Abstract
Recent studies have identified causal links between altered gut microbiome, chronic inflammation, and inflammation-driven conditions such as diabetes and cardiovascular disease. Childhood cancer survivors (CCS) show late effects of therapy in the form of inflammaging-related disorders as well as microbial dysbiosis, supporting a hypothesis that the conditions are interconnected. Given the susceptibility of the gut microbiome to alteration, a number of therapeutic interventions have been investigated for the treatment of inflammatory conditions, though not within the context of cancer survivorship in children and adolescents. Here, we evaluate the potential for these interventions, which include probiotic supplementation, prebiotics/fiber-rich diet, exercise, and fecal microbiota transplantation for prevention and treatment of cancer treatment-related microbial dysbiosis in survivors. We also make recommendations to improve adherence and encourage long-term lifestyle changes for maintenance of healthy gut microbiome in CCS as a potential strategy to mitigate treatment-related late effects.
Background
In recent years, research has illuminated the critical role of the host microbiome in a number of essential physiological operations, including but not limited to digestion, immune regulation, and central nervous system function. 1 Microbiota composition can be perturbed by various external factors. In cancer patients, changes in the gut and oral microbiota may result from exposure to chemotherapeutic agents, radiotherapy, and antibiotics.2–8 Not unexpectedly, microbiota profile disparities between children and adolescents with cancer compared to healthy peers have been observed. Numerous studies reported reduced microbial diversity and treatment-induced alterations in the abundance of certain bacteria in the cancer group, particularly for the gut microbiome.9–12 For example, Fijlstra et al 2 demonstrated in rat models that methotrexate, a cytotoxic agent associated with gut mucositis, depletes gut bacteria, reduces microbial diversity, and modifies bacterial composition. Additionally, patients with cancer are predisposed to episodes of neutropenic sepsis which are often treated with broad-spectrum antibiotics. The use of antibiotics has a long-term impact on gut microbiota, including increased prevalence of antibiotic-resistant microorganisms.5,13 Thus, the cumulative effect of cancer treatment is a significant dysbiosis in gut bacteria.
Early childhood constitutes a “critical period” during which the microbiome is particularly susceptible to alteration; gut flora then stabilizes over the ensuing decades. 14 Children undergoing cancer treatment within this critical period may therefore develop microbial dysbiosis which remains long after completion of cancer therapy. This observation has been reported in children with acute myeloid leukemia, in whom Bacteroides and Bifidobacterium remained depleted 6 weeks after completing anti-leukemia treatment when compared to controls, despite apparent restoration of other genera. 15 We conducted a longitudinal observational study in 7 children with acute lymphoblastic leukemia and demonstrated similar findings: gut microbiota profiles remained marginally different (both in beta diversity and abundance) 3 months after chemotherapy cessation. 9 A follow-up study revealed that gut microbiome dysbiosis persisted into adulthood, suggesting a more chronic pathology. 16 Thus, this review will focus on gut microbiome reconstitution as a potential therapeutic intervention to mitigate late effects in childhood cancer survivors (CCS).
Link Between Microbiome Dysbiosis and Chronic Inflammation
Gut microbiota play a key role in immune activation through a variety of mechanisms, many of which are metabolic. The interaction between nutrition, gut flora, and regulation of inflammatory pathways is mediated by short-chain fatty acids (SCFAs), which are produced via the metabolism of complex carbohydrates by gut bacteria. Certain SCFAs have known anti-inflammatory effects. For example, the SCFA butyrate is a metabolite that increases lipolysis and promotes mitochondrial activity in adipocytes, decreasing risk of obesity and ultimately blocking the generation of pro-inflammatory cytokines.17–20 Butyrate and several other SCFAs also play a crucial role in the maintenance of the gut mucosal barrier, which prevents the translocation of inflammation-inducing microbes/microbial components into the host bloodstream. 21
Given their crucial role in maintaining immune balance, changes in bacterial metabolites and microbial translocation actively contribute to systemic immune activation and inflammation. Both these latter conditions are increasingly recognized in CCS.22–24 Gut microbiota dysbiosis and subsequent translocation have also been linked to elevated levels of systemic inflammatory markers (serum interleukin 6 [IL-6] and C-reactive protein [CRP]) in rats, thought to be caused by the presence of pro-inflammatory pathogen-associated molecular patterns.25–27 Finally, unmethylated CpG motifs, which are abundant in bacterial DNA, as well as lipopolysaccharides (LPS) in the bacterial cell wall have been shown to promote host immune activation.28–31 In short, these studies support the current understanding that microbial dysbiosis, disruption of gut mucosa, and chronic inflammation are intimately related as components of a self-perpetuating loop.
Association Between Microbial Dysbiosis, Chronic Inflammation, and Late Effects in Childhood Cancer Survivors
Given the cumulative evidence of treatment-mediated microbial dysbiosis in patients with cancer, as well as the link between gut microbiome perturbations and chronic inflammation, it is unsurprising that a large proportion of CCS goes on to develop inflammation-driven diseases in adulthood. Indeed, CCS are highly susceptible to late effects, a term that refers to therapy-related complications or adverse outcomes that persist or arise years after completion of treatment for a pediatric malignancy. 32 CCS have elevated risks of metabolic and cardiovascular disease, neurological complications, and endocrine dysfunction compared to their peers (Figure 1).33–37 A large cohort study in the United States which included over 10 000 adult survivors of childhood cancer found that approximately 60% of these survivors reported at least 1 chronic health condition within 30 years of diagnosis, of which 28% were classified as severe, life-threatening, or disabling. 38 Prevalent health conditions within the CCS cohort included obesity, diabetes mellitus, and various cardiovascular conditions, with a large increase in the risk of ischemic heart disease in particular. 38

Potential drivers and late effects in childhood cancer survivors (Figure created using BioRender under Academic License).
While there have been large advances in our understanding of microbiome-mediated chronic inflammation and thus, late effects in CCS, there have not yet been compelling studies looking at interventions focused on gut flora restitution in this population. However, a myriad of studies has evaluated gut microbial interventions for cardiometabolic conditions such as obesity, diabetes mellitus, and ischemic heart disease, all of which constitute prevalent late effects in CCS. Trends of microbial dysbiosis in CCS and patients with cardiometabolic disease appear to overlap, including changes in the relative ratio of Bacteroidetes/Firmicutes, the 2 most prevalent phyla in the gut. 39 Our group reported a significant decrease of Bacteroidetes in CCS cohorts compared to controls. 16 Similarly, studies have documented a decreased Bacteroidetes/Firmicutes ratio in obesity, coronary artery disease, and ischemic stroke.39–41 Several other microbes have additionally been implicated in childhood cancer survivorship and inflammation-related diseases. For example, Faecalibacterium, which has been called a “master regulator” of inflammatory factors IL-2, IL-6, and IL-8 in the gut, has been found to be depleted both during therapy and as a long-term effect of cancer survivorship.42–44 Reduced Faecalibacterium has also been documented in coronary artery disease and was found to be inversely correlated with levels of HbA1c in patients with type 1 diabetes mellitus (T1DM). 45 Notably, Faecalibacterium depletion has also been associated with ischemic stroke. 46 In comparison to healthy peers, our cohort of 73 young adults who survived childhood leukemia had depletion of Faecalibacterium, and this finding was significantly correlated with elevated inflammatory and immune activation markers, namely plasma IL-6, CRP, as well as HLA-DR+ CD4+ and HLA-DR+CD8+ T cells. 16 On the other hand, both CCS cohorts and patients with type 2 diabetes mellitus (T2DM) have been shown to have an enrichment of Actinobacteria, although the direct consequences of this are less well known.16,47 Given these shared associations, dysbiosis resulting from cancer treatment is a likely driver of inflammation and late effects in CCS.
Therapeutic Approaches to Modulate Microbial Dysbiosis
The studies reviewed above suggest a shared mechanism of gut microbial dysbiosis between CCS and patients with inflammation-related diseases. As previously discussed, many of these conditions are also highly prevalent as late effects in CCS cohorts. Therefore, we posit that microbiome-focused interventions which have been successful in adults with cardiometabolic diseases may also help ameliorate late effects in CCS (Figure 2).
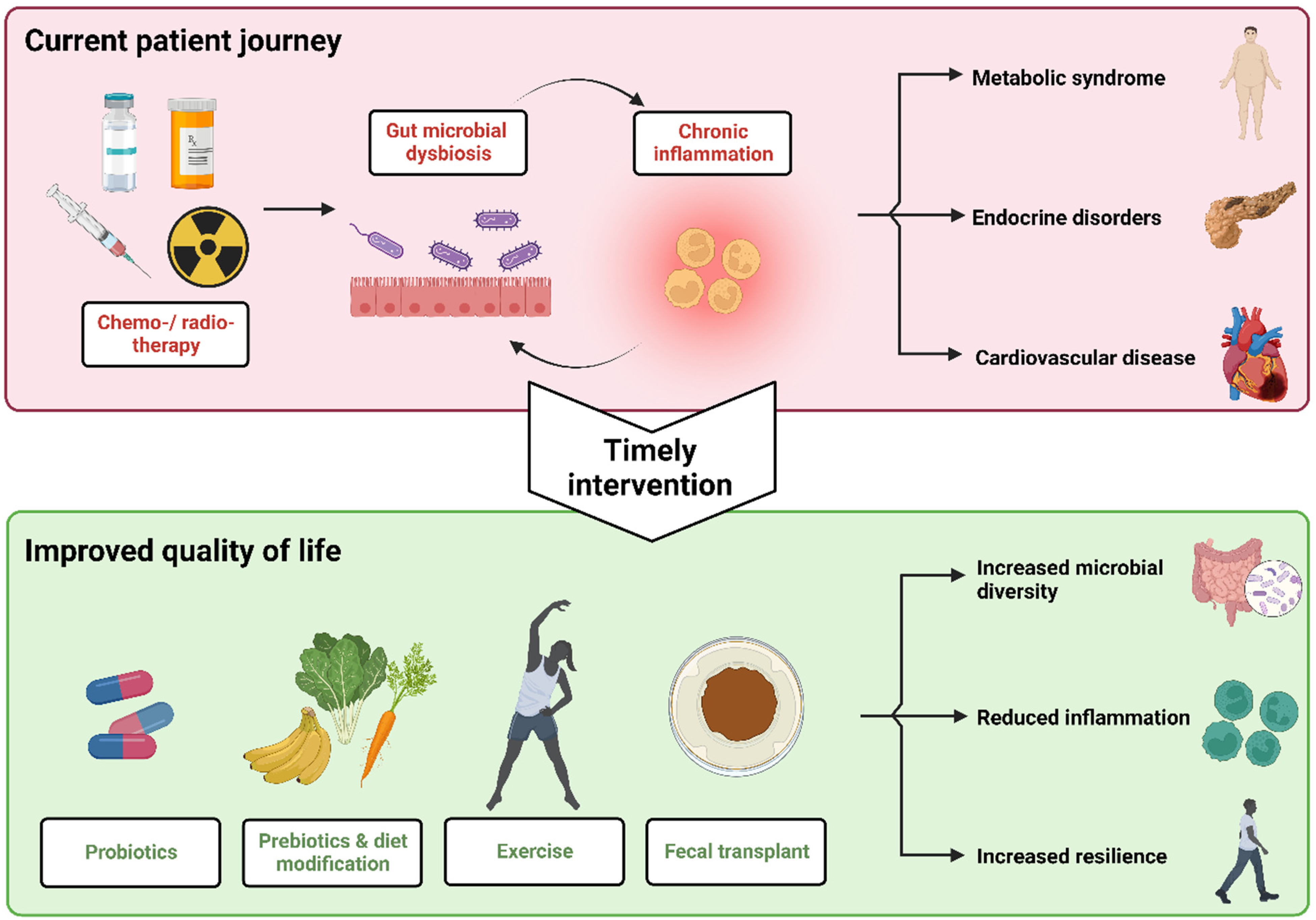
Potential health journey of childhood cancer survivors using interventions to address microbial dysbiosis and late effects (Figure created using BioRender under Academic License).
Reviewing recent literature, we identified 4 primary approaches which have shown promising therapeutic applications to address CCS late effects, namely supplementation with oral probiotics, consumption of a ‘healthy’ diet rich in prebiotics, exercise, and fecal microbial transplantation.
Probiotic Supplementation
Probiotics are a promising intervention for the restitution of dysbiotic microbial communities. Probiotic strains confer health benefits through a number of mechanisms, including the restoration of host microbial communities, the competitive exclusion of pathogens, improvements to gut barrier integrity, and the generation of metabolites that can influence host immunity and metabolism.48,49 Typical probiotic supplements consist of either homogenous or heterogenous Bifidobacterium, Lactobacillus, and Saccharomyces spp. Several additional species—including Faecalibacterium prausnitzii, Akkermansia muciniphila, and Clostridia spp.—are currently under evaluation for therapeutic use. 48
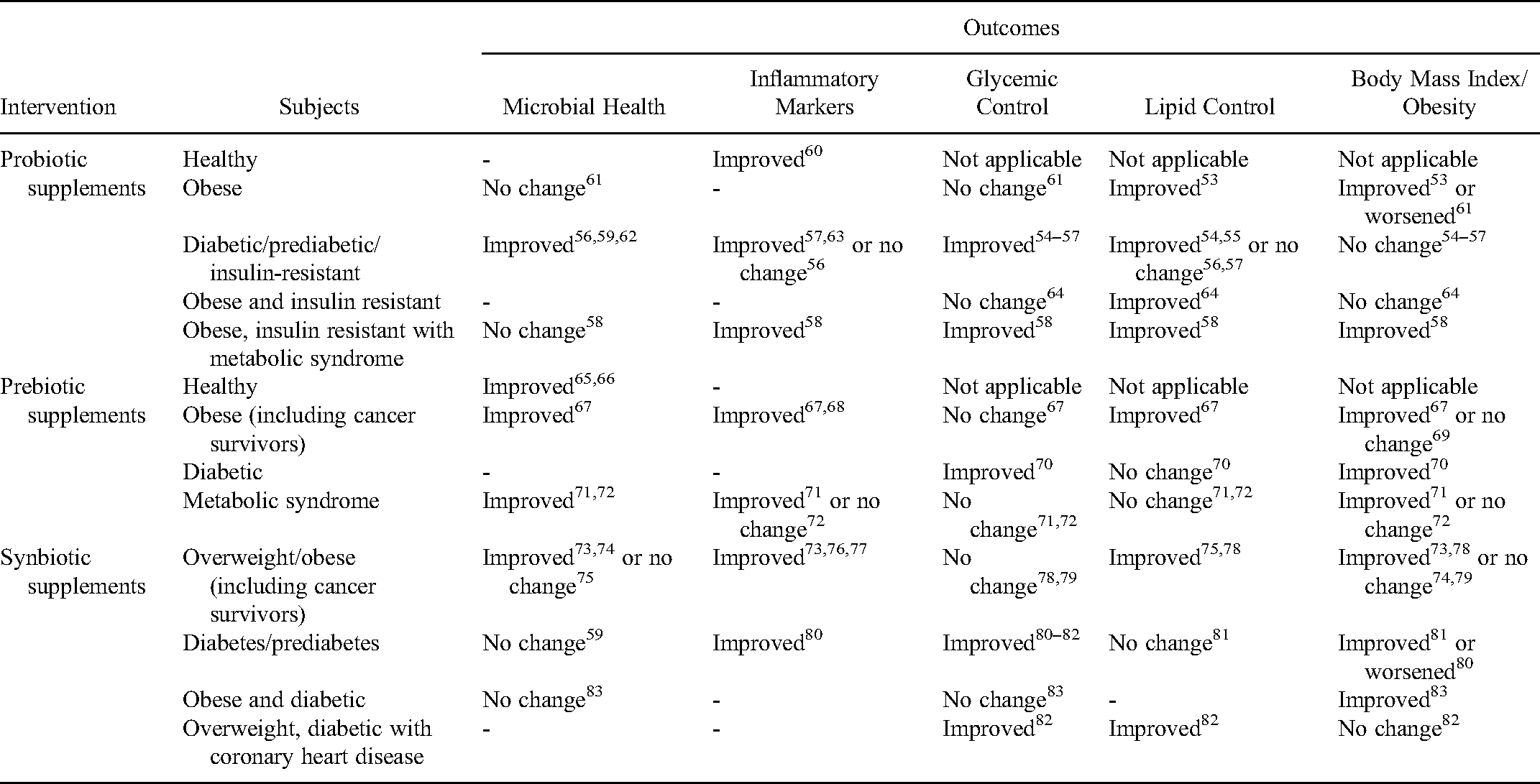
Probiotics have demonstrated therapeutic potential for a number of inflammation-related metabolic conditions including obesity, insulin resistance, and T2DM.50–52 Numerous randomized controlled trials (RCT) investigating the effect of probiotic supplementation in patients with cardiometabolic risk factors have reported reduction in fasting glucose and HbA1c in diabetic subjects, as well as body fat reduction and improved lipid metabolism in obese/overweight subjects (Table 1 and Supplemental material, Appendix 1).52–58 For example, probiotic supplementation of Lactobacillus acidophilus, Bifidobacterium lactis, and Lactobacilus casei over the course of 8 weeks significantly reduced body fat and increased fat-burning adropin in 30 obese adults. 53 Similarly, a 6-month intake of L. acidophilus, B. lactis, B. bifidum, and B. longum in prediabetic adults (n = 40) increased Bacteroidetes/Firmicutes ratio, reduced blood glucose and blood pressure, and increased high-density lipoprotein (HDL).54,59 Many studies also reported a significant reduction in inflammatory markers in treatment cohorts, a finding which is particularly relevant for inflammaging-prone CCS populations. 52 Considering the increased prevalence of metabolic disorders in CCS, these interventions are highly promising for the treatment of late effects. Recent clinical trials are summarized in Supplemental material, Appendix 1.
Outcomes of probiotic, prebiotic and synbiotic oral supplementation in healthy adults and in patients with cardiometabolic conditions.
Diet and Prebiotics
Animal studies indicate that diet—particularly intake of fats and complex carbohydrates—may modulate gut microbiota composition and consequently, metabolic health.84,85 Human observational studies across globally distinct populations have demonstrated that diets high in animal protein and saturated fats correlate with reduced microbiota richness and diversity. 86 Alternatively, plant-based diets high in complex carbohydrates were associated with high microbial diversity and increased abundance of Prevotella and butyrate-producing bacteria (Faecalibacterium, Roseburia sp., Eubacterium rectale, Ruminococcus bromii). 87 Within the context of the gut microbiome, these complex carbohydrates are often referred to as prebiotics, or “a substrate that is selectively utilized by host microorganisms, conferring a health benefit.” 88 Prebiotics such as inulin, fructo-oligosaccharides (FOS), and galactooligosaccharides (GOS) work symbiotically to nurture the growth of beneficial bacteria/probiotics. 89
Sufficient prebiotic intake can improve microbial environment through promotion of Bifidobacteria, Lactobacillus, and butyrate-producing bacteria (Table 1).65–67,71,72,90,91 Such microbial modulation has been correlated with an increase in SCFAs and decrease in inflammatory response, as evident in overweight/obese children and adults with metabolic risk factors or T2DM (Table 1 and Supplemental material, Appendix 2).67,71,91–97 In a RCT of 22 overweight/obese children, 16-week FOS-enriched inulin supplementation reduced levels of IL-6 while ameliorating obesity-related microbial dysbiosis. 67 Likewise, an 8-week whole grain diet reduced serum CRP, IL-1B, and IL-6 in adults with metabolic risk factors (n = 50). 71 Similar effects were also shown in individuals with T2DM.94–97
With regards to the efficacy of prebiotics in directly improving clinical outcomes, studies have been generally promising, albeit varied. Prebiotics such as FOS and/or inulin were shown to reduce body fat and body mass index (BMI) while improving satiety in obese/overweight individuals.98,99 Moreover, prebiotic-mediated reduction in serum triglycerides, cholesterol, and low-density lipoprotein, as well as elevation of HDL has been reported in individuals with T2DM.94,100 Conversely, some studies noticed modulation in microflora compositions without changes in metabolic parameters.71,72,101,102 The diverse impacts of prebiotic supplementation could be explained by variations in basal metabolic alteration within patient cohorts, as well as type of prebiotic intervention, dosing, and duration.
Recently, there has been an increasing interest in whether synbiotics—a combination of probiotics and prebiotics—may serve as a potential therapeutic approach for inflammation-related and metabolic diseases. Results from animal studies and clinical trials demonstrated that synbiotics may improve obesity, diabetes, and cardiovascular conditions (Table 1 and Supplemental material, Appendix 3).76–83,103 Of note, the positive effect of synbiotic supplementation in improving glycemic control among individuals with diabetes appears to surpass the benefit achieved with probiotics or prebiotics alone.55,64,67,71,72,79,80,82,100,104
Exercise
Exercise has been shown to alter both the gut microbiome and intestinal permeability; which in turn, control immune-inflammatory pathways and energy homeostasis.105–107 Sohail et al 108 reported that across several studies, exercise produced consistent global changes at a phylogenetic level (improvement in Bacteroidetes/Firmicutes ratio) with increased bacterial diversity and species richness. In a case-control experiment comparing sedentary to physically active rats, Queipo-Ortuno et al 109 reported increases in Lactobacillus, Bifidobacterium, and Blautia coccoides-Eubacterium rectale species with concomitant decrease in Clostridium and Enterococcus in the exercise group. Notably, both Bifidobacterium and B. coccoides-E. rectale have been associated with anti-inflammatory butyrogenic effects in the colon.109,110 Studies have also repeatedly demonstrated increased production of the beneficial SCFAs acetate, propionate, and butyrate by bacteria following exercise.111,112
In humans, a few observational studies have looked at differences in bacterial composition, inflammatory, and metabolic markers between athletes and controls, or among individuals with different exercise patterns. Clarke et al 113 demonstrated increased gut microbial diversity in professional rugby players compared to controls, and a significantly greater abundance of Akkermansia muciniphila in athletes with low BMI. Akkermansia is a mucin-degrading bacteria whose abundance is inversely correlated to obesity and metabolic syndrome. 114 In a large study involving 1493 participants from the American Gut Project, individuals who exercised regularly had significantly greater alpha diversity and increased prevalence of Faecalibacterium. 115 Comparing exercise interventions in healthy elderly women, Morita et al 116 found that aerobic exercise training through brisk walking (1 hour daily for 12 weeks) resulted in a significant increase in the relative abundance of intestinal Bacteroides compared to those who received trunk muscle training. In summary, the studies reviewed above support the use of exercise to improve gut microbial health and present a particularly attractive intervention for CCS.
Fecal Microbiota Transplantation
Gut microbiota may be directly altered through a therapeutic method known as fecal microbiota transplantation (FMT). The successful use of FMT was first reported in adult patients with Clostridium difficile infection (CDI) with restoration of healthy microbial flora in recipients and subsequent resolution of symptoms.117,118 Beyond CDI, FMT has also been implemented in patients with functional gut disorders, inflammatory bowel disease, and conditions associated with alteration in gut microbiota such as obesity, metabolic syndrome, and T2DM.119–123 Studies on the use of FMT in patients with obesity and metabolic syndrome have shown a positive association between increased abundance of butyrate-producing bacteria and insulin sensitivity, although neither were sustained long term.119,120 Additionally, a recent study examined the use of washed microbiota transplantation (WMT)—a method involving microfiltration and repeated centrifugation and suspension. A significant short and medium-term improvement in lipid profile was reported in 90 human subjects with hyperlipidemia without additional dietary interventions. 124
A synergistic effect of FMT with diet alteration appears promising in sustaining engraftment of donor fecal microbiota. Mocanu et al 125 showed that low-fermentable fiber supplementation following a single dose of oral FMT in 17 individuals with severe obesity and metabolic syndrome significantly improved insulin sensitivity at 6 weeks, resulting in increased microbial diversity and composition. Interestingly, patients who received high-fermentable fiber supplementation alone had reduced TNF-α and IL-8, but these markers returned to baseline after fiber cessation and there was no association with glycaemic outcomes. 125 In a randomized study in which 16 patients with T2DM received a specially designed diet of prebiotics, probiotics, and whole grains, either with or without the addition of FMT for 90 days, Su et al 126 successfully demonstrated improvements in glycaemic control and blood pressure as well as early weight loss in the treatment cohort. The authors also noted a change in the dominant intestinal microbiota genera from Bacteroides to Prevotella, regardless of dominant bacterium genera in donors. The overall effect was an increase in the abundance of Bifidobacterium and a decrease in prevalence of Bilophila and Desulfovibrio, which have been positively correlated with hyperglycemia. In summary, FMT with the addition of a healthy diet appears to be promising in addressing gut microbial dysbiosis and its consequences, particularly for CCS populations experiencing metabolic late effects.
Conclusion
Late effects faced by CCS pose a major health challenge. In the United States, for example, approximately 1 in 640 young adults in the 20- to 39-year age group are survivors of childhood cancer. 127 Thus, management of chronic comorbidities arising from late effects presents an enormous burden to national healthcare expenditure as well as personal well-being.
Basic lifestyle advice, such as exercise and healthy diet, may seem redundant to patients who have completed their cancer journey. However, microbial reconstitution as a targeted therapeutic intervention requires a highly nuanced understanding of the mechanisms driving chronic health problems in CCS. Inflammaging may start well before the diagnosis of CCS-prevalent diseases: for example, in a CCS cohort with a median age of 25, we detected evidence of inflammaging and significant leukocyte telomere shortening in the majority of patients despite the absence of overt T2DM or hypertension. 128 These findings suggest that the optimal window of intervention may be significantly earlier than the current standard, which is instituting treatment upon clinical manifestation of late effects.
Since the timeline of microbiome-driven inflammaging in CCS remains largely unknown, the possibility of gut bacterial dysbiosis should be considered from the moment of diagnosis. Moreover, early intervention—even while undergoing treatment—could preempt late effects. Thus, we recommend implementing all feasible interventions across the timeline of cancer treatment and recovery: for example, judicious use of broad-spectrum antibiotics, as well as encouraging patient consumption of prebiotic-rich foods from the onset of cancer diagnosis. For safety reasons, low-risk interventions such as exercise and probiotic regimens may be better implemented after the completion of cancer therapy. Moreover, these practices have the potential to improve treatment-related dysbiosis in other parts of the alimentary canal: the oral cavity, for example.129–135
There is the potential that treatment-induced microbial dysbiosis could eventually be avoided altogether. Many emerging cancer therapeutics promise targeted mechanisms and a reduction in adverse effects, which may eventually include perturbations to gut flora. Notably, bacteria-based cancer therapy (BBCT) has the potential to target tumor microenvironments and increase the precision of drug delivery.136,137 Genetic engineering of nonpathogenic bacteria in particular may lower host pathogenicity and increase antitumor efficacy without driving late effects in CCS.138,139 On the other hand, BBCT inherently activates host antitumor immune response, which may trigger inflammation and disturb gut microbiota.136,137 Furthermore, 2 relevant complications of BBCT—high levels of bacterial toxins and undesired colonization of implanted medical devices 140 —may drive increased use of antibiotics which exacerbate microbial dysbiosis in cancer patients. 141 Long-term clinical trials are needed to evaluate BBCT as well as other targeted cancer therapeutics to determine impact on late effects.
The main challenge of microbiome-focused interventions is that of sustainability, which was a common theme for all 4 approaches examined. Changes to the microbiome through probiotic and prebiotic supplementation, healthy diet, exercise, and FMT all require long-term commitment in order to achieve clinical benefits.59,65,68,75,115,119,120 Additionally, the success of each approach was found to be highly dependent on a number of host factors, including age, ethnicity, and health conditions, as well as methodological factors: for example, type and volume of exercise, or quantity/strain of probiotic.61,65,68,108,116,142 In many cases, these factors could explain the variability that we noted in study outcomes, particularly when it came to pre-, pro-, and synbiotic supplementation. Altogether, these findings suggest that restoration of the microbiome requires a treatment plan that is intentional, highly targeted, and with the long term in mind.
As such, measures would need to be taken to encourage strict treatment plan adherence by CCS. Exercise and a healthy diet, which promote gut health, are typically considered “lifestyle preferences,” as opposed to “therapy,” and thus may not compel adherence. Specificity in treatment plans—such as the optimal amount of time/type of exercise (eg, ≥30-minute sessions, thrice weekly) and the number of servings of fruits and vegetables as well as components such as flavonoids may encourage the formation of long-term habits, particularly in adolescents or young adults.143,144 Monitoring/tracking apparatuses, such as electronic diet diaries and wearable tracking devices, should appeal to young adults and may prove more accurate than self-reporting.145,146 Overall, we believe that reviewing diet, exercise, and biomarkers of inflammaging such as plasma CRP could be the new paradigm for late effects’ surveillance in CCS attending long-term follow-up clinics. 128 Focused strategies to address microbial dysbiosis, most of which are easily implementable, should be incorporated into the holistic management of children who have been diagnosed with or survived malignancy.
Supplemental Material
sj-docx-1-tct-10.1177_15330338221149799 - Supplemental material for Manipulating the Gut Microbiome as a Therapeutic Strategy to Mitigate Late Effects in Childhood Cancer Survivors
Supplemental material, sj-docx-1-tct-10.1177_15330338221149799 for Manipulating the Gut Microbiome as a Therapeutic Strategy to Mitigate Late Effects in Childhood Cancer Survivors by Lixian Oh, PhD, Syaza Ab Rahman, MBBCh, Kailey Dubinsky, BSc, Mohamad Shafiq Azanan, PhD, and Hany Ariffin, MBBS, PhD in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by a grant from the Ministry of Higher Education, Malaysia (grant number FRGS/FP006-2021).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
