Abstract
Objective
To prepare S2.2/DOX magnetic nanoliposomes by combining the potential benefits of MNPs in MRI and the targeted performance of nano-drugs as an innovative method for integrated diagnosis and treatment of breast cancer (BC).
Methods
We created a S2.2-PEG-MZF/DOX molecular probe by using a lipid material to encapsulate PEG-MZF-NPs and doxorubicin (DOX), and a S2.2 aptamer to target MUC1 to conjugate with PEG-MZF/DOX nanoliposomes. The potential of probe for cell-specific targeting and magnetic resonance (MR) molecular imaging was evaluated by MR scanner and Prussian blue staining. Additionally, we explored the feasibility by using nanoliposome magnetic induction heating to interfere with MCF-7 (MUC1+) BC cells under the influence of an alternating magnetic field (AMF).
Results
PEG-MZF-NPs were biologically safe. The T2 relaxation rate of PEG-MZF-NPs was found to inhibit T2 signal in a concentration-dependent manner, and the T2 signal of the S2.2-PEG-MZF molecular probe in MCF-7 cells was significantly lower than that in PEG-MZF-NPs group. Moreover, the T2 signal reduction was more pronounced in MCF-7 cells than in the hepatoma cell line HepG2 (MUC1-), suggesting a strong MRI potential of the S2.2-PEG-MZF molecular probe. The S2.2-PEG-MZF/DOX nanoliposome was able to achieve the desired temperature range for tumor hyperthermia (42-44 °C) in vitro. The S2.2-PEG-MZF/DOX nanoliposome accompanied by magnetic fluid hyperthermia (MFH) could inhibit proliferation and invasion and induce apoptosis of MCF-7 cells. The effects of this approach were significantly higher than those observed in the other groups.
Conclusion
We successfully developed a novel technique for BC diagnosis and treatment using thermochemotherapy under the guidance of MR molecular imaging. This approach holds great potential for improving the management of this devastating disease in the future.
Keywords
Introduction
In 2020, there were approximately 2.26 million new cases of breast cancer (BC) reported globally, making it the most prevalent type of cancer and the leading cause of cancer-related deaths in women. 1 Early diagnosis and treatment are crucial in reducing the death rate of BC. It is therefore of great significance to explore new non-invasive, safe, sensitive and specific methods for early diagnosis and treatment of BC. Molybdenum target X-ray, ultrasound, and MRI are the most common imaging methods for BC diagnosis at present. However, these methods can detect the disease only when anatomical changes are present in the middle or advanced stage of the disease. 2 As a result, it is an urgent problem to be solved to enhance early BC diagnostic sensitivity and accuracy and to develop a detection approach with high safety and efficacy. Molecular imaging can diagnose BC at cellular, molecular and genetic levels, thus providing a new approach to early detection. 3
Magnetic nanoparticles (MNPs) have been demonstrated as efficient imaging agents for molecular imaging due to their unique physical and chemical properties, including good biocompatibility and magnetic properties. Compared with traditional contrast agents, MNPs have the advantages of good safety, long blood half-life, and targeted imaging as a new type of magnetic resonance (MR) contrast agent. 4 By targeting MNPs to tumor cells, Magnetic fluid hyperthermia (MFH) can minimize thermal damage caused by hyperthermia and improve the treatment efficacy. 5 Recent studies have shown that MFH can also significantly increase the cytotoxicity of chemotherapy drugs, resulting in a synergistic effect with hyperthermia. This approach offers a new strategy for the diagnosis and treatment of cancer. 6
Doxorubicin (DOX), a chemotherapy drug widely used in clinical practice, has shown significant efficacy against BC. But as DOX lacks selectivity for tumor cells, it may also cause harm to healthy cells, leading to toxic adverse effects such as myelosuppression, cardiotoxicity, nausea and vomiting. 7 To address these issues, new targeted chemotherapies that can reduce systemic adverse effects and improve local efficacy are required. Consequently, changing the DOX dose form and administration mode is clinically significant. Apart from it the local administration route, persistent drug release, target localization and dosage form must also be taken into consideration. Several studies have demonstrated the potential of targeted nanotechnology-based chemotherapy for BC treatment. 8
Researchers have also tried to include more properties by modifying the surface of MNPs for targeted drug delivery and thermotherapy.9,10 In recent decades, coupling of biomolecules such as antibodies, aptamers or peptides to their surfaces can enhance the targeting of MNPs, allowing them to target and enrich in tumor cells. Mucin 1 (MUC1) is a transmembrane protein specifically and highly expressed in various BC tissues.11–13 MUC1 plays a crucial role in the development and prognosis of BC, 14 making it an important target for imaging and therapeutic interventions in BC. 15 Nucleic acid aptamers (Apts) are short oligonucleotide fragments, including single-stranded DNA or small molecule RNA, which have shown great potential for targeted diagnosis and treatment of tumors.16,17 Compared with traditional antibodies, Apts have numerous advantages, such as easy screening and synthesis, lower molecular weight, high stability, low immunogenicity, and rapid blood clearance.18–20 A recent study showed that a single-stranded DNA aptamer called S2.2 (5′-GCAGTTGATCCTTTGGATACCCTGG-3′) could specifically bind to MUC1 for BC diagnosis and targeted therapy. 21
Compared to other nanoparticles, nanoliposomes exhibited distinct advantages in terms of stability, biocompatibility, and controlled drug release. In our previous study, remarkable progress has been made in using MZF nanoliposomes for tumor diagnosis and treatment.22–24 The present study focused on preparing S2.2-PEG-MZF-NPs molecular probe (S2.2-PEG-MZF) by integrating MZF MNPs after PEG modification and S2.2 aptamer (targeted to MUC1) to conduct MRI, thus making it more sensitive and accurate to diagnose early BC. Meanwhile, MZF nanoparticles were utilized as drug carrier to construct the S2.2-PEG-MZF/DOX nanoliposomes. Besides, we applied hyperthermia-related magnetic parameters to conduct thermochemotherapy together with MRI on BC. During the progress, MUC1 was used as the target and PEG-MZF-NPs were used as the drug carrier and magnetic hyperthermia medium. Thus, diagnosing and treating BC guided by MRI were conducted. In this study, we attempted to provide a new integrated approach for BC diagnosis and treatment by combining MRI guided DOX chemotherapy and magnetic induction hyperthermia (Figure 1).

Schematic illustration of S2.2-PEG-MZF/DOX preparation and application.
Materials and Methods
Main Materials and Cells
Materials used in this study were DSPE-PEG2000, DSPE-PEG-MAL and MZF (Ruixi Biological Technology, Xi’an, China); Annexin-V-FITC apoptosis kit (BD, San Diego, USA); anti-BCL-2 (3498S, 1:1000), anti-Bax (5023S, 1:1000) and anti-MMP9 (3852S, 1:1000) antibodies (Cell Signaling Technologies, Danvers, USA); DOX, S2.2-Aptamer, secondary antibodies, cell counting kit-8 (CCK-8) kit, Prussian blue staining kit, crystal violet and the other reagents (Sangon Biotech, Shanghai, China).
Human BC cell line MCF-7 (accession number: SCSP-531), human hepatoma cell line HepG2 (accession number: SCSP-510), and Mouse fibroblast cell line L-929 (accession number: SCSP-5039) were obtained from Shanghai Institute of Cell Science, Chinese Academy of Sciences. The cells were cultured in MEM/DMEM medium containing 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin and in a standard condition (37 °C, 5% CO2) incubator.
Preparation and Characterization of NPs
Preparation of PEG-MZF-NPs
The MZF-NPs (Mn-Znferrite-nanoparticles) were prepared using a hydrothermal method by dissolving ferric nitrate hydrate (5 mmol), manganese chloride (1.25 mmol), and zinc chloride (1.25 mmol) in 20 mL deionized water, followed by addition of 5 mL trimethylolamine and 20 mL sodium hydroxide solution. The mixture was stirred for 15 minutes and then reacted at 200 °C for 5 hours. After cooling to room temperature, the mixture was centrifuged three times and washed. The final product was dispersed in deionized water and kept aside for later use. To improve the stability of MZF-NPs, their surface was modified with PEG-MAL to obtain (PEG-MAL)-MZF-NPs, which is referred to as PEG-MZF-NPs hereafter. The surface conjugation step using PEG-MAL can be described in detail as follows: Sodium citrate solution was added to 100 mg pre-prepared MZF-NPs and sonicated for 2 minutes. Following this, the resultant mixture underwent centrifugation, allowing the removal of the supernatant and subsequent washing of the MZF-NPs three times with deionized water, resulting in the production of carboxylated MZF-NPs. To activate the carboxylated MZF-NPs, a combination of EDC and NHS reagents was introduced, and the ensuing reaction was allowed to proceed for a duration of 2 hours. Subsequently, 100 mg of solid NH2-PEG2000-Mal powder was accurately weighed and subjected to an overnight reaction with the activated MZF-NPs. The resulting mixture was then subjected to centrifugation (12,000 rpm, 10 minutes) and washed three times, ultimately yielding the desired precipitate.
Preparation of S2.2-PEG-MZF
The covalent bond was formed by reacting the Mal group on NH2-PEG2000-Mal and the sulfhydryl group (-SH) modified at the end of the S2.2-aptamer. First, 12.6 mg PEG-MZF-NPs and 9OD S2.2-aptamer were mixed and reacted at room temperature overnight. The resulting mixture was then dialyzed in deionized water to remove any free aptamer, and the precipitate was obtained by centrifugation at 12,000 rpm for 10 minutes. The precipitate was then referred to as S2.2-PEG-MZF, which is the S2.2-PEG-MZF-NPs molecular probe.
Characterization of NPs
The morphology of the PEG-MZF-NPs and PEG-MZF/DOX nanoliposomes were observed by transmission electron microscopy (TEM). To do this, an appropriate amount of anhydrous ethanol was added to the sample, ultrasonicated for 15 minutes, and then dropped onto a copper mesh with a membrane. The hydrodynamic diameter and surface Zeta potential of the test samples were detected by dynamic light scattering (DLS) after placing 1 mL sample in the cuvette. Fourier transform infrared spectroscopy (FTIR, Waltham, MA, USA) was used to validate the coupling of surface functional groups of NPs before and after modification. To evaluate the stability of the prepared NPs, they were dissolved in distilled water, MEM cell medium, and phosphate buffer solution (PBS, pH = 7.3), and placed in a 4 °C refrigerator for 24 hours. Changes in water solubility and stability were observed, and the delamination and precipitation of the NPs were recorded daily for 2 months.
Biocompatibility Assessment of S2.2-PEG-MZF
Cytocompatibility assessment involved sterilizing S2.2-PEG-MZF and diluting them with MEM culture medium to achieve iron concentrations of 30 µg/mL, 60 µg/mL, and 90 µg/mL as three experimental groups. The control group was intervened with MEM culture medium only. After 24 hours, the growth state of L929 cells was observed in each group and recorded using a microscope. Then, 10 μL CCK-8 solution was added to each well. After 1-hour incubation, the plate was measured at 450 nm using a microplate reader. The relative growth rate (RGR) of cells was calculated using the following formula: RGR (%) = OD value of the experimental sample/OD value of the control group × 100%. RGR values were converted into 6-level toxicity grades. Results of 0 to 1 level grades (RGR ≥ 75) were considered qualified, while 2-level grade (RGR ≥ 50) required evaluation in combination with cell morphology assessment. Results of 3–5 level grades (RGR < 50) were considered unqualified.
The biocompatibility of S2.2-PEG-MZF was assessed by hemolysis test and cytocompatibility assessment. For the hemolysis test, fresh anticoagulant blood was diluted with normal saline (NS) solution at a 1:1.5 ratio. NS was used as a negative control (NC), and distilled water was used as a positive control (PC). S2.2-PEG-MZF-NPs were diluted with NS to an iron concentration of 30 µg/mL, 60 µg/mL, and 90 µg/mL, and 10 mL of the diluted NPs was added to the test tube as the experimental group. Each group had three parallel test tubes. After preheating the test tubes in a 37 °C water bath, 0.5 mL diluted anticoagulant was added to each test tube. After 1-hour incubation, the test tube in each group was centrifuged (3000 rpm, 5 minutes), and the absorbance of the supernatant was measured at 545 nm using a Synergy HT microplate reader (BioTek, USA). The hemolysis rate was calculated as follows: Hemolysis rate (%) = (OD value of the tested sample − OD value of the NC group) / (OD value of the PC group − OD value of the NC group) × 100. A hemolysis rate of less than 5% was considered non-hemolytic.
MRI Potential of S2.2-PEG-MZF
S2.2-PEG-MZF samples were prepared by diluting them with ultrapure water to create magnetic fluid solutions with Fe concentrations of 0.5 µM (≈30 µg/ mL), 1 µM (≈60 µg/ mL), 1.5 µM (≈90 µg/ mL), 2 µM (≈120 µg/ mL) and 2.5 µM (≈150 µg/ mL). Pure water was used as the control group. Each sample was placed in a 5 mL tube and fixed with agarose for later use. A Siemens 3.0 T MRI scanner was used to scan and photograph the NPs at different Fe concentrations. The Axial FSE T2WI sequence scanning parameters were set as follows: TR 5000 ms, TE 146.80 ms, matrix 256 × 256, field of view 160 mm × 160 mm, slice thickness 3.0 mm, and slice spacing 1.5 mm. Regions of interest (ROI), which were the same size and larger than 20 mm2, were drawn in the central region of the images obtained from each group. After scanning, the T2 value of each image was measured to obtain the relaxation rate of each concentration sample.
In Vitro Targeting Test of S2.2-PEG-MZF
The sterilized solutions of PEG-MZF-NPs and S2.2-PEG-MZF were diluted with DMEM complete culture medium to an iron concentration of 90 µg/mL, which was used as the culture medium of the experimental group, and the DMEM complete culture solution was used as the control. These solutions were added to well-grown MCF-7 (MUC1+) and HepG2 (MUC1-) cells, respectively. After 24 hours, the cells were collected, fixed using agarose, and then scanned in a Siemens 3.0 T magnetic resonance scanner using the T2WI sequence scanning parameters described above. The T2WI signal differences were observed.
An equal concentration of sterilized PEG-MZF-NPs and S2.2-PEG-MZF solution was added to MCF-7 (MUC1+) and HepG2 (MUC1-) respectively. Cells were incubated for 6 hours, and then washed 3 times with PBS, fixed with 1 mL of 4% paraformaldehyde for 30 minutes, washed again 3 times and stained with 1 mL Prussian blue staining solution for 30 minutes. Subsequently, they were washed 3 times again, stained with nuclear fast red solution for 5 min, washed 3 times with PBS, and finally observed under an inverted microscope.
Magnetic Hyperthermia Experiment of S2.2-PEG-MZF in AMF
Different concentrations of S2.2-PEG-MZF (30, 60, 90, and 120 µg/mL) were each mixed with 5 mL solution and placed in test tubes separately. The test tubes were then subjected to an alternating magnetic induction instrument (SPG-10 (A)-11, Shenzhen, China) for 1 hour, with the magnetic induction coil positioned about 0.5 cm above the bottom of the test tube under the output frequency of 230 kHz and the output current of 30 A. The temperature was recorded every 10 min to plot the heating curves of S2.2-PEG-MZF with different concentrations. Based on the heating curves, an appropriate Fe concentration was selected for use in subsequent experiments.
Preparation and Characterization of S2.2-PEG-MZF/DOX
First, a membrane was formed by dissolving DOX (2.5 mg), DSPE-PEG2000 (1.5 mg), and phosphatidylcholine (15 mg) in chloroform (3 mL) and drying with a rotary evaporator. The resulting sample was then mixed with 2 mL deionized water to form uniform-sized liposomes through ultrasonication. PEG-MZF-NPs were added, and ultrasonication was performed for 30 minutes until the potential remained unchanged. The resulting mixture was dialyzed to obtain PEG-MZF/DOX nanoliposomes (briefly referred as PEG-MZF/DOX), which was then mixed with the S2.2-aptamer and reacted overnight at room temperature. The mixture was dialyzed to remove the free S2.2, and the S2.2-PEG-MZF/DOX nanoliposomes (which were referred to as S2.2-PEG-MZF/DOX hereafter) were obtained by centrifugation (12,000 rpm for 10 minutes). Finally, their hydrodynamic size and surface zeta potential were measured using DLS.
DOX Loading and Aptamer Coupling Efficiency of S2.2-PEG-MZF/DOX
The efficiency was measured by a UV–vis spectrophotometer. First, a solution of DOX was prepared at various concentrations (3.125, 6.25, 12.5, 25, and 50 μg/mL). The absorbance of DOX at 485 nm was measured to generate the standard concentration (µg/mL) curve. Next, 120 mg S2.2-PEG-MZF /DOX nanoliposomes containing 2.5 mg DOX were diluted fourfold and homogenized with Triton X-100 to obtain the supernatant. The absorbance of the supernatant was measured, and the mass of DOX in the sample was calculated using the standard curve. To construct the S2.2 standard curve, a solution of S2.2 with concentrations of 2.0625 µg/mL, 4.125 µg/mL, 8.25 µg/mL, 16.5 µg/mL, 33 µg/mL, and 66 µg/mL was prepared, and the absorbance of S2.2 was measured at 260 nm. Next, 9 OD (approximately 9 × 33 µg) of the S2.2 was reacted to obtain S2.2-PEG-MZF/DOX nanoliposomes. The absorbance of the supernatant, which was diluted fourfold, was measured. Finally, the mass of the S2.2 in the sample was determined using the standard curve. The drug loading efficiency and the aptamer coupling efficiency of the S2.2-PEG-MZF/DOX were calculated according to the following formula, respectively: Drug loading capacity (%) = (amount of DOX in NPs/total amount of NPs) × 100; Aptamer coupling efficiency (%) = (amount of S2.2 in NPs/total amount of S2.2 added) × 100
In Vitro Antitumor Effects of NPs
Cell Viability Assay
To determine the effect of S2.2-PEG-MZF/DOX on MUC1-overexpressing MCF-7 cells, cell viability was assessed using the CCK8 assay kit by seeding cells overnight in a 96-well plate in four groups: Group A, serving as the negative control using DMEM medium; Group B, receiving DOX; Group C, intervened with S2.2-PEG-MZF/DOX (Fe concentration of 90 µg/mL); Group D, intervened with S2.2-PEG-MZF/DOX accompanied by MFH (Fe concentration of 90 µg/mL). The cells in group D were placed 0.5 cm above the high-frequency magnetic induction flat coil with a power of 4 kW, a frequency of 230 kHz, and an output current of 30 A for 45 minutes and then cultured for 24 hours. Afterward, 10 µL CCK8 reagent was added to each well and incubated for an additional 2 hours at 37 °C. The OD value of the supernatant was measured at 545 nm using the microplate reader (Synergy HT, BioTek, USA). The rate of cell viability (%) = OD value of the experimental group/OD value of the control group × 100.
Cell Apoptosis Detection
Apoptosis analysis was performed using Annexin-V-FITC Apoptosis Detection Kit (Beyotime, Shanghai) according to the manufacturer's instructions. The cells were grouped as described above. After being intervened 24 hours, cells were collected and mixed with 5 μL Annexin-V and 2.5 μL propidium iodide (PI), and then incubated at room temperature in the dark for 15 minutes. The apoptosis rates of different groups were checked with BD FACS and analyzed by FlowJo X software (Franklin Lake, NJ, USA).
Cell Invasion Assay
The invasion of MCF-7 cells was conducted using transwell chambers coated with Matrigel (8 μm pore; Millipore, Billerica, USA). Approximately 5 × 104 pretreated MCF-7 cells were suspended in 100 µL serum-free DMEM medium, and seeded into upper chamber. Simultaneously, the bottom chamber filled 500 µL DMEM medium containing 20% FBS as chemoattractant. After 24-hour incubation, cells were fixed by 4% paraformaldehyde and then stained with 0.1% crystal violet for 20 minutes. The invaded cells were observed under the light microscope (Zeiss, Jena, Germany).
Western Blot Assay
Total proteins from MCF-7 cells were extracted with RIPA buffer and the concentration was quantified via a BCA Kit (Beyotime, Shanghai, China). Subsequently, 20 µg of the total protein was loaded onto 10% SDS-PAGE and transferred to PVDF membranes (Millipore, America). After being blocked via 5% skim milk, the membranes were incubated with corresponding primary antibodies at 4 °C overnight, washed with PBST, and then incubated with HRP conjugated secondary antibody at room temperature for 1 hour. Finally, target protein bands were developed using a chemiluminescence system and quantified with Image J software (NIH, Bethesda, MD, USA), using GAPDH as control.
Statistical Analysis
All statistical analyses were performed using GraphPad Prism 8.0 software. Quantitative data were exhibited as mean ± standard deviation (SD). Two-group comparisons were assessed by the Mann–Whitney
Results
Characterization of NPs
The diameters of PEG-MZF-NPs and PEG-MZF/DOX nanoliposomes both averaged approximately 6 nm, and their size was uniformly distributed, as seen in Figure 2A. FTIR spectra indicated the presence of the carbonyl group (C=O) stretching vibration and C–N stretching vibration on Dox at 1621.8 cm−1 and 1201.4 cm−1, respectively. The absorption band at 2925 cm−1 was attributed to the stretching vibrations of C-H bonds of S2.2-aptamer (Figure 2B). The successful conjugation of the DNA aptamer S2.2 with negatively charged phosphate residues was confirmed by DLS results, which showed a slight increase in size and a change in zeta potential from −9.97 mV to −15.73 mV in nanoliposomes conjugated with the aptamer (Figure 2C and D). The successful synthesis of S2.2-PEG-MZF/DOX was confirmed by these results. The S2.2-coupling efficiency was 84.98%, and the DOX-loading efficiency was 1.18%.

Characterization of nanoparticles. (A) TEM of nanoparticles. (B) FTIR spectra of (a) PEG-MZF-NPs, (b) DOX, (c) PEG-MZF/DOX nanoliposomes, and (d) S2.2-PEG-MZF/DOX nanoliposomes. C. Hydrated particle size of nanoparticles (mean ± SD;
Stability and Biocompatibility of NPs
The stability of PEG-MZF-NPs magnetic NPs was evaluated by observing and recording their appearance over the course of a month when they were stored in a 4 °C refrigerator. No sign of layering or precipitation was observed, indicating that the NPs were stable under these storage conditions.
The growth of L-929 cells co-cultured with S2.2-PEG-MZF at different Fe concentrations (30, 60, 90 μg/mL) was observed under an inverted microscopy after 24 hours, and no significant difference was observed compared with the control group (Figure 3A). According to the toxicity evaluation criteria, S2.2-PEG-MZF at all three Fe concentrations (30 μg/mL, 60 μg/mL, and 90 μg/mL) showed grade 1 cytotoxicity after co-cultivation with L-929 cells, meeting the requirements for medical biomaterials (Figure 3B). The results of the hemolysis test are presented in Figure 3C. The hemolysis rate of S2.2-PEG-MZF at a Fe concentration of 90 µg/mL is only 3.4%, which is less than the acceptable limit of 5% for medical biomaterials.
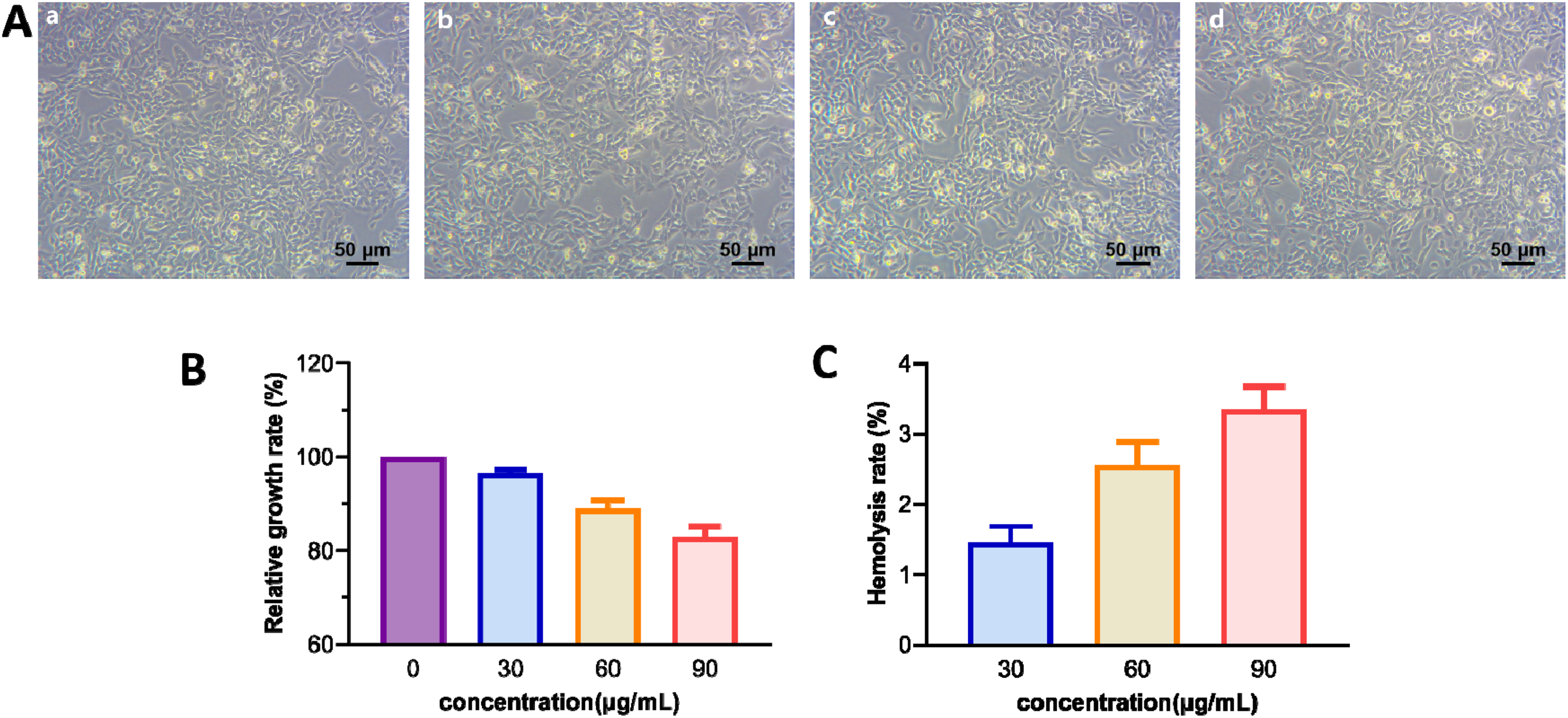
Stability and biocompatibility of nanoparticles. (A) Morphological observation of L-929 cells after co-culture with S2.2-PEG-MZF-NPs (a, control group; b-d, Fe concentration of 30, 60, and 90 µg/mL). (B) Relative growth rate (mean ± SD;
MRI and MFH Potential of S2.2-PEG-MZF
The MRI was performed on different Fe concentrations of S2.2-PEG-MZF and ultrapure water based on set parameters. A stepwise decrease in T2 signal with increasing S2.2-PEG-MZF concentration, at a relaxivity of 331.45 mM−1s−1, was observed (Figure 4A and B). To induce hyperthermia, different Fe concentrations (30, 60, 90, and 120 µg/mL) of S2.2-PEG-MZF were subjected to a high-frequency alternating magnetic field (AMF) with a rated power of 4 kW, a frequency of 230 kHz, and an output current of 30 A at room temperature. Within 30 minutes, the temperature of S2.2-PEG-MZF increased rapidly. At the Fe concentration of 90 µg/mL, the temperature of S2.2-PEG-MZF increased gradually and finally stabilized at around 42 to 44 °C, an ideal temperature for cancer hyperthermia treatment (Figure 4C).

MRI and MFH potential of S2.2-PEG-MZF. (A) MR images of S2.2-PEG-MZF with different Fe concentrations. (B) The measured lateral relaxation time (R2) of S2.2-PEG-MZF is 331.45 mM−1s−1. (C) In vitro magnetic induction heating curve of S2.2-PEG-MZF with different Fe concentrations.
In Vitro Targeting Test of S2.2-PEG-MZF
The effects of PEG-MZF-NPs and S2.2-PEG-MZF on MCF-7 and HepG2 cells were investigated by MR imaging (Figure 5A). Both molecular probes were observed to inhibit the T2 of MCF-7 and HepG2 cells to some extent when compared with the control group. The T2WI signal of MCF-7 cells was more significantly inhibited by S2.2-PEG-MZF as compared with HepG2 cells (

In vitro targeting test of S2.2-PEG-MZF. (A) Images obtained by magnetic resonance scanning after PEG-MZF-NPs and S2.2-PEG-MZF were incubated with MCF-7 cells and HepG2 cells (mean ± SD;
After staining with Prussian blue and nuclear fast red and 6-hour incubation of MCF-7 cells with S2.2-PEG-MZF at the Fe concentration of 90 µg/mL, significantly more blue particles were observed in thecytoplasm as compared with PEG-MZF-NPs. On the other hand, fewer blue particles were observed in the cytoplasm of HepG2 cells incubated with S2.2-PEG-MZF, and there was no significant difference in the PEG-MZF-NPs group (Figure 5B).
In Vitro Antitumor Effects of S2.2-PEG-MZF/DOX
Cell Viability Assay
After the successful synthesis and characterization of S2.2-PEG-MZF/DOX, inhibition of MCF-7 cell viability after intervention of S2.2-PEG-MZF/DOX accompanied by MFH was determined in vitro. Compared with the control group, varying degrees of toxicity and MCF-7 cell activity inhibition were observed in DOX, S2.2-PEG-MZF/DOX and S2.2-PEG-MZF/DOX/MFH group, among which the cell viability of the S2.2-PEG-MZF/DOX/MFH group was only (39.75 ± 1.26) %, which was significantly lower than the other groups (
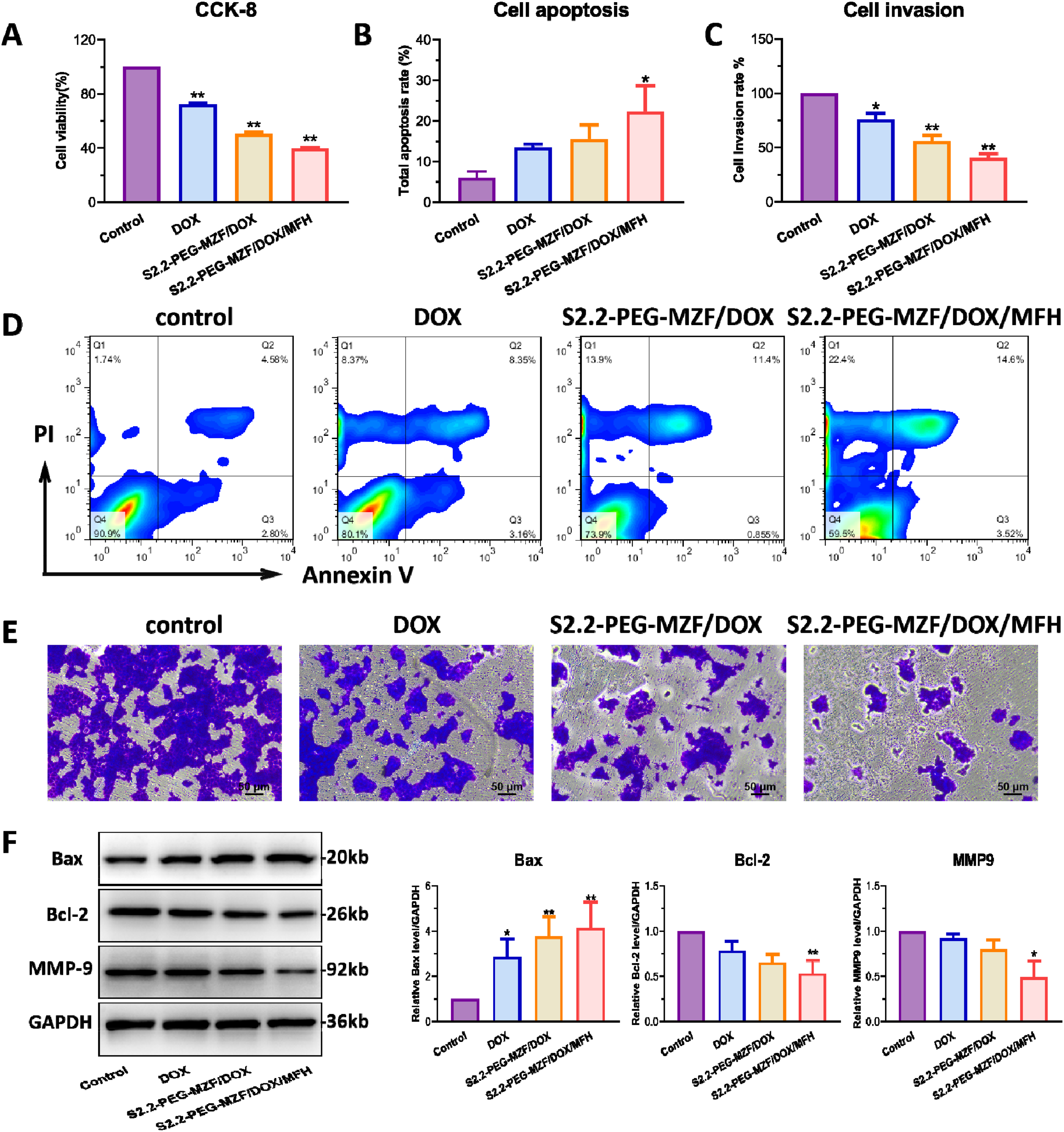
In vitro anti-tumor effects of S2.2-PEG-MZF/DOX. (A) Cell viability of MCF-7 cells after 24-hour intervention (mean ± SD;
Cell Apoptosis Detection
To further confirm the inhibitory effects of S2.2-PEG-MZF/DOX on MCF-7 cells under AMF, flow cytometry was used to detect apoptosis induction. As shown in Figure 6B and D, the apoptosis rate of the S2.2-PEG-MZF/DOX group was (16.8 ± 7.83) %, which was significantly higher than the single DOX chemotherapy group (
Cell Invasion Assay
After intervening MCF-7 cells with the different experimental groups, cell invasion ability was assessed via the transwell system. The cell invasion rate in S2.2-PEG-MZF/DOX/MFH group was lower than that in S2.2-PEG-MZF/DOX and DOX groups [(40.56 ± 6.58) % versus (55.65 ± 9.60) % and (75.63 ± 10.45) %,
Western Blot Assay
The expressions of apoptosis and invasion related proteins were examined by Western blot. As shown in Figure 6F, the expression of MMP9 and Bcl-2 was significantly decreased in each group, while the expression of Bax was significantly increased (
Discussion
Thanks to the continuous advancements in nanotechnology in recent years, nanomedicine has made significant progress in cancer diagnosis and treatment. This article focused on the successful construction of nanomaterials as the key step of nanomedicine realization. An ideal nanomaterial should possess certain characteristics, such as small size, good biocompatibility, and a large surface area. 25 As an important type of nanoparticles, Magnetic nanoparticles (MNPs) have several additional merits. According to several studies, modification of MNPs with polyethylene glycol (PEG) can improve their stability and prolong the cycle half-life. They can be easily modified, allowing for the conjugation of drugs and small molecule ligands. Additionally, MNPs have superparamagnetic properties, making them useful for MR imaging, and can serve as magnetic thermal therapy medium for tumor hyperthermia in alternating magnetic fields (AMF). 26
In this study, we successfully prepared MZF using the hydrothermal method and then enhanced their stability by modifying them with PEG. TEM observation revealed that the NPs, with a size about 6 nm, were well dispersed. Chemical bonds were used to couple the S2.2-aptamer to create the molecular probe S2.2-PEG-MZF. DLS results demonstrated that the average hydrodynamic size of S2.2-PEG-MZF was 50 nm, meeting the requirements for medical nanomaterials. Additionally, hemolysis tests confirmed that the hemolysis rate of S2.2-PEG-MZF was less than 5%, making them suitable for medical use. CCK-8 results showed that the probe had no significant cytotoxicity. These promising results lay a foundation for future research.
Molecular imaging is a vital tool for the ultra-early diagnosis of tumors as it can detect abnormalities in disease-related genes or proteins before anatomical changes occur. 27 Magnetic resonance molecular imaging is a rapidly advancing field and an essential branch of molecular imaging. The development of specific, efficient, and safe nanocontrast agents is critical for achieving magnetic resonance molecular imaging. 28 The molecular probe S2.2-PEG-MZF developed in this study showed excellent imaging potential in MRI. As the concentration of the molecular probe increased, the corresponding image's T2WI signal intensity gradually decreased. In addition, the S2.2-aptamer conjugated in the molecular probe is able to target and bind MUC1, which is highly expressed on the surface of BC cells. Our further in vitro MRI study demonstrated that the T2WI signal inhibition intensity of the S2.2-PEG-MZF molecular probe was significantly higher in MCF-7 cells (MUC1+) than that in HepG2 cells (MUC1−). These findings demonstrate the potential of the S2.2-PEG-MZF as a targeted contrast agent for MR molecular imaging of BC. Prussian blue staining, also known as ferric-hematoxylin staining, is a technique that uses ferricyanide potassium and dilute acid to react with ferric ions (Fe3+) in the cytoplasm or interstitium, resulting in a blue color. The results of the Prussian blue staining indicated that the number of iron particles deposited in MCF-7 cells in S2.2-PEG-MZF group was significantly greater than that in PEG-MZF-NPs group. However, there was no significant difference in the uptake of S2.2-modified NPs by HepG2 cells. These findings suggested that S2.2-mediated endocytosis is the main route of phagocytosis by MCF-7 cells, whereas HepG2 cells primarily rely on passive phagocytosis. The Prussian blue staining results also demonstrated that the S2.2-modified NPs have a high specificity for binding to MCF-7 cells with a high expression of MUC1.
MFH is a promising therapeutic approach known as “green therapy.” MNPs are injected near the tumor, and an AMF is applied to induce a temperature increase that causes cancer cell apoptosis and necrosis. 29 In 2011, MFH was approved for clinical treatment of glioblastoma. Hyperthermia has been shown to trigger apoptosis through mechanisms such as protein denaturation and DNA damage. 30 Besides, tumors have inefficient blood flow and oxygen supply, leading to an acidic and nutrient-deficient environment, making cancer cells more vulnerable to sudden temperature increases as compared with healthy cells. 31 Hence, the use of MNPs to induce tumor hyperthermia through the heating properties under an external AMF has gained significant attention. In this study, the prepared S2.2-PEG-MZF exhibited good heating ability under an AMF. The NPs showed gradual warming after 30 minutes, and finally stabilized at an optimal temperature of 42 to 44 °C, which is ideal for tumor hyperthermia. Additionally, the S2.2-aptamer conjugated in the MNPs allows for targeted heating, which potentially reduces damage to normal tissues, thus improving the efficiency of cancer thermal therapy. However, further animal experiments are necessary to verify these results.
Nanomedicine, utilizing the advancement of nanotechnology, offers a novel approach for combining tumor diagnosis and treatment. 32 MNPs have also been shown to have good performance as drug carriers in several studies. They have the advantages of integrated diagnosis and treatment, such as early diagnosis, dynamic monitoring of efficacy, and adjustment of treatment strategies.5,6 In this research, the S2.2-PEG-MZF molecular probes were used as a basis for drug conjugation to create the S2.2-PEG-MZF /DOX nanoliposome. Subsequently, in vitro experiments were conducted to study the therapeutic effects of this nanoliposome on BC cells.
The results of the experiment showed that the survival rate of MCF-7 cells after targeted hyperthermia treatment was only (35.45 ± 3.69) %, with a high apoptosis rate. Additionally, the invasion ability of tumor cells is closely related to the metastasis and infiltration of tumors. Therefore, the study further investigated the impact of targeted nanoliposome combined with thermochemotherapy intervention on the invasion ability of MCF-7 cells. The transwell results indicated that the S2.2-PEG-MZF/DOX/MFH group had the strongest inhibitory effect on the invasion ability of BC cells, which was significantly better than the other three groups. Finally, the expressions of apoptosis and invasion related proteins were examined using Western blot methods after the intervention of each group on MCF-7 cells. Notably, the S2.2-PEG-MZF/DOX/MFH group exhibited the most significant changes in these proteins, indicating its strong ability to induce apoptosis and inhibit invasion of tumor cells. These experimental results demonstrate the potential of S2.2-PEG-MZF/DOX as an ideal targeted therapy for MCF-7 cells, and provide a new approach for the integrated diagnosis and treatment of BC.
The combination of hyperthermia and chemotherapy using PEG-MZF nanoliposomes showed promising results in terms of enhanced therapeutic efficacy. These findings are consistent with previous research, further highlighting the potential of PEG-MZF nanoliposomes as a versatile platform for synergistic cancer treatment.23,24 However, it is important to acknowledge the limitations of our study. Firstly, the in vitro experiments conducted may not fully capture the complexity of the tumor microenvironment and physiological conditions in vivo. Future studies should include in vivo experiments to validate the efficacy and safety of PEG-MZF nanoliposomes in a more realistic setting. Secondly, further elucidation is needed regarding the cellular uptake mechanisms and intracellular fate of PEG-MZF nanoliposomes to enhance our understanding of their therapeutic mechanisms. Additionally, optimizing the loading efficiency of DOX and APT in PEG-MZF nanoliposomes is crucial to maximize their therapeutic potential. Various parameters such as drug-to-lipid ratio, lipid composition, and preparation methods should be systematically studied to achieve optimal drug loading and release profiles.
Conclusion
The S2.2-PEG-MZF molecular probe constructed in this study has proven as having good biocompatibility, MR imaging potential, and magnetic induction heating properties. In addition, the probe has good targeting and MR imaging effect on BC cells (MUC1+), which is expected to become a new contrast agent for MR molecular imaging of BC. Given the good targeting and MRI effect, we successfully prepared S2.2-PEG-MZF/DOX nanoliposomes for thermochemotherapy of BC cells, and the results showed that they could inhibit BC cell proliferation and invasion and induce their apoptosis, which may provide a new idea for integrated diagnosis and treatment of BC.
Footnotes
Abbreviations
Acknowledgements
The authors appreciate the technical support of Dr Dengyun Nie for this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Natural Science Foundation of Nanjing University of Chinese Medicine (XZR2020093), Taizhou People's Hospital Medical Innovation Team Foundation, China (CXTDA201901), Postdoctoral research program of Taizhou Clinical Medicine School of Nanjing Medical University (TZBSHKY202204), and the project of Taizhou People's Hospital (ZD202031).
Taizhou People’s Hospital Medical Innovation Team Foundation, the project of Taizhou People's Hospital, Postdoctoral research program of Taizhou Clinical Medicine School of Nanjing Medical University, Natural Science Foundation of Nanjing University of Chinese Medicine (grant numbers CXTDA201901, ZD202031, TZBSHKY202204, XZR2020093).
