Abstract
Introduction
According to the 1993 revised classification system for human immunodeficiency virus (HIV) infection, there are 2 parts of HIV-associated cancers: acquired immune deficiency syndrome (AIDS)-defining cancers and non-AIDS-defining cancers. 1 Non-Hodgkin's lymphoma (NHL) was considered as one of the AIDS-defining cancers; Hodgkin's lymphoma (HL) was considered as one of the non-AIDS-defining cancers in patients with HIV. 2 Although the introduction of combined antiretroviral therapy (cART) has greatly reduced the mortality rate of HIV-infected patients, cancer remains a significant cause of death. Among HIV-infected patients, NHL is considered as one of the most common cancers and as a frequent cause of death,3-5 causing 3.5% of deaths per year (ie, 396.9 per 100 thousand populations) due to HIV-associated cancers as recorded in the United States during the 2001 to 2015 time period. 6 Due to the use of cART,7-9 the incidence of diffuse large B-cell lymphoma was reduced, but it showed no significant effect on Burkitt lymphoma (BL), which remained or even increased over time.10-12 Adult BL accounts for 35% of HIV-related lymphomas,13,14 and AIDS-related BL (AR-BL) usually occurs very early and is often referred to as the earliest AIDS-defining disease.15-17
The international prognostic index (IPI) was first proposed in 1993 to predict long-term survival. 18 IPI score consists of 5 variables: age over 60 years, Eastern Cooperative Oncology Group Performance Status (ECOG PS) 2 to 4, advanced stage, elevated lactate dehydrogenase (LDH), and ≥ 2 extra-nodal (EN) location. 19 IPI has also been shown to correlate with the prognosis of HIV-associated lymphoma. 20
Owing to the lack of a standard prognostic model for BL, BL international prognostic index (BL-IPI) was proposed according to a recent study as a novel prognostic model to aid risk stratification with BL patients. The population of the study included HIV-positive patients and the BL-IPI contains 4 variables: age over 40 years, ECOG PS 2 to 4, LDH > 3 × ULN, and CNS involvement. 21 However, there is still a lack of a specific prognostic model for AR-BL patients. The data regarding prognostic factors and outcomes in AR-BL patients treated in the cART era remain scarce. Therefore, the clinical characteristics and outcomes of patients with AR-BL from our center were analyzed and we conducted a novel risk stratification model named GZ8H model to predict prognosis.
Methods
Patients and Study Design
We retrospectively collected the data on AR-BL patients initially treated in the Guangzhou Eighth People's Hospital, Guangzhou Medical University from January 2011 to December 2021. All patient details were de-identified. Those over 18 years old were included in this survey. A definite diagnosis of BL by pathological examination was made according to diagnostic criteria 22 in May 2022 and the diagnosis of AIDS met the standards of HIV/AIDS according to the 2021 Chinese guidelines. 23 Patients with other active malignancies were excluded.
Detailed information about clinical data was collected from medical records. Patients were stratified by IPI score with 4 levels of risk and BL-IPI score with 3 groups of risk. Combined with the Ann Arbor system with physical examination, routine laboratory tests, and imaging tests, lymphoma staging was confirmed. 24 The reporting of this study conforms to STROBE guidelines. 25
Study Endpoint
Patients were followed up through hospitalization or telephone interviews. Overall survival (OS) and progression-free survival (PFS) were the primary study points. OS was defined as the time from BL diagnosis to the last follow-up or death from any cause. PFS was defined as the time from BL diagnosis to disease progression, relapse, or death from any cause. The follow-up endpoint of this study was May 31, 2022.
Statistical Analysis
Continuous variables were summarized as the medians and interquartile ranges (IQR) with the Mann-Whiney U-test or t-test, and categorical variables were expressed as frequencies and percentages with the Chi-square test. The Kaplan-Meier method was used to plot the survival curves and the differences between curves were compared using the log-rank test. Variables in the univariate analysis with a significant P value (P < .05) were selected for the multivariate analysis using the Cox hazard model using SPSS Version 25.0. Then the screened factors were used with the assignment method to calculate the prediction probability of an outcome and to draw the receiver operating characteristic curve (ROC). The area under the curve (AUC) and C-index were compared using GraphPad Prism 8. The nomogram and calibration curve were drawn to predict the probability and accuracy using R software.
Ethical Consideration
The research protocols received ethical approval from the Ethics Committee of Guangzhou Eighth People's Hospital, Guangzhou Medical University (No. 202210222).
Results
Baseline Characteristics of AR-BL Patients
A total of 34 AR-BL patients were collected in the present retrospective study. Detailed patient characteristics are shown in Table 1. Most patients were male, which accounts for 91.18% of the patients, and 3 (8.82%) were female. The age of these patients was with a median age of 42 years old and 4 patients were over 60 years old (11.76%). Overall, HIV and AR-BL diagnoses were synchronous in 27 cases (79.41%). The median time between lymphoma diagnosis and HIV diagnosis was 37 months, ranging between 9 and 121 months. The main route of HIV infection was through sexual transmission in 18 cases (52.94%), while in 15 cases (44.12%), it was unknown. Regarding the Ann Arbor stage, there were 2 stage I cases (5.88%), 15 stage II cases (44.12%), 2 stage III cases (5.88%), and 15 stage IV cases (44.12%). Twenty-three cases (67.65%) had an ECOG PS 2 to 4 and LDH was elevated in 73.53% of the patients (n = 25) with LDH > 3 × ULN in 32.35% (11/34) and > 5 × ULN in 14.71% (5/34). 29.41% (n = 10) of the patients had ≥ 2 EN sites of disease. The most common EN sites involved in diagnosis were the liver, lung, bone, and gastrointestinal tract. Thirteen patients (38.24%) had B symptoms, most of whom were loss of weight and fever. Nine cases (26.47%) had bulky disease. Regarding HIV characteristics, the median CD4 count was 176 cells per microliter (range, 25-597 cells per microliter) with 19 patients (55.88%) presenting with a CD4 count < 200 cells/µL. At the time of AR-BL diagnosis, HIV viral load was detectable in 67.65% and undetectable in 32.35%.
Demographical and Clinical Features at AR-BL Diagnosis.
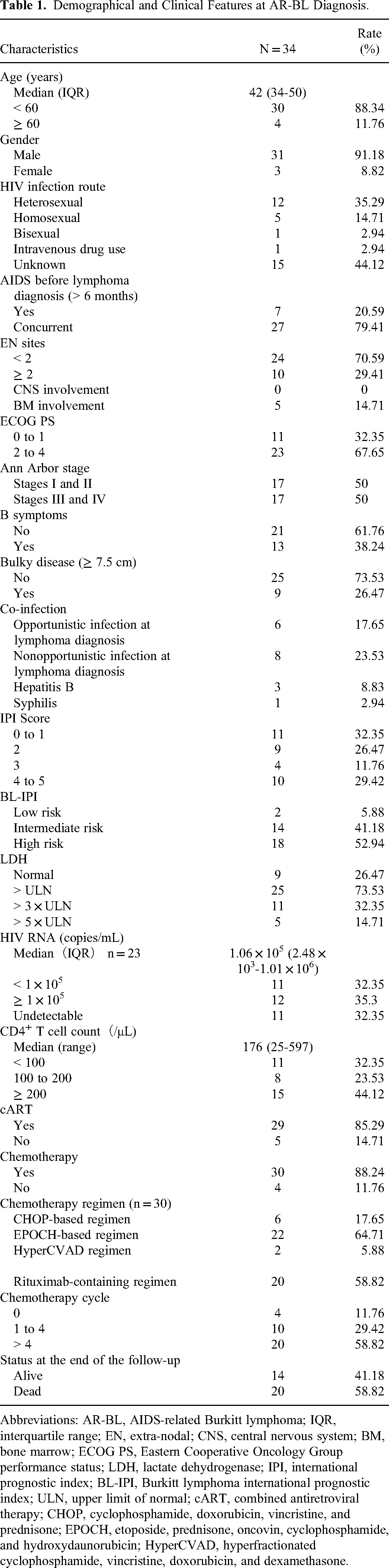
Abbreviations: AR-BL, AIDS-related Burkitt lymphoma; IQR, interquartile range; EN, extra-nodal; CNS, central nervous system; BM, bone marrow; ECOG PS, Eastern Cooperative Oncology Group performance status; LDH, lactate dehydrogenase; IPI, international prognostic index; BL-IPI, Burkitt lymphoma international prognostic index; ULN, upper limit of normal; cART, combined antiretroviral therapy; CHOP, cyclophosphamide, doxorubicin, vincristine, and prednisone; EPOCH, etoposide, prednisone, oncovin, cyclophosphamide, and hydroxydaunorubicin; HyperCVAD, hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone.
Treatment and Survival Analysis
In this study, most patients received chemotherapy and cART treatment. Twenty-nine patients received cART treatment (85.29%). The majority of the initial cART regimens were 2 NRTIs (nucleoside reverse transcriptase inhibitors) plus 1 INSTIs (intergrase strand transfer inhibitors) (n = 14), followed by 2 NRTIs plus 1 NNRTIs (nonnucleoside reverse transcriptase inhibitors) (n = 12), and 2 NRTIs plus 1 PIs (protease inhibitors) (n = 3). Thirty cases (88.24%) received chemotherapy, while 4 patients refused the treatment. Twenty-seven patients received both chemotherapy and cART treatment. Among the treated patients, 64.71% received an EPOCH-based regimen. Twenty cases (58.82%) received more than 4 cycles of chemotherapy. Twenty patients (58.82%) died while 14 patients (41.18%) were alive at the last follow-up (Table 1).
The median PFS and OS for AR-BL patients were 14 and 15 months, respectively. The overall 2-year PFS and OS rates were 40.50% and 36.18%, respectively (Figure 1A and B). The OS was better for patients who received chemotherapy compared with those who did not (Figure 1C, P = .0012), whereas no significant difference was observed between patients who received cART treatment and those who did not (Figure 1D, P = .8577). The differences in terms of OS between AR-BL patients who received no-cART, cART-only, and cART plus chemotherapy were significant (Figure 1E, P = .0003).
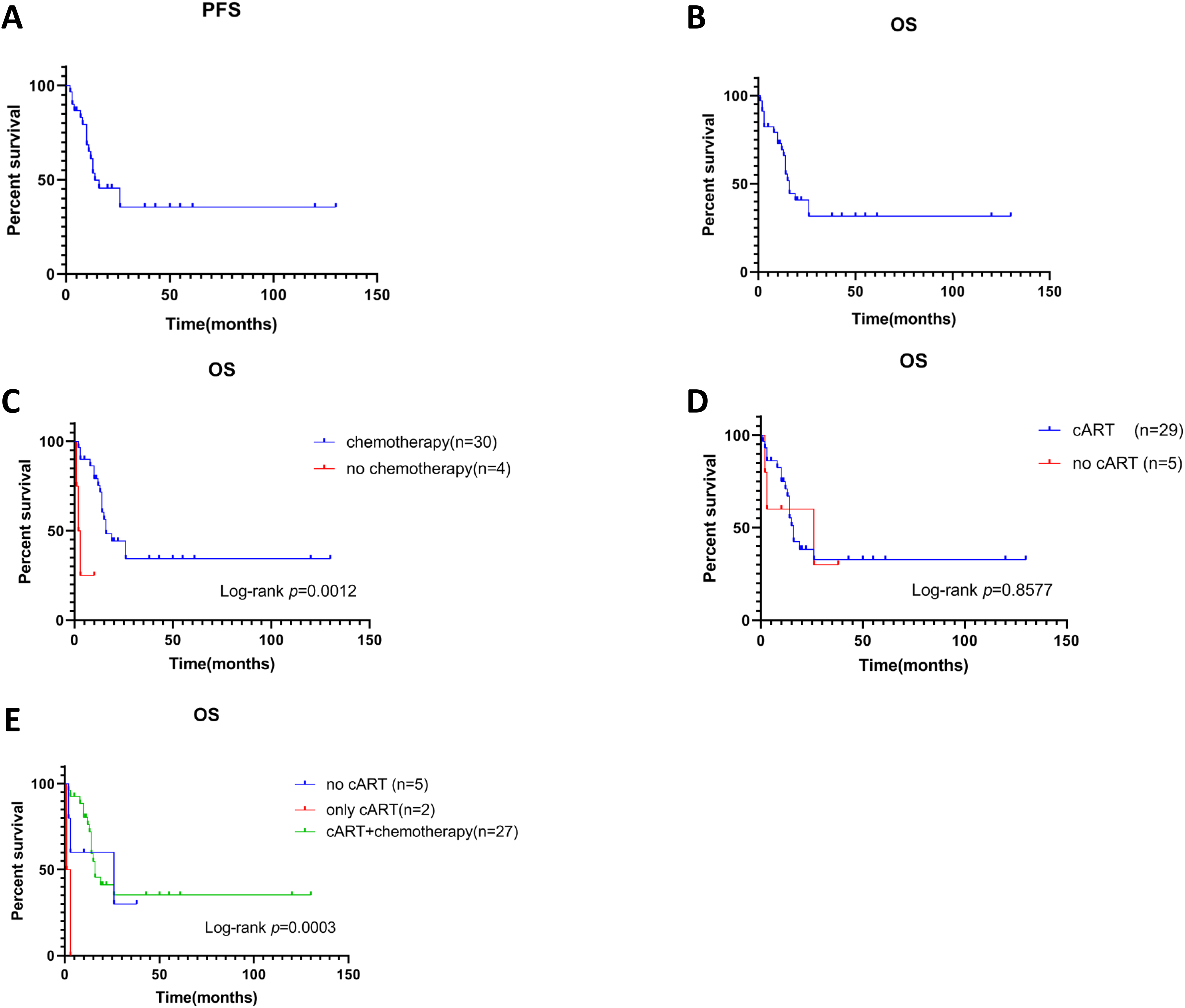
Survival and survival by received treatment. Kaplan-Meier curves of (A) PFS (n = 30) and (B) OS (n = 34) for AR-BL patients. Kaplan-Meier curves of different treatment (C-E) for OS in AR-BL patients. OS for AR-BL based on (C) chemotherapy (P = .0012) and (D) cART treatment (P = .8577); (E) OS for AR-BL patients with no cART, only cART, and cART plus chemotherapy (P = .0003).
When the relationship between treatment regimens and clinical outcomes was evaluated, differences in terms of OS and PFS between patients who received an EPOCH-based regimen, CHOP-based regimen, and HyperCVAD regimen were significant (Figure 2A, P = .0002; Figure 2B, P = .0158). Outcome using rituximab-containing regimens (n = 20) was not significantly superior compared to no rituximab-containing regimens (n = 10) for either OS (Figure 2C, P = .2366) or PFS (Figure 2D, P = .2096). According to treatment duration, median OS were 2.5, 12, and 19 months who received 0 cycle, 1 to 4 cycles, and > 4 cycles, respectively. In particular, patients who received > 4 cycles showed a significantly longer OS than those who received no chemotherapy (Figure 2E, P = .0036). Treatment with an EPOCH-based regimen was associated with longer OS (HR = 0.109; P = .001) and PFS (HR = 0.237; P = .009) (Table 2).

Survival by chemotherapy regimens and cycles. Kaplan-Meier curves of different chemotherapy regimens and cycles for OS and PFS in AR-BL patients. (A) OS and (B) PFS for AR-BL patients with EPOCH-based regimen, CHOP-based regimen, and HyperCVAD regimen (OS, P = .0002; PFS, P = .0158); (C) OS and (D) PFS for AR-BL patients with rituximab-containing regimen and without rituximab-containing regimen (OS, P = .2366; PFS, P = .2096); (E) OS for AR-BL patients with 0 cycle, 1 to 4 cycles, and > 4 cycles (P = .0036).
Multivariate Cox Hazard Regression for Risk Factors of OS and PFS in Patients With AR-BL.

Bold values means P < 0.05, which indicate the results were statistically significant.
Abbreviations: OS, overall survival; PFS, progressive free survival; HR: hazard ratio; CI, confidence interval; B symptoms, consisting of fever > 38°C, night sweats, and/or unintentional weight loss of > 10% of the body weight over a period of up to 6 months; Ref, reference; ECOG PS, Eastern Cooperative Oncology Group performance status; CHOP, cyclophosphamide, doxorubicin, vincristine, and prednisone; EPOCH, etoposide, prednisone, oncovin, cyclophosphamide, and hydroxydaunorubicin; HyperCVAD, hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone; LDH, lactate dehydrogenase; IPI, international prognostic index; BL-IPI, Burkitt lymphoma international prognostic index; ULN, upper limit of normal; cART, combined antiretroviral therapy; EN, extra-nodal; BM, bone marrow.
Independent Prognostic Factors Associated With AR-BL Patients
In univariate analysis, EN involvement (P = .03); ≥ 2 EN sites (P = .022); ECOG PS 2 to 4 (P = .007); LDH > 3 × ULN (P = .002); LDH > 5 × ULN (P = .022); and no chemotherapy (P = .006) were associated with shorter OS. EN involvement (P = .048), ECOG PS 2 to 4 (P = .017), and LDH > 3 × ULN (P = .003) were associated with shorter PFS in univariate analysis. In multivariate analysis, chemotherapy (HR = 0.075; 95% CI, 0.009-0.614) and ECOG 2 to 4 (HR = 4.738; 95% CI, 1.178-19.061) were independent prognostic variables of OS. Besides, only LDH > 3 × ULN (HR = 3.007; 95% CI, 1.036-8.73) was an independent predictor of worse PFS in patients with AR-BL (Table 2). Chemotherapy (P = .0012) and ECOG PS (P = .0019) had a significant impact on OS as shown in Figures 1C and 3A.
Prognostic Value of the GZ8H Model
Chemotherapy and ECOG PS were used to create a novel prognostic model stratifying the entire cohort into 3 groups with an assignment method. Consequently, 10 patients were assigned to the low-risk group (chemotherapy and ECOG PS 0-1), 21 patients to the intermediate-risk group (chemotherapy and ECOG PS 2-4 or no chemotherapy and ECOG PS 0-1), and 3 cases to the high-risk group (no chemotherapy and ECOG PS 2-4). The median OS in the low-risk, intermediate-risk, and high-risk groups were not reached, 14 and 2 months, respectively (P < .0001) (Table 3 and Figure 3B). According to the IPI and BL-IPI models, different risk groups stratified for OS are shown in Figure 3C and D. The novel risk prognostic model showed better distinguishing ability than both IPI and BL-IPI.
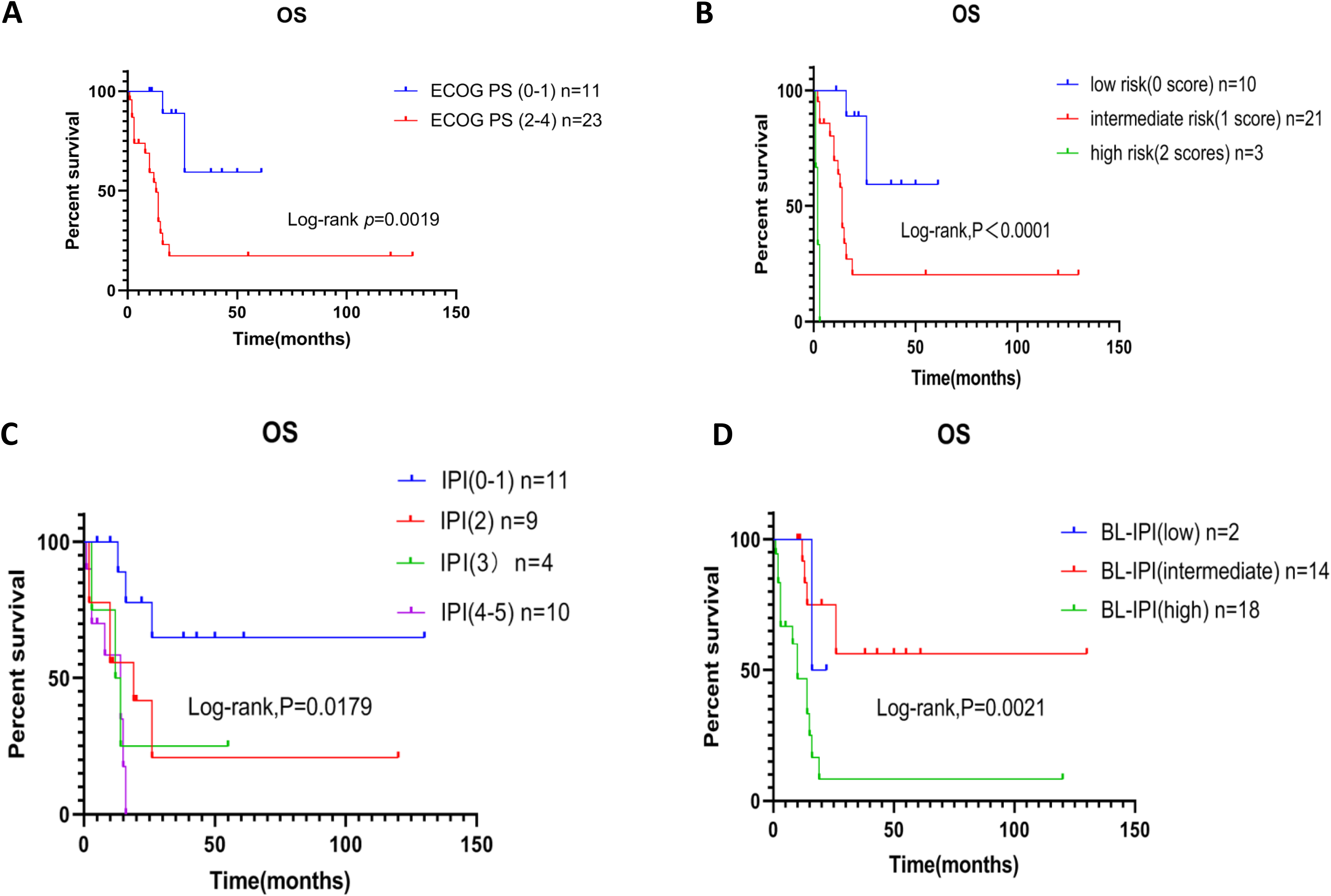
Survival by different prognostic stratification models. Kaplan-Meier curves of (A) ECOG PS (P = .0019) for OS in AR-BL patients; Kaplan-Meier curves of (B) GZ8H model (P < .0001), (C) IPI (P = .0179), and (D) BL-IPI (P = .0021) for OS in AR-BL patients.
GZ8H Model Consisting of Chemotherapy and ECOG PS.

Abbreviations: N, patients’ number; OS, overall survival; HR, hazard ratio; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; Low risk, EOCG PS 0-1 and chemotherapy; Intermediate risk, no chemotherapy and ECOG PS 0-1 or chemotherapy and ECOG PS 2-4; High risk, no chemotherapy and ECOG PS 2-4.
The predictive accuracy of the proposed prognostic model for OS, as measured by AUC was 0.713 (95% CI, 0.535-0.89), compared with 0.741 (95% CI, 0.566-0.916) of risk stratified by IPI, and 0.700 (95% CI, 0.517-0.883) of risk stratified by BL-IPI (Figure 4A). The C-index values based on the present GZ8H model, IPI, and BL-IPI were 0.76, 0.696, and 0.714, respectively (Figure 4B).

Prognosis of GZ8H model. Comparison of (A) AUC and (B) C-index in different prognostic risk stratification models; (C) nomogram to predict 1-, 3- and 5-year OS for AR-BL patients; (D) calibration curve of nomogram for predicting 1-and 3-year OS for AR-BL patients.
Nomogram of GZ8H Model
A nomogram is used to predict the probability of a particular outcome event occurring in a population with specific traits. 26 A nomogram to predict OS in patients with AR-BL was constructed based on chemotherapy and ECOG PS, which can predict the probability of OS at 1, 3, and 5 years (Figure 4C). The C-index of the developed nomogram was 0.884 in the entire cohort. The calibration plots were drawn to evaluate the consistency of the nomogram, which showed excellent agreement between the predicted and actual results of OS (Figure 4D).
Discussion
The study showed important real-world data on the clinical characteristics and outcomes of AR-BL in a center in China, analyzing the clinical features, outcomes of the specific treatment regimens, and the factors affecting prognosis in 34 AR-BL patients, thus providing information that would facilitate the research on HIV-associated BL.
Previous research presented that the median age of AR-BL patients ranged from 38 to 44 years old, 27 which is consistent with our data. Compared with non-AIDS related lymphoma, the age of onset is earlier in ARL patients, a finding related to the following factors: T cell immune surveillance dysfunction, cell pathway signaling disturbances, and persistent EBV infection after HIV infection, leading to the acceleration of disease development and progression. 28
Regarding treatment, in our cohort, most patients (64.71%) received an EPOCH-based regimen achieving a longer survival than patients treated with other regimens. The study conducted by Alderuccio et al 29 showed that the DA-EPOCH regimen could be selected for AR-BL patients with a lower risk of CNS involvement, while the benefits of this regimen in AR-BL require further study. Though Evens et al 30 revealed that CNS involvement acts as a vital adverse prognostic factor in BL, underscoring the critical role in adequate CNS staging in BL; in our series, no patients had CNS involvement, which could have been prevented by the intrathecal injection of methotrexate and cytarabine. In accordance with our findings, Sparano et al 31 revealed that R-EPOCH was an effective regimen in HIV-associated NHL with a CR rate of 73%. Additionally, the impact of rituximab was evaluated in a randomized study of 260 BL patients and was shown to be associated with improved PFS. 32 Although in our series the use of anti-CD20 treatment was not associated with a different outcome, this finding could be due to the small sample size.
Current evidence about prognostic factors in AR-BL patients is scarce. Alderuccio et al 29 showed that ECOG PS 2 to 4 (HR = 1.87; P = .007), LDH > 5 × ULN (HR = 2.09; P < .001), and > 1 EN (HR = 1.58; P = .043) were significant predictors of OS in AR-BL patients according to multivariate analysis. High LDH level (HR = 2.32, 95% CI 1.09-4.92, P = .029) and chemotherapy (HR = 0.16; 95% CI 0.06-0.39, P < .001) were significant independent predictors of OS in previous AIDS-related lymphoma research. 33 In the present study, univariate analysis showed that EN involvement ≥ 2 EN sites, ECOG PS 2 to 4, LDH > 3 × ULN or > 5 × ULN, and chemotherapy were significantly associated with the OS of AR-BL patients. However, only chemotherapy and ECOG PS were confirmed as independent predictors of OS by multivariate analysis. In fact, the significance of chemotherapy and ECOG PS could indicate that it's critical for patients to receive treatment in time and actively. As for PFS, EN involvement, ECOG PS 2 to 4, LDH > 3 × ULN were prognostically significant in univariate analysis, but only LDH > 3 × ULN was confirmed to be an independent predictor of worse PFS in the multivariate analysis.
Concerning the available prognostic models, some studies have shown that IPI is strongly correlated to the prognosis of AR-NHL.34,35 Age and LDH level are used for patients’ stratification according to the IPI scoring, but most patients with ARL are younger than 60 years old and generally have a high LDH level, which might affect their IPI scores. 36 In the present study, there were only 4 (11.76%) patients older than 60 years old, and most of the patients, 25 (73.53%) had elevated levels of LDH, and 17 (50%) were stratified into the low or moderate-to-high risk groups according to the IPI scoring system.
A large survey evaluated the characteristics, treatment patterns, and prognosis of adult patients with BL from 2009 to 2018 across 30 US cancer centers, including HIV-positive patients. Four baseline factors were selected for the prognostic model since they were independently associated with inferior survival (age ≥ 40 years, ECOG PS 2-4, LDH > 3 × ULN, and CNS involvement) to create a prognostic score which was called BL-IPI. 21
As shown by these efforts, new markers are needed to reliably assess the prognosis and identify the high-risk population for treatment tailoring. In our series, the currently available models showed some limitations. The Kaplan-Meier curve for OS based on IPI could not distinguish significantly different risk groups. In terms of AUC, IPI was found to have a larger area than the proposed GZ8H model and the C-index for the latter was better than IPI and BL-IPI. Moreover, it has to be considered that due to the lack of necessary validation data, IPI is not commonly used for HIV patients. Moreover, it is interesting to note that only patient- and lymphoma characteristics-, but not HIV-related factors have been found to be predictive of poor outcomes in the cART era.37-39
Regarding the biological relevance of the observed prognostic markers, elevated serum LDH has been recognized as a poor prognostic indicator for hematological malignancies and solid tumors for a long time; moreover, LDH is one of the IPI risk factors. 40 The metabolism of tumor cells has some peculiar characteristics, like energy production through glycolysis. LDH, as one of the key enzymes of the glycolytic pathway, is involved in this energy metabolism process, which can convert pyruvate to lactic acid. 41 LDH, found in the cytoplasm, is significantly elevated in patients with malignancies, including lymphoma. The elevation of LDH is closely related to tumor aggressiveness and tumor burden, and it is due to the rapid growth of tumor cells, which can easily lead to hypoxia in the tumor microenvironment. 40 LDH value is considered to be one of the most independent and important prognostic factors in patients with lymphomas and can be easily obtained during clinical practice. 42
ECOG PS score ranges from 0 to 5, it was ranged between 1 and 4 in our data. This is because 0 indicates normal mobility and 5 indicates death of the patient. In fact, the onset of this disease is insidious and the clinical symptoms are diverse, so the patient is already in the middle and advanced stages at the time of treatment, which can explain the ECOG PS in our study. Owing to the data we collected being limited, the calibration curve of the nomogram could only predict the 1- and 3-year OS for AR-BL patients.
We acknowledge that the present research has some important limitations. Firstly, it was a single-center retrospective study. Secondly, the sample size was relatively small, resulting in a low statistical power. Lastly, given the rarity of the disease, the sample size was not sufficient to create a validation cohort. Despite its retrospective nature and the mentioned limitations, this study is particularly valuable because it describes a population for which little data is currently available in the literature. Therefore, it's essential for a broader patient population to validate the results of this study.
Conclusion
In conclusion, we analyzed the clinical characteristics of a series of patients with AR-BL and designed a novel risk stratification model that might be easily used in clinical research after validation in an independent cohort.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 81911530169), CQMU Program for Youth Innovation in Future Medicine (W0202), Talent Program of Chongqing Health Commission, Chongqing Science and Technology Bureau (CSTB2022NSCQ-MSX0227 and cstc2022jxjl120022), and the Science and Technology Research Program of Chongqing Municipal Education Commission (Grant No. KJZD-K202300408). This work was also supported by Municipal and University (Hospital) joint funding project of Guangzhou Municipal Science and Technology Bureau (No. 202201020250 and No. 202201020253, No.2023A03J0796) and Medical Key Discipline Program of Guangzhou‐Viral Infectious Diseases (2021−2023).
Author Contributorship
HY designed the research and contributed the manuscript revision. HLH guided on clinical data collection and paper review. RQL performed the research, collected the data, analyzed the data, and wrote the paper. HZ and GYX participated in data collection, data review, and interpreted the data. YT, XPT, LZF, BLL, BL, JLG, LHL, and ZMC provided clinical suggestions. All authors contributed to the article and approved the final version of the manuscript.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
