Abstract
Although single-drug chemotherapy regimens were used as second-line chemotherapy for advanced lung squamous cell carcinoma (LSCC) patients, there are still no standard guidelines for second-line chemotherapy. The purpose of this study was to compare the efficacy and safety of docetaxel combined with nedaplatin or carboplatin in the second-line treatment of advanced LSCC patients. One hundred and ninety-six LSCC patients receiving docetaxel plus nedaplatin (DN, n = 96) or carboplatin (DC, n = 100) were retrospectively collected until disease progression or unacceptable toxicity. The progression-free survival (PFS), overall survival (OS), objective response rate (ORR), disease control rate (DCR), and adverse events (AEs) were analyzed in the two groups. The ORR was 18.8% versus 16.0%, and the DCR was 39.6% versus 34.0% in DN group and DC group (P > .05 and P > .05), respectively. The PFS was 5.3 versus 3.8 months, and the OS was 8.5 and 6.7 months in DN group and DC group (P = .013 and P = .404), respectively. The rate of digestive reaction and hepatotoxicity was similar in DN and DC groups, whereas more patients in DC group than in DN group suffered from leucopenia (P < .05). Docetaxel combined with nedaplatin is an effective regimen for advanced LSCC patients. Compared with a similar regimen with carboplatin, the response rate was similar; however, nedaplatin regimen shows some superiority as regards survival and some treatment side effect.
Introduction
Lung cancer is the most common cancer in China and the world, and it is also the main cause of cancer death. 1 More than 80% of lung cancer cases are nonsmall cell lung cancer (NSCLC), of which about 20% to 30% is lung squamous cell carcinoma (LSCC). 2 Due to the lack of drugs targeting oncogenic drivers, the median overall survival (OS) of the advanced LSCC patients is less than 1 year.3,4 Poor prognosis has become the main challenge in clinical treatment of patients with Stage III to IV LSCC. The standard first-line treatment for advanced NSCLC without a drive-gene mutation is a platinum-based chemotherapy.5,6 Some monotherapies, such as docetaxel and erlotinib, were used as second-line treatment for patients with advanced LSCC.7,8 However, there is still no standard protocol for second-line treatment.
Nedaplatin is a new second generation of organic platinum antitumor drugs. It is widely used in head and neck cancer, ovarian cancer, lung cancer, and so on.9–15 The anticancer mechanism of nedaplatin is similar to that of cisplatin. Its water solubility is about 10 times that of cisplatin. There is no need for large-dose hydration. The nephrotoxicity and gastrointestinal toxicity are lower than that of cisplatin. Moreover, nedaplatin has no complete cross-resistance with other platinum drugs. Nedaplatin can still obtain certain curative effect when platinum is drug-resistant. However, there is insufficient evidence for the efficacy of nedaplatin in second-line chemotherapy in advanced LSCC. Therefore, we performed this retrospective clinical study to assess the efficacy and safety of docetaxel combined with nedaplatin as a second-line treatment for advanced LSCC.
Materials and Methods
Patients
This study is a retrospective study. In total, 240 are advanced LSCC patients, 196 of whom had complete medical information and follow-up in Nanjing Brain Hospital from February 2020 to December 2021 (Figure 1). Patients with advanced LSCC were stratified into two groups, docetaxel plus nedaplatin (DN) group (96 cases) and the docetaxel plus carboplatin (DC) group (100 cases). Inclusion criteria are as follows: LSCC diagnosed by histopathology; Stage IIIB and IV patients who progressed after first-line chemotherapy; response was assessed according to response assessment criteria for solid tumors (Response Evaluation Criteria in Solid Tumors [RECIST] version 1.1); there were measurable target lesions before treatment; the systemic functional status score of the Eastern Cooperative Oncology Group performance status (ECOG PS) was 0–2; blood routine, hepatic, renal, or bone marrow functions were basically normal; life expectancy no shorter than 3 months; and patients completed at least 2 cycles of chemotherapy.
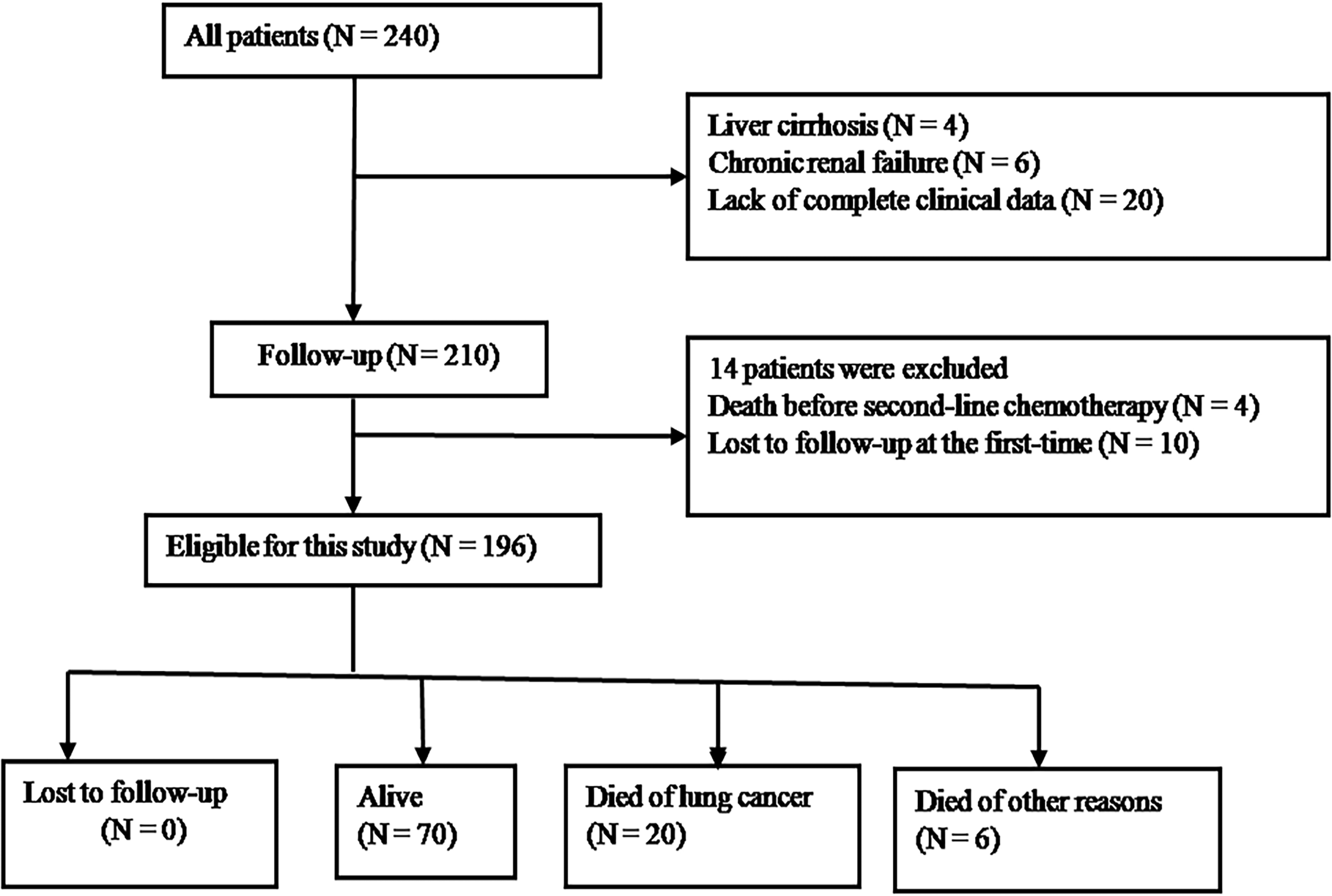
Flow of patients in the study.
The study was approved by the Ethics Committee of Affiliated Nanjing Chest Hospital, Nanjing Medical University (NJXK202026), Nanjing, China. All patients provided written informed consent before enrollment.
Treatment
Docetaxel (75 mg/m2) was given on the first day of each treatment cycle (1 cycle for 3 weeks). Nedaplatin group received 80-mg/m2 nedaplatin for at least 1 h. The control group received carboplatin (area under the curve = 5) infusion, which was completed within 2 h. The infusion was given on the first day of each cycle. In principle, treatment must be completed or can be stopped in cases of definite disease progression, and researchers believe that chemotherapy is ineffective or potentially harmful to patients.
Efficacy and Toxicity Assessment
Tumor response was assessed according to the RECIST version 1.1 at intervals of 4 weeks until disease progression.
The efficacy was divided into complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). (CR + PR)/(CR + PR + SD + PD) was calculated as objective response rate (ORR), and (CR + PR + SD)/(CR + PR + SD + PD) was calculated as disease control rate (DCR).
Weekly complete blood count and serum chemistry tests were performed at baseline and during treatment. Clinical adverse events (AEs) and changes in laboratory parameters were assessed according to the National Cancer Institute Common Terminology Criteria for AEs, version 4.0. All patients with tumor-associated AEs received supportive therapy.
Statistical Analysis
SPSS version 18.0 software was used (SPSS Inc, Chicago, IL, USA) for statistical analyses. OS was defined as the time from randomization until death or last follow-up. Progression-free survival (PFS) was defined as the time from randomization until progression or death whichever occurred first. Descriptive statistics were used to summarize the demographics and AEs. Fisher's exact test was used to compare ORR, DCR, and demographic data between subgroups. Student's t-test was used to compare means. Kaplan–Meier and log-rank tests were used to estimate PFS and OS. Multivariate Cox analyses were employed to estimate the factors affecting the efficacy of treatment. P < .05 was considered statistically significant.
Results
Clinicopathologic Characteristics
One hundred and ninety-six advanced LSCC patients who met the study criteria were divided into the DN group and the DC group. Males accounted for 70.8% and females 29.2% in DN group and 60.0% and 40.0% in DC group. The average age was 58.2 years in DN group and 59.5 years in DC group, respectively. More than 90% of LSCC patients were ECOG PS 0 to 1, nearly half of the patients were current or former smokers. After first-line chemotherapy, 33.3% of patients in DN group and 32.0% of patients in DC group showed responses. The SDs of DN group and DC group were 50.0% and 54.0%, respectively. There was no significant difference in clinical features between the two groups (Table 1).
The Clinical Characteristics of Advanced LSCC Patients.

CR, complete response; ECOG PS, Eastern Cooperative Oncology Group performance status; PD, progressive disease; PR, partial response; SD, stable disease.
Efficacy
Patients in DN group completed 348 cycles of chemotherapy, including 16 cases in 3 cycles and 10 cases in 2 cycles. Patients in DC group completed 368 cycles of chemotherapy, including 20 cases in 3 cycles and 16 cases in 2 cycles. The responses for patients were presented in Table 2. The CR was low in both groups, estimated as 2.1% in DN group and 0.0% in DC group. The PR was 16.7% in DN group and 16.0% in DC group. The ORR was improved, from 16.0 in DC group to 18.8% in DN group (P > .05). Moreover, the SD was improved, from 34.0% in DC group to 39.6% in DN group. The differences were not statistically significant (P > .05).
Efficacy of Advanced LSCC Patients.
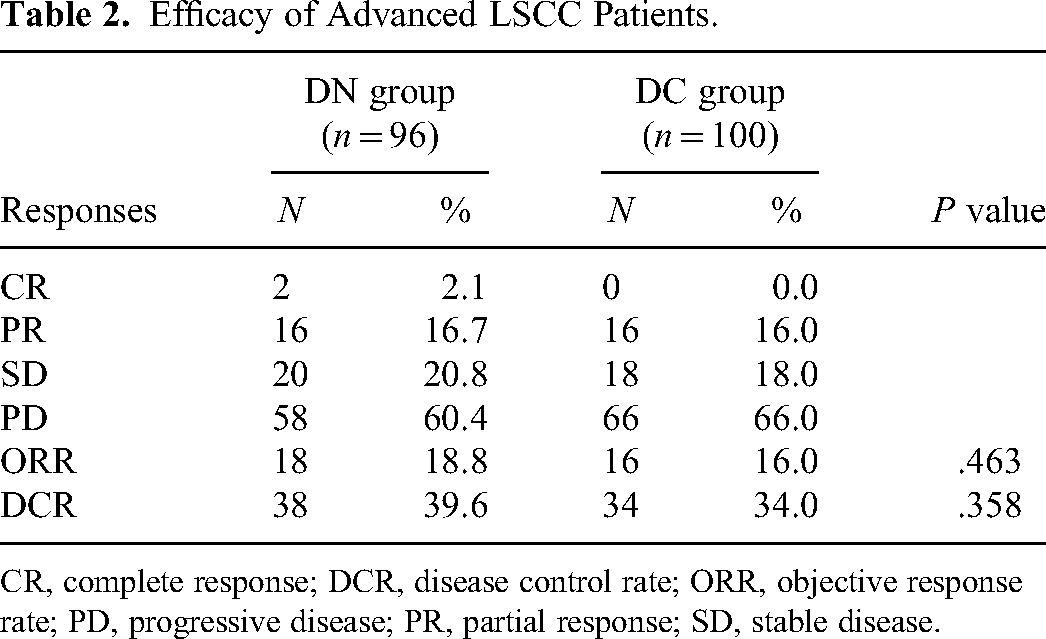
CR, complete response; DCR, disease control rate; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
The median PFS and OS were 5.3 and 8.5 months in DN group, compared with 3.8 and 6.7 months in DC group (Figure 2).

PFS and OS curves of DN (green) and DC (blue) groups. (A) Kaplan–Meier survival curves showing the PFS of patients in the DN group and DC group (median PFS, 5.3 vs 3.8 months; P = .013); (B) Kaplan–Meier survival curves showing the OS of patients in the DN group and DC group (median OS, 8.5 vs 6.7 months; P = .404). Abbreviations: DC, docetaxel plus carboplatin; DN, docetaxel plus nedaplatin; OS, overall survival; PFS, progression-free survival.
Multivariate Cox regression analysis showed that chemotherapy regimen (P = .043), disease stage (P = .015), and distant metastasis (P = .017) were independent risk factors for the PFS in LSCC patients (Table 3), reaffirming that docetaxel combined with nedaplatin improved the PFS of advanced LSCC patients.
Univariate and Multivariate Cox Analysis of OS and PFS in all Patients.

CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; PFS, progression-free survival; OS, overall survival.
Adverse Events
The overall incidence of Grade 3 and 4 AEs rates was 8.3% for DN and 12.0% for DC, respectively. The highest rate of Grade 3 or 4 AEs was leukopenia, accounting for 10.4% of total patients in DN group and 36.0% of total patients in the DC group, respectively. There was significant difference between the two groups (P < .05). The prevalence of other AEs, including Grade ≥3 AEs in the two groups was similar. In DN group, other Grade 3 and 4 AEs were nausea (4.2%), fatigue (2.1%), abnormal liver function (2.1%), and anemia (2.1%). In DC group, other Grade 3 and 4 AEs were thrombocytopenia (4.0%), anemia (6.0%), fatigue (4.0%), and nausea (6.0%). The differences were not statistically significant (P > .05) (Table 4).
AEs of Patients with Advanced LSCC.

AEs, adverse events; DC, docetaxel plus carboplatin; DN, docetaxel plus nedaplatin.
Discussion
In this study, we conducted a retrospective study to explore the efficacy and safety of docetaxel combined with nedaplatin in the second-line treatment of advanced LSCC. Our results indicated that the ORR was increased from 16.0% in DC group to 18.8% in DN group. We also found a significant improvement in PFS in the DN group, from 3.8 to 5.3 months. The median OS was also improved, from 6.7 months in DC group to 8.5 months in DN group. Multivariate analyses showed that docetaxel combined with nedaplatin was an independent beneficial factor of PFS. This study demonstrated that docetaxel combined with nedaplatin was safe and effective for LSCC patients.
Platinum combined with the third generation cytotoxic drug chemotherapy is the standard regimen for the treatment of advanced NSCLC and has achieved good therapeutic effect. However, due to the complex biological characteristics such as pathological type and differentiation of lung cancer, it is easy to relapse. Patients who relapse and progress after first-line treatment need to receive second-line treatment. After the first-line chemotherapy, the patient's physical condition and immunity are poor, and the drug tolerance is poor, which makes the second-line treatment relatively difficult. Therefore, it is very important to select appropriate drugs and regimen.
In order to find the best way of second-line treatment, docetaxel combined with other drugs has been used in second-line treatment. A clinical trial of second-line treatment for Stage IIIB and IV NSCLC showed that the survival risk of docetaxel combined with gemcitabine was reduced by 11% compared with docetaxel alone.
The median survival was 11.3 and 10.1 months for patients treated with docetaxel plus gemcitabine and docetaxel monotherapy, respectively, and the 1-year survival was 47% and 38%, and the median PFS was 3.1 and 2.1 months. 16 Another study comparing docetaxel alone with docetaxel combined with irinotecan in the second-line treatment of NSCLC relapsed after platinum chemotherapy showed that the effective rate, median survival time, and 1-year survival rate of docetaxel combined with irinotecan did not exceed docetaxel alone. 17
Nedaplatin is a cell cycle nonspecific platinum anticancer drug. It mainly reacts with nucleoside to form nucleoside platinum conjugate, binds to DNA in the same way as cisplatin, and inhibits DNA replication. Studies have shown that nedaplatin and other platinum drugs have no complete cross-resistance, low toxicity, and wide antitumor spectrum. It still showed a certain curative effect in some platinum-resistant patients.18–20
Previous studies have reported that nedaplatin combined with docetaxel as a second-line treatment for advanced NSCLC. 21 The results showed that the total effective rate of 20 patients is 50.0%, PFS is 4.35 months, and the 1-year survival rate is 29.0%, which is basically consistent with the results of this study. In this study, the DCRs of docetaxel combined with nedaplatin or carboplatin were 39.6% and 34.0%, respectively, and the PFSs were 5.3 and 3.8 months, respectively. All of the patients in this study were first-line platinum-containing regimen failures. The second-line treatment of docetaxel combined with nedaplatin achieved satisfactory DCR. Previous study has shown that for NSCLC patients who failed to be treated with platinum-containing regimen, the response rate of nedaplatin-containing regimen is 17%, indicating that the cross-resistance between nedaplatin and other platinum classes is not complete.
It is reported that the main side effects of nedaplatin are thrombocytopenia, leukocyte, and hemoglobin reduction, and other side effects are nausea, vomiting, and loss of appetite. 22 In this study, the main side effect of docetaxel combined with nedaplatin group was neutropenia, the incidence was lower than that of docetaxel combined with carboplatin group, the difference was statistically significant, and other toxic and side effects were similar to that of docetaxel combined with carboplatin. Lu et al. showed a higher incidence of Grade III or IV AEs and Grade 3 and 4 nausea and fatigue in the CD group compared with the ND Group. This is not consistent with our findings, possibly because our study population was second-line treatment and there were individual differences. 23
Our study has some limitations. Firstly, this is a single-center retrospective study with a small sample size, which needs to be verified in a larger prospective data set; secondly, the patient's pathological specimens were not tested for gene mutation. Next, we will conduct a study to explore the efficacy and safety of targeted drugs such as afatinib and erlotinib in the second-line treatment of advanced LSCC.
Conclusion
In conclusion, our results show that docetaxel combined with nedaplatin can be used as a second-line chemotherapy reagent for patients with advanced LSCC.
Footnotes
Abbreviations
Availability of Data and Materials
The data used to support the findings of this study are included within the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Ethics Committee of Nanjing Brain Hospital, Nanjing, China (NJXK202026). All patients provided written informed consent before enrollment.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Major Program of Nanjing Medical Science and Technique Development Foundation (ZKX16064).
