Abstract
Anaplastic thyroid cancer (ATC) is one of the most lethal human malignancies, and it is rapidly falling without any effective therapeutic options. Although radical surgery, radiotherapy, and chemotherapy are performed on patients, the curative effect is suboptimal, and the survival rate is still poor. The discovery of altered gene pathways involved in this aggressive disease has advanced, but molecular targeted drugs targeting these pathways are still in clinical trials. To date, there is no effective way to treat this disease, so it is particularly urgent to find new treatments. At present, multimodal therapy is gradually being applied in clinical practice, which provides a new possibility for prolonging the survival time and improving the prognosis of anaplastic thyroid carcinoma. In this study, we retrospectively analyzed the current clinical multimodal therapy for patients with anaplastic thyroid cancer to evaluate its effect on improving the survival of patients with anaplastic thyroid cancer at different stages.
Introduction
Anaplastic thyroid carcinoma (ATC) is a rare endocrine malignancy with poor prognosis, accounting for only 1–2% of all thyroid cancers. ATC is a highly invasive tumor. Its tumor cells can infiltrate adjacent thyroid and other tissues, lymph and blood vessels and have strong potential for distant metastasis. 1 Approximately half of anaplastic thyroid cancer patients have distant metastasis, and the lung, bone and brain are the most common metastatic sites. Even with active treatment, only a few patients can survive for a long time, and many patients die in a short time.2,3 Generally, the median survival time from diagnosis to death is only 1–6 months. According to the TNM staging guidelines of the 8th edition of the American Joint Commission on Cancer (AJCC), all ATC patients were stage IV: the primary lesions in stage IVA were limited to the thyroid IV a = T1-T3b, N0/NX, M0); local lymph node involvement can be seen in IVA/IVB stage (IV B: T1-T3a, N1, M0 or T3b-T4, any N, M0); distant metastasis in IVC stage (IV C: any T, any N, M0) 4 the clinical manifestations of ATC are related to the rapidly growing neck mass. When the mass grows beyond the thyroid gland, it can invade the adjacent organs, trachea, esophagus, and recurrent laryngeal nerve and can appear dyspnea, dysphagia, neck pain, and hoarseness.5,6 Approximately 15% of ATC patients show extensive local invasion and distant metastasis through blood and lymphatic channels. For local lesions diagnosed after initial surgical treatment, adjuvant chemotherapy plus radiotherapy is recommended, but the prognosis is still very poor, and the 1-year overall survival rate (OS) is only between 20 and 50%. In addition, undifferentiated thyroid cancer lacks the expression of sodium iodide symporter (NIS), which leads to no response to radioactive iodine 131 treatment or endocrine hormone treatment.6,7 With the development of science and technology, researchers have a deeper understanding of the gene expression and molecular mechanism related to undifferentiated thyroid cancer. Therefore, researchers are actively trying to use molecular drug targeted therapy as a single therapy or as part of multimodal therapy, combined with immunotherapy in clinical trials. 8 Based on the current situation of poor curative effect and low survival rate of undifferentiated thyroid cancer after treatment, a multimode treatment scheme is formed by combining single operation, radiotherapy, chemotherapy, molecular targeted drug therapy, and immunotherapy, which may provide a new direction for the treatment and management of undifferentiated thyroid cancer.
Trimodal of Treatment: Surgery, Radiotherapy, and Chemotherapy
The traditional trimodal treatment includes surgical resection combined with radiotherapy and chemotherapy. 9 The initial surgical method of ATC patients evaluated as resectable lesions depends on many factors. For patients with IVA and IVB ATC who are willing to receive active treatment, the guidelines issued by the American Thyroid Association (ATA) strongly recommend radical surgical resection. For IVC patients who break through the capsule and invade adjacent structures or even transfer to distant places, palliative surgery can be considered to control local diseases and alleviate or prevent possible complications in the future (invasion or obstruction of the airway, esophagus and throat).10–12 Studies have shown that preoperative or postoperative adjuvant therapy combined with external beam radiation therapy (EBRT) and chemotherapy (CTX) may be beneficial to improve the prognosis of ATC patients, especially local recurrence-free survival (LPF). At the same time, considering the local invasiveness of ATC, adjuvant therapy can improve the quality of life of patients. For patients with unresectable diseases, nonsurgical palliative treatment can be provided, that is, EBRT combined with CTX. Chemotherapy is usually used in the treatment of advanced ATC, including doxorubicin, paclitaxel, cisplatin, carboplatin, and other drugs. The clinical activity of doxorubicin was low, and the response rate was 22%. Therefore, more active compounds need to be used in the treatment of ATCs.9,13–15 Although doxorubicin, docetaxel/paclitaxel and cisplatin have been recognized by ATA guidelines in the treatment of ATC, the survival rate of advanced ATC has not improved. 16 Many current clinical trials are trying to apply various combinations of targeted therapy and immunotherapy to the treatment of ATC. 17
Molecular Targeted Drug Therapy
Molecular targeted therapy (MTT) refers to a kind of therapeutic based on tumor molecular biology that regards the specific structural molecules of tumor tissues or cells as targets and uses some antibodies and ligands that can specifically bind to these target molecules to achieve the purpose of direct treatment or targeted treatment. Molecular targeted therapy is the key receptor in the process of tumor development and corrects its pathological process. Because it takes diseased cells as the target, it has a “curative” effect compared with traditional treatment methods such as surgery, radiotherapy, and chemotherapy. Broad molecular targets mainly include cell molecules involved in tumor cell differentiation, cycle change, apoptosis, cell migration, infiltration, lymphatic metastasis, and systemic metastasis from the DNA to protein (enzyme) level. With the discovery of some potential targets for the pathogenesis of ATC, the most commonly mutated genes in ATC include the oncogenes BRAF, NRAS, KRAS, and HRAS, as well as the tumor suppressor genes TP53, NF1, and PTEN. BRAFV600E is the most common mutation gene. Epidermal growth factor receptor (EGFR), histone deacetylases (HDACs), β-catenin, Aurora kinase, cyclin, and platelet-derived growth factor receptor (PDGFR) are also common in ATC cells. Similarly, the RAS/Raf/MEK/ERK and PI3 K/AKT/mTOR pathways are activated in ATC. These findings suggest that there are high gene mutations and abnormal expression of a large number of molecules in ATC, resulting in the imbalance of multiple signaling pathways.13,15,18 For drugs through different molecular pathways, see Figure 1, mechanism and therapeutic efficacy of different drugs are shown in Table 1. Further randomized clinical trials are also ongoing.
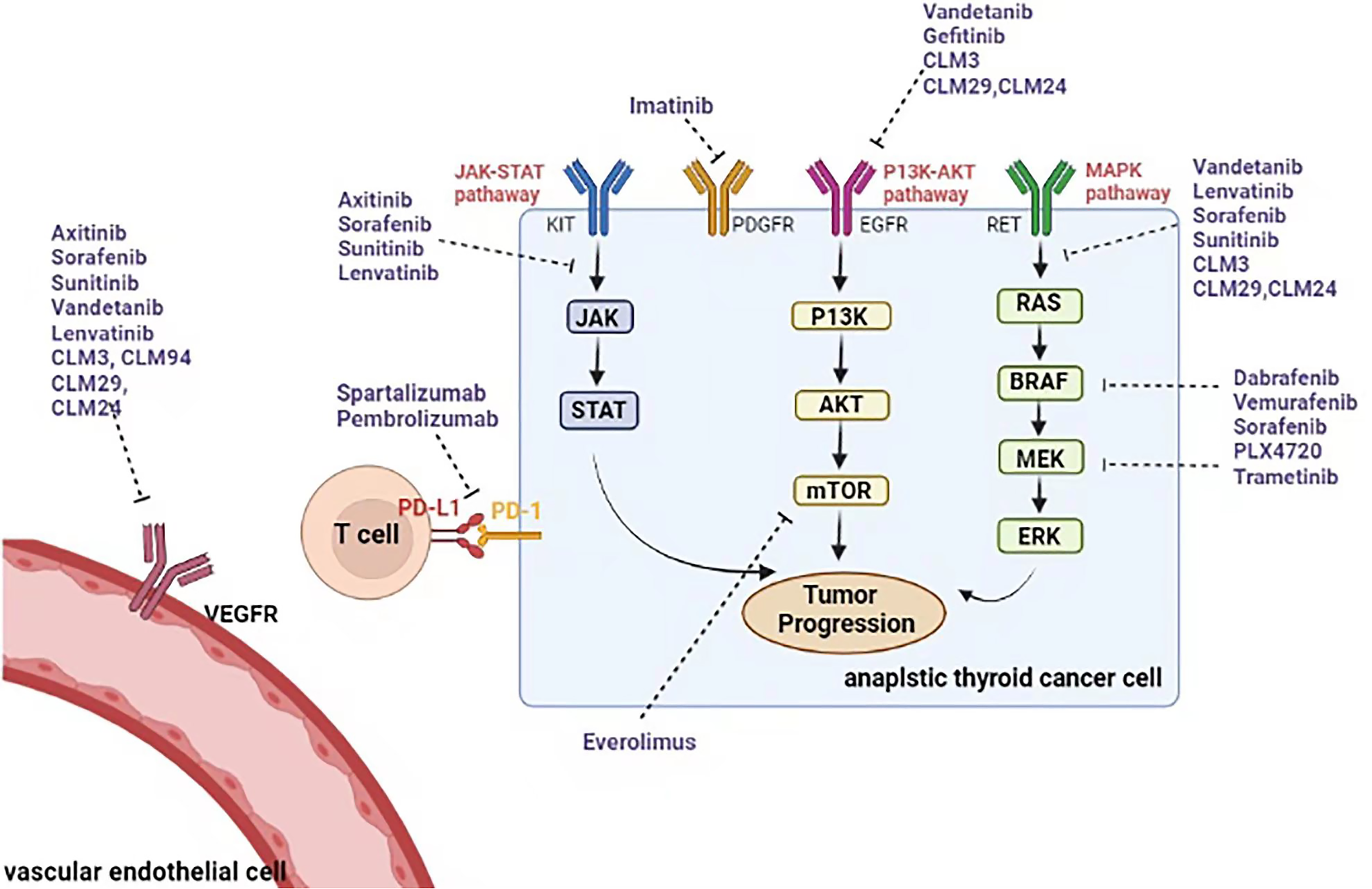
Molecular targets and molecular targeted agents in anaplastic thyroid carcinoma.
Trials on Molecular Target Therapies Used in Anaplastic Thyroid Cancer Treatment

Abbreviations:P, pre-clinical: *; Orthotopic mouse model: **; ATC cell lines; MPFS, median progression free survival; PR, partial response; SD, stable disease; DCR, disease control rate; MOS: median overall survival; m, months; VEGFR, vascular epidermal growth factor receptor; EGFR, epidermal growth factor receptor; uPA, urokinase-type plasminogen activator; PPAR γ, peroxisome proliferator-activated receptor γ; SAHA, suberoylanilide hydroxamic acid.
Tyrosine kinase inhibitors include multiple processes regulating a series of cell growth, survival, migration, and invasion. It has been confirmed that it is overexpressed in a variety of tumor cells, including undifferentiated thyroid cancer cells. It has become a new molecular therapeutic target. gefitinib is a selective EGFR tyrosine kinase inhibitor with high selectivity. It inhibits tumor growth, metastasis and angiogenesis by preventing EGF-stimulated EGFR autophosphorylation and EGFR-mediated downstream signal transduction and increases tumor cell apoptosis. It is the first EGFR tyrosine kinase inhibitor approved by the FDA. In vitro, gefitinib was shown to inhibit the growth of thyroid cancer cells. 19 Sunitinib is a new multitarget oral tyrosine kinase inhibitor that mainly acts on vascular epidermal growth factor receptor (VEGFR) 1 and 2 and platelet-derived growth factor receptor (PDGFR) α,β,c-KIT and RET. It can resist tumors by blocking the tumor blood supply and directly attacking tumor cells. 20 Vandetanib is active against the EGFR family, VEGF receptor, RET, protein tyrosine kinase 6 (BRK), tyrosine kinase with immunoglobulin and EGF domain-2 (TIE2), members of the renin (EPH) receptor kinase family and members of the Src tyrosine kinase family. Vandetanib can increase the apoptosis of tumor cells in vitro, reduce the migration and invasion of tumor cells, and reduce the growth of tumors in vivo through its antiangiogenic effect. 21
Proteasome Inhibitors
The proteasome is a kind of ATP-dependent polymer complex that reduces intracellular proteins through ubiquitination. Some key regulators of cell cycle regulation, transcription, growth and apoptosis, such as nuclear factors-κB (NF-κB), p53, c-myc, and JNK, are all derived from the degradation of the proteasome. 22 Bortezomib is a proteasome inhibitor approved by the FDA for the treatment of multiple myeloma. At present, it is widely studied for the treatment of other malignant tumors. In addition, it was also found to have a synergistic effect with the chemotherapeutic drug doxorubicin. 23 Carfilzomib: Mehta Amit et al conducted quantitative high-throughput screening (qHTS) on ATC cell lines (sw-1736, 8505c and c-643) by using the compound library of 3282 drugs. QHTS identified 100 compounds that were active in all three ATC cell lines. According to enrichment analysis, proteasome inhibitors are one of the most effective drugs. Among the three selected proteasome inhibitors, the second-generation proteasome inhibitor carfilzomib is the most active. Carfilzomib can inhibit the proliferation of ATC cells by upregulating the expression of p27 (CDKN1B) and downregulating the expression of the antiapoptotic protein ATF4 to induce G2/M cell cycle arrest and caspase-dependent apoptosis. 24
Histone Deacetylase Inhibitors (HDIs)
Histone deacetylase inhibitors are a class of compounds that interfere with the function of histone deacetylase. It can effectively upregulate the expression of tumor suppressor genes, block tumor growth and induce selective apoptosis of tumor cells. HDACis inhibit tumor cell proliferation and induce cell differentiation and/or apoptosis by increasing the degree of histone acetylation and the expression of p21 and other genes. HDACis have become a research hotspot in tumor-targeted therapy. Its inhibitory effect on tumor cell migration, invasion, and metastasis and antitumor angiogenesis have also been confirmed. It not only has good therapeutic effects on a variety of hematological tumors and solid tumors but also has the advantages of high selectivity and low toxicity to tumor cells. According to their different structures, they can be divided into four categories: (1) fatty acids, such as butyrate, phenyl butyrate and valproic acid; (2) hydroxamate, such as TSA and SAHA; (3) cyclic peptides, such as the natural products phenolic acid peptide FK-288, apicidin and epoxy oxime acid; and (4) benzamides, such as MS-275 and MGCD0103. 25 SAHA (suberoylanilide hydroxamic acid) was approved by the U.S. Food and Drug Administration (FDA) in October 2006. It is the first drug for the treatment of skin T-cell lymphoma that continues, worsens, or relapses during or after other drug treatments. SAHA can nonspecifically bind histone deacetylases (HDAC1, HDAC2, HDAC6) to promote the increase in acetylated histones, resulting in abnormal transcription processes in cancer cells and then inducing cancer cell cycle arrest, differentiation, and even death. 26
Multikinase Inhibitors
The results of clinical trials show that multitarget inhibitors are better than single target inhibitors in the treatment of tumors. Multitarget combined blockade of signal transduction is the development direction of tumor treatment and drug development. Sorafenib is a multikinase inhibitor that can simultaneously inhibit a variety of kinases inside and outside cells, including BRAF kinase, vascular epidermal growth factor 2 and 3 (VEGFR2 and 3) and platelet-derived growth factor-β (PDGFR- β), FCT-3, KIT, and other kinases, showing that sorafenib has dual antitumor effects. On the one hand, it can directly inhibit tumor growth by inhibiting the Raf/MEK/ERK signaling pathway. On the other hand, it can block the formation of tumor neovascularization by inhibiting VEGFR and PDGFR and indirectly inhibit the growth of tumor cells. Imatinib is a TKI that inhibits BCR/ABL, PDGFR, c-fms, ckit, and RET; it has been approved by the FDA for the treatment of chronic myeloid leukemia, GIST, and prominent cutaneous fibrosarcoma. Preclinical studies have shown potential benefits from imatinib treatment of patients because it has proven effective in inhibiting ATC cell lines as a single treatment, as part of combination therapy, together with docetaxel (whose effect is to enhance imatinib inhibition through NF-KB activity) and gefitinib.27–29
BRAFV600E inhibitor
The tumor protein BRAFV600E inhibitor tumor protein BRAF gene is located on chromosome 7. Statistics show that mutation of the BRAF gene can be observed in approximately 25%–38% of ATC patients. PLX4720 is a selective small molecule inhibitor of BRAFV600E and an ATP analog. It blocks the ATP binding site of carcinogenic BRAFV600E by stabilizing the active conformation of the tumor protein BRAFV600E. 30
VEGFR inhibitor
Fosbretubulin is an antiangiogenic compound that is different from the VEGFR kinase inhibitor mentioned above. Its principle of action is to combine with tubulin to destroy the formed microvessels to block the blood supply of the tumor. 31 Axitinib is a potent selective inhibitor of vascular endothelial growth factor receptor (VEGFR-1, - 2, - 3). In addition, it also inhibits platelet-derived growth factor receptor (PDGFR) and c-KIT. However, due to limited clinical data, its role in ATC cannot be accurately evaluated. 32
EGFR inhibitor
The epidermal growth factor receptor inhibitor (EGFR inhibitor) gefitinib is an EGFR inhibitor that has been approved for advanced non-small-cell lung cancer (NSCLC). 33 Cell and experimental studies of ATCs overexpressing EGFR showed that gefitinib had a significant antitumor activity in this tumor. In vitro studies showed that gefitinib could inhibit the proliferation and increase apoptosis of the ATC cell line. In the mouse model, the drug effectively inhibited tumor growth in a dose-dependent manner. A preclinical in vitro study showed that radiation-induced inhibition of ATC cell line proliferation increased before gefitinib: This suggests that combination with gefitinib allows the use of lower doses of ionizing radiation, thereby minimizing radiation toxicity. Gefitinib has also been tested in combination with other drugs. Imatinib combination therapy works well in vivo and in vitro because both molecules have been shown to be effective, but when the two drugs are used in combination, their activity is even greater. Studies have also shown that gefitinib can enhance the toxicity of doxorubicin to ATC cells. A phase II trial was conducted to evaluate the efficacy of gefitinib (250 mg/day) in patients with advanced thyroid cancer. Of the 27 patients with ATC, 5 were enrolled. There was no ORR in the evaluated patients, and one patient had SD = 12 months. 32
mTOR inhibitor
Everolimus is a sirolimus-derived mTOR inhibitor. Constitutive activation of the phosphatidylinositol-3-kinase (PI3 K)/AKT/mTOR pathway has been reported in the pathogenesis of thyroid cancer. Its activity against several thyroid cancer cell lines has been confirmed in vivo and in vitro. In the experimental study, everolimus inhibited the growth of ATC cell lines carrying the PI3 K mutation, making these cells resistant to gefitinib, suggesting that its efficacy may be related to the change in the PI3 K/Akt/mTOR signaling pathway. In a phase II study conducted by Hanna GJ et al, among 7 ATC patients, 1 had a partial response and was progression-free until 17.9 months after study entry, and one had disease stability for 26 months. 34
Molecular Targeted Drugs Combined with Immunotherapy
The T-cell family expresses programmed cell death receptor 1 (PD-1) on its surface, which can interact with ligands in normal tissues. Tumor cells express programmed cell death receptor ligand 1 (PD-L1). In the tumor microenvironment, PD-1 binds to PDL-1 and inhibits the function of T cells through a mechanism called “adaptive resistance.”35,36 PD-1 and PDL-1 play a crucial role in the ability of tumor cells to escape the host immune system. Therefore, in preclinical models, blocking their binding in vitro can enhance the immune response and antitumor activity.13,16 One study revealed the effect of PD-L1 and anti-PD-L1 antibody immunotherapy alone or in combination with BRAF inhibitors on the intratumoral immune response; that is, the combination of PD-L1 antibody and PLX4720 can reduce the tumor volume and increase tumor infiltrating lymphocytes. 37 In addition, the study published by LIN Gui et al reported that a 67-year-old ATC patient had a rapid local recurrence after radical thyroidectomy. Because it was not suitable for reoperation, immunohistochemical analysis and target next-generation sequencing of the resected tumor tissue, targeted treatment could not be carried out, and the patient could not tolerate chemotherapy. Based on the high expression of PD-L1, this research team adopted an optimal strategy. That is, the combination of the antiangiogenic drug anlotinib and the immunotherapy drug sintilimab. The tumor volume of the patient decreased significantly, reaching a continuous remission period of 18.3 months. This is an effective case of antiangiogenic drugs and immunotherapy for ATC, which provides a feasible and novel treatment option for ATC patients. 38
Radiotherapy Combined with Immunotherapy
Radiotherapy is usually used to treat various cancers to achieve local or palliative tumor control. In addition, low-dose or low-dose radiotherapy can regulate tumor cells in a variety of ways, making tumor cells more vulnerable to immune cell reactivity. 39 The combination of radiotherapy and immunotherapy has been shown to have synergistic effects on preclinical models and cancer in selected patients by regulating immunity in the microenvironment. Experiments have proven that immune checkpoint inhibitors against PD-1 or PD-L1/2 alone or in combination with chemotherapy are effective in a variety of cancer types, especially in tumors or microenvironments with high expression of PD-L1. Yang S-R et al reported a 63-year-old female ATC patient who received radioactive iodine treatment after total thyroidectomy and left neck lymph node dissection. After that, the left neck mass was found, and the biopsy showed ATC, with 95% PD-L1 positivity. Low-grade radiotherapy was performed on the recurrent area, and pembrolizumab was given at the same time 2 days after radiotherapy. A total of 10 cycles of pembrolizumab (2 mg/kg) were given every 3 weeks. CT after radiotherapy and 3 cycles of pembrolizumab showed a reduction in cervical lymph nodes. Continuous follow-up CT showed further reduction of lymph nodes. After receiving combined radiotherapy and pembrolizumab, the patient continued to respond for 26 months with only mild toxicity. This result suggests the potential of combining RT and immune checkpoint inhibitors in the treatment of ATC, but further research is still needed. 40
125I Seed Implantation Combined with Molecular Targeted Drugs
Given the poor response to conventional therapy, ATC patients are suitable candidates for neoadjuvant therapy, including 125I seed implantation and molecular targeted drugs, which have been proven to be effective for other tumors. Apatinib is a small molecule and widely targeted antitumor drug. It is a selective inhibitor of vascular endothelial growth factor receptor-2 (VEGFR-2). 41 Although ATC has high resistance to radiation, it has been reported that postoperative radiotherapy may be beneficial to regional control. On the other hand, traditional EBRT patients suffer from radiation with high dose, high frequency and long duration, and the side effects are obvious. 125I seed implantation can overcome these problems to a certain extent. The radioactive particles directly implanted into the tumor continue to cause radiation damage, while the reduction of radiation dose does less damage to adjacent tissues. Niu y et al reported that a patient with ATC underwent two operations, two 125I seed implantations and apatinib administration. 125I seed implantation and the molecular targeted drug apatinib can locally alleviate the tumor size and show a strong antitumor effect. After 13 months of follow-up since the initial diagnosis of anaplastic thyroid cancer, the patient's condition was pathologically stable. This scheme can be proven to be a feasible alternative treatment for ATC patients who cannot undergo surgery. 42
Drug Carrier: Nanoparticles
Malignant ATC cells often develop resistance to chemotherapeutic drugs such as adriamycin, resulting in treatment failure. Therefore, overcoming cell resistance is very important to improve the prognosis of ATC. A key mechanism of ATC resistance is related to the expression of the multidrug resistant 1 (MDR1) transporter. The MDR1 transporter pumps adriamycin molecules out of cells, reduces the intracellular concentration of drugs, and hinders the effect of chemotherapeutic drugs. In recent years, nanoparticles have been used as drug carriers to overcome the drug resistance of cancer cells. Nanoparticles can carry high concentrations of drugs and improve the intracellular drug concentration by increasing cell uptake or inhibiting drug efflux.43,44
Discussion
Due to the high degree of malignancy, strong local invasiveness and distant metastasis of ATC, the treatment and management of patients is still a difficult problem in the clinic. Surgery, radiotherapy, chemotherapy, and systemic therapy are the recommended first-line therapies. Although they have been proven to alleviate the local symptoms of patients to a certain extent, they cannot effectively prolong progression-free survival and improve the survival rate. With the further understanding of the occurrence and development mechanism of ATC, more mutant genes and molecular pathways have been identified, which has broad application prospects. At present, multimodal therapy has been gradually put into preclinical experiments, clinical trials, and clinical practice. Surgery, radiotherapy, chemotherapy, molecular targeted drug therapy, and immunotherapy are combined to varying degrees, aiming to give full play to the advantages of each treatment method, alleviate local symptoms on the general level, inhibit tumor cell proliferation, and promote tumor cell apoptosis on the molecular level. In addition, the combination of two or more molecules can inhibit several molecular pathways at the same time, improve drug response, and reduce toxicity. We should put more emphasis on personalized treatment, customize ATC patients according to their clinical and genetic characteristics, flexibly combine and apply known therapies, and provide reliable treatment schemes for patients.
Footnotes
Abbreviations
Acknowledgements
Not applicable.
Availability of Data Material
All data generated or analyzed during this study are included in this published article.
Contributions
All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants of Chongqing Provincial Science and Technology department (No. cstc2021jcyj-msxmX0006).
Patient Consent for Publication
Not applicable.
Ethics Statement
Not applicable. This is a review article, so do not have to go through the ethics committee.
