Abstract
Keywords
Introduction
Immunotherapy combined with chemotherapy has become a first-line option for treating many types of tumours, including lung cancer.1,2 Immune checkpoint networks are distributed in various human tumours, and these immune checkpoint molecules transmit signals that stimulate or suppress T-cell responses. Human endogenous retrovirus-H long terminal repeat associating 2 (HHLA2) was identified as a novel immune checkpoint in the field of tumour immunity. 3 As a ligand, HHLA2 binds to the putative receptor CD28H (also called TMIGD2, transmembrane and immunoglobulin domain containing 2) and leads to suppression of the T-cell response. Moreover, as a gland, HHLA2 binds to another receptor of KIR3DL3 (Killer cell immunoglobulin like receptor, three Ig domains and long cytoplasmic tail 3) expressed on activated T cells. HHLA2-KIR3DL3 promotes T-cell exhaustion and influences macrophage polarity. The KIR3DL3-HHLA2 and TMIGD2-HHLA2 pathways may serve as crucial immune checkpoints within the tumour microenvironment.3,4 Sadiye et al 5 demonstrated the role of HHLA2 in negative immune regulation; HHLA2 inhibits T-cell proliferation and activation and cytokine secretion by affecting TCR and CD28 signalling. KIR3DL3-HHLA2 and TMIGD2-HHLA2 pathways blockage may provide better insights into the development of novel therapeutic drugs against human malignancies. 4 The emergence of targeted immune checkpoint therapy has led to major developments in malignant tumour treatment. However, new reliable immune biomarkers are needed to improve the response to immunotherapy. 6 Chemotherapy combined with immunotherapy is the classic first-line treatment for lung cancer. Immunotherapy has shown considerable promise in non-small cell lung cancer (NSCLC) patients. Therapy targeting PD-L1/PD-1, such as pembrolizumab and nivolumab, and antibodies targeting cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), such as tremelimumab and ipilimumab, have been developed. 7 Small cell lung cancer (SCLC) is the most common and lethal type of lung cancer. Because of its rapid progression, early widespread metastasis and limited therapeutic options, SCLC still has a poor prognosis. Although the PD-L1 inhibitors durvalumab and atezolizumab combined with standard chemotherapy as first-line strategies have been shown to improve progression-free survival (PFS) and overall survival (OS) in patients with advanced-stage SCLC, 8 we still need to explore other unknown immune checkpoint molecules in SCLC to further improve survival outcomes. HHLA2 has been detected in various human tumours, including NSCLC, 9 but its role in SCLC remains unknown. Therefore, further exploration of the importance of HHLA2 in SCLC is needed. In the future, therapies targeting HHLA2 and its receptors CD28H and KIR3DL3 may constitute promising immunotherapy strategies for SCLC.
Materials and Methods
Lung Cancer Specimen Collection
SCLC specimens from 91 patients and normal lung tissues from 50 patients were obtained from two hospitals. Verbal informed consent was obtained from the study participants. All patient data were deidentified. All SCLC specimens were obtained by surgery or bronchoscopy biopsy, and none of the SCLC patients received any chemotherapy, radiotherapy or targeted therapy. Relevant clinical data, including the survival time of the SCLC patients, were collected retrospectively. The Academic Ethics Committees of the hospital approved the study protocol (LS2021019, LS2022098; [2021]-K-73). The TNM classification of SCLC patients was determined according to the International Society for International Lung Cancer [IASLC] (8th edition). 10 Paraffin-embedded SCLC specimens were assessed via immunohistochemistry (IHC). There were 66 limited stage (LS) SCLC patients and 25 extensive stage (ES) SCLC patients. Among the SCLC patients, 71 were males and 20 were females. The median age was 64 years. Because of the short survival time of SCLC patients, a survival endpoint of 24 months was selected. Moreover, we retrospectively investigated the other clinical data of SCLC patients, including carcinoembryonic antigen (CEA) and neuron-specific enolase (NSE) levels. The reporting of this study conforms to the REMARK guidelines. 11
Determination of Serum Cancer Markers
Venous blood was obtained from 91 SCLC patients and centrifuged at 3000 r/min for 10 min. The supernatants were obtained and analysed. CEA was visualised by chemiluminescence with an Abbot i2000 or Beckman DXI800 chemiluminescence immunoassay instrument. NSE was assessed by electrochemiluminescence with a Roche e601 instrument. The standard cut-off value for CEA (5.0 ng/mL) was used. The standard serum cut-off value of NSE (17 ng/mL) was used. The results of the serum cancer marker analyses were recorded in hospitals.
Immunohistochemical Detection of HHLA2 and Ki-67
SCLC specimens and adjacent normal tissues embedded in paraffin were sectioned into 4 µm slices. The tissue slides were dewaxed and dehydrated, followed by antigen retrieval with EDTA in a high-pressure cooker for 3 min. Hydrogen peroxide was used to block the endogenous peroxidase activity of the slices for 30 min. Rabbit anti-HHLA2 polyclonal antibody (1:500, ab214327, IgG, Abcam, Cambridge, UK) was added to the slides overnight. A rabbit IgG Ab1 antibody (1:1000; Merck & Millipore, FCMCS) was used as a control. Finally, a secondary antibody (goat anti-rabbit IgG, Abcam, UK) was added for 15 min of incubation, and the sections were stained with diaminobenzidine (DAB) to observe the images. All slices were independently assessed by two pathologists, and the clinical status of the patients was determined by two clinicians. The procedure used for Ki-67 immunochemistry was the same as that used for HHLA2 analysis. Mouse anti-human Ki-67 was used as the primary antibody (1:500; Novus, NBP2-22112), and rabbit anti-mouse IgG was used as the secondary antibody (CST, #58802).
IHC Evaluation of SCLC Specimens
The immunohistochemical staining intensity of HHLA2 was determined according to the H-score. H-Score = ∑(PI × I)= (percentage of unstained cells × 0) + (percentage of weakly stained cells × 1) + (percentage of moderately stained cells × 2) + (percentage of strongly stained cells × 3). PI represents the percentage of positive cells among all cells in the slice; I represents the intensity of staining.12,13 The H-score ranged from 0 to 300. The staining intensity of the tumour tissues was scored as 0-3 (0, no staining; 1, weak staining; 2, intermediate staining; 3, strong staining). All stained slices were independently examined by two pathologists. The average H-score was calculated for further statistical analysis. Immunohistochemical staining for Ki-67 was observed via light microscopy, and the percentage of Ki-67-positive cells was determined according to the percentage of Ki-67-positive cells in the whole slice of the SCLC specimen.
Statistical Analysis
Pearson's Chi-squared test and Fisher's Chi-squared test were used for data analysis. K‒M analysis was used to compare survival rates. A log-rank test was carried out to analyse the statistical significance of the differences between groups. All the statistical analyses were performed with SPSS version 21. P < .05 was considered to indicate a significant difference.
Results
HHLA2 Expression Status in SCLC Specimens
In this cohort, 91 SCLC samples and 55 lung tissue samples were used to detect HHLA2 expression via IHC. Sixty-seven SCLC samples were found to be positive for HHLA2. Seventy-four percent of SCLC patients expressed HHLA2. HHLA2 expression was indicated mainly by moderate or weak positive staining. HHLA2 was localised within the nucleus of SCLC cells, whereas normal lung tissue specimens did not show HHLA2 staining (Figure 1).

HHLA2 expression in SCLC HHLA2 staining is observed in nucleus of SCLC: (A) HHLA2 strong staining in SCLC tissues; (B) HHLA2 moderate staining in SCLC tissues; (C) HHLA2 weak staining in SCLC tissues; (D) HE staining in SCLC tissues (40×).
Correlations Between HHLA2 Expression and Clinical Parameters in SCLC Patients
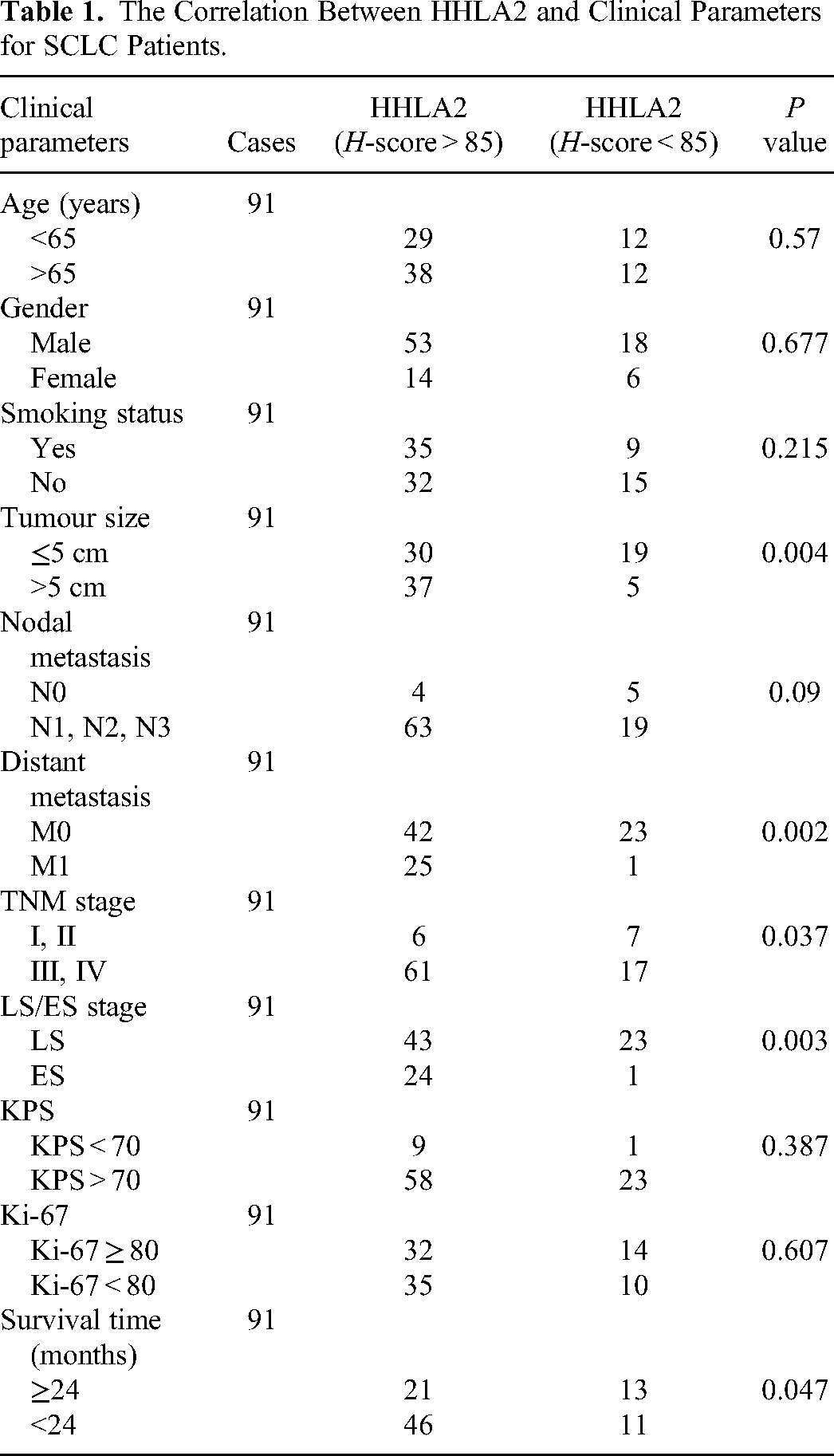
The association between HHLA2 staining and the clinical status of SCLC patients was also investigated. The results demonstrated a strong correlation between HHLA2-positive staining and clinical features, such as tumour size, distant metastasis, TNM stage and LS/ES status. In particular, a greater proportion of LS SCLC samples than ES SCLC samples exhibited HHLA2 staining. Compared with SCLC patients with metastasis, SCLC patients without distant metastasis accounted for a much greater proportion of HHLA2-positive patients. A greater percentage of stage III and IV SCLC patients had HHLA2-positive staining than did stage I and II SCLC patients. HHLA2 expression was more frequently detected in the group with a tumour size greater than 5 cm than in the group with a tumour size less than 5 cm. We also investigated the 2-year survival status of SCLC patients with different HHLA2 expression levels. Due to the high degree of malignancy and limited available therapies, SCLC patients have a short survival time. There was a significant difference in the 2-year survival rate between the HHLA2-positive group and the HHLA2-negative group. A high proportion of SCLC patients with HHLA2-positive staining had a survival time <2 years. There were no significant differences in other clinical factors, such as age, sex (male, female), smoking status (smoking, non-smoking), nodal metastasis status (N0, the N1, the N2, N3), Karnofsky Performance Scale (KPS) grade, or Ki-67 expression rate, between the HHLA2-positive group and the HHLA2-negative group (Table 1). We also evaluated the OS rate, and there was a significant difference between the HHLA2-positive group and the HHLA2-negative group (Figure 2).

Comparison of the overall survival rate in HHLA2-positive and HHLA2-negative group.
The Correlation Between HHLA2 and Clinical Parameters for SCLC Patients.
Relationships Among NSE, CEA and Ki-67 in SCLC Patients With Different HHLA2 Expression Statuses
Moreover, NSE, CEA and Ki-67 expression was measured (Table 2, Figure 3). The CEA and Ki-67 levels were not significantly different between the HHLA2-positive and HHLA2-negative patients. However, patients in the HHLA2-positive group had higher NSE levels than patients in the HHLA2-negative group did. NSE is a classic SCLC biomarker. The HHLA2-positive group exhibited a greater tumour burden than the HHLA2-negative group; therefore, HHLA2 may act as an immune inhibitor to promote SCLC progression and lead to higher NSE levels.

Ki-67 expression in SCLC: (A) Ki-67 expression is observed in SCLC; (B) HE staining in lung tissues (20×).
The Relationship Among NSE, CEA and Ki-67 in SCLC Patients With Different HHLA2 Expression Statuses.

Discussion
SCLC is a lethal disease for humans worldwide. The importance of immune checkpoints and their value as therapeutic targets have attracted increasing amounts of attention in the field of human malignancies. HHLA2 is a newly identified immune checkpoint molecule. HHLA2 was found in the majority of tumour tissues. 3 Masataka et al 14 detected HHLA2 expression in the plasma of human gastric cancer patients. High expression levels of HHLA2 were associated with distant metastasis, tumour stage and tumour invasion depth. HHLA2 expression was not found in blood specimens from the healthy group. Zhu et al 15 detected HHLA2 expression in human colorectal cancer (CRC) patients. HHLA2 expression was found in colorectal carcinoma and was correlated with tumour invasion depth and CD8+ T-cell infiltration within tumour tissues. Patients with high HHLA2 expression were shown to have a worse prognosis. HHLA2 has prognostic value in human CRC. In an in vitro study, HHLA2 silencing enhanced the chemosensitivity of an osteosarcoma cell line to paclitaxel, and HHLA2 was reported to be an inhibitory immune molecule in osteosarcoma. 16 HHLA2 was also found to be expressed in advanced gallbladder cancer (GBC) and to promote GBC growth both in vivo and in vitro. 17 In contrast, pancreatic ductal adenocarcinoma (PDAC) patients with high HHLA2 expression had better survival outcomes than did those with low HHLA2 expression. HHLA2 overexpression was demonstrated to be a good marker of PDAC progression. 18 Qi et al 19 analysed the function of HHLA2 in malignant glioma and revealed that HHLA2 was a favourable prognostic marker and therapeutic target in glioma patients.
Regarding NSCLC, Chen Yan et al 9 reported HHLA2 and B7-H4 expression in EGFR-mutant and wild-type lung adenocarcinomas. HHLA2 expression was observed more frequently in the EGFR-mutant population than in the EGFR wild-type population. Moreover, patients with low HHLA2 expression had a relatively longer disease-free survival (DFS) than patients with high HHLA2 expression. Haiying Cheng et al 20 compared HHLA2 expression and its clinical value in PD-L1-positive and PD-L1-negative populations of lung cancer patients and demonstrated that HHLA2 suppressed T-cell proliferation and reduced cytokine secretion. HHLA2-Ig inhibited T-cell proliferation more potently than PD-L1-Ig. In addition, HHLA2 expression was observed in PD-L1-negative tumours. Moreover, NSCLC tissue has been shown to be positive for HHLA2 expression, and an EGFR mutation status and a high density of tumour-infiltrating lymphocytes have been found to be associated with HHLA2 expression in lung adenocarcinoma. 21 Farrag et al 22 reported that HHLA2 overexpression was observed in NSCLC and was associated with increased tumour stage and EGFR overexpression. HHLA2 could serve as a predictive biological marker of progression and distant metastasis in NSCLC. Sun et al 23 demonstrated that, in terms of biological function, HHLA2 acts as an inhibitory checkpoint gene in NSCLC. Knockdown of HHLA2 inhibited NSCLC cell proliferation, migration and invasion by inactivating the EGFR/MAPK/ERK signalling pathway. Knockdown of HHLA2 also inhibited the M2 polarisation of tumour-associated macrophages (TAMs). In vivo, knockdown of HHLA2 suppressed tumorigenesis in nude mice. These findings preliminarily indicate that HHLA2 acts as an inhibitory checkpoint in NSCLC. However, studies of HHLA2 in SCLC are still limited. Although Farrag et al 22 detected HHLA2 expression in 8 of 14 SCLC patients, the association between HHLA2 expression and the clinical status of SCLC patients was not further investigated in detail. However, the importance of HHLA2 in SCLC remains unknown. However, further investigations of the clinical importance of HHLA2 in SCLC are needed.
In this observational study, we found HHLA2 expression in most SCLC tissues, but HHLA2 expression was not observed in normal lung tissues. HHLA2 expression was closely associated with tumour size, distant metastasis status, TNM stage, LS/ES status and survival period. The proportion of patients with HHLA2 expression was greater among SCLC patients without distant metastasis than among SCLC patients with distant metastasis. Among patients with high HHLA2 expression, LS SCLC was also more common than ES SCLC. Moreover, more patients in the SCLC group with a tumour size greater than 5 cm had HHLA2 expression than did those with a tumour size less than 5 cm. The stage III and IV SCLC group of patients had a greater percentage of HHLA2-positive patients than did the stage I and II SCLC groups of patients. For tumour biomarkers in plasma, NSE expression differed significantly between the HHLA2-positive and HHLA2-negative groups, while CEA and Ki-67 levels were not significantly different. Finally, we investigated the difference in 2-year survival in SCLC patients with different HHLA2 expression statuses. The SCLC patients with HHLA2 expression accounted for a greater percentage of the population with a survival time < 2 years. Moreover, patients with HHLA2-positive tumours had a worse OS rate. The above results showed that HHLA2 is expressed in the earlier stages of SCLC, and SCLC patients with HHLA2 expression may have a worse prognosis. NSE is a specific classic biomarker for SCLC, and NSE expression is linked to a high tumour burden and shorter survival time in SCLC patients.24,25 SCLC patients with HHLA2 expression had higher levels of NSE than patients without HHLA2 expression, and high NSE levels indicate a worse survival outcome. Our study indicated that HHLA2 is correlated with advanced disease stage and shorter survival time. HHLA2 may contribute to SCLC progression and affect the survival period of patients with SCLC. HHLA2 may serve as a potential immunotherapy target in SCLC. These results may reveal an inhibitory effect of HHLA2 on SCLC, which is the same as previous findings showing that HHLA2 functions in NSCLC. The clinical significance of HHLA2 in SCLC may provide additional insights into the development of novel therapeutic drugs against SCLC, such as immune checkpoint blockade agents. However, our study has several limitations. The number of SCLC specimens enrolled was not sufficient. Moreover, our study did not explore the biological function of the HHLA2 gene or the relevant mechanisms in SCLC either in vitro or in vivo. Further research is needed to determine the biological function of the HHLA2 gene and clarify its relevant mechanisms in SCLC.
Conclusion
Immunotherapy is the hallmark of malignant tumour therapy. Blockade of immune checkpoints (PD-L1/PD-L2/PD-1, B7-1/B7-1/CTLA-4) has been a very important strategy for both NSCLC therapy and SCLC therapy. 26 HHLA2 is overexpressed in SCLC. HHLA2 has important clinical value in SCLC patients. The HHLA2 checkpoint may serve as a promising biomarker for predicting progression in SCLC patients, and it may also be a novel potential target in SCLC immunotherapy, such as immune checkpoint blockade and antibody‒drug conjugate treatment.
Footnotes
Abbreviations
Acknowledgments
The authors thank Mr Weiguo Xie for manuscript revision and statistical analysis for this project. They also thank the First People's Hospital of Yancheng Affiliated with Nantong University for SCLC samples and clinical data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Standards
This study was approved by the Affiliated Hospital Ethics Boards of Jiangnan University (LS2021019, LS2022098) and The First People's Hospital of Yancheng Affiliated with Nantong University ([2021]-K-73). Verbal informed consent was obtained from the study participants. All patient details have been dedentified.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
