Abstract
Introduction
Conventional transbronchial needle aspiration (TBNA) was introduced by Wang with a flexible bronchoscope to provide tissue diagnosis of lesions adjacent to the main airway. 1 With the use of ultrasound in bronchoscopy, conventional TBNA has been replaced by endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA), which has been extensively used for the evaluation of mediastinal and hilar lymph nodes, as well as peribronchial lesions, and has become standard practice in an increasing number of institutions.2–4 The most frequent application of EBUS-TBNA is for the diagnosis and accurate staging of lung cancer and other metastatic tumors.5–8 In addition, the procedure is also utilized for the assessment of mediastinal or hilar lymphadenopathy caused by benign lesions, such as sarcoidosis and infection.9–11 Although EBUS-TBNA is superior to conventional TBNA in diagnosing intrathoracic lymphadenopathies,12,13 the pathologic findings are sometimes nonspecific, and distinguishing malignant from benign lesions is challenging and imperative.
An EBUS-TBNA procedure usually includes several aspirations. When aspirations are all-shot, 2 to 3 aspirations are generally enough. However, one or more aspirations without a tissue core (punctures that aspirate no solid substance in the EBUS-TBNA needle) are common in clinical practice, and more aspirations are necessary in this case. To the best of our knowledge, no study has systemically compared aspirations with a tissue core and aspirations without a tissue core in the diagnosis. This study aimed to evaluate the value of all-shot aspirations in EBUS-TBNA to distinguish malignant from benign lesions.
Patients and Methods
Study Design and Participants
This single-center retrospective study was conducted at a tertiary hospital between January 2017 and March 2021. Operating record charts of bronchoscopy were screened, and patients fulfilling the following inclusion criteria were enrolled: (1) age ≥18 years; (2) undergoing EBUS-TBNA because of pulmonary abnormality for the first time during the study period; (3) the number of aspirations and biopsy specimens were recorded, and at least 2 aspirations were performed in an EBUS-TBNA; and (4) a pathologic diagnosis and a clinical diagnosis associated with the pulmonary abnormality was obtained. Patients were divided into 2 groups: patients with aspirations all with a tissue core (all-shot group), and patients with one or more aspirations without a tissue core (no-tissue-core group).
EBUS-TBNA was performed under local anesthesia by pulmonologists using the Olympus BF-UC180F system (Olympus Inc.) at our hospital. The 22-G aspiration needle (model no. NA-201SX-4022) and 20-mL aspiration pressure were routinely utilized.
Pathologic Diagnosis and Clinical Diagnosis
Pathologic diagnoses after EBUS-TBNA were divided into 3 groups: (1) neoplasms; (2) granuloma or features indicative of granuloma (epithelioid cells, or multinuclear giant cells); and (3) nonspecific pattern without features of malignancy or granuloma. Medical records of all included patients were read through to make a clinical diagnosis. For pathologic nonspecific subjects, if medical records failed to produce a diagnosis, follow-ups by clinic or telephone were undertaken to obtain a clinical diagnosis.
Statistical Analysis
Data were analyzed using the Statistical Package for the Social Sciences (SPSS, version 22.0 software, SPSS Inc.). Age and EBUS-TBNA were reported in numbers and expressed as mean ± standard deviation, and a Student t test was used for comparison. Patient numbers were reported in numbers and percentages. The χ2 test was applied for categorical variables in subgroups. P values of less than .05 were considered to be statistically significant.
Ethics Approval
The study was approved by the Ethics Committee of our hospital. Due to its retrospective nature, patient informed consent was waived. The raw data were first extracted from the hospital information system, and patient identities, including names, screening IDs, patient IDs, and mobile phone numbers, were de-identified. This retrospective study was conducted in accordance with the STROBE guidelines. 14
Results
During the study period, 1108 EBUS-TBNA procedures were performed. According to inclusion criteria, 557 EBUS-TBNAs were excluded because of repetition or lacking a description of aspiration numbers. Five patients had only one aspiration in an EBUS-TBNA and were excluded. During follow-up, 41 patients were lost to follow-up or failed to achieve a clinical diagnosis and were also excluded. Therefore, 505 patients were enrolled in the study for analysis (Figure 1).
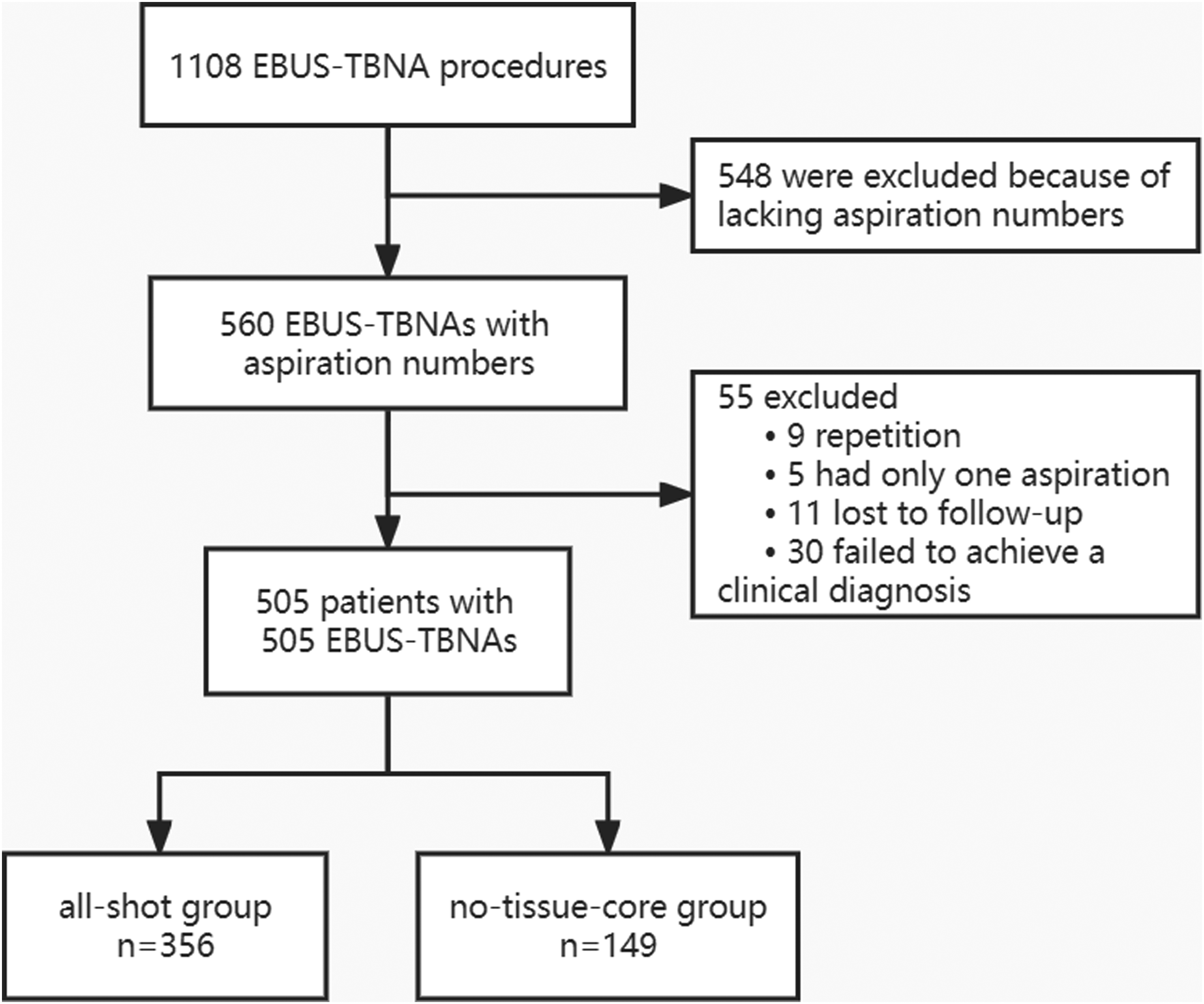
Flow diagram for patient inclusion.
Patient Characteristics
Patient characteristics are shown in Table 1. All 505 patients underwent 1402 aspirations, 1184 (84.5%) of which had a tissue core. All the patients had a mean age of 55.0 ± 12.3 years, with a slight male prevalence (52.1%). Pathological examinations of all 505 patients revealed neoplasms in 214 (42.4%) patients, granulomas in 158 (31.3%) patients, and nonspecific pathological findings in 133 (26.3%) patients. All patients with pathological neoplasms proved to have a clinical diagnosis of malignancy, and all patients with pathological granulomas proved to have a clinical diagnosis of benign disease. All 133 patients with pathologic nonspecific findings were followed up, and a clinical diagnosis of malignancy was proven in 31 patients and benign disease in 102 patients. Therefore, in all 505 patients, a clinical diagnosis of malignancy was proven in 245 patients, 88.2% of whom had lung cancer; and a clinical diagnosis of benign disease was proven in 260 patients, 53.1% of whom had sarcoidosis and 27.3% had a pulmonary infection.
Characteristics of Patients Undergoing EBUS-TBNA and the Diagnosis.

Abbreviation: EBUS-TBNA, endobronchial ultrasound-guided transbronchial needle aspiration.
When 2 or more stations are aspirated, each is included.
P values less than .05 is indicated in bold.
Comparison of All-shot Group and No-tissue-core Group
The all-shot group had 356 (70.5%) patients with aspirations all with a tissue core, and the no-tissue-core group had 149 (29.5%) patients with at least one aspiration without a tissue core. No difference was observed between the all-shot group and the no-tissue-core group in terms of age and sex. The all-shot group had statistically fewer aspirations, but more biopsy specimens for each patient than the no-tissue-core group. When aspiration locations were considered, a statistical difference was not observed in lymph node stations between the 2 groups except 4R and 11R (Table 1).
In terms of pathologic diagnosis after EBUS-TBNA, neoplasms were noted in 164 of 356 (46.1%) of all-shot patients, but in only 50 of 149 (33.6%) of no-tissue-core patients (odds ratio [OR], 1.69; 95% confidence interval [CI], 1.14-2.52; P = .009). Regarding final clinical diagnosis, malignancy was noted in 189 of 356 (53.1%) of all-shot patients, but in only 56 of 149 (37.6%) of no-tissue-core patients (OR, 1.88; 95% CI, 1.27-2.78; P = .001). As to 133 pathologic nonspecific patients after EBUS-TBNA, a clinical diagnosis of malignancy was noted in 25 of 79 (31.6%) of all-shot patients, but only 6 of 54 (11.1%) of no-tissue-core patients (OR, 3.70; 95% CI, 1.40-9.79; P = .006).
Sensitivity Analysis
Because more specimens were collected in the all-shot group (Table 1), the specimen number was a possible confounding factor in this study. We conducted a comparison of biopsy specimen numbers in patients with malignancy disease and benign disease and discovered that the number of biopsy specimens was similar between the 2 groups (2.40 vs 2.29 per patient, P = .066). Additionally, we compared all-shot patients and no-tissue-core patients in the subgroups with 2 specimens (n = 315) and 3 specimens (n = 147). In EBUS-TBNAs with 2 specimens, malignancy was found in 106 of 209 (50.7%) of all-shot patients, but only in 39 of 106 (36.8%) of no-tissue-core patients (P = .019). In EBUS-TBNAs with 3 specimens, malignancy was found in 70 of 129 (54.3%) of all-shot patients, but only in 6 of 18 (33.3%) of no-tissue-core patients (P = .096).
Discussion
No-tissue-core aspirations in TBNA are not rare in clinical practice. Lee et al 15 evaluated the number of aspirations required for non-small cell lung cancer and showed that 24.3% of aspirates did not contain solid substances. Our study revealed that 15.5% of aspirations did not have a tissue core, and only 80.5% of the patients had all-shot aspirations in an EBUS-TBNA. More pathological neoplasms and clinical malignancies were observed in all-shot patients than in no-tissue-core patients.
No-tissue-core aspirations are affected by various factors and are usually due to failure to access lymph nodes in conventional TBNA. However, real-time EBUS-TBNA allows visualization of the TBNA needle within the ultrasound plane and enables easy and reliable access to lymph nodes, even in less experienced hands. 2 Despite this, no-tissue-core aspirations are still commonly seen in EBUS-TBNA. Except for the operator's skill level, size of the punctured lymph nodes or lesions, and selection of the puncture area, the texture of lymph nodes is also a probable explanation for no-tissue-core aspirations. Heterogeneity is a feature of malignancy, reflecting areas of high cell density, necrosis, hemorrhage, and myxoid change. 16 Computed tomography (CT) texture analysis for quantifying heterogeneity accurately differentiates malignant and benign mediastinal nodes in lung cancer 17 and is useful for discriminating between mediastinal lymphadenopathy caused by sarcoidosis and that caused by small cell lung cancer. 18 The texture of lymph nodes may influence the amount of tissue obtained by TBNA, and further studies are necessary to compare the association between the CT features of lymph nodes and all-shot or no-tissue-core aspirations.
The specimen number may influence the tissue adequacy and the pathological finding. Studies have reported the optimal results for non-small cell lung cancer need 3 EBUS-TBNA aspirations per lymph node station 15 and 4 to 5 aspirations for sarcoidosis. 19 However, the specimen number was not associated with the diagnosis of malignant or benign disease in our study. Furthermore, we found that in subgroups with 2 or 3 specimens, the all-shot group also had a higher incidence of malignancy (although no statistical significance was found in the subgroup with 3 aspirations due to its small patient number). Therefore, although more specimens were collected in the all-shot group, the specimen number did not influence our conclusion that patients with all-shot aspiration may have more malignancy. Furthermore, our study showed that the incidence of malignancy in all-shot patients was approximately 3 times that in no-tissue-core patients for patients with pathologic nonspecific findings after EBUS-TBNA. Therefore, when an EBUS-TBNA revealed pathologic nonspecific findings, all-shot aspirated patients are at higher risk for malignancy and more measures should be taken to confirm the diagnosis.
This study had some limitations. First, due to its retrospective nature, the sample size was not calculated and justified. The data in some cases were incomplete, and some patients were lost to follow-up. Second, patient inclusion bias was an issue since half of the cases were excluded because their operating record charts did not mention aspiration numbers. Third, although we excluded patients with only one aspiration in the EBUS-TBNA, the aspiration number in an EBUS-TBNA still varied from 2 to 7, which influenced the number of no-tissue-core aspirations. Nevertheless, we included a relatively large number of patients and found statistical differences between the all-shot and no-tissue-core groups in the outcomes.
Conclusion
In conclusion, this study demonstrated that patients with all-shot aspirations in EBUS-TBNA are more likely to have pathological neoplasm and clinical diagnosis of malignancy than those with no-tissue-core aspirations. More measures should be taken to exclude malignancy in all-shot patients when the EBUS-TBNA was nondiagnostic.
Footnotes
Abbreviations
Authors’ Note
XS, TZ, and MW designed the work, XS, TZ, CS, PW, MC, and JZ acquired data and drafted the manuscript; XS, TZ, and MW analyzed the data and revised the manuscript. The final version of the manuscript was approved by all authors. The study was approved by the Ethics Committee of Peking Union Medical College Hospital (approval number: S-K1868, approval date: 6/12/2021, location: Beijing China). Due to its retrospective nature, patient informed consent was waived. The raw data were first extracted from the hospital information system, and patient identities, including names, screening IDs, patient IDs, and mobile phone numbers, were de-identified. All the data will be available to other researchers on reasonable requests to the corresponding author after publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National High Level Hospital Clinical Research Funding (2022-PUMCH-B-106) and the CAMS Innovation Fund for Medical Sciences under grant 2018-I2M-1-003.
