the platelet membranes. The presence of WBC membranes reduces the interaction with homologous WBCs. The cell separation efficiency of HM-IMBs increased from 66.68% to 91.77% and cell purity increased from 66.53% to 96.98% compared with commercial magnetic beads. This novel composite membrane coating strategy is expected to be open new possibilities for overcoming the limitations of current therapeutic diagnostics. Die et al 52 designed gelatin NPs loaded with pioglitazone and vitamin E, which were then modified with platelet and neutrophil complex membranes. Inheriting the properties of naturally derived cells with surface markers containing many signaling factors, immune-evasion ability, and dual inflammatory enrichment due to specific surface adhesion molecules, NPs can enhance their targeting of inflammatory sites and are enriched in liver tissue. The high expression of matrix metalloproteinase-9 at the site of nonalcoholic steatohepatitis enables gelatin NPs to respond intelligently to degradation and then release vitamin E and pioglitazone for drug treatment. Taken together, this novel composite membrane-coated nanoplatform can enhance nonalcoholic fatty liver disease therapy in the complex liver microenvironment and may provide the basis for future translational research. It provides new technical support and methods for drug development and clinical application, improves the efficacy and safety of drugs, and promotes drug development and clinical transformation.
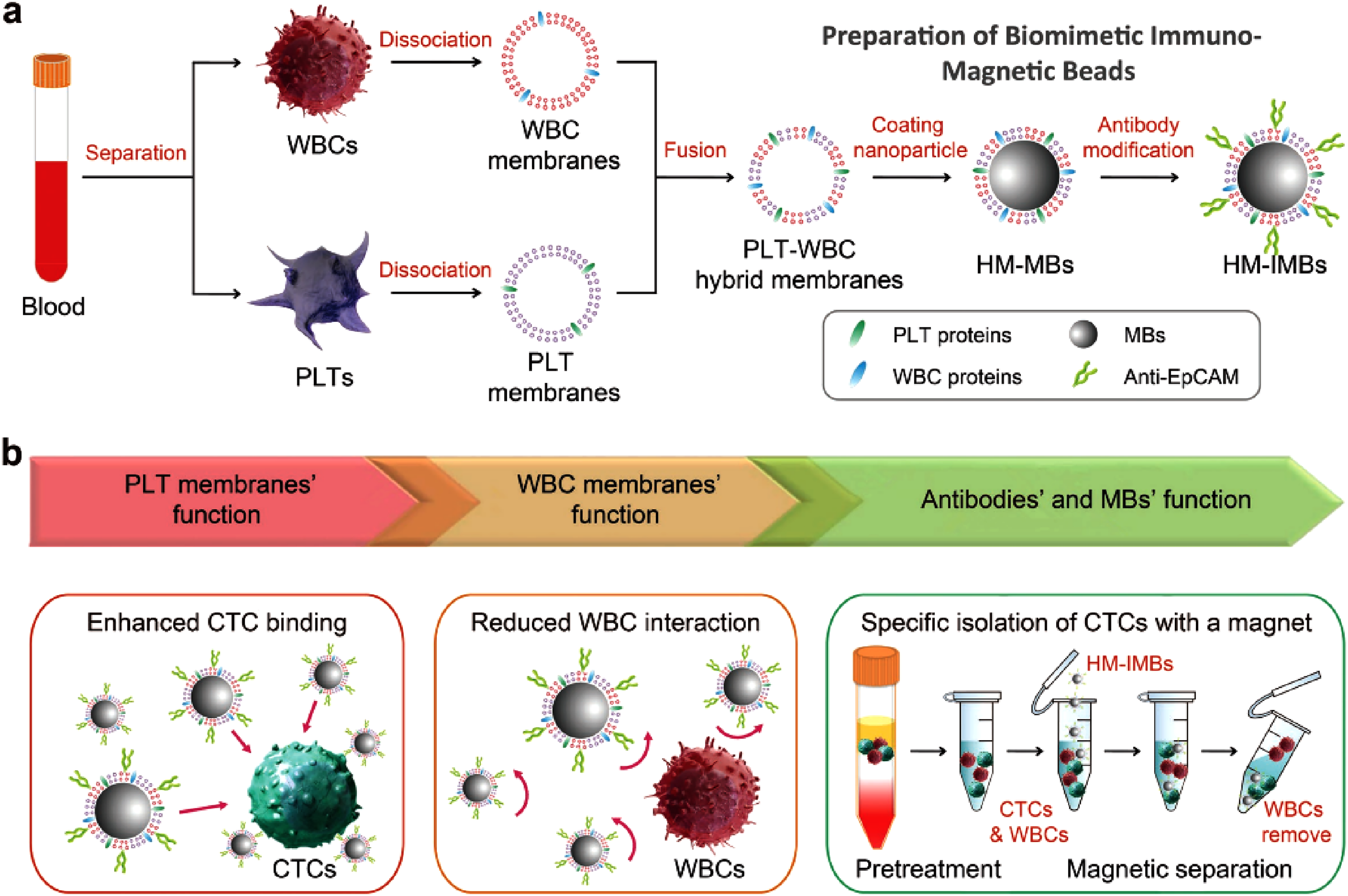
Schematic of the preparation of HM-IMBs for high-performance isolation of CTCs. (a) WBC and PLT membranes, along with their associated proteins, were independently separated from blood samples, fused, and coated onto MBs. Then, the resulting PLT–WBC HM-coated MBs were surface-modified with antibodies to form HM-coated immuno-MBs. (b) HM-IMBs inherited enhanced CTC binding from PLTs and the property of reduced interaction with homologous WBCs from WBCs were used for high-efficiency and high-purity isolation of CTCs.(Copyright© 2018 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim).
The noncancer cell membrane–noncancer cell membrane (NCM/NCM) composite biofilm biomimetic nanodrug delivery system shows high biocompatibility and long circulation time in vivo. 53 It enhances accumulation in the tumor region, but it lacks targeting.54,55 To address this issue and achieve personalized drug delivery and precision treatment according to the characteristics of the patient's disease, researchers have extracted CC membranes to further achieve modification.56,57
NCM/CM Composite Biofilm Biomimetic Nanodrug Delivery System
Erythrocyte–Cancer Cell Composite Biofilm Biomimetic Delivery System
CC membrane-encapsulated nanodrug delivery systems exhibit good self-recognition and internalization through parental CC types. They have the properties of prolonged circulation time and improved selective drug accumulation for tumor therapy. 58 Moreover, the presence of target recognition molecules on CC membranes allows for the precise treatment of tumors. 59 However, the formation of CC membranes may lead to disadvantages such as loss of membrane protein integrity and incomplete immune surveillance evasion. 60 Thus, by fuzing erythrocytes and CCs to form a composite biofilm, researchers have found that the above problems can be solved, whereas the original advantages of both cells can be amplified. Wang et al 61 fused RBC and melanoma cell (B16-F10) membranes to form a composite membrane biomimetic delivery system (RBC-B16) (Figure 5). They used a composite membrane modified with hollow copper sulfide NPs containing doxorubicin (DCuS@[RBC-B16] NPs) for combined chemotherapy/photothermal therapy (PTT) of melanoma. Characterization of DCuS@[RBC-B16] NPs showed that they retained the characteristic features of 2 cell sources. The RBC membrane significantly enhanced immune escape, the self-labeling proteins on the membrane inhibited macrophage phagocytosis, and the B16 membrane enhanced homologous targeting of melanoma. Compared with bare copper sulfide NPs, DCuS@[RBC-B16] NPs exhibit higher specific self-recognition of the B16-F10 cell line in vitro, thereby significantly prolonging the circulating lifespan and enhancing the homologous targeting ability in vivo. In vitro results showed that more than 94.5% of B16-F10 cells treated with DCuS@[RBC-B16] combined with near-infrared (NIR) irradiation died, and approximately 65% of B16-F10 cells treated with DOX combined with NIR irradiation. Synergistic photothermal/chemotherapeutic treatment can effectively inhibit tumor growth of melanoma with an inhibition rate of approximately 100%. Taken together, the fusion of membrane-coated biomimetic NPs from multiple cell types contributes to the personalized treatment of different tumors. Zhao et al 62 designed a cell membrane biomimetic nanodrug delivery system (RBC-H@DOX/3-HF@MSN) for efficient combination therapy for cancer. This system incorporates composite membrane-camouflaged mesoporous silica NPs of HepG2 cells and RBCs and is coloaded with the chemotherapeutic drug DOX and photoCORM 3-hydroflavone (3-HF). The composite membrane camouflage confers immune evasion ability to this delivery vehicle, thereby prolonging the blood-circulation delivery time (from RBCs) and the homologous tumor-targeted delivery (from HepG2 cell membranes). 3-HF is an ideal candidate for nongaseous carbon monoxide (CO) releasing molecules that can control CO release with precise spatial and temporal control under light, enabling combined CO gas therapy and CO-sensitizing chemotherapy, with high anticancer efficiency and low side effects on normal cells. This system increased DOX therapeutic efficacy from 29.0% to 82.4%. Similarly, Han et al 63 fused RBC and tumor cell membranes, which can effectively reach the spleen and activate T cell immune responses in vivo. In a mouse B16F10 tumor model, combined immune checkpoint blockade therapy significantly inhibited melanoma recurrence and metastasis after surgery. Yang et al 64 fused RBC membranes with MCF-7 cell membranes, and in vitro cellular uptake analysis showed that RBC-M exhibited significant targeting of MCF-7 compared with the other 3 cell lines. This finding is attributed to the fact that the RBC-M composite cell membrane shell contains MCF-7 cell membrane fragments, which contain homologous self-recognition biomarkers, such as n-calmodulin, galactoglucan lectin-3, and epithelial cell adhesion molecule (EpCAM). The RBC-M NP platform possesses excellent isotype targeting, long cycling, good biocompatibility, controllability, and photothermal properties, making it an ideal prospect for clinical applications. Erythrocyte–CC composite biofilms have attracted considerable attention for their extended circulation time, immune escape mechanisms, and homologous targeting properties. Because of specific proteins and other properties inherited from the source cells, biomimetic nanosystems derived from different types of cell membranes can perform increasingly complex tasks in dynamic biological environments. This biomimetic delivery system combines the characteristics of RBCs and CCs and takes advantage of the compatibility and targeting of biofilms to achieve precise drug delivery. This innovative drug delivery technology brings new ideas and methods to the field of drug delivery and is expected to promote its development. This provides a new way and the possibility for the realization of personalized treatment, which helps to better meet the treatment needs of patients. 65

(A) Schematic of membrane fusion and coating. Membrane materials were derived from RBCs and B16-F10 cells and fused. The resulting hybrid membrane was used to camouflage DOX-loaded hollow copper sulfide nanoparticles (DCuS NPs) to produce DCuS@[RBC-B16] NPs. (B) Synergistic photothermal/chemotherapy of melanoma. (C) Photographs of melanoma-bearing mice and (D) tumors for each tested group (1: NS, 2: CuS@[RBC-B16], 3: DOX, 4: NIR laser (1064 nm, 1 W/cm2), 5: DCuS@[RBC-B16], 6: CuS@[RBC-B16] with NIR laser (1064 nm, 1 W/cm2), 7: DCuS@[RBC-B16] with NIR laser (1064 nm, 1 W/cm2). (E) Relative tumor volume and (F) Relative body weight of melanoma-bearing mice that received different treatments. (Copyright © 2018, American Chemical Society).
Dendritic Cell–Cancer Cell Composite Membrane Bionic Delivery System
Dendritic cells (DCs) play a key role in inducing the immune response of T cells against pathogens and malignant cells.66,67 Because of the major histocompatibility complex (pMHC) of antigenic peptides on DC membranes, they contribute to the uptake, processing, and presentation of tumor antigens. 68 Thus, DCs and CCs form a composite membrane bionic delivery system that can be combined with the immune response to treat cancer more effectively and precisely. For example, Hao et al 69 fused glioma cell membranes with DC membranes as a delivery vehicle for chemoimmunotherapy (Figure 6). This delivery system can target homologous CCs through the homologous targeting mechanism of glioma cell membranes. In contrast, an immune response is achieved based on the specialized antigen-presenting properties of DC membranes, which carry abundant and intact tumor cell membrane antigens and promote cell membrane-bound tumor antigen uptake for efficient presentation and downstream immunity, thereby significantly improving the therapeutic efficacy of glioma treatment. The growth of glioma in the preparation group with fusion of glioma cell membrane and DC membrane was significantly inhibited, indicating that because of the homotypic targeting ability of glioma cell membrane, the preparation can be accurately delivered to the brain tumor site and more effectively inhibit the rapid growth of glioma. The antitumor efficacy of the preparation group of glioma cell membrane fused with DC membrane was better than that of single cell membrane, which may be due to the immune response induced by DC membrane. The combination of drug delivery and antigen delivery has resulted in better chemoimmunotherapy efficacy in gliomas. Liu et al 70 designed a cancer-specific vaccine that was dependent on DC and 4T1 cell membranes. This composite biofilm maintains DC- and CC-associated functional proteins, such as pMHC and costimulatory molecules, conferring various biological capabilities, including lymph node homing and antigen-lifting ability. Thus, this delivery system directly activates T cells and stimulates DC maturation and the subsequent generation of the T cell immune response. Thus, a robust antitumor immune response is observed. In contrast, pretreatment with membrane-coated nanovaccines extended tumor-free time to varying degrees. At 36 days, the tumor-free rate of the composite membrane group was as high as 60%, whereas that of the 4TI and DC membrane groups was 20% and 0%, respectively. In vitro and in vivo studies confirm that the composite biofilm biomimetic drug delivery system can serve as a vaccine against tumor growth. Moreover, Xu et al 71 constructed a polymeric multicellular nanoconjugate for synergistic NIR-II photothermal immunotherapy. It comprises a NIR-II-absorbing polymer acting as a photothermal core, which is camouflaged as a cancer vaccine shell with composite membranes originating from CCs and DCs for synergistic photothermal immunotherapy. In mice irradiated with NIR-II light, primary tumors are gradually ablated, and antitumor T-cell immunity is further promoted. This combination therapy also generates immune memory for long-term immune surveillance. This combination of photothermal and immunotherapy provides a new immunotherapy strategy for tumor treatment. It provides a potential research method for designing tumor treatment nanocellulators with better therapeutic effects and biocompatibility by combining nanomaterials with different functional components.

Schematic illustration of the effects of biomimetic nanosuspensions directly and indirectly, including homologous targeting chemotherapeutics for direct treatment of glioma and enhanced immunotherapy that kills tumors indirectly by activating the body's immune system to maximize synergistic chemo/immunotherapy (Copyright©2021 Springer Nature).
Bacterial–Cancer Cell Composite Biofilm Biomimetic Nanodrug Delivery System
Bacteria have natural adhesion proteins, antigens, or specific molecules on the surface of their protein shells that have a targeting effect on tumors and can enhance the immune function of autologous tumor-antigen immunogenicity. For example, Chen et al 72 constructed a combined eukaryotic–prokaryotic vesicle-based therapeutic nanoplatform by fuzing melanoma cell membrane vesicles and attenuated Salmonella outer membrane vesicles (OMVs). Melanoma cell membrane vesicles provide tumor antigens, and attenuated Salmonella OMVs act as natural adjuvants. The 2 vesicles serve as shell carriers that cover the photothermal nuclei. In the nanovaccine and laser therapy group, a 96.9% maturation ratio of bone marrow-derived DCs, a significant increase in the secretion levels of TNF-α and interleukin 12 by 18% and 56%, respectively, and a high rate of T-cell proliferation (80.7%) were observed. These findings indicate a synergistic antitumor effect as a therapeutic vaccine. Ni et al 73 developed a cotransported NP vaccine that fuzes tumor cell membranes with the cytoplasmic membrane of the bacterium Escherichia coli, which acts as an exogenous adjuvant. The bacterial membrane fragment can be rapidly recognized by the immune system, leading to DC maturation and T cell initiation. In multiple mouse cancer models, the vaccine induces a robust tumor-specific immune response, effectively inhibits tumor recurrence, induces tumor regression, and prolongs postoperative survival. Wang et al 74 reported an OMV-CC hybrid membrane comprising a bacterial OMV and a B16-F10 CC membrane (Figure 7). It is used to modify hollow polydopamine (HPDA) NPs to take advantage of OMV immunotherapy and HPDA-mediated PTT and thus improve antitumor efficacy against melanomas in a mouse model. HPDA@[OMV-CC] NPs target melanoma and activate the immune response by stimulating the maturation of DCs in mouse lymph nodes. Their results show that the antitumor immune response and PTT mutually enhance the therapeutic ability and eradicate melanoma without significant adverse effects. This therapeutic strategy can not only directly attack melanoma cells but also enhance the immune response by activating the immune system, which provides a new therapeutic strategy for the treatment of melanoma and other tumors and is expected to achieve important breakthroughs in clinical practice.

(A) schematic of the membrane derived from OMV and CC fusion and the resulting fused membrane camouflaged with HPDA NPs to produce HPDA@[OMV-CC]NPs. (b) Synergistic Photothermal/Immunotherapy of Melanoma Copyright © 2020, American Chemical Society.
Platelet–Cancer Cell Composite Biofilm Biomimetic Nanodrug Delivery System
This system uses the specific adhesion and recognition ability of platelets and CCs to wrap drug particles on the biofilm of platelets, resulting in better biocompatibility and targeting. It can effectively deliver drugs into the interior of CCs, reduce damage to normal cells, and improve drug efficacy and safety. Sun et al 75 proposed for the first time the use of platelet-cancer stem cell (CSC) composite membrane-coated iron oxide magnetic NPs (MN) ([CSC-P]MN) for the enhanced PTT of head and neck squamous cell carcinoma (HNSCC) (Figure 8). Platelet membranes inherit the properties of primitive cells and have immune-evasion ability because of the large amount of signaling factors contained on their surface. Conversely, CSC membranes have homologous targeting ability because of specific surface-adhesion molecules. Compared with single cell membrane-encapsulated MNs, [CSC-P]MNs prolong the cycling time and enhance the targeting ability. [CSC-P]MN also has superior photothermal ability and can effectively inhibit HNSCC tumor growth. Overall, this composite biofilm biomimetic nanoplatform may enhance the antitumor effect in the complex tumor microenvironment, which can provide new ideas and methods for personalized and precise treatment. It is favored in the targeted therapy of tumors. It also provides new directions and ideas for research in the fields of nanomedicine and biomedicine.

Fabrication of [CSC-P]MNs and their application in cancer therapeutics. The membrane material is derived from both CSCs and platelets and then fused. The resulting fused membrane is further coated onto Fe3O4 MN cores to produce [CSC-P]MNs. Subsequently, the resulting [CSC-P]MNs were injected intravenous (i.v.) back into the donor mice. After systematic circulation and because of the homotypic targeted effect and high concentration of aggregation of inflammatory factors from both cell membranes, [CSC-P]MNs were enriched in the tumor site. To exploit the magnetic properties and optical absorption ability of MNs, biomimetic [CSC-P]MNs were also used for enhanced in vivo tumor magnetic resonance imaging (MRI) and photothermal therapy. Copyright© 2019 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
WBC–CC Composite Biofilm Biomimetic Nanodrug Delivery Systems
CTCs play a key role in tumor metastasis and progression, and capturing and detecting CTCs from blood samples with high purity and sensitivity remains a major challenge. Ding et al 76 developed a nanoplatform for the efficient separation and ultrasensitive detection of CTCs by combining Ag2S nanodots functionalized with NIR multivalent aptamers with magnetic NPs in WBC and tumor cell membranes. The nanoplatform enhances the antiinterference and binding ability of CTCs, resulting in sensitive recognition, efficient capture, easy separation, and sensitive detection of CTCs. The nanoplatform has been successfully applied to detect CTCs in blood samples. It provides a new nanoplatform for the efficient isolation and ultrasensitive detection of CTCs, solves the limitations of existing technologies, and brings important progress and enlightenment to the field of tumor diagnosis, treatment, and research. Gong et al 77 prepared composite membrane-modified adriamycin-loaded poly (lactic acid ethanolic acid) by fuzing macrophage and 4T1 cell membranes for the treatment of lung metastases caused by breast cancer (Figure 9). This study demonstrates that composite membranes derived from macrophages and CCs have the advantages of massive accumulation at sites of inflammation, specific targeting for metastasis, and homologous tumor-targeting ability in vitro. These membranes can significantly enhance the multitargeting ability in an in vivo lung metastasis model. These results demonstrate the potential advantages of composite membranes derived from macrophages and CCs in tumor therapy and provide important ideas for the use of composite membranes derived from macrophages and CCs as a new strategy for tumor therapy. By combining membrane fusion technologies from different cell sources, nanodrug delivery systems with better targeting and therapeutic effects can be designed, providing a new approach to the treatment of tumors such as breast cancer.

Formation and release of RAW-4T1 hybrid membrane coated with doxorubicin (Dox)-loaded PLGA nanoparticles (DPLGA@[RAW-4T1] NPs) Copyright © 2020 Springer Nature.
NCM-cancer cell membrane (NCM/CM) composite biofilm biomimetic nanodrug-delivery systems show superior properties over single membrane-encapsulated NPs in immune evasion, active tumor targeting, magnetic resonance imaging, personalized treatment of different tumors, and PTT therapy. 78 It can improve the efficiency of drug delivery, expand the scope of treatment, reduce drug toxicity, and promote the progress of biofilm research. This provides new enlightenment and the possibility for the innovative development of drug delivery technology, improvement of therapeutic effect, and in-depth discussion in the field of biofilm research. 79 However, intact cell membranes on the surface of NPs may prevent the release of drugs. Moreover, whether the tumor cell membrane retains carcinogenicity and other biological safety and mechanisms of action are not very clear.
At present, all kinds of composite biofilm bionic research and development and the application of nanodrug-delivery systems have become the focus of tumor diagnosis and treatment. Composite biofilms can combine the characteristics of different types of cell membranes to form multifunctional delivery systems, which can improve the targeted delivery efficiency of drugs. 80 At the same time, because of the characteristics of different types of cell membranes, it can be applied to the treatment of a variety of diseases, expanding the scope of treatment and providing new possibilities for the treatment of a variety of diseases. 81 This provides new enlightenment and the possibility for the innovative development of drug delivery technology, improvement of therapeutic effect, and in-depth discussion in the field of biofilm research. 82 In the future, we can further explore the interaction and synergistic effects of different types of cell membranes, reveal more mysteries and mechanisms of biofilms, and promote the in-depth development of biofilm research. A summary of all types of composite biofilm bionic NP delivery systems and key literature are shown in Tables 1 and 2. However, some challenges and problems must be faced. First, the extraction and separation process of the biofilm is not mature enough. Many extraction methods have been proposed, but the extraction and purification processes are complicated, with cumbersome routes, inconsistent standards, and poor reproducibility. Thus, achieving large-scale preparation is difficult, and pollution and impurities emerge. Developing a cell membrane preparation method that saves time and labor and has high product purity is still an urgent problem to be solved. 83 Second, the characterization methods are limited, and the success of membrane encapsulation is verified only by particle-size detection and morphological observation. Protein-blotting analysis can only demonstrate whether the surface composition of the composite biofilm mimetic nano is similar to that of the source cell membrane, but it fails to verify whether the membrane is partially disrupted after membrane encapsulation. Whether the stability and functionality of membrane proteins are affected also remains unclear; therefore, identifying a suitable method of maintaining their integrity and activity is necessary. 84 Third is the issue of biofilm safety. Although numerous studies have proven that biofilm biomimetic nanomedicine has low immunogenicity, validation experiments have been conducted only in animals. For humans, biofilm biomimetic nanomedicine remains a foreign substance, and whether any adverse reactions occur in the long term remains unknown. Meanwhile, whether biofilm introduces a heat source and viruses when wrapping NPs also needs to be considered. Fourth, whether tumor cell membranes retain oncogenic factors need to be investigated. Many problems need to be solved from the laboratory to the clinic, but the natural advantages and application potential of composite biofilm biomimetic nanodrug-delivery systems are still undeniable. 85 Our findings provide a new perspective for the design and application of nanodrugs, which can become a breakthrough point for researchers as well as a new direction for tumor treatment. With the deepening of research, more diseases will be gradually overcome with the development of new drugs.
Types of Common Composite Biofilm Biomimetic Nanoformulations and Their Advantages and Disadvantages.

Abbreviations: WBC, white blood cell; CTC, circulating tumor cell.
Summary of the key Literature on Biomimetic Nanodelivery Systems for Composite Biofilms.

Abbreviations: WBC, white blood cell; CTC, circulating tumor cell.
Prospect and Discussion
Composite biofilm biomimetic technology has made significant progress in recent years, bringing new directions for the treatment of many diseases. It has great significance in the fields of drug delivery, disease mechanism research, and biosensors. 86 In biomedical research, composite biofilms can better simulate the complex structure and function of biological cell membranes. These biofilms can help to study the biological properties of cell membranes, signaling mechanisms, and drug interactions, as well as provide more accurate models and targets for disease treatment and drug development. In terms of drug delivery and targeted therapy, the study of composite biofilms can help design and develop more effective drug-delivery systems. 87 It can realize targeted delivery of drugs by simulating the characteristics of different cell membranes and improving the bioavailability and therapeutic effects of drugs. In particular, in the field of tumor-targeting therapy, composite biofilm-mimicking technology has advantages that traditional drugs cannot match, especially in retaining the functions and characteristics of the source cell membrane. It has low immunogenicity, high targeting, good biocompatibility, and the ability to avoid being cleared by the immune system, thereby greatly extending the blood circulation time in the body. 88 In addition, composite biofilms can be used to construct biosensors and biomimetic diagnostic technologies, which can realize the highly sensitive detection of biomolecules by mimicking the characteristics of cell membranes and provide a more reliable method for the early diagnosis and treatment of diseases. In the field of bioenergy and environmental protection, by using the characteristics of composite cell-membrane mimicking biofilm, bioenergy conversion technology and biofilm pollution treatment technology can be developed to provide new solutions in the field of renewable energy and environmental protection. 89 The application of biofilm in more fields awaits researchers to explore and discover.
Conclusions
This article reviews the progress of several different types of composite biofilm biomimetic nanodrug delivery systems in cancer therapy. Various cell membranes have naturally unique functions and can be applied in a wide range of fields. With the continuous progress of technology and in-depth research, we believe that composite biofilm biomimetic nanodrug-delivery systems will realize precisely targeted therapy for many types of diseases in the future and will also be more extensively applied in other fields with great potential value.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This is a review article therefore, an ethics statement is not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Changzhou Social Development Project, Social Development Project of Jiangsu Provincial Key Research & Development Plan, Jiangsu Provincial Medical Key Discipline Construction Unit (Oncology Therapeutics [Radiotherapy]), Natural Science Foundation of Jiangsu Province, and General Project of Jiangsu Provincial Health Commission (Grant Nos. CE20235063, BE2022720, JSDW202237, BK20231190, and M2020006).
