Abstract
In recent years, extracellular vesicles (EVs) have gained significant attention due to their tremendous potential for clinical applications. EVs play a crucial role in various aspects, including tumorigenesis, drug resistance, immune escape, and reconstruction of the tumor microenvironment. Despite the growing interest in EVs, many questions still need to be addressed before they can be practically applied in clinical settings. This paper aims to review EVs’ isolation methods, structure research, the roles of EVs in tumorigenesis and their mechanisms in multiple types of tumors, their potential application in drug delivery, and the expectations for their future in clinical research.
Keywords
Introduction
Extracellular vesicles (EVs) are a diverse group of bilayer lipid particles with varying sizes, secretion of cellular origin, biological functions, and release pathways. 1 They are comprised of three subtypes: microvesicles (MVs) (100-500 nm), exosomes (30-150 nm), 2 and apoptotic bodies (500-2000 nm), each containing a range of contents 1 such as DNA, mRNA, small RNA, long non-coding RNA (lncRNA), lipids, proteins, and sugars. 1 In this paper, we will mainly focus on exosomes due to the growing research interest in this subtype as well as the overlap between MVs and EVs.
EVs were first identified as microvesicles, which are exfoliated membrane vesicles from normal or neoplastic cell lines. 3 The proteins in EVs were identified in S. cerevisiae in 1997 and were later recognized as the part of the highly conserved RNA processing complex. 4 EVs play an essential role in physiology and pathology, including maturation of reticulocytes by shedding the membrane proteins, 5 cell communication, antigen presentation,6,7 synaptic activities by controlling neurotransmitter release, 8 tumor suppression, 9 and tumor facilitation. 10 Therefore, EVs have the potential for various applications, such as delivery vehicles for siRNA in tumor therapy, 11 modulated exosomes for the treatment of pancreatic cancer, 12 vaccines, 13 and as diagnosis markers,2,14 With cancer being one of the leading causes of death globally, causing about 10 million deaths in 2020, the role of EVs is of significant interest. 15 This paper will focus on current methods for EV isolation, their role in tumor regulations, and their promising applications in the field of clinical research.
EVs’ Isolation and Identification
Various methods are currently available for isolating EVs. The most common method is ultracentrifugation-based, which is still considered the gold standard. However, other methods have been developed to overcome the limitations of ultracentrifugation, such as size-based techniques, immunoaffinity capture-based techniques, precipitation, and microfluidic-based isolation techniques. Unfortunately, none of these methods alone can achieve the high recovery and high specificity of EVs required according to Minimal Information for Studies of Extracellular Vesicles (MISEV2014), which was proposed by the International Society prompted for Extracellular Vesicles (ISEV) in 2014 and updated MISEV2018.16,17 Therefore, investigators must combine different methods to meet the requirements of their research and applications. This paper provides a summary of the most widely applied methods and compares their recovery, specificity, advantage, and drawbacks (Table 1).
Summary of Widely Used Methods of EVs Isolation.

Ultracentrifugation remains the most commonly accepted method for isolating EVs. However, it cannot effectively separate the non-specific particles, which has led to the development of additional methods to exclude unwanted particles. 1 One such method is ultrafiltration. 18 Another widely used method is precipitation, which can be achieved through immune and chemical precipitation. Both methods demonstrate high purity but low yield. 19 However, since EVs from different origins exhibit different marker expressions and heterogeneity, 21 the immune precipitation may not suitable for all isolation work. Other methods have been developed for EV isolation, such as microfluid tools are developed to separate EVs and to synthesize EV mimetics for drug delivery applications, 20 or based on size-exclusion chromatography, charge-based techniques, hydrophobic interaction chromatography to isolate EVs. However, each of these methods has its own limitations. 22
Since none of the method for EV isolation are perfect and each has advantages and drawbacks. Therefore, it is essential to choose the appropriate method for the specific application. According to the recommendation of MISEV2014 and MISEV2018, it is better to use a combination of methods for EV identification and characterization, such as physical exosome enriched proteins like TSG101, CD63, CD81, CD9, and non-expected proteins. Analysis can be done using Western blot, flow cytometry, or mass spectrum. It is also recommended to identify EVs using at least two methods to ensure accuracy and reliability.
Structural Studies
Direct structure determination of a full EV by biophysical approaches, including cryo-electron microscopy (cryo-EM) or cryo-electron tomography (cryo-ET) remains challenging due to its variable sizes and heterogeneous compositions, and the technical limitations in biochemical isolation and purification of a specific EV. Depending on the origin of EVs, the lipid surface can contain different types of membrane proteins, which commonly include tetraspanins (primarily CD9, CD63, and CD81) and integrins, which play diverse roles in EV function.23–25 Structures of CD9 26 and CD81 27 reveal a similar 4-transmembrane (TM)-helices architecture of tetraspanins. Remarkably, the large extracellular loop (LEL)/TM3/TM4 of CD9 is involved in binding to a partner membrane protein EWI-2, 26 whereas CD81 engages the CD19 receptor through the LEL/TM1/TM2. 28 These observations highlight a central role of the LEL domain while the 4 TMs of tetraspanins may create different interaction interfaces to accommodate many other membrane proteins. Integrins are heterodimers comprising α and β subunits, and function as cell adhesion receptors that mainly mediate cell-cell and cell-extracellular matrix interactions. 29 Crystallographic and cryo-EM studies in recent years have started to unravel the complete picture of the ligand-binding mechanisms of integrins. The structures of pro-TGF-β1 bound integrin αVβ6 30 and L-TGF-β bound αvβ8 31 provide insights into how integrins engage their macromolecular ligands. And the structure of the LM511-integrin α6β1 complex deciphers the molecular basis of laminin recognition by integrins. 32 Furthermore, the structure of a fibronectin–integrin complex was determined in lipid nanodiscs, which comprises integrin α5β1 purified from the human placenta and its ligand FN7-10. 33 Both the laminin and fibronectin-bound integrin structures suggest that conformational changes of integrin are not necessarily linked to the ligand-binding affinity. Notably, these ligand-bound integrins share similar extended-open conformations except αvβ8 which retains an extended-closed conformation. Interestingly, EVs have also been found to carry TGF-β1, laminin, or fibronectin,34–38 this may provide a unique mechanism for EVs to enhance their targeting to integrin-enriched recipient cells in addition to engaging their own integrins with diverse ligands of target cells.
There are accumulating evidence indicating that cytokines and chemokines are packaged within EVs or associated with the EV surface,39–41 highlighting a specific role of EVs in delivering these critical factors to target cells. Cytokines and chemokines generally exhibit high affinities for their receptors, which may allow efficient recognition and uptake of EVs even after long-distance trafficking to the recipient cells. Previous structural studies have provided valuable insights into how these EV-associated proteins recognize their binding partners or receptors. The structure of VEGFR-1 in complex with VEGF- reveals how the full extracellular domains (ECDs) of the receptor dimers specifically recognize the VGEF-A cytokine. 42 Structures of IL-cytokines, including IL6, IL12/IL23 and IL17, bound to the receptors have been determined,43–45 which reveal similarities and diversities in the coupling mechanisms of different IL family members. Also, chemokines activate the G protein-coupled receptors (GPCRs), which belong to the largest family of membrane proteins. Notably, one chemokine can bind several GPCRs, and a single receptor can also bind multiple chemokines. Recent cryo-EM studies unravel the molecular mechanisms of receptor activation and G-protein coupling by chemokines, including CCL20/CCR6/Go, 46 CXCL8/CXCR2/Gi, 47 CCL5/CCR5/Gi.48,49 These structures could facilitate the rational design of novel drugs for the chemokine-receptors, which might use engineered EVs as cargo carriers. Also, syncytin proteins are reported to be carried by placenta exosomes 50 and syncytiotrophoblast extracellular microvesicles, 51 and the cryo-EM structure of human MFSD2A–SYNC2 complex 52 suggests that EVs could exert an inhibitory effect via recognizing the membrane proteins of the target cells. Notably, many other molecules related to the immune system, such as FasL and PD-L1 or MHC molecules 53 could be incorporated within EVs, and the related structures are not reviewed here.
EV formation and cargo-sorting into exosomes could be dependent on the endosomal sorting complex transport (ESCRT) pathway which requires a number of proteins such as ESCRT complex proteins, ALIX, Rab5, and Rab27a. 54 The cryo-EM structure of CHMP1B in complex with IST1 advanced our understanding of how human ESCRT-III subunits form spiral filaments to remodel membranes. 55 The structural information of ALIX and Rab27a is also available.56,57 A recent cryo-ET study determined the structure of VPS34 complex II on Rab5–GTP-coated membranes, which might also have implications in EV biogenesis. 58 EV formation also involves lipid ceramide and neutral sphingomyelinase (nSMase). 59 The structure of human nSMase2 was reported, 60 revealing potential activation mechanisms of nSMase for generating ceramide, which is highly enriched in EV. Another important aspect of EV function is vesicle fusion into recipient cells, which is mainly mediated by the SNARE complex. A wealth of structural information provided valuable insights into SNARE assembly catalyzed by Sec1–Munc18 proteins61–64 and SNARE disassembly mediated by NSF and SNAPs.65,66
EV research has been a rapidly developing field in these years, but direct structural studies of EV subunits are very limited. Further comprehensive investigations are needed to clarify how EV function is coupled to the subunit composition in different cell contexts. Future cryo-EM and cryo-ET studies are expected to elucidate the detailed molecular mechanisms underlying EV generation, cargo loading, trafficking, and uptake, which would aid in structure-based drug discovery, EV engineering, and developing EV-based therapeutic strategies.
EVs and Tumor
Brain Tumor, Leukemia, and Reproductive Tumor
Exosomes are critical mediators of communication within the tumor microenvironment, enabling crosstalk between cellular components and neighboring active cells. 67 The bioactive cargoes carried by exosomes make them a unique mechanism of intercellular communications. Notably, tumor-derived exosomes have been showed to play a role in the reprograming of immune regulation and the phenotype of immune cells, with particular emphasis on macrophage repolarization. This phenomenon has been widely studied, as it has significant implications for the understanding and treatment of cancer. 68
Brain Tumor
Glioma is a prevalent primary brain tumor characterized by its aggressive nature and poor prognosis despite multidisciplinary treatment. The unique tumor microenvironment of Glioma is highly immunosuppressive due to the presence of the blood–brain barrier (BBB). 69 Therefore, BBB creates a heightened intercellular communication via growth factors, cytokines, and exosomes. 70 Tumor-derived exosomes play a crucial role in immunosuppression by promoting M2 polarization of naive macrophages with high PD-L1 expression,71,72 and contributing to immune escape by infiltrating B cells. These observations highlight the high plasticity of traditional immunocytes in adapting to the malignant environment. 73 Challenges related to neuroradiology, biopsy, and surgery are exacerbated by the brain unique anatomical structure and BBB. However, a growing body of literature suggest that exosomes provide a liquid biopsy option that is both convenient and capable of representing the tumor's complex heterogeneity. 74 In glioma, exosomes play a critical role in radioresistance status via Wnt/ß-catenin signaling pathway. 75 Exosomal miR-21 is a promising indicator for glioma diagnosis and prognosis, 76 while exosomal miR-151a is a potential therapeutic target in TMZ-resistant glioma. 77 In addition to non-coding RNA, coding RNA transported by exosomes is also plays a role in tumorigenesis. 78 The abundant exosomes secreted by glioma cells lead to the expansion of regulatory T-cells (Tregs), 79 and enrichment of vital markers such as epidermal growth factor receptor (EGFR) and isocitrate dehydrogenase 1 (IDH1), ultimately favoring tumor transformation or progression. Exosomes also contain elevated level of monocyte cells and programmed death-ligand 1 (PD-L1, 80 while proteins associated with EIF2, mTOR, and ephrin-B signaling pathways are abundant in the exosomes derived from glioma-derived stem cells (GSCs). 72
Leukemia
Leukemia, unlike solid tumors, has not been extensively studied in terms of the role of exosomes. Leukemia patients are defined by T-cell deficiency and exosomal miR-155 can be transferred to Treg cells, leading to immune escape via PD-L1 expression. 81 This finding highlights the potential significance of exosomes in leukemia research. Additionally, exosome-encapsulated microRNA has been found to induce Hedgehog signaling, 82 and the malignant microenvironment may promote tumor-derived exosomes by affecting phenotype of cancer-associated fibroblasts (CAF) in bone marrow stromal cells. 83 Another subtype of leukemia, multiple myeloma, has been identified as having a pro-angiogenic function that increases the tumorigenic profile of myeloid-derived suppressor cells (MDSCs). Cytokines play a role in the malignant microenvironment and are mediated and executed through function of exosomes. Moreover, tumor antigen recognition and immune escape are additional roles that exosomes play in leukemia.84,85
Reproductive Tumor
Breast Cancer
Breast cancer (BC) is the most prevalent cancer in females, and drug resistance remains a major challenge despite the development and clinical application of various novel drugs. Exosomes, which mediate cell communication have been identified as significant contributors to drug resistance in BC by facilitating the exchange and metabolism of drugs, as well as delivering proteins or ncRNAs. 86 However, due to the heterogeneous of BC subtypes, understanding the role of exosomes in drug resistance is complex. Various ncRNAs, such as miR-423-5p, miR-155, miR-1246, miR-221/222, miR-567, lncRNA APAP2-AS1, and lncRNA-SNHG14, lncRNA-H19, lncRNA-UCA1, as well as coding proteins, including GSTP1, UCH-L1, P-gp, TRPC5, and Survivin, have been found to mediate drug resistance in BC through exosomes. 87 Moreover, exosome cargos induce CSC phenotype and expression of stemness-associated genes . 88
Exosomes can also polarize macrophages into M1 or M2 sub-phenotypes, depending on the tumor type.89,90 The STAT3 signaling pathway in bone marrow-derived macrophages (BMDMs) is stimulated, followed by cytokines secretion, such as IFNγ and IL-1β, then represent M1, while M2 phenotype is stable. 91 In triple-negative breast cancer (TNBC), exosomes contribute to M2 polarization, creating a favorable niche for lymph node metastasis. 90 However, miR-16, derived from malignant exosomes, decreases the IKKα expression and promotes NF-κB signaling pathways, 92 it has the potential to suppress infiltration potential and M1 polarization, leading to reduced tumor growth.
Moreover, exosomes can be stimulated by radiation and transmit non-targeted effects (NTEs), including immunostimulatory radiation is a necessary technique in BC treatment, and recently, NTEs have been identified in addition to the direct ionization damage. One of the mechanisms through which NTEs are transmitted is the increased exosome production by cells after being stimulated by radiation. Exosomes derived from irradiated cells have been identified as a vehicle for transferring IFN-stimulatory dsDNA to dendritic cells.93–95 BC has poor immunogenic. 96 And so far, no effective immunotherapy is widely employed clinically; the study of exosome-mediated mechanisms in BC is promising for developing effective immunotherapy.
Renal Cancer, Prostate Cancer, and Bladder Cancer
Renal cell carcinoma (RCC) is a common malignancy of the urinary system, ranking second in prevalence among urological cancers after prostate and bladder cancers. RCC is the most lethal form of renal cancer, and renal clear cell carcinoma is its most common pathological type. The standard treatment for localized primary kidney cancer is radical nephrectomy. However, approximately 25% of patients who undergo surgery will develop distant metastases, while 30% of primary kidney cancer patients present with local progression or distant metastases at the time of diagnosis. Systemic drug therapy, including chemotherapy, targeted therapy, and immunotherapy, is the main treatment option for patients with local progression or distant metastases. 97 Targeted drug therapy have been shown to significantly improve the objective response rate and prolong the survival time and progression in metastatic kidney cancer patients. The discovery of new immunotherapeutic drugs has ushered in a new era in kidney cancer drug therapy. However, advanced kidney cancer remains challenging to treat, and drug resistance is a significant issue. Recent research has focused on new drug discovery and drug resistance. Tumor cells are metabolically active and secretory, and exosomes have been implicated in tumor occurrence, development, and drug resistance in various urological tumors, including renal cancer. In bladder cancer, exogenous overexpression of exosome miR-23 can affect macrophage phenotype and promote tumor metastasis, indicating that miR-23 may have a pathogenic role in bladder tumorigenesis. 98 Furthermore, Franzen et al found that co-culturing muscle-invasive bladder cancer-derived exosomes with uroepithelial cells resulted in mesenchymal cell-like phenotypes in uroepithelial cells, which may contribute to the invasive and metastatic behavior of bladder cancer. 99 Similarly, exosomes have been implicated in prostate cancer metastasis and drug resistance.100–102 Exosomes derived from the sera of prostate cancer patients can influence the response to docetaxel. 103 During prostate cancer bone metastasis, exosomal miR-214 found to enhance osteoblast differentiation through the Notch 1 signaling pathway, and high expression of exosomal miR-940 significantly enhanced prostate cancer bone metastasis.104,105
The exosomes produced by kidney cancer are easily accessible through blood and urine, making them an ideal source for clinical studies. Several studies have identified specific components of exosomes, including proteins (eg, MMP9, PODXL, DKK4, CAIX), miRNAs (eg, miRNA-451, miRNA-378, miRNA-1233, miRNA-210), and lncRNAs, that have high relevance to kidney cancer 106 and are significantly different in kidney cancer patients compared with normal subjects. In addition to being used as serological markers for early diagnosis and prognostic assessment of kidney cancer, exosomes may play a role in its development, progression, and drug resistance.
Recent studies have shed light on the crucial involvement of exosomes in the malignant biological behavior of certain tumors and drug therapy resistance. Tumor cells secrete exosomes into the microenvironment, where they promote tumor cell proliferation and distant metastasis, although the exact mechanism of action remains poorly understood. Exosomes have been found to wrap around anti-cancer drugs and release them outside of the cell, thereby reducing their effectiveness. Moreover, exosomes can compete with anti-cancer drugs for binding to the target site and can weaken the effects of the drugs by interfering with drug-carrying lysosomes. Additionally, drug-resistant cells can secrete exosomes that transmit drug-resistant information. Emerging evidence suggests that exosomes may contribute to drug resistance in a variety of cancer types, including myeloma, hepatocellular carcinoma (HCC), lung cancer, breast cancer, bladder cancer, and prostate cancer.107,108
Target therapy and immunotherapy are the main treatment options for RCC, including TKIs (tyrosine kinase inhibitors), mTOR inhibitors, and PD-1 and PD-L1 inhibitors. Sorafenib, sulforaphane, pegaptanib, cabozantinib, everolimus, pabrolizumab, and nabulizumab are commonly used. Even though existing drugs have enabled some kidney cancer patients to gain survival benefits, there is still a giant gap in improving the overall effect. Researches have therefore focus on understanding and combating drug resistance in kidney. Exosomal miRNA, lncRNA, and protein are among the most common research targets for studying the mechanisms of drug resistance. In one study, researchers discovered that the renal cancer exosome lncARSR could increase the expression of AXL and c-MET, both of which are associated with drug resistance in kidney cancer, by competitively binding to Mir-34/Mir-449. This interaction promotes the patients’ resistance response to sunitinib, and activated lncARSR can be transported to sensitive cells through exosomes, thereby propagating sunitinib resistance. 109 Targeting AXL/c-MET inhibitors or locking lncARSR-targeted nucleic acids has been shown to restore sunitinib sensitivity in renal cancer. Therefore, efforts to overcome drug resistance through these mechanisms may lead to improved outcomes for patients with RCC.
Digestive Tumors
Digestive system tumors mainly include gastric, colorectal, esophageal, liver, and pancreatic tumors. Exosomes have been studied for their roles in intracellular communication during tumor development. Exosome-associated RNAs, miRNAs, proteins, DNAs, and even metabolites can change the fate of recipient cells by ECM communication. 110 Exosomes play a key role in building a supportive microenvironment in the organs to support cancer growth.
Gastric Cancer
Emerging evidence indicates that exosomes are directly involved the development and progression of gastric cancer (GC). Gastric cancer exosomes have been found to promote tumor cell proliferation by activating key pathways such as PI3K/Akt and MAPK/ERK. 111 Meanwhile, Exosome-mediated transfer of EGFR can lead to suppression of miR26a/b expression and upregulation of hepatocyte growth factor (HGF) expression, which creates a favorable microenvironment for liver-specific metastasis. 112 Some exosomes from other cells also can facilitate GC growth. For example, p53 mutation mesenchymal stem cell (MSC) can secrete ubiquitin protein ligase E3 component n-recognin2 (UBR2) rich exosomes to promote GC growth and metastasis by activating the wnt/β-catenin signaling pathway. 113 GC cells derived MiR-21-5p can induce peritoneal mesothelial cells to undergo mesothelial-to-mesenchymal transition by targeting SMAD7, ultimately promoting GC peritoneal metastasis. 114 Overall, these findings highlight the significant role of exosomes in the pathogenesis and progression of gastric cancer, suggesting that targeting exosomal signaling pathways may hold promise as a potential therapeutic strategy.
Colon Cancer
Colorectal cancer (CRC)-derived exosomes contain various biomolecules such as proteins (eg, TGFβ, VEGF-A, αvβ5 integrin, PAD4, angiopoietin-like protein 1 (ANGPTL1), WNT4), RNA (eg, miR-25-3p, miR-21, miR-27a, miR-10b, miR-2149-5p, miR-6737-5p, miR-6819-5p), and DNA. For example, CRC cells can induce CAFs formation by stimulating macrophage to secret TGF-beta-containing exosomes.115,116 Exosomes from CRC can also reprogram the proteome of CAFs, leading to distinct phenotypes in primary or metastatic CRC within the same patient. 117 In hypoxic conditions, CRC cells can promote endothelial cell migration and angiogenesis through secreting exosomes containing growth factor, such as VEGF-A. 118 Exosomal WNT4 from CRC cells can also activate endothelial cells via the Wnt/β-catenin signaling pathway. 119 Additionally, Proteomic studies have shown that in colon cancer, the exosomal αvβ5 integrin was associated with liver metastases, while the α6β4 and α6β1 integrins were associated with lung metastases. 120 Exosomal PAD4 can affects the citrullination of ECM, which promote colon cancer EMT and increase CRC cells metastasis to the liver. 121 Exosomal ANGPTL1 downregulated MMP9 level in Kupffer cells by inhibiting the JAK2-STAT3 signaling pathway, which prevented the liver vascular leakiness and attenuated liver metastasis. 122
CRC cells secrete Exosomal miR-25-3p to regulate the expression of VEGFR2, ZO-1, occluding, and Claudin5 in endothelial cells by targeting KLF2 and KLF4, consequently promoting vascular permeability and angiogenesis. 123 Exosomal miR-21 promotes colon cancer cells proliferation, invasion, and therapy resistance through target PDCD4, and engineered exosomes deliver miR-21 inhibitors can reverse drug resistance. 124 MiR-27a derived from gastric and colon cancer can transform fibroblasts into cancer associate fibroblast (CAFs). 125 MiR-10b secreted from CRC cells can inhibit fibroblasts’ PIK3CA expression and reduce PI3K/Akt/mTOR pathway activity, promotes expression of TGF-β and α-SMA, and enable fibroblasts to acquire the characteristics of CAFs. 126 Exosomal miR-2149-5p, miR-6737-5p, and miR-6819-5p can inhibit the expression of TP53 in fibroblasts to promote tumor proliferation. 127
Exosomal DNA regulates tumor-associated immunity. Hippo pathway kinase Lats1/2 knock out cells can release nucleic acid-rich exosomes, which induce antitumor immunity by stimulating the TLR-IFN pathway. 128 After being treated with topotecan, cancer cells release DNA-containing exosomes, triggering dendritic cell (DC) activation and subsequent CD8+ T cell activation via CGAS-STING pathway. 129
Non-CRC original exosomes remodel CRC cells as well. The miR-92a-3p from stromal cell exosomes can induce CRC cells epithelial mesenchymal transition through activates Wnt/β-catenin pathway and inhibits mitochondrial apoptosis by directly inhibiting FBXW7 and MOAP1. 130 Some exosomes can suppress cancer growth, for example, bone marrow mesenchymal stem cells (BMSCs) derived-exosomal miR-22-3p can inhibit CRC cells proliferation and invasion via mediating RAP2B/PI3K/AKT pathway. 131
Pancreatic Cancer
Exosome-delivered CD44v6 promotes pancreatic and CRC cell motility, invasion, and cancer-initiating cell marker expression. 132 Exosome-delivered CD44v6/C1QBP complex drives pancreatic cancer liver metastasis through promote fibrotic liver microenvironment. 133
Tetraspanins, a family of proteins that cross the membrane four times with a short amino-and carboxy-terminal tail, are abundant in exosomes. The tetrapanins CD82 and CD9 mostly suppress tumor progression. CD151 and tetraspanin 8 are overexpressed in several human tumors and seem to support tumor progression. 134 Macrophage migration inhibitory factors (MIF) are high expression in pancreatic cancer-derived exosomes, which regulate tumor metastasis to the liver. 135 Pancreatic cancer distant liver metastasis has a significantly higher MIF expression level in exosomes than in primary tumor. So, identifying MIF in exosomes might be a potential prognostic marker of pancreatic cancer. In pancreatic ductal adenocarcinoma, Myoferlin has a high expression level, and it can regulate VEGFA secretion through exosome and promote tumor-associated angiogenesis as well. 136
miR-27a has been reported to have a high expression in pancreatic cancer. Pancreatic cancer derived-exosomes containing miR-27a promote tumor associated endothelial cells’ proliferation, invasion, and angiogenesis by suppressing B-cell translocation gene 2 (BTG2), which promotes pancreatic cancer survival and growth. 137 MiR-222 is often overexpressed in highly metastatic pancreatic cancer; it can be released to ECM through exosome. miR-222 can decrease tumor cells’ P27 expression via PPP2R2A/AKT axis. As a result, those poorly metastatic pancreatic cancer cells can gain of high proliferation and invasion ability. 138 MiR-301a from pancreatic cancer-derived exosome can be uptake by macrophages, it can be polarized into M2 phenotype due to miR-301a activate cells’ PTEN/PI3K signaling cascade. 139 Circular RNA also plays an important role in exosome besides miRNAs. Exosomal circular RNA IARS (circ-IARS) can promote tumor metastasis by increasing endothelial cell activation and down-regulating miR-122 and Zonula occludens-1 (ZO-1). 140
Liver Cancer
HCC, CAFs can secrete cytokines through exosomes, such as vascular endothelial growth factor (VEGF), bFGF, TGF-β, matrix metalloprotein 2 (MMP2) and MMP9, therefore to promote cancer proliferation and metastasis. 141 Proteomics results have demonstrated that HCC cells tend to secret more sugar metabolism regulatory proteins through exosomes, which aid cancer cells in forming a tumor microenvironment. 142
HCC-derived exosomal miR-21 can convert fibroblast to CAFs, through depressing PTEN, upregulating PDK1/ATK pathway. 143 Exosomal miR-93 can stimulate HCC proliferation and in vitro invasion by suppressing CDKN1A, TP53INP1 and TIMP2 function. 144 External original miR-122 promotes the chemosensitivity of HCC cells. 145 MiR-1247-3p induces CAF to secret pro-inflammatory cytokines, including IL-6 and IL-8, to foster lung metastasis of HCC. 146
EVs for Drug Delivery
As precise therapy and targeted therapy have expanded, nano-enabled drug delivery/imaging techniques have significantly contributed to the development of intelligent carriers in recent years. 147 Of note, lipid-based, and polymer-based nanocarriers provide versatile platforms for drug encapsulation, resulting in the clinical translation of multiple formulations. Aside from artificial nanocarriers, cell-derived extracellular-vesicle carriers have attracted substantial interest. EV-based cell-cell communications are conserved across all realms of life. Compelling evidence has suggested that EVs are involved in major pathological and/or physiological processes, including cancer development. From the drug delivery perspective, EVs are advantageous over synthetic carriers, which have opened new opportunities for drug delivery. However, the clinical translation of EV-based therapy remains challenging. The basic biology of EV-based therapy focusing on understanding the biogenesis, release of these vesicles, and the fate of these natural carriers upon interaction with target cells is crucial. The genotypic responses that EVs induce and the mechanisms by which EVs mediated cell communications also need to be clarified.148,149 EVs can encapsulate hydrophilic drugs in their cores and contain hydrophobic molecules in their lipid bilayers due to their specific structure. There are several ways to incorporate charges into EVs, such as repeat extrusion through membranes, co-incubation, sonication, free/thaw cycles, electroporation, and permeabilization by involving surfactants, etc. (Figure 1).

After isolation of the exosome vesicles, various drugs/imaging agents can be loaded through multiple methods.
The biogenesis of exosomes starts from the endosomal system. Early endosomes develop into late endosomes/multivesicular bodies (MVBs), which invaginate the endosomal membrane to create intraluminal vesicles (ILVs) in the lumen of organelles. 150 ILVs, also known as exosomes, are released into the extracellular environment by MVBs fusing with the cell's plasma membrane. In both normal and pathological conditions, diverse cell types may secrete exosome vesicles involved in distinct biogenesis pathways. 151 Recognition of EVs by cells has been defined by surface receptor interaction, direct membrane fusion, micropinocytosis, and phagocytosis; 149 these processes may be cell-type dependent, for which the communication of surface molecules between specific cells and EVs is critical for recipient cell recognition and adhesion.148,152 In addition, like many other nanocarriers, EVs must escape endosomes to release their cargo. As a result of endosomal escape, acidic compartments of the lysosomal pathway are degraded, which could affect the integrity of EV charges. 153 Despite the initial speculation that EVs could facilitate direct membrane fusion and cytosolic delivery, vesicle uptake has been found to be an extremely complex process, requiring more in-depth analysis using high-resolution imaging or novel live-cell reporters.154,155
From the perspective of EVs’ targeting abilities of drug delivery in cancer treatment, as opposed to lipid nanoparticle carriers that are mainly passively deposited in tissues unless additional surface ligands are present on their surfaces. EVs may have inherent targeting abilities from the parent cells, such as homologous targeting of cancer cell-derived EVs to certain cancer cells 156 and the capability to deliver functional RNAs across various biological barriers, such as the blood–brain barrier. EVs may be a natural route for efficient drug transport, as synthetic drug delivery systems have proven to be substantially less targeted than natural drug delivery systems. 157 In fact, different mammalian cancer cell EVs have shown tumor homing.158,159 In addition, there are safety concerns regarding these cancer EVs as drug carriers due to the possibility of tumor migration and epithelial–mesenchymal conversion, which could lead to tumor resistance. 160
The biodistribution pattern of EVs derived from different cell lines was compared, and the findings showed that, like lipid and polymer nanoparticles, when systematically delivered, EVs accumulate non-specifically in the liver, spleen, and lungs; despite significantly being influenced by the vesicle source and the route of administration (Figure 2), such as EVs from dendritic cells were preferentially accumulated in the spleen, while EVs from BC cells were more prominently accumulated in the liver.159,161 Interestingly, intranasal delivery of ligand-modified EVs showed substantial accumulation in orthotopic glioma tissue. 158 However, half-lives of EVs are notably shorter than that of lipid or polymer nanoparticles. The terminal half-life of EVs, even when modified with polyethylene glycol for enhanced stealth property, was a maximum one hour, while that of modified lipid/polymer nanoparticles could be as long as several hours.162,163 Noteworthy, parallel biodistribution studies between EVs and lipid/polymer nanoparticles are required, especially under the same experimental conditions. Moreover, the stiffness factor of the target cell environment is another important yet underestimated parameter indicating the effectiveness of EV nanocarriers.
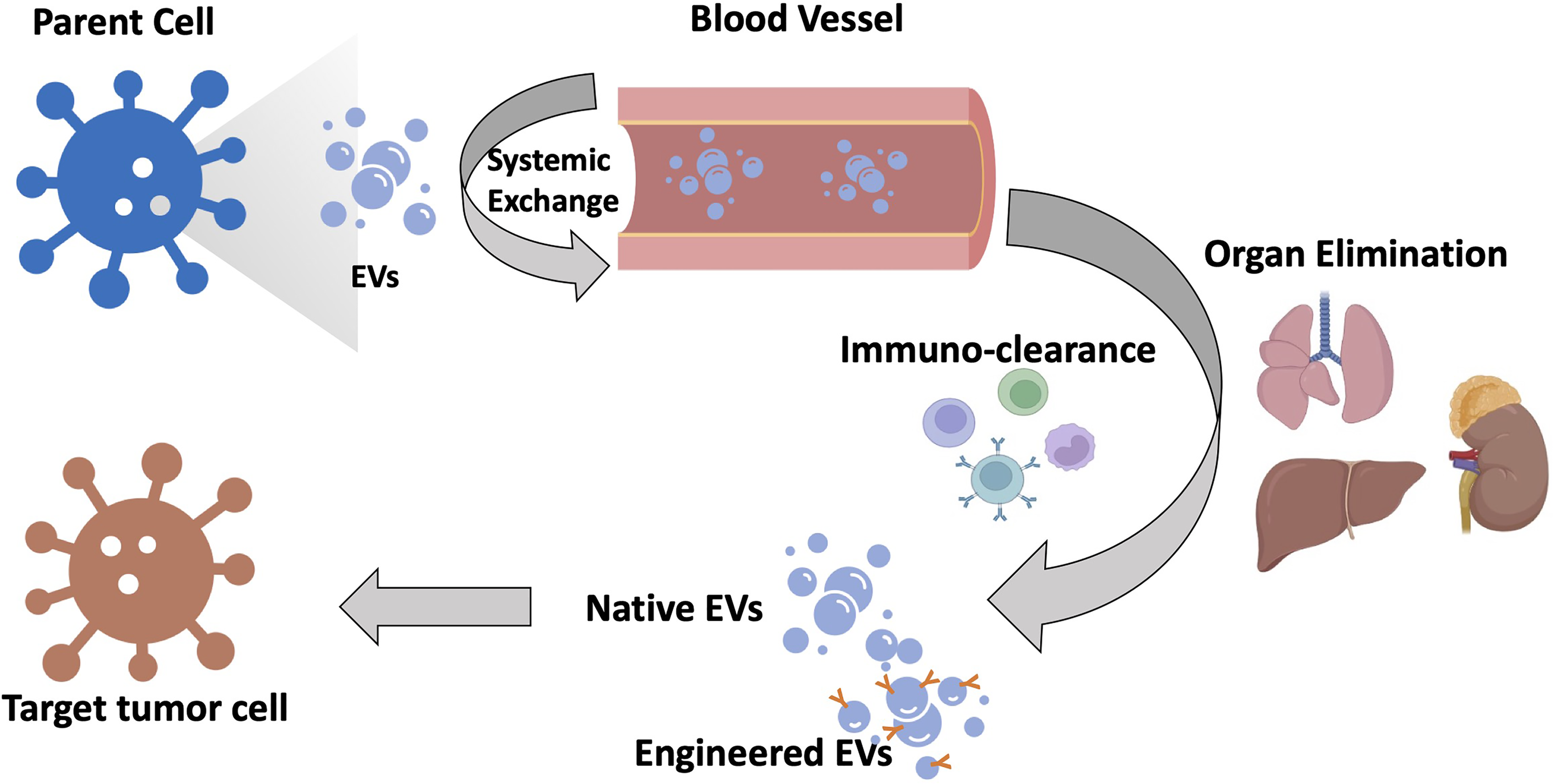
Schematic illustration of EVs’ biodistribution. When EVs are secreted from their parental cells, they may engage in cell-to-cell exchange; once they reach circulation, they should bypass the immune system and accumulate in tumors. Targeting efficiency may be determined by the inherited or engineered functionalization of EVs and the interaction between EVs and their targets.
Although EVs have been exploited to develop intelligent drug delivery systems that exhibit promising advantages over synthetic nanocarriers in terms of targeting, pharmacokinetics, and safety; clinical translation remains a challenge due to the natural complexity of the EVs as drug carriers, such as size heterogeneity, and batch-to-batch fluctuations occurring during the manufacturing process, etc Patients’ cells can be transferred into a culture medium, and isolated vesicles can then be re-administered to them as autologous EVs. Autologous EV use is possible under certain conditions, including the delivery of therapeutic agents to tumor tissue using vesicles from cancer patients. 164 However, it may be limited in its application to other acute diseases, such as cardiovascular incidents and sepsis. 165 Nevertheless, non-autologous EVs are likely to be used for most applications mainly for safety reasons and the regulatory necessity of having a streamlined, exceptionally qualified product. To date, mesenchymal stromal cell-derived EVs for regeneration and DC-derived EVs for vaccine delivery have been found to be promising in phase I clinical trials.166,167
Overall, EVs can be used for a variety of drug delivery applications. When administered systemically to animal models, EVs delivered functional cargos with reduced immuno-clearance compared with conventional delivery methods. However, it is still necessary to conduct further testing in clinically relevant scenarios and perform unbiased comparisons with lipid/polymer nanocarrier-based alternatives to systematically evaluate the potential benefit-risk. To translate EVs successfully, cost-effective and mass production, precise isolation, and characterization techniques for measuring batch-to-batch variations and conveniently available packaging methods are required. Innovative approaches, such as the fusion of liposomes with EVs to improve drug loading and targeting efficiency have been explored.168,169 Specifically, microfluidics has recently been reported to produce more uniform EVs.158,161 Further understanding of how EVs infiltrate cells and evade immune detection is necessary to unlock the full potential of this promising therapeutic platform.
EVs and Immune Response
More and more researchers have reported the EVs’ role in the immune response. EVs may not only act in tumorigenesis and immune suppression but also in immune activation and tumor suppression. Tumor cells may evade immune surveillance by the elevated expression of programmed death-ligand 1 (PD-L1) 170 and suppress the inflammation response. 171 Other mechanisms were also been reported, such as via miRNA, 172 promoting tumor cells’ epithelial–mesenchymal transition, inducing angiogenesis, establishing the pretransfer microenvironment, immune escape, and so on. 173 While EVs from other types of cells can suppress tumor growth and metastasis. They may activate T cells by direct or indirect antigen presentation and transfer naïve antigens to antigen presenting cells (APCs). 174 Numerous reports have proven the therapeutic roles of EVs. 175 Therefore, one of the promising methods is to develop a novel tool to suppress tumor EVs’ role or activate the immune response to suppress the tumor growth or combinate with engineered EVs to delivery drugs to the target tumor cells.
EVs and Clinical Applications
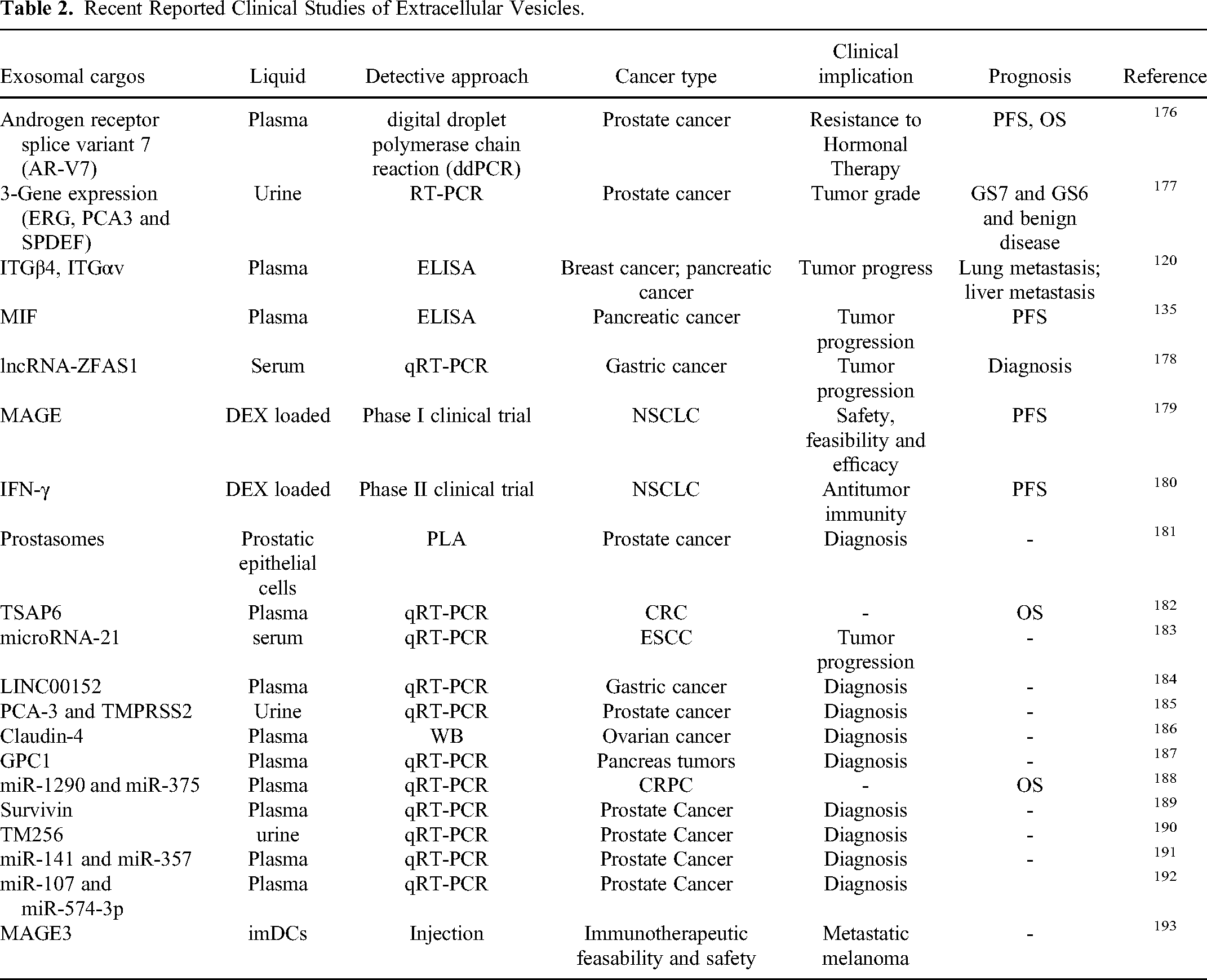
Currently, there are more than 130 clinical trials related to EVs that have either been completed or are still ongoing in the United States alone. No specific criteria have been established to define the potential applications of EVs in various diseases, detection or therapy. Therefore, most studies and clinical trials refer to the MISEV2018 guidelines to identify and characterize EVs. Tables 2 and 3 illustrate recent clinical studies focusing on EVs, drug delivery, and clinical applications, respectively.
Recent Reported Clinical Studies of Extracellular Vesicles.
Recent Undergoing Clinical Studies of Extracellular Vesicles.

Conclusion
Although there have been some breakthroughs in the study of EVs, much work still needs to be done to apply them to clinical and industrial applications. We cannot expect practical applications in the near future due to the limited research on EVs. In order to improve the sensitivity and specificity of tumor patient diagnosis in the future using EVs, extensive work is required to transfer bench work to clinical practice. However, EVs have a promising future in cancer diagnosis, therapy (including not only targeted therapy, but also chemotherapy and immunotherapy), and drug delivery (as shown in Figure 3).

Graphical abstract of EVs’ release, functions, and applications.
Footnotes
Author Contributions
MT wrote the introduction, EVs isolation methods, and immunosuppression, and organized the whole paper. XW wrote RCC, YG wrote brain tumor, leukemia, BC, and clinical trials. HT wrote digestive tumor. YW and FH wrote EVs structure, YL wrote EVs release, drug delivery, and draw all figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by Postdoctoral Fund of People's Government of Jiangsu Province, China (200730000107 to YG).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
