Abstract
Purpose
Compared and analyzed the MRI imaging features of brain invasion otherwise benign (BIOB) meningiomas and WHO grade 1, grade 2 meningiomas, discussed the WHO grading of BIOB from the perspective of imaging.
Materials and Methods
A retrospective analysis was performed on 675 meningiomas patients who carried on MRI examination from January 2006 to February 2022. Setting the 2022 Central nervous system (CNS) WHO Guidelines as the gold standard for pathological diagnosis. Statistical analysis of age, gender, and MRI features of meningiomas in relation to WHO grade and brain invasion.
Results
Among 675 cases meningiomas, 543 (80.4%) were WHO grade 1, 123 (18.2%) were WHO grade 2, and 9 (1.3%) were WHO grade 3. There were 108 cases meningiomas with brain invasion (BI) (16.0%) and 567 cases without BI (84.0%). Among BI cases, 67 cases were BIOB. Compared the MRI features between BIOB and WHO grade 1 meningiomas, multivariate analysis demonstrated that the most strongly factors associated with distinguish them were enhancement degree, peritumoral edema, tumor–brain interface, fingerlike protrusion, mushroom sign, and bone invasion (AUC: 0.925 (0.901∼0.945), sensitivity: 0.925, specificity: 0.801). Compared the MRI features between BIOB and WHO grade 2 meningiomas, multivariate analysis demonstrated that the most strongly factors associated with distinguish them were enhancement degree and the tumor–brain interface (AUC: 0.779 (0.686∼0.841), sensitivity: 0.746, specificity: 0.732), their efficacy was slightly weaker.
Conclusions
BIOB is more similar to WHO grade 2 meningiomas in clinical and imaging features than WHO grade 1, so we think that it may be reasonable to classify BIOB as WHO Grade 2 meningiomas in the guidelines.
Introduction
Meningiomas is one of the most common brain tumors of the central nervous system, accounting for 37.6% of primary brain tumors. 1 According to the statistics of the World Health Organization (WHO), the total incidence of brain invasion is about 5% to 78%. Among them, WHO grade 2 atypical meningiomas are 13% to 70%, and WHO grade 3 anaplastic meningiomas are 70% to 100%. 2 However, in a recent meta-analysis, the incidence of brain invasion otherwise benign meningiomas (BIOB) was 4.14% (2.73%–6.23%). 3
BIOB specifically refers to benign tumors with brain invasion. In 1993, Perry's 4 team found that 23% of WHO grade 1 benign meningiomas had brain invasion, and the recurrence rate may increase. In 1997, they conducted another experiment to compare BIOB with atypical meningiomas without brain invasion. 5 They found that the recurrence rate and fatality rate of BIOB were similar to atypical meningiomas without brain invasion. Later, many studies believed that the recurrence rate of BIOB was higher than non-brain invasion meningiomas.6,7 Therefore, the brain invasion as one of the diagnostic criteria for atypical meningiomas was included in the fourth edition of the WHO guidelines in 2016. 8 This variation has increased the proportion of WHO grade 2 meningiomas by 1% to 10%. 9 In recent years, Brokinkel et al. 2 have done a systematic review and in the most studies they found that brain invasion is associated with tumor progression. But some researchers disagree with this view, they found that BIOB's prognosis was not significantly different from benign meningiomas.3,10 In the meta-analysis by Nakasu et al., 3 the overall recurrence rate of brain-invasion meningiomas exceeds non-brain-invasion meningiomas, but this does not hold true in separate discussions at all grades of the WHO. So, was the decision to classify BIOB as atypical meningiomas too hasty? Especially since molecular detection is difficult to be popularized due to its high price, the determination of WHO grade and brain invasion still depends on pathological morphology and immunohistochemistry, whether BIOB should be classified as WHO grade 1 or WHO grade 2 atypical meningiomas is still a controversial issue. 11 At present, there are very few imaging reports regarding brain invasion as an independent study, and no clear standard for the imaging diagnosis of brain invasion.12–15 Similarly, the imaging studies on BIOB have not been mentioned in the literature. Therefore, incomplete clinical surgical resection of BIOB patients may result in an increased risk of recurrence and death.16,17 This study focused on the above-mentioned argumentation of BIOB, compared the imaging and clinical characteristics of BIOB with WHO grade 1 and WHO grade 2, and discussed the grading opinions of BIOB from the perspective of imaging.
Materials and Methods
Research Subjects
Retrospective analysis of 675 patients with meningiomas who underwent MRI examination at our hospital from January 2006 to February 2022, including 210 males and 465 females, aged 11 to 90 years, with a mean age of (50.83 ± 13.23) years. This study was a retrospective clinical study, and the study protocol was reviewed and approved by the ethics committee of our hospital, with the approval number: 2023-036-01PJ. All participants provided written informed consent for the images collected and used in this study.
Inclusion criteria: (1) Preoperative MRI examination; (2) Surgical treatment was performed after examination; (3) The surgically resected tissue was confirmed to be meningioma by routine pathology and immunohistochemistry; (4) Complete medical records and imaging data.
Exclusion criteria: (1) 135 cases did not meet the following MRI scan criteria; (2) 46 patients with unsatisfactory image quality determined by two neuroradiologists with more than 10 years of experience; (3) Pathological diagnosis of meningioma brain invasion, but not described including brain organizers in nine cases; (4) 26 patients underwent traditional Chinese medicine therapy, bio-targeted therapy, and neoadjuvant chemotherapy before surgery; (5) Two patients with recurrent meningioma underwent reoperation.
The reporting of this study conforms to STROBE guidelines. 18
Scanning Solutions
The specific scan parameters are as follows Table 1.
Equipment and Sequence and Scan Parameters

T1WI, T1-weighted imaging; T2WI, T2-weighted imaging; T2Flair, T2-fluid attenuated inversion recovery; T1C, T1-weighted imaging contrast; DWI, diffusion-weighted imaging; TR, repetition time; TE, echo time; NEX, number of excitations; FOV, field of view; THK, thickness; ETL, echo train length, BW, bandwidth; Number of patients, Number of patients examined by the machine.
Image Data Collection
We have de-identified patient details such that the identity of any person may not be ascertained in any way.
Criteria for MRI Signal Evaluation of Brain Tumors. 19
T1-weighted imaging (T1WI) signal intensity (SI) scoring system: 1 = significantly lower than the gray matter and equal to the cerebrospinal fluid; 2 = mildly lower than the gray matter signal; 3 = equal to the gray matter signal; 4 = mildly higher than the gray matter signal; 5 = significantly higher than the gray matter signal and equal to the fat signal.
T2-weighted imaging (T2WI) signal intensity (SI) scoring system: 1 = significantly lower than the gray matter and equal to the bone cortex; 2 = mildly lower than the gray matter signal; 3 = equal to the gray matter signal; 4 = mildly higher than the gray matter; 5 = significantly higher than the gray matter and equal to the cerebrospinal fluid.
T1-weighted imaging contrast (T1C) enhancement grading: 1 = significant enhancement with similar to fat signal; 2 = moderate enhancement with slightly lower enhancement than fat signal; 3 = mild enhancement with lower enhancement than fat signal and higher than gray matter and white matter signal.
Morphological Evaluation Criteria
Tumor size: 20 PACS measured the longest axis of the tumor in the transverse, coronal, and sagittal directions; and its volume was estimated using the formula: V = r1×r2×r3×π×4/3.
Peritumoral edema: a patchy area of low-SI T1WI, high-SI T2WI, high-SI T2 Flair, and unenhanced low-SI in the brain surrounding the tumor. Described as yes or no.
Lobulation sign: Tumor surface edge uneven, arc depression, or convex change. Tumors are described as lobulated or non-lobulated.
Flowing void effects: refers to the fact that MRI cannot collect signals from the blood flowing in the tumor blood vessels, and it appears as a strip low signal on T1WI and T2WI sequences, indicating that there are abundant tumor blood vessels in the tumor tissues. 21
Dural tail sign: T1C is most clearly shown, showing enhancement, thickening, and distal thinning of the meninges adjacent to the tumor. 22
Sinus invasion: Assessed on T2WI and T1C, doctors can observe tumor tissue adhering to the venous sinus, invading the venous sinus, or completely blocking the venous sinus. 23
Tumor-brain tissue interface:14,15,20 The tumor and brain tissue were separated by cerebrospinal fluid-vascular space and arachnoid interface, T1C showed a low signal ring. When the tumor invaded the brain parenchyma, the low signal ring disappeared, indicating that the tumor–brain interface was poorly displayed.
Finger-like protrusion or pseudopod sign:14,15,20 The T1C shows clearly, and it can be observed that tumor tissue protrudes into the adjacent brain parenchyma like a finger or pseudopodia.
Mushroom sign:14,15,20 Appeared as an enhancing band of spherical tumor invading peripherally along the dural attachment, extending farther, thicker, and longer than the commonly seen in dural tail sign, and often with a more coarse and uneven proximal cerebral surface.
ADC Value
The raw images of DWI were uploaded to the workstation (Syngo) and the ADC images were obtained by image post-processing and analysis using software (Siemens Healthcare, Erlangen, Germany). Five regions of interest (ROIs) were selected from the tumor parenchyma based on the ADC images, selecting the largest dimension of the lesion, and avoiding areas of necrosis, hemorrhage, cystic lesions, or calcifications. ROIs should not be too large, each ROI was set to 5 cm2 (approximately 200 pixels), and the average value was calculated and proceeded statistical analysis at a later stage.
Pathological Image Interpretation
All pathological findings were reinterpreted by two neuropathologists according to the 2022 CNS WHO guidelines, and all specimens were subjected to HE staining and immune-histochemical examination. Morphologic diagnosis was made using the “933” typing model, ie, 9 WHO grade 1 subtype, 3 WHO grade 2 subtypes, and 3 WHO grade 3 subtypes to determine the pathologic typing of meningiomas and to diagnose cases of brain invasion. The controversial cases were determined after discussion by the whole department.
Pathological diagnostic criteria of brain invasion2,24: (1) HE staining of the tumor–brain interface reveals irregular, tongue-like invasion of tumor cells into the brain parenchyma without soft meningeal involvement. (2) There was glial cell proliferation and neuronal degradation in the invaded brain tissue. (3) Positive immune-histochemical staining for SPARC, GFAP, CD44, etc. in paraffin sections. Positive for any of the above criteria is considered as brain invasion of the tumor. In this study, the pathological report is required to describe brain tissue, otherwise excluded.
Statistical Methods
SPSS software (v.26.0, IBM, USA) was used for statistical analysis and Medcalc software (v.20.0.22, Solvusoft Corporation, USA) was used to make the ROC curves.
Meningioma grading, typing, and brain invasion diagnosis were based on pathology as the gold standard, and the results were descriptive statistics.
Patients’ age, gender, and MRI imaging features were evaluated by an associate senior and a full senior central nervous system radiologist without knowledge of the pathological findings. The results of their evaluations were tested for consistency. ICC statistics were used for measurement data, and Kappa coefficient was used for enumeration data.
Results
Pathological Classification of Meningioma
Among the 675 meningiomas, 108 (16.0%) were brain invasion meningiomas and 567 were non-brain invasion meningiomas. There were 543 cases of WHO grade 1 meningiomas, 123 cases of WHO grade 2 (67 cases of BIOB, 56 other cases), and 9 cases of WHO grade 3.
Consistency Test Results of Two Central Nervous System Imaging Specialists
Two senior (associate and senior) neuroradiologists (11 years/16 years of working experience) judged the MRI images. The data of the two groups were tested for consistency, and the intra-group correlation coefficient (ICC) ranged from 0.844 to 0.997, which can be statistically considered as a good agreement of the results between the two individuals.
Compared the BIOB with WHO Grade 1 and Grade 2 Meningiomas
The clinical and MRI characteristics of 67 cases BIOB were compared with 543 cases WHO grade 1 meningiomas and 56 cases WHO grade 2 meningiomas what was using logistic univariate analysis, the results are shown in Table 2. Ten characteristic parameters as gender, enhancement degree, tumor size, peritumoral edema, flowing void effects, dural tail sign, bone invasion, tumor–brain interface, finger-like protrusion, and mushroom sign were statistically significant in distinguishing WHO grade 1 meningiomas from BIOB. Three imaging characteristics parameters, including enhancement degree, bone invasion, and tumor–brain interface, were statistically significant in discriminating WHO grade 2 meningiomas from BIOB (P = 0.002, P = 0.014, P = 0.001).
Logistic Univariate Regression Analysis of wHO Grade 1 and WHO Grade 2 Meningiomas Compared with BIOB Characteristics

BIOB, Brain invasion otherwise benign meningioma; WHO, World Health Organization.
T1WI, T1-weighted imaging; T2WI, T2-weighted imaging; T1C, T1-weighted imaging contrast; ADC, apparent diffusion coefficient
Logistic multivariate regression analysis was performed on the ten characteristics and the results are shown in Table 3. The six characteristics that enhancement degree, peritumoral edema, tumor–brain interface, finger-like protrusion, mushroom sign, and bone invasion were independent risk factors for differentiating WHO grade 1 meningiomas from BIOB (P < 0.05), resulting in a regression equation: Y = −2.960 + 2.912Xtumor−brain interface + 2.685X mushroom sign + 1.864X finger−like protrusion + 1.351X bone invasion −0.967X enhancement degree −0.886Xperitumoral edema. Six variables were used to make ROC curves to identify and distinguish BIOB and WHO grade 1 meningiomas (Figure 1

ROC curves of six independent risk factors for WHO grade 1 meningiomas compared with BIOB.
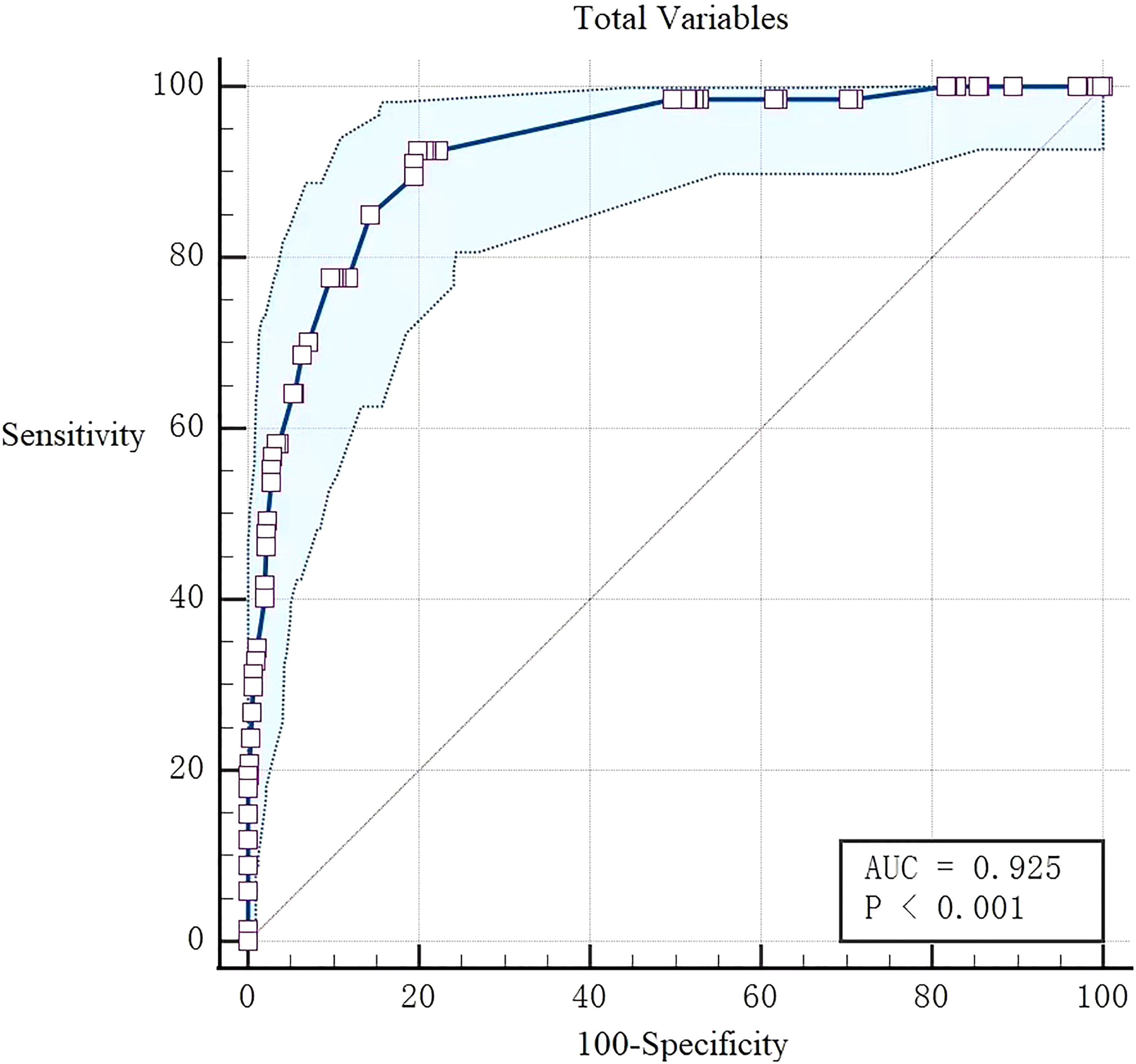
ROC curves of total variables for the comparison of WHO grade 1 meningiomas and BIOB.
Logistic Multivariate Regression Analysis of WHO Grade 1 Meningioma Compared with BIOB

ROC Curve of WHO Grade 1 Meningioma Compared with BIOB

Logistic multivariate regression analysis was done for the characteristic parameters of T1C SI, bone invasion, tumor–brain interface, dural tail sign, and mushroom sign, the results were shown in Table 5. The enhancement degree and tumor–brain interface were independent risk factors for distinguished WHO grade 2 meningiomas from BIOB, resulting in a regression model of Y = −0.300 + 1.602Xtumor−brain interface −1.421Xenhancement degree + 0.956Xdural tail sign + 0.702Xbone invasion + 0.217Xmushroom sign.
Logistic Multivariate Regression Analysis of WHO Grade 2 Meningioma Compared with BIOB

Two variables, the enhancement degree and the tumor–brain interface were used to make ROC curves to differentiate BIOB from WHO grade 2 meningiomas (Figure 3). Their AUC, sensitivity and specificity were calculated, and the results are shown in Table 6.

ROC curves of two independent risk factors and total variables for WHO grade 2 meningiomas compared with BIOB.
ROC Curve of WHO Grade 2 Meningioma Compared with BIOB

The AUC of enhancement degree to identify BIOB and WHO grade 2 meningiomas was 0.647 (0.556-0.731), a sensitivity was 0.866 and a specificity was 0.429. The AUC differentiating BIOB from WHO grade 2 meningiomas at the tumor–brain interface was 0.640 (0.548−0.724), the sensitivity was 0.851, and the specificity was 0.429 (Figure 3).
All features were formed into fitting variables to make ROC curves to distinguish them (Figure 3), resulting an AUC was 0.779 (0.686-0.841), a sensitivity was 0.746, and a specificity was 0.732. The differences in MRI imaging features between the two were not significant.
The preoperative imaging study of a BIOB patient histopathological diagnosed as WHO grade 1 transitional meningioma found that the tumor material had a uniform signal with clear borders but large edema of the surrounding brain parenchyma. T1C showed that the tumor–brain interface was blurred, suggesting that the clinical surgical resection area was extensive, including the tumor–brain interface, and the local brain tissue was infiltrated by the tumor under the microscope. The patient had a good prognosis with no recurrence (Figure 4).

a. Axial T1-weighted sequence in a 38-year-old man with a 2-month history of Blurred vision with dizziness and vomiting, no-lobulated mass located in the parietal lobe, a well-circumscribed. b and e. Axial T2-weighted sequence and Coronal T2 – flair sequence showed the tumor is to be isointense to gray matter with massive peritumoral edema. c. T1WI enhancement sequence showed blurred tumor–brain interface. d. DWI signal of the lesion was not high. f. Pathological HE staining (20 × 10 magnification): the patient was diagnosed as transitional meningioma with brain invasion. The arrow shows the infiltrated brain tissue.
Discussion
In 1975, meningioma brain invasion was first proposed, which may be a sign of malignant meningioma. 25 In 1997, Perry, 5 one of the guideline authors, found that 23% of benign meningiomas had brain invasion. He subsequently found that benign meningioma with brain invasion had very similar recurrence and mortality rates to atypical meningioma, and that affected the prognosis of benign meningioma. 26 Therefore, Perry et al. 8 included brain invasion as a diagnostic criterion for atypical meningioma in the fourth edition of the WHO guidelines in 2016. In recent years, Brokinkel 2 and Nakasu 3 reviewed all previous studies on brain invasion of meningiomas and found that in most studies, brain invasion was associated with tumor progression. However, some researchers have found that although the prognosis of BIOB is better than the other atypical meningioma, it's not much different from benign meningioma.3,10 They did not support the classification of BIOB as atypical meningioma. Until now, the controversy over brain invasion remains unresolved. In this study, the author took BIOB as the entry point to comprehensively compare and analyze the MRI imaging characteristics of BIOB between WHO grade 1 and 2 meningioma. Discussed the classification of BIOB, the core issue that scholars questioned the guidelines, from the imaging perspective.
It is different from the previous results of Kandemirli et al., 12 in this study, the AUC of MRI features comparison between BIOB and WHO grade 1 meningiomas was 0.925 (0.901∼0.945), the sensitivity was 0.925, and the specificity was 0.801, indicating excellent diagnostic efficiency. The differences between them are mainly reflected in the six characteristics that is enhancement degree, peritumoral edema, tumor–brain interface, finger protrusion, mushroom sign, and bone invasion, the tumor–brain interface is the most effective.
The BIOB enhancement degree was mainly degree 1, and the proportion was higher than WHO1 and WHO 2 meningiomas (P = 0.034 & P = 0.002). In Adeli's 20 report, the proportion of brain-invasion meningiomas showing heterogeneous enhancement was higher than non-brain-invasion tumors, regardless of WHO grade. The authors believe that the enhancement degree of meningiomas depends on the content of cell components in the tumor and the abundance of nourishing blood vessels in the surrounding tumor, but the enhancement homogeneity depends on the complexity of the internal components of the tumor. For example, hemangiomatous meningiomas have significant enhancement (signal intensity is equivalent to fat) because the cells in the tumor are more abundant than other types, while the enhancement of gravelly meningiomas is more uneven due to more calcification components in the tumor.27,28
In the current studies on brain invasion of meningiomas, most scholars believe that peritumoral edema is the characteristic sign of brain invasion.2,14,20,29 In this study, it was a differentiating role for benign meningiomas with or without brain invasion. Unfortunately, when included the pathological classification of WHO grade 2 and 3 meningiomas, this conclusion was not valid. The results showed that peritumoral edema had low efficacy in differential BIOB and WHO grade 1 meningiomas. While having no statistically significant difference between BIOB and WHO grade 2 meningiomas in peritumoral edema. The author supports that the etiology of peritumoral edema is closely related to tumor volume and tumor angiogenesis, and may be also related to increased expression of vascular endothelial growth factor (VEGF), but the relationship between the tumor and brain invasion is weak.7,30–32
In recent 2 years, some radiologists have paid attention to the significance of the tumor–brain interface in the diagnosis of brain invasion.14,20 The results of previous study models constructed in the tumor–brain interface region suggest that the tumor–brain interface is very effective in diagnosing brain invasion meningioma.13,15 In our study, the tumor–brain interface has high diagnostic performance in differentiating BIOB and WHO grade 1 meningiomas, and is a more significant imaging characteristic between them. However, for differentiating BIOB from WHO grade 2 meningiomas, there was little difference between them. Correct preoperative image sign interpretation has important guiding reference value for intraoperative resection and pathological tissue sampling, and helps reduce the recurrence rate. Most scholars believe that the incidence of brain invasion has been underestimated by current surgery and pathology.7,33–35
Fingerlike protuberance and mushroom sign have also been reported by many scholars as the characteristic of malignant meningiomas.5,26,36 The pathological basis is caused by tumor invasion of the adjacent dural surface, dura mater, arachnoid membrane, subarachnoid space, pia mater, and brain.33,37 These are usually accompanied by an unclear and continuous tumor–brain interface. Notably, in the analysis, fingerlike protuberance and mushroom sign may be more sensitive in display detail about differentiating BIOB from WHO grade 1 meningiomas and have a higher diagnostic capacity than identification of BIOB from WHO grade 2 meningiomas. Therefore, the writers believe that fingerlike protrusion and mushroom sign are the MR characteristic imaging features to distinguish BIOB from WHO grade 1.
The incidence of bone invasion was statistically different between BIOB and WHO grade 1 meningiomas. However, it is important to note that, on pathology, bone and meningeal invasion of meningiomas cannot be considered brain invasion. 29 In this study, bone invasion had a high proportion in benign tumors (11.6%) and a certain proportion in non-invasive meningiomas (11.2%). Therefore, the authors hold that bone invasion can be used as a reference to distinguish BIOB from WHO grade 1 meningiomas, but cannot be used as a characteristic sign to judge WHO grade and brain invasion.
In comparison of all clinical and imaging features between BIOB and WHO grade 2 meningiomas, AUC was 0.779 (0.686∼0.841), sensitivity was 0.746, and specificity was 0.732. The enhancement degree and the tumor–brain interface are the distinguishing features, but their effectiveness is not strong. The MRI characteristics of BIOB and WHO grade 2 meningiomas were similar.
Conclusion
Preoperative MRI can accurately determine the WHO grade and extent of meningioma brain invasion. The mushroom sign, tumor–brain interface, finger-like process, bone invasion, and lobulation sign are all independent risk factors for determining meningioma WHO grade. The tumor–brain interface, finger protrusion, mushroom sign, bone invasion, lobular sign, tumor size, and ADC value were all independent risk factors for meningioma infiltration diagnosis. The tumor–brain interface is critical in determining WHO grade and brain invasion.
Comparative analysis of multiple groups of MRI characteristics showed that BIOB and WHO grade 2 meningiomas were more similar in clinical and imaging features than grade 1 meningiomas. Therefore, we think that it may be reasonable to classify BIOB as WHO Grade 2 meningiomas in the guidelines.
Limitation
In this study, MR examinations were performed with devices with varying magnetic field strengths, resulting in partial heterogeneity in image resolution and ADC value, as well as a few statistical errors in the results. This study yielded a single data center with certain limitations. The next step is to conduct multi-center data research. We will conduct quantitative data analysis based on the findings of our study. Computer-aided diagnosis can assist doctors in evaluating meningiomas brain invasion and WHO classification, allowing us to work more efficiently.
Footnotes
Acknowledgements
I can successfully complete this paper, because many friends and teachers had given me many help from topic selection, data collection, statistics, writing, polishing to the final draft.
First of all, I would like to extend my sincere gratitude to MD Juan Yu, for her instructive advice and useful suggestions on my thesis. I am deeply grateful of her help in the completion of this thesis.
I am grateful MD. Hong Jiang and MM Zhang Zhu for their guidance and help on statistical methods, and whose earnest attitude tells me how to learn more statistical.
I am also deeply indebted to MM Xia Jing Liu and MM Jun Jiang in data collection their direct and indirect help to me.
Special thanks should go to MD Fan Lin and MD YuLi Wang who have put considerable time and effort into their comments on the Image diagnosis.
High tribute shall be paid to Kan Deng, whose profound knowledge of English triggers my love for this beautiful language English.
Finally, I am indebted to My colleagues and my family for their continuous support and encouragement.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
This study was a retrospective clinical study, our study was approved by The Shenzhen Second People's Hospital Research Ethics Committee (approval no. 2023-036-01PJ). All patients provided written informed consent prior to enrollment in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Shenzhen Basic Research Project (Natural Science Foundation) (Grant No. JCYJ20220530150416036)
