Abstract
Objective
This study intends to examine the anticipatory power of clinical and radiological parameters in detecting clinically significant prostate cancer in patients demonstrating Prostate Imaging Reporting and Data System 3 lesions.
Methods
This was a retrospective study. The study included participation from 453 patients at the First Affiliated Hospital of Soochow University, sampled between September 2017 through August 2022. Each patient underwent a routine 12-core prostate biopsy followed by a 2 to 5 core fusion-targeted biopsy. We utilized both univariate and multivariate logistic regression analyses to identify the parameters that have a correlation with clinically significant prostate cancer. The predictive ability of these parameters was assessed using the receiver operating characteristic curve, leading to the creation of a nomogram.
Results
Clinically significant prostate cancer was detected in 68 out of 453 patients with Prostate Imaging Reporting and Data System 3 lesions (15.01%). Among Prostate Imaging Reporting and Data System 3a and 3b patients, 4.78% (3.09% of the total) and 33.75% (11.92% of the total), respectively, had clinically significant prostate cancer. Systematic biopsy improved prostate cancer and clinically significant prostate cancer detection rates by 7.72% and 3.09%, respectively, compared to targeted biopsy. Without systematic biopsy, there would be an undetected rate of 15% for prostate cancer and 8.13% for clinically significant prostate cancer in Prostate Imaging Reporting and Data System 3b patients. Several clinical parameters, including age, prostate-specific antigen density, lesion volume, apparent diffusion coefficient, and digital rectal examination, were statistically significant in the logistic regression analysis for clinically significant prostate cancer. The individual diagnostic accuracies of these parameters for clinically significant prostate cancer were 0.648, 0.645, 0.75, 0.763, and 0.7, respectively, but their combined accuracy improved to 0.866. A well-fit nomogram based on the identified risk factors was constructed (χ2 = 10.254, P = .248).
Conclusion
The combination of age, prostate-specific antigen density, lesion volume, apparent diffusion coefficient, and digital rectal examination presented a higher diagnostic value for clinically significant prostate cancer than any single parameter in patients with Prostate Imaging Reporting and Data System 3 lesions. Systematic biopsy proved crucial for biopsy-naive patients with Prostate Imaging Reporting and Data System 3 lesions and should not be omitted.
Introduction
Prostate cancer (PCa) is a prevalent disease with an escalating global incidence rate, particularly in China,1,2 where it ranks as the second most common cancer among males. The current methods of treating PCa primarily include surgery and androgen deprivation therapy. Although there are some new treatment options for late-stage PCa in clinical practice, 3 early detection and treatment of PCa can greatly benefit the patient's prognosis. Some studies propose that systematic biopsy (SB) may be unnecessary in patients who do not show suspicious lesions on multiparametric magnetic resonance imaging (mpMRI). This suggestion is primarily relevant to those with low probabilities of possessing clinically significant PCa (csPCa), as determined by additional clinical variables. 4 However, the feasibility of omitting SB in men displaying MRI suspicious lesions remains an unresolved query.
Recently, mpMRI guidance for prostate biopsy (PB) has gained popularity. To streamline the interpretation and reporting of mpMRI, the Prostate Imaging Reporting and Data System (PI-RADS) was conceived as the predominant classification framework for prostate mpMRI. Based on the PI-RADS, prostate lesions are classified from 1 to 5, reflecting the level of associated malignancy risk. 5 Lesions characterized as PI-RADS 1 and 2 are generally dismissed on mpMRI, showing a 90% negative predictive value, whereas the presence of PI-RADS 4 and 5 lesions typically signifies malignancy. 6 However, PI-RADS 3 lesions pose interpretational challenges due to their equivocality. MRI fusion biopsy studies have identified PCa in 35% of PI-RADS 3 lesions, and csPCa in only 17% of this category. 7 Therefore, these lesions particularly exacerbate the challenge of biopsy decisions.
Given these challenges, recognizing reliable distinguishing factors for PI-RADS 3 lesions is a critical step. By customizing treatment options to each patient's needs, we can improve the diagnostic accuracy of csPCa, and consequently decrease the rate of overdetection of clinically insignificant PCa (cisPCa).
Materials and Methods
Clinical Data
The reporting of this study conforms to STROBE guidelines. 8 This study conducts a retrospective evaluation of patients who received transperineal PB in the Urology Department of the First Affiliated Hospital of Soochow University, within the period from September 2017 to August 2022. The total pool of participants included 453 men, all bearing PI-RADS 3 lesions. Criteria for exclusion were as follows: (1) patients who had an mpMRI screening more than 6 months prior to the biopsy; (2) men who previously underwent any medical or surgical treatment for PCa; (3) cases of repeated biopsy; and (4) patients with a history of prostate surgery. All study participants underwent real-time transperineal MRI/ultrasound fusion targeted biopsy (TB) and SB. The study was sanctioned by the Institutional Review Board of the First Affiliated Hospital of Soochow University, adhering to the stipulations prescribed in the Declaration of Helsinki (No.389). All patients provided their signatures on the consent forms prior to the commencement of the study, and we have taken strict measures to deidentify all patient-related details.
Multiparametric Magnetic Resonance Imaging and its Parameters
An mpMRI was performed with a 3 T system (MAGNETOM Skyra; Siemens Healthineers), which included 3 primary scanning sequences: axial T2-weighted image, diffusion-weighted imaging, and dynamic contrast enhancement images. As per the PI-RADS v2.1 guidelines, the apparent diffusion coefficient (ADC) value was calculated using the uppermost b-value, less than or equal to 1000 s/mm2, to bypass the diffusion kurtosis effect. 9 Subsequently, a semiautomatic 3-dimensional segmentation image of the index lesion was procured, and autocalculated lesion volume (LV) was obtained in milliliters. 10
Transperineal PB Procedure
Executing TB required the importation of mpMRI images’ DICOM data into the Real-time Virtual Sonogra (RVS) ultrasound system (Preirus). The target lesion was identified and classified as a region of interest (ROI). A urologist performed the TB, taking 2 to 5 core samples from each ROI. The same urologist continued with SB (12-core) following the conclusion of TB. 11 All biopsy specimens were 10% formalin-fixed and subsequently sent for pathological analyses.
Statistical Analysis
SPSS (version 25.0; IBM Corp) statistical software and R (version 4.2.1) were employed for data analysis. Measurement data that followed a normal distribution were represented as mean ± standard deviation, and an independent sample t test was utilized for cross-group comparisons. For variables with a non-normal distribution, values were presented as medians (interquartile range, IQR) and differences across groups were evaluated using the Mann-Whitney test. Categorical variables were illustrated as numbers (ratios) and examined using a Chi-square test. Both univariate and multivariate binary logistic regressions were conducted to identify independent risk factors of csPCa. Package “rms [version 6.4.0],” and package “Resource Selection [version 0.3-5]” were used for nomogram construction and calibration analysis. The Youden index was utilized to establish a threshold value. Statistical significance was denoted by a 2-tailed P value of less than .05. The sample size required to demonstrate significance in our study, as computed by R software (version 4.1.0), was 103. 12 To ensure accuracy, all data were meticulously reviewed by 2 separate senior statisticians.
Results
Variability Under Investigation in the Groups of Benign/cisPCa and csPCa
Overall, 453 patients qualified based on the inclusion/exclusion benchmarks. The media (IQR) age was 66 years (60-72), prostate-specific antigen density (PSAD) 0.17 ng/mL3 (0.11-0.24), prostate volume (PV) 45 mL (33-58), lesion size 10 mm (7-14), ADC 996 mm2/s (776.5-1189), LV 0.41 mL (0.24-0.66), number of cores 14 (13-15.55), and number of positive cores 4 (2-6).
Prostate cancer was detected in 99 (21.85%) patients, as follows: 31 (6.84%) had cisPCa and 68 (15.01%) csPCa. Patients with csPCa had higher median PSA (10.1 vs 8.74 ng/mL, P = .14), PSAD (0.3 vs 0.2 ng/mL2, P = .02), age (69.18 vs 64.87, P = 0), and lower PV (43.69 vs 50.59 mL, P = .04) than those with benign or cisPCa (Table 1).
Demographics of Patients With PIRADS 3 and MRI-Informed Prostate Biopsy.

Abbreviations: ADC, apparent diffusion coefficient; csPCa, clinically significant prostate cancer; cisPCa, clinically insignificant prostate cancer; fPSA, free prostate-specific antigen; PCa, prostate cancer; PSAD, prostate-specific antigen density; tPSA, total prostate-specific antigen; DRE, digital rectal examination; ADC, apparent diffusion coefficient; ISUP, International Society of Urological Pathology; PI-RADS, prostate Imaging Reporting and Data System; MRI, magnetic resonance imaging.
Variability Under Investigation in PI-RADS 3 Category and Subcategories
Results of MRI and histopathological analysis of PI-RADS v2.1 score 3 category and subcategories (PI-RADS 3a and PI-RADS 3b) are resumed in Table 2. Of the 453 investigated patients, 293 (64.68%) were classified as PI-RADS 3a and 160 (35.32%) as PI-RADS 3b. In PI-RADS 3a patients, 279 (95.22%) patients included 259 benign (88.40%) and 20 cisPCa (6.82%). By contrast, 14 patients (4.78%) were diagnosed as csPCa (corresponding to 3.09% of entire PI-RADS v2.1 score 3 risk category). In PI-RADS 3b patients, 95 patients (59.37%) were diagnosed as benign disease and 11 patients (6.88%) were diagnosed as cisPCa, whereas 54 patients (33.75%) were diagnosed as csPCa (corresponding to 11.92% of the entire score 3 risk category). The detection rate of csPCa was higher in PI-RADS 3b than PI-RADS 3a patients.
Demographics of Patients With PIRADS 3 and MRI-Informed Prostate Biopsy According to Lesion Volume.
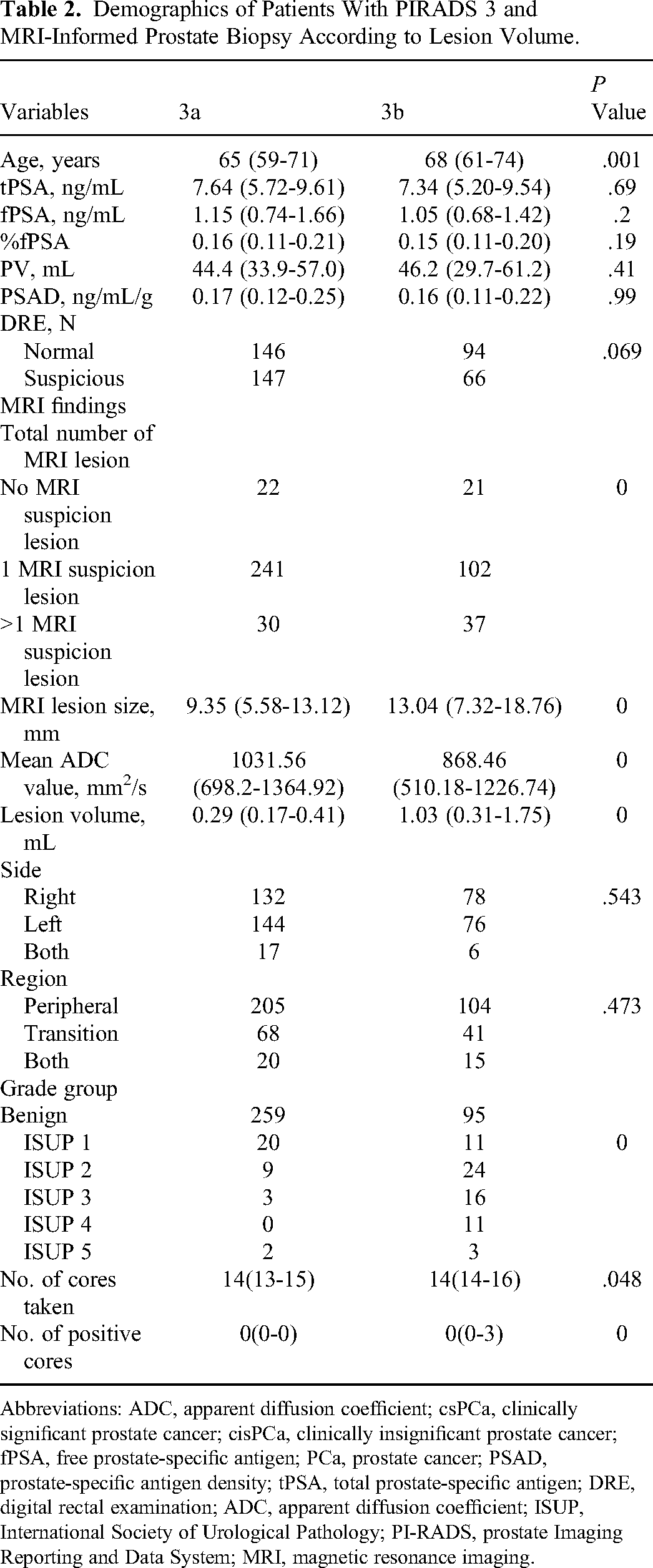
Abbreviations: ADC, apparent diffusion coefficient; csPCa, clinically significant prostate cancer; cisPCa, clinically insignificant prostate cancer; fPSA, free prostate-specific antigen; PCa, prostate cancer; PSAD, prostate-specific antigen density; tPSA, total prostate-specific antigen; DRE, digital rectal examination; ADC, apparent diffusion coefficient; ISUP, International Society of Urological Pathology; PI-RADS, prostate Imaging Reporting and Data System; MRI, magnetic resonance imaging.
Detection Rate of SB and TB in PI-RADS 3 Category and Subcategories
Overall, 453 patients underwent combined SB + TB. The detection rate of PCa for SB, TB, and TB + SB was 18.54%, 14.13%, and 21.85%, for csPCa it was 10.82%, 9.05%, and 12.14%, respectively. Systematic biopsy added 7.72% PCa and 3.09% csPCa detection to TB (Table 3). A total of 160 patients with PI-RADS 3b underwent combined SB + TB. The percentage in which PCa is detected for SB, TB, and TB + SB are 35%, 25.63%, and 40.63%, respectively. Similarly, the detection percentage for csPCa is 25%, 20%, and 28.13%, respectively. Within this population, 15% of PCa and 8.13% of csPCa may go undetected if SB is not performed (Table 3).
Prostate Cancer Detection in Patients With PIRADS 3 and MRI-Informed Prostate Biopsy.
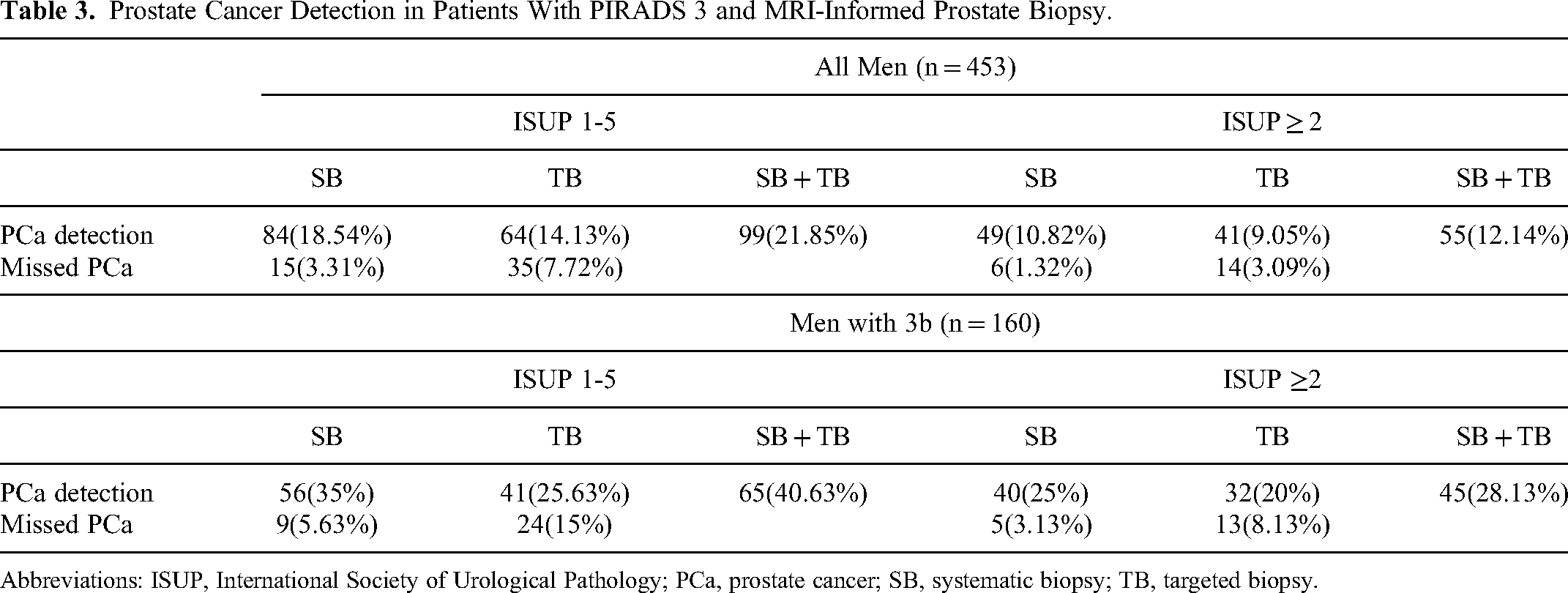
Abbreviations: ISUP, International Society of Urological Pathology; PCa, prostate cancer; SB, systematic biopsy; TB, targeted biopsy.
The detection rates of PCa and csPCa for patients with a lesion PI-RADS 3 improved when LV increased for both stand-alone SB, stand-alone TB, and the combination of SB + TB. Histology cross-tabulation is shown in Tables 4 and 5 for TB versus SB. Overall, the same Gleason Grade (GG) was found in both SB and TB for 88.7% (402/453) of the subjects. However, TB alone resulted in GG upgrading by 3.31% (15/453), compared to a 7.95% (36/453) upgrade by SB alone. In patients with PI-RADS 3b, both SB and TB showed the same GG in 78.75% (126/160) of instances. However, TB alone resulted in a GG upgrade in 6.25% (10/160) of cases, compared to a 15% (24/160) upgrade by SB alone.
Cross-Tabulation of Systematic Versus Target Biopsy in Patients With PIRADS 3 and MRI-Informed Biopsy.

Cross-Tabulation of Systematic Versus Target Biopsy in Patients With PIRADS 3, MRI-Informed Biopsy and 3b.

Bivariate and Multivariate Logistic Regression Analysis and Receiver Operating Characteristic Curve of Age, PSAD, LV, ADC, and digital rectal examination as Predictors of csPCa in PI-RADS 3 Category
Table 6 illustrates the treatment methods applied to the whole group, as per the risk criteria of PCa established by the National Comprehensive Cancer Network. A total of 79 patients at our institutions underwent radical prostatectomy (RP), hence, we had their RP pathology/histology reports available (Table 7). Any upgrading on SB, TB, and SB + TB in comparison to RP specimen were 12.66%, 3.8%, and 16.46% for overall population (N = 79); and 16.98%, 5.66%, and 15.09% in those with PI-RADS 3b (N = 53), respectively (Table 8). In the bivariate and multivariate logistic regression analyses, age, PSAD, LV, ADC, and digital rectal examination (DRE) were significantly associated with the presence of csPCa at biopsy. On the contrary, PV was not significantly associated with the presence of csPCa (P = .946). Area under the curve (AUC) of age, PSAD, LV, ADC, and DRE is 0.648, 0.645, 0.75, 0.763, and 0.7 for detection of csPCa, respectively, by univariate receiver operating characteristic (ROC) curve analysis (Figure 1A). Multivariate ROC curve analysis with age, PSAD, LV, ADC, and DRE as multivariate statistically significant parameters showed a sensitivity for detection of PCa of 86.2% with a specificity of 74.3% and AUC of 0.866 (Table 9, Figure 1B).

Receiver operating characteristics (ROC) curves and area under the curves (AUC) of age, DRE, PSAD, ADC, and lesion volume. (A) ROC curves of age, DRE, PSAD, ADC, and lesion volume; (B) ROC curves of combination of these parameters. PSAD, prostate-specific antigen density; ADC, apparent diffusion coefficient; DRE, digital rectal examination.
Treatment According to National Comprehensive Cancer Network Prostate Cancer Risk Classification.

Demographics and Radical Prostatectomy Specimen Pathology for Patients With PIRADS 3 and MRI-Informed Systematic and Target Biopsy.

Abbreviations: ADC, apparent diffusion coefficient; csPCa, clinically significant prostate cancer; cisPCa, clinically insignificant prostate cancer; DRE, digital rectal examination; fPSA, free prostate-specific antigen; ISUP, International Society of Urological Pathology; MRI, magnetic resonance imaging; PCa, prostate cancer; PSAD, prostate-specific antigen density; PI-RADS, Prostate Imaging Reporting and Data System; tPSA, total prostate-specific antigen; RPS, radical prostatectomy specimen.
Concordance of Prostate Cancer Grade Group on Systematic Biopsy, Target Biopsy, and Combined Systematic and Target Biopsy According to RPS in Patients With PIRADS 3 and 3b.

Abbreviations: RPS, radical prostatectomy specimen; SB, systematic biopsy; TB, targeted biopsy.
Univariable and Multivariable Binary Logistic Regression Analysis Testing Variables and Independent Risk Factors of csPCa.

Abbreviations: ADC, apparent diffusion coefficient; csPCa, clinically significant prostate cancer; PSAD, prostate-specific antigen density; DRE, digital rectal examination; ADC, apparent diffusion coefficient; ROC, receiver operating characteristic; PV, prostate volume; LV, lesion volume; AUC, area under the curve.
Nomogram Construction and Evaluation
The nomogram was established based on the outcomes of multivariate logistic regression analysis, which screened out the variables: Age, PSAD, LV, ADC, and DRE. Through the application of 1000 bootstrap resamples, our model demonstrated strong discrimination with a C-index of 0.890. This indicates a 95% confidence interval ranging from 0.851 to 0.929 for csPCa, as illustrated in Figure 2 and Figure 3.

A nomogram model for predicting clinically significant prostate cancer in biopsy-naive patients with PI-RADS 3 lesions.

Calibration curve of the nomogram.
Discussion
The utilization of mpMRI for PCa assessment has gained widespread popularity. 13 Considered an excellent diagnostic tool for PCa, PI-RADS is a standardized MRI evaluation methodology for examining prostate lesions. 14 While it is commonly agreed that biopsies are unnecessary for PI-RADS 1 and 2 lesions and unquestionably required for PI-RADS 4 and 5 lesions, consensus is lacking regarding the necessity for biopsy in patients with PI-RADS 3 lesions, 15 placing these cases in a “gray zone.” However, radiologists with limited experience might have inaccuracies in their PI-RADS scoring. As reported by Pepe et al, 16 out of 241 patients with a PI-RADS score of 3 assessed by an affiliated radiology center, 122 cases had their scores adjusted—either upgraded or downgraded—after review by experienced radiologists, representing 50.62% of the total. Therefore, in this study, all PI-RADS scores were evaluated and reviewed by 2 experienced radiologists.
The contribution of mpMRI to increasing the detection of csPCa and potentially eliminating the need for SB in biopsy-naive patients. In this study, the detection rates of overall PCa and csPCa were 21.85% and 12.14%, respectively, somewhat lower than the 25.3% and 22.2% reported in a previous study. 17 Concordantly, our study also mirrored findings from the MRI-FIRST, PRECISION, and 4 M trials, achieving a csPCa detection rate of 9.05% for TB among biopsy-naive patients with PI-RADS 3 lesions. The indispensability of SB in such cases was underscored by the improvement in detection rates of csPCa from 12% in the MRI-FIRST trial to 17% following the inclusion of SB.18–20 Our study demonstrated a similar trend, elevating the overall PCa and csPCa detection rates from 14.13% and 9.05% at TB to 21.85% and 12.4% in combination with SB. Therefore, SB was indispensable and could not be omitted in biopsy-naive patients with PI-RADS 3 lesions.
The objective of our study was to explore and identify potential radiological characteristics that may aid clinicians in deciding on the need for biopsy in PI-RADS 3 lesions. Preliminary studies suggest MRI parameters including LV, ADC, lesion diameter, and PV may also be used in the prediction of csPCa in patients with PI-RADS 3 lesions. Apparent diffusion coefficient value ≥ 900 mm2/s was a good indicator to distinguish prostatitis from PCa. 21 It has been reported that an ADC value threshold ranging from 750 to 900 mm2/s can aid in distinguishing between BPH and PCa. If the ADC value falls below this threshold, it is typically associated with csPCa. 22 Previous research indicated that an ADC value of ≤900 mm2/s was not considered an independent risk factor in binary logistic regression. However, an ADC value of ≤750 mm2/s did serve as an independent risk factor. 23 Hermie et al discovered that the combined condition of a median PV under 44 mL and a ratio below 70% for the lesion ADC value over the contralateral prostate ADC value resulted in a detection sensitivity of 59% and a specificity of 88% for csPCa, with an AUC value of 0.78. 24 In this study, ADC value was an independent risk factor in the binary logistic regression for detection of csPCa with AUC 0.763. This finding was consistent with the results from the study conducted by Pepe et al. 25
In recent times, multiple studies have precisely assessed the alignment of mpMRI LV with the histological tumor volume found in RP specimens. These have affirmed that while MRI slightly underestimates the actual tumor volume, it still remains within an acceptable range.26–28 Previous research has shown a robust correlation between the LV in MRI and the surgical specimen. Thus, a cutoff value of 0.5 mL can be applied to pinpoint suspicious lesions that may be at risk of csPCa. 26 Eugenio et al 29 indicate that the LV classification based on 0.5 mL cutoff may represent an effective pretreatment tool to easily discriminate csPCa from iPCa. In our study, we arrived at a similar conclusion concerning the usefulness of LV in predicting malignant outcomes. We found that 54 patients (33.75%) were diagnosed as csPCa in PI-RADS 3b patients (corresponding to 11.92% of the entire score 3 risk category), significantly higher than in PI-RADS 3a patients. The AUC (0.732) of LV indicated a good diagnostic accuracy in identifying csPCa at biopsy. 29 Lesion volume was an independent risk factor in the multivariable binary logistic regression for detection of csPCa with AUC of LV 0.75 in this study.
It should be emphasized that several factors beyond the MRI appearance may affect a patient's clinical management. These include diagnose at the previous biopsy, age, family history, DRE, PSA, PSAD, and PSA velocity.30–34 Many previous studies have regularly used age and DRE to predict PCa and csPCa. In multivariate analysis, it appeared that suspicions raised during DRE independently triggered biopsies. 25 A risk stratification of PI-RADS 3 lesions was formulated by Sheridan et al, wherein age and DRE were recognized as risk factors for csPCa. 35 Meanwhile, Radtke et al built a nomogram (AUC 0.84) containing age and DRE by evaluating a cohort of 1159 PI-RADS patients. 28 In the present study, there were significant difference between cisPCa and csPCa for age and DRE and also independent risk factors for detection of csPCa (AUC = 0.648, 0.7, respectively).
PSA is widely employed as the primary screening and effectiveness assessment index for PCa. However, its use continues to be controversial because of its low diagnostic specificity. 36 In recent years, several new PSA-related parameters have been proposed to enhance the diagnostic effectiveness of PCa, with PSAD being the most prominent among them.37,38 According to a report by Roscigno et al, 39 a PSAD of ≥0.20 ng/mL2 could enhance the predictive accuracy of mpMRI results for reclassifying patients in active surveillance. On the other hand, a PSAD of <0.10 ng/mL2 might assist in identifying patients who are at a lower risk of harboring csPCa. A recent study has shown that when total PSA is between 4 and 10 ng/mL, PSAD is an independent predictor of PCa. 40 In our previous study, when setting 0.15 ng/mL/mL as a cutoff, the sensitivity, specificity, PPV, and NPV for csPCa were 0.82, 0.46, 0.16, and 0.96, respectively, in our “double gray zone.” 23 A multicenter study conducted in China demonstrated that the optimal PSAD cutoff value for diagnosing PCa was determined to be 0.33 ng/mL/mL when the PSA levels ranged between 10.1 and 20.0 ng/mL. 41 The result of this study showed that PSAD had a high diagnostic value (AUC = 0.645) for csPCa at an optimal cutoff value was 0.12 ng/mL/mL.
Eugenio et al 29 have demonstrated that the predictive ability of LV increased if it was included, along with age and PV in a multivariate model. The resulting AUC (0.865) of multivariate model was superior than those of single variables. 30 Similarly, multivariate ROC curve analysis with age, PSAD, LV, ADC, and DRE as multivariate statistically significant parameters showed AUC 0.866 for detection of csPCa, obviously higher than single variables in our study.
There were certain constraints in this study that are worth noting. Initially, this study was a retrospective single-center research, inherently susceptible to selection bias. This paves the path for conducting additional prospective studies across multiple centers. Moreover, our investigation was carried out in a tertiary hospital, under the guidance of a 10-year experienced radiologist who examined the MRI scans and a skilled urologist who executed all MRI fusion biopsies. While these factors ensured study consistency, they also bring into question the potential relevance of our findings to nontertiary hospitals. Furthermore, the definition used for csPCa may not cover all clinically significant diseases. In relation to this, studies have recently surfaced that 68 Ga-PSMA PET/CT exhibits superior diagnostic value for PI-RADS score 3 patients in identifying csPCa, compared to combined clinical prediction models involving mpMRI.42,43 Unfortunately, our institution currently lacks this equipment. As this hardware is procured in the future, further studies will be conducted. Lastly, suggestions have been made that urinary cytology markers and serum biomarkers could revolutionize PCa detection. 44 We intend to delve deeper into the predictive impact of these biomarkers on csPCa for patients presenting a PI-RADS score of 3 in future studies.
Conclusion
This study has found a csPCa detection rate of 15.01% among all PI-RADS 3 lesions, and as high as 33.75% within PI-RADS 3b lesions. A combined approach with age, PSAD, LV, ADC, and DRE variables exhibited a higher predictive value for csPCa on PB. Further, SB significantly increased the overall PCa and csPCa detection rates compared to TB alone, emphasizing the indispensability of SB in biopsy-naive patients with PI-RADS 3 lesions.
Footnotes
Acknowledgments
Thank all patient's participation and grant from the National Natural Science Foundation of China (Grant No.32200533) and (Grant No.82002685).
Authors’ Note
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The Institutional Review Board of First Affiliated Hospital of Soochow University approved this study (Approval No.389 (2023)), which is in accordance with the Declaration of Helsinki. And written informed consent was taken from all individual participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number 32200533, 82002685).
